Abstract
Biodiesel is an alternative to petroleum-based fuels derived from a variety of feedstocks, including vegetable oils, animal fats, and waste cooking oil. At present, biodiesel is mainly produced from conventionally grown edible oils such as soybean, rapeseed, sunflower, and palm. The cost of biodiesel is the main obstacle to commercialization of the product. Biodiesel produced from edible oils is currently not economically feasible. On the other hand, extensive use of edible oils for biodiesel production may lead to food crisis. These problems can be solved by using low-cost feedstocks such as non-edible oils and waste cooking oils for biodiesel production. This paper reviews numerous options of non-edible oils as the substantial feedstocks, biodiesel processing, and effect of different parameters on production of biodiesel.
Introduction
Interest in the use of biofuels worldwide has grown strongly in recent years due to the limited oil reserves, concerns about climate change from greenhouse gas emissions and the desire to promote domestic rural economies (Balat and Balat, 2009; Ozturk, 2014). The term biofuel is as such referred to solid, liquid, or gaseous fuels that are produced from plant matter and residues, agricultural crops, municipal wastes, and agricultural and forestry by-products (Aburas and Demirbas, 2015a; Qin et al., 2009, 2010; Altun and Yasar, 2013). Liquid biofuels can be used as an alternative to traditional fossil fuels in the transportation sector (Aburas, 2015).
Globally, the transportation sector is a major source of energy consumption and carbon dioxide (CO2) emissions. This sector is responsible for 18% of primary energy consumption (Wang et al., 2014) and 23% of all energy-related CO2 emissions globally (Du Ploy and Nel, 2012). The world energy demand continues to increase. This growth will affect the stability of ecosystems and global climate as well as global oil reserves. The most feasible way to meet this growing demand is by utilizing alternative fuels (Demirbas et al., 2015a, 2015b). Production of biofuel from biomass is one way to reduce both the consumption of crude oil and environmental pollution (Ayanoglu and Aksoy, 2015).
Global production of biofuels has been growing rapidly in recent years, rising from about 18 billion liters in 2000 (Kemausuor et al., 2013) to 110 billion liters in 2013 (IEA, 2014). It is expected to reach to 140 billion liters by 2018 (IEA, 2014). Current consumption of biofuels globally constitutes about 3.5% of transportation fuels by energy content, and this could expand to about 4% by 2018 (IEA, 2014).
Majority of liquid biofuels is produced in the form of bioethanol (around 80%) and the rest constitutes biodiesel (Figiel and Hamulczuk, 2014). The global biodiesel industry is one of the fastest-growing markets. Since 2004, global biodiesel production increased 15-fold, while bioethanol production grew nearly fourfold (REN21, 2014). This paper reviews different processing techniques for biodiesel production from common non-edible vegetable oils and determines the most suitable one.
Raw materials for biodiesel production
Biodiesel production is a very modern and technological area for researchers due to the relevance that it is winning every day because of the increase in the petroleum price and the environmental advantages (Aburas and Demirbas, 2015b; Marchetti et al., 2007). Biodiesel can be derived from a variety of sources including vegetable oils, animal fats, and waste cooking oil.
Fuel-related properties of selected vegetable oils.
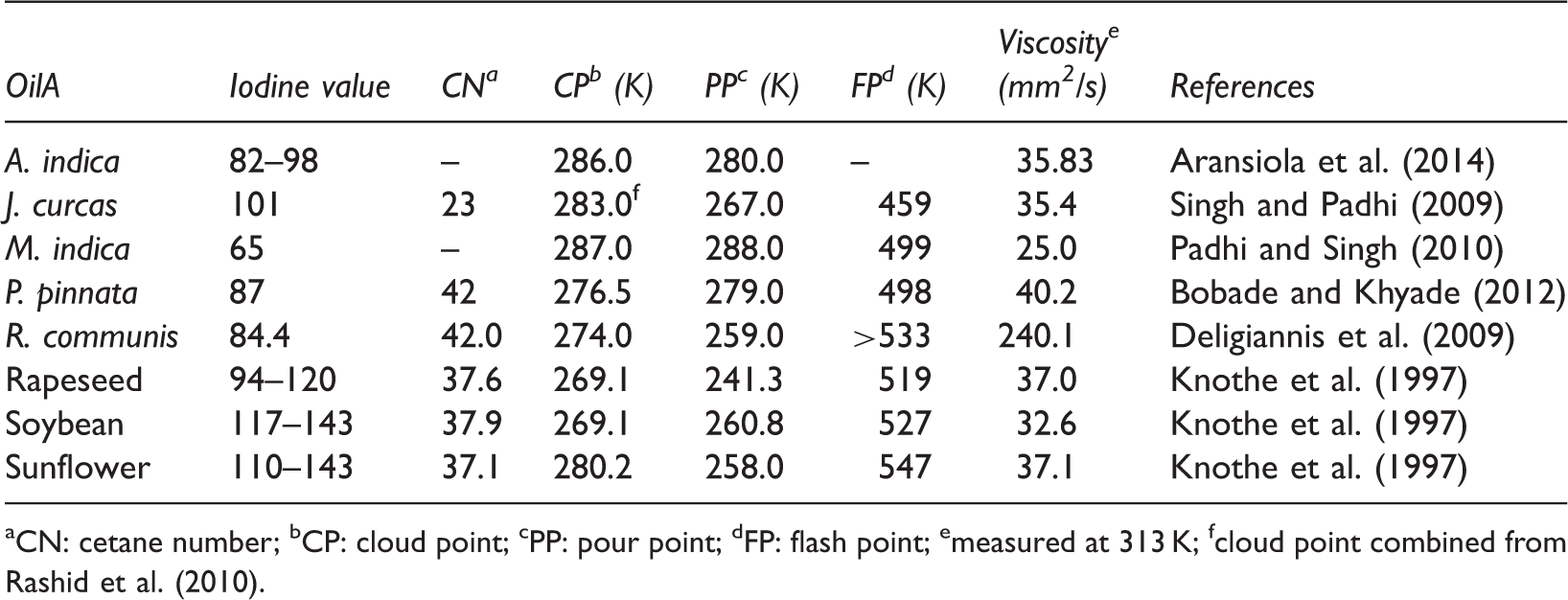
CN: cetane number; bCP: cloud point; cPP: pour point; dFP: flash point; emeasured at 313 K; fcloud point combined from Rashid et al. (2010).
Most of the biodiesel is currently made from soybean, rapeseed, sunflower, and palm oils (Demirbas, 2015b). New plant oils that are under consideration include mustard seed, peanut, sunflower, and cotton seed. Soybean oil is commonly used in the United States and rapeseed oil is used in many European countries for biodiesel production, whereas, coconut oil and palm oils are used in Malaysia and Indonesia for biodiesel production (Ghadge and Raheman, 2005; Srivastava and Verma, 2008). About 80% of the European Union’s total biofuel production is comprised of biodiesel produced from rapeseed and sunflower seeds (Ahmad et al., 2010). Soybean oil accounts for approximately 90% of the biodiesel produced in the United States (Balat and Balat, 2010). Rapeseed oil has 59% of total global biodiesel raw material sources, followed by soybean (25%), palm oil (10%), sunflower oil (5%), and other (1%) (Pahl, 2008).
Food versus fuel
The major obstacle for commercialization of biodiesel is its cost approximately 70%–90% of biodiesel cost is arises from the cost of raw materials (Shikha and Rita, 2012). Therefore, biodiesel produced from edible vegetable oils is currently not economically feasible. Non-edible oil plants are easily available in developing countries and are very economical comparable to edible plant oils. The production of biodiesel from different non-edible oilseed crops has been extensively investigated over the last few years.
Extensive use of edible oils may cause other significant problems such as starvation in developing countries. Using edible oils to produce biodiesel raises major nutritional and ethical concerns. About 13% of global vegetable oil supplies were used for biodiesel production in 2012. World annual petroleum consumption and vegetable oil production is about 4.018 and 0.107 billion tons, respectively (Demirbas, 2010). Biodiesel production accounted for 25.6% of the global demand for rapeseed oil, 16.1% of the global soybean oil demand, and 5.9% of global palm oil demand in 2012.
On the other hand, extensive use of edible vegetable oils may cause other significant problems such as starvation in developing countries. There are concerns that biodiesel feedstock may compete with food supply in the long term (Chhetri et al., 2008). Biodiesel produced from non-edible vegetable oil has good potential as an alternative diesel fuel. The use of non-edible plant oils when compared with edible plant oils is very significant because of the tremendous demand for edible oils as food, and they are far too expensive to be used as fuel at present (Mahanta et al., 2006). Non-edible oil plants can be grown in waste lands that are not suitable for food crops and the cost of cultivation is much lower because these plants can still sustain reasonably high yield without intensive care (Fatah et al., 2012). Non-edible oil plants are well adapted to arid, semi-arid conditions and require low fertility and moisture demand to grow (Atabani et al., 2013).
Non-edible vegetable oils as raw material
Comparison of fatty acid composition between various edible and non-edible vegetable oils.
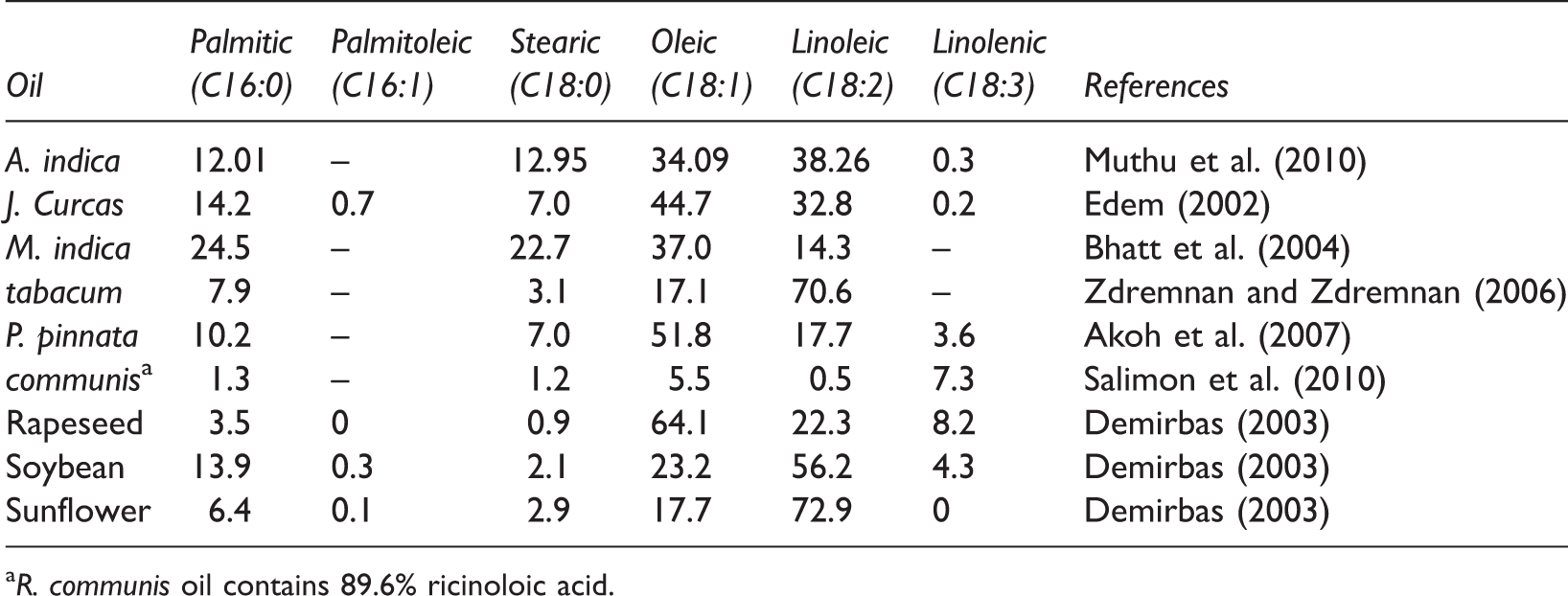
R. communis oil contains 89.6% ricinoloic acid.
The oil content in seeds and kernels of some non-edible plants.
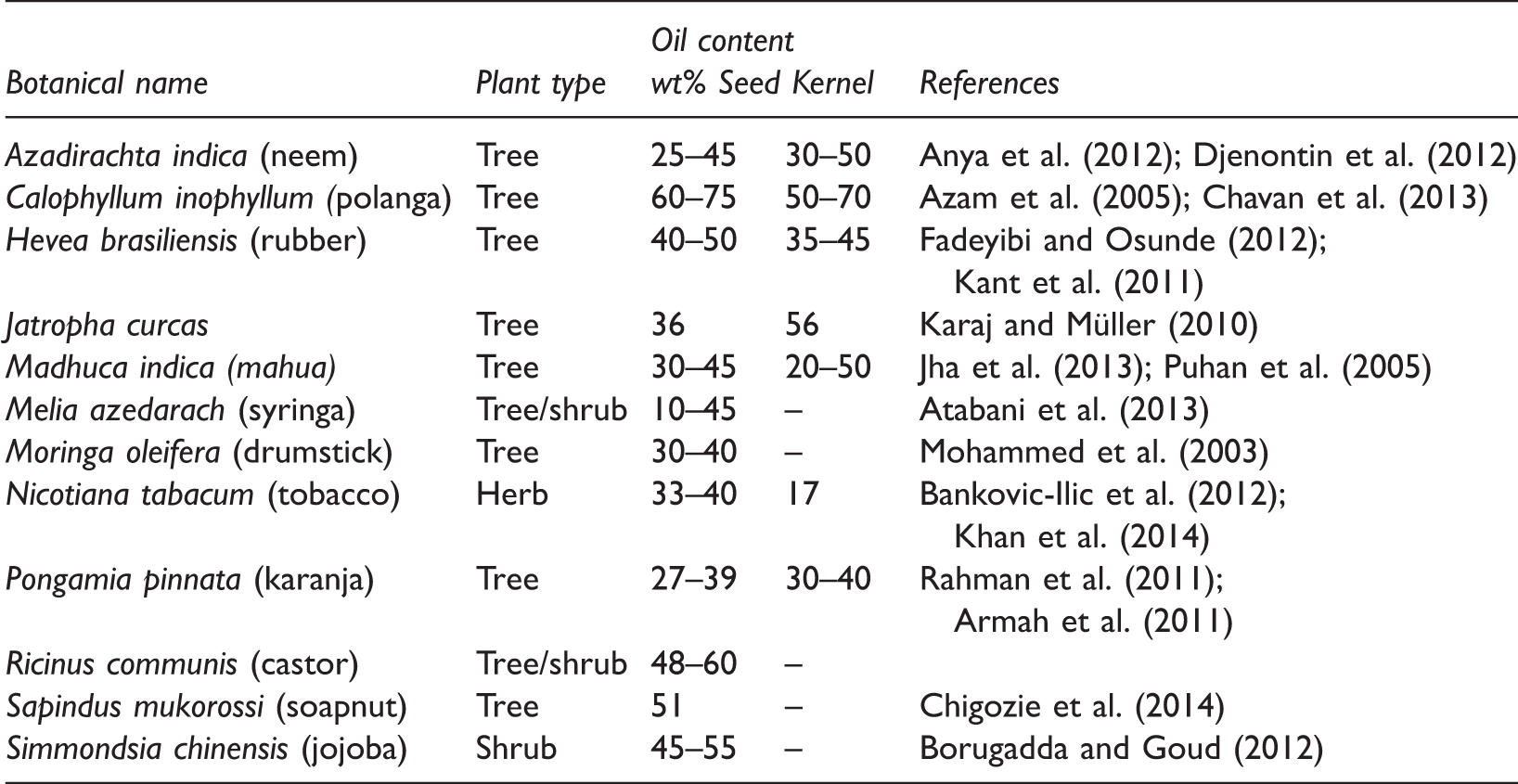
Jatropha curcas
Jatropha curcas is a tall bush or small tree, up to 5–7 m tall, belonging to the Euphorbiaceae family (Atabani et al., 2013). Originally from Central America, Jatropha curcas is found throughout the tropics including much of Africa and Asia (Nahar and Sunny, 2014). It has been planted in several arid regions, in these regions it only yields about 0.5 ton per hectare (Balat and Balat, 2010). The plant is important for climate change issues as a mature plant or tree absorbs CO2 at a rate of 18 lbs per year (Nahar and Sunny, 2014). A research study (Becker et al., 2013) showed that one hectare of Jatropha curcas could capture up to 25 tons of CO2 from the atmosphere every year (over a 20-year period). Jatropha curcas oil is a leading candidate for the commercialization of non-edible vegetable oils (No, 2011). Jatropha curcas seeds have an oil content ranging between 30% and 40% (Pandey et al., 2012). Jatropha curcas oil contains approximately 24.60% of crude protein, 47.25% of crude fat, and 5.54% of moisture content (Akintayo, 2004). Most of the non-edible oils including Jatropha curcas carry a high level of FFAs. The oil fraction of Jatropha curcas consists of both saturated (14.2% palmitic acid and 7.0% stearic acid) and unsaturated fatty acids (44.7% oleic acid and 32.8% linoleic acid) (Table 2).
Pongamia pinnata (Karanja)
Pongamia pinnata, commonly known as karanja, is a medium-sized glabrous, perennial tree grows in the littoral regions of South Eastern Asia and Australia (Satyavati et al., 1987). It is a hardy tree of 12–15 m height, branches spread into hemispherical crown of dense green leaves (Naik and Katpatal, 2013). The yield of oilseed per tree is between 8 and 24 kg (Doshi and Srivastava, 2013). The seeds of Pongamia pinnata contain around 30%–40% of oil (Scott et al., 2008). Pongamia pinnata oil is considered to be less toxic and cheaper than Jatropha curcas oil, so it has become the subject of biodiesel research (Khayoon et al., 2012). Most of the physical and chemical properties of the Pongamia pinnata oil are similar to those of the diesel fuel, however this oil is more viscous and produce higher carbon residue (Meenakshi, 2014). Pongamia pinnata oil contains oleic acid (51.8%) as the major fatty acid followed by linoleic (10.7%), palmitic (10.7%), and stearic (7%) acids (Table 2).
Madhuca indica (Mahua)
Madhuca indica, commonly known as mahua or butternut tree, is a middle-sized large deciduous tree, which grows to a height of 10–15 m (Chidrewar et al., 2010). It belongs to the family Sapotaceae. The Madhuca indica tree starts producing seeds after 10 years and continues for up to 60 years (Pandey, 2008). An average yield of 800 kg/ha can be expected in a Mahua plantation after a decade (Kant et al., 2011). Each tree yields about 20–40 kg of seed per year (Borugadda and Goud, 2012). Madhuca indica seed contains 35% oil and 16% protein (Panigrahi et al., 2014). Fresh Madhuca indica oil from properly stored seeds is yellow, while commercial oils are generally greenish yellow with an offensive odor and disagreeable taste (Puhan et al., 2005). The Madhuca indica oil contains a high level of FFAs (up to 20%) (Ghadge and Raheman, 2005) and a procedure for converting this oil to biodiesel is very much required. FFA composition of Madhuca indica oil is shown in Table 2.
Ricinus communis (Castor)
Ricinus communis, commonly known as castor oil plant, is a plant species of the family Euphorbiaceae. It originates in Africa but it is found in both wild and cultivated states in all the tropical and subtropical countries of the world (Forero, 2004). Ricinus communis is a small wooden tree that can reach a height of about 6 m. The comparative advantage of Ricinus communis is that its growing period is much shorter than that of Jatropha curcas and Pongamia pinnata, and there is considerably greater experience and awareness among farmers about its cultivation (Rajagopal, 2007). Ricinus communis is an ideal candidate for production of high value, industrial oil feedstocks because of the very high oil content (48%–60%) of the seed, and the extremely high levels of potential oil production (500–1000 l of oil/acre). The main constituent of Ricinus communis oil is ricinoleic acid (90%), which contains 18 carbon atoms with a hydroxyl group position 12. Therefore, Ricinus communis oil contains more oxygen than other oils and, therefore, Ricinus communis oil and its derivatives are more soluble in alcohols during the transesterification reactions. The main disadvantage of Ricinus communis oil is its high viscosity (240.12 mm2/s at 313 K) (Deligiannis et al., 2009). The high viscosity of Ricinus communis oil to its poor atomization of the fuel, incomplete combustion, choking of the fuel injectors, and ring carbonization (Baroutian et al., 2010).
Azadirachta indica (Neem)
Azadirachta indica (commonly known as “neem tree”), a member of the family Meliaceae, is a majestic, evergreen, tropical forest tree, with a broad crown and a height of approximately 25 m. Most parts of the Azadirachta indica plant such as leaves, bark, flower, fruit, seed, and root have applications in the field of medicine. Azadirachta indica is well established plant in at least 30 countries worldwide, in Asia, Africa, and Central and South America. Some small scale plantations are also reportedly successful in Europe and United States of America (Kumar and Navaratnam, 2013). Azadirachta indica tree normally begins to bear fruit between 3 and 5 years and becomes fully productive in 10 years. From the 10th year onwards, it can produce up to 50 kg of fruits annually (Girish and Shankara, 2008). It requires little water and plenty of sunlight and rainfall in the range of 450 to 1200 mm with wide temperature range of 273 to 322 K (Heroor and Bharadwaj, 2013). Azadirachta indica seeds contain about 45% of a brownish-yellow of fixed oil (Radha and Manikandan, 2011), mainly constituted by oleic acid (34.01%), palmitic acid (12.1%), stearic acid (12.95%), linoleic acid (38.26%) (Muthu et al., 2010). Traditionally, Azadirachta indica oil has been used as fuel in lamps for lighting purpose in rural areas and it is used on an industrial scale for manufacturing of soaps, cosmetics, pharmaceuticals, and other non-edible products (Gahadwal and Yadav, 2014).
Hevea brasiliensis (Rubber)
The yields of various non-edible oil feedstocks.
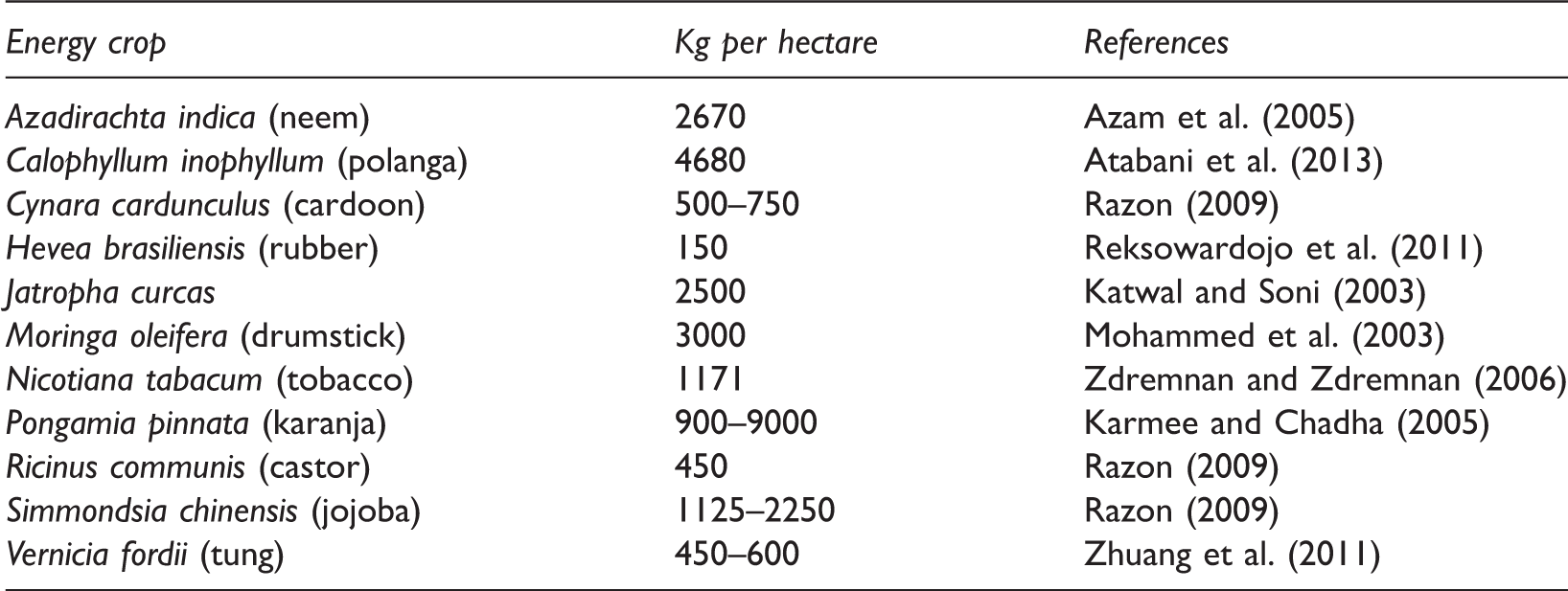
Nicotiana tabacum (Tobacca)
Nicotiana tabacum, commonly referred to tobacco, is an annually grown herbaceous plant belonging to the Solanaceae family, widespread in North and South America, commonly grown for the collection of leaves. The highest seed production is found in the Nicotiana tabacum varieties used to obtain the chewing Nicotiana tabacum, reaching 1171 kg seeds per hectare, which corresponds to 432.9 kg oil per hectare (Zdremnan and Zdremnan, 2006). The seed oil content ranges between 33 and 40 wt% (Khan et al., 2014). The major fatty acids in seed triacylglycerols are linoleic acid (70.6%), oleic acid (17.1%), palmitic acid (7.9%), and stearic acid (3.1%) (Table 2).
Rice bran
Rice is the main cultivation in subtropical Southern Asia, and it is a staple food for a large part of the world’s human population especially in East, South, and South Eastern Asia, making it the most consumed cereal grain (Subbaiah et al., 2010). Rice bran is a low value co-product of rice milling, which contains approximately 15%–23% oil (Sinha et al., 2008). The oil fraction of rice bran consists of both saturated (15.0% palmitic acid and 1.9% stearic acid) and unsaturated fatty acids (42.5% oleic acid and 39.1% linoleic acid) (Mohanty, 2013). Rice bran oil is an underutilized non-edible vegetable oil, which is available in large quantities in rice cultivating countries like China and India, and very little research has been done to utilize this oil as a replacement for mineral diesel (Sinha et al., 2008).
Moringa oleifera (Drumstick tree)
Moringa oleifera, also called drumstick tree, is a softwood perennial tree. This plant belongs to the monogeneric family Moringaceae and is an extremely fast growing tree that can get to 10 m. Moringa oleifera is most commonly cultivated in South India, Ethiopia, Philippines, Sudan, and has been grown in West, East and South Africa, tropical Asia, Latin America, the Caribbean, Florida, and the Pacific Islands (Fahey, 2005). The fast growing, drought-tolerant Moringa oleifera can tolerate poor soil, a wide rainfall range (25 to 300 + cm per year), and soil pH from 5.0 to 9.0 (Rashid et al., 2008). It has been reported that some 3000 kg of Moringa oleifera seeds could be obtained from one hectare, equivalent to 900 kg oil per hectare, comparable to soybean which also yields an average of 3000 kg seeds per hectare but with only 20% oil yield (Mohammed et al., 2003; Orhevba et al., 2013). Moringa oleifera seed has an oil content of between 30% and 40%, depending on the plant variety and climate (Mohammed et al., 2003). Moringa oleifera oil contains oleic acid (67.7%) as the major fatty acid followed by stearic (8.3%), behenic (7.4%), arachidic (4.7%), palmitic (6.9%), linoleic (10.7%), and eicosenoic (2.3%) acids (Sunga and Whitby, 1995).
Calophyllum inophyllum (Polanga)
Calophyllum inophyllum, commonly known as polanga or honne, is a large evergreen, belongs to the Clusiaceae family, widespread in East Africa, India, South East Asia, and Australia (Silitonga et al., 2013). Calophyllum inophyllum is a medium and large-sized evergreen sub-maritime tree that averages 8–20 m in height with a broad spreading crown of irregular branches (Ong et al., 2011). Each tree yields about 20–100 kg of whole fruits per year (Venkanna and Reddy, 2009). Trees begin to bear significantly after 4–5 years (Chavan et al., 2013). The nut kernel contains 50%–70% oil and the mature tree may produce 1–10 kg of oil per year depending upon the productivity of the tree and the efficiency of extraction process (Chavan et al., 2013). Traditionally, Calophyllum inophyllum oil has been used as a medicine, soap, lamp oil, hair grease, and cosmetic in different parts of the world (Ong et al., 2011).
Simmondsia chinensis (Jojoba)
Simmondsia chinensis, commonly known as jojoba, is a perennial shrub belonging to the Simmondsiaceae family (Moser, 2011). This plant is native to the Mojave and Sonoran deserts of Mexico, California, and Arizona. A 10-year old tree yields on an average 1 kg of seeds per year (Kant et al., 2011). Simmondsia chinensis is unique among plants in the fact that its seeds contain about 50% of oil by weight, which is more than amount in soybean and somewhat more than in most oil seed crops (Sharma and Singh, 2011). Simmondsia chinensis oil is practically colorless and odorless and it is composed mainly of straight chain monoesters of C20 and C22 acids and alcohols with two double bonds (Zaher et al., 2004).
Sapindus mukorossi (Soap nut)
Sapindus mukorossi (soap nut) is a perennial tree belonging to the family Sapindaceae, indigenous to northern India (Philomina and Rao, 2000). This plant grows very well in deep loamy soils and leached soils so cultivation of Sapindus mukorossi in such soil avoids potential soil erosion (Chhetri et al., 2008). Sapindus mukorossi tree can be used for rural building construction, oil and sugar presses, and agricultural implements (Chhetri et al., 2008). Sapindus mukorossi seeds contain 23% oil of which 92% is triglycerides (Atabani et al., 2013).
Melia azedarach (Syringa)
Melia azedarach (syringa) is a deciduous tree that grows between 7 and 12 m in height in the mahogany family of Meliaceae that is native to Pakistan, India, Bangladesh, Southeast Asia, and Australia (Moser, 2011; Sarin, 2012). The oil content of dried syringe berries is around 10 wt% (Atabani et al., 2013). Melia azedarach oil is characterized by a high percentage of unsaturated fatty acids such as oleic (21.8%) and linoleic (64.1%) acids (Moser, 2011). Other constituents that are present in greater than 1% are saturated species such as palmitic (10.1%) and stearic (3.5%) acids (Moser, 2011).
Vernicia fordii (Tung)
Vernicia fordii, commonly known as tung tree, is an oil-bearing woody plant belonging to the Euphorbecaeae family that is native to China, Burma, and Vietnam. It is a small- to medium-sized deciduous tree, which grows up to 20 m in height. The oil content of Vernicia fordii seeds and whole nuts is approximately 21 and 41 wt%, respectively (Yu et al., 2013), and the average yield of oil is about 450–600 kg per hectare (Zhuang et al., 2011). Its seed oil had been conventionally used in lamps for lighting, as well as an ingredient for wood paint and varnish (Wang et al., 2011). Tung oil principally contains the unusual conjugated fatty acid, eleostearic acid (9,11,13-octadecatrienoic acid; 63.8%), with linoleic (11.5%), oleic (8.6%), and behenic (8.4%) acids also present in significant quantities (Moser, 2011).
Schleichera oleosa (Kusum)
Schleichera oleosa, also called as kusum, is medium-sized (up to 40 m in height) deciduous or nearly evergreen tree belonging to the Sapindaceae family that is native to South and South East Asia. The fruits, seeds, and young leaves of this plant are edible and used for medicinal and dye purposes (Kant et al., 2011). The oil content of kusum seeds is 51%–62% but the yields are 25%–27% in village ghanis (oil mills) and about 36% oil in expellers (Acharya et al., 2011). Iodine value of the oil is 215–220 and its total fatty acid content is 91.6% (Acharya et al., 2011).
Derivatives of non-edible oils as diesel fuels
Non-edible vegetable oils can be used as fuels for diesel engines. The primary problems associated with using straight these oils as alternative fuel in compression ignition (CI) engines are caused by very high fuel viscosity and low volatility (Basavaraja et al., 2005). The high viscosity of non-edible vegetable oils can be reduced by several techniques which include dilution, microemulsion, pyrolysis, and transesterification.
Dilution of non-edible vegetable oils
The oil–diesel fuel blend is a simple way to reduce the viscosity of non-edible vegetable oil. This method does not require any chemical process. A number of researchers have examined the use of blends of conventional diesel fuel with non-edible vegetable oils, such as Jatropha curcas oil (Pramanik, 2003), Pongamia pinnata oil (Agarwal and Rajamanoharan, 2009), Azadirachta indica oil (Prasad and Agarwal, 2012), etc.
Pramanik (2003) studied the effect of temperature on viscosities of Jatropha curcas oil and its various blends at a temperature range from 298 to 348 K. Results of this study showed that viscosity values of 50:50 Jatropha curcas oil/diesel fuel and 40:60 Jatropha curcas oil/diesel fuel are close to diesel in the range of 328–333 K and at about 318 K, respectively, whereas the blend containing 30:70 Jatropha curcas oil/diesel fuel has viscosity close to diesel at the range of 308–313 K. In another study, Agarwal and Rajamanoharan (2009) investigated the performance and emission characteristics of a CI engine fueled with karanja (Pongamia pinnata) oil and diesel fuel blends and the effect of preheated oil on engine performances. They reported that karanja oil blends with diesel (up to 50% v/v) without preheating as well as with preheating could be replaced diesel for operating the CI engine with lower emissions and improved performance.
Using microemulsions for non-edible vegetable oils
Microemulsions are transparent, thermodynamically stable colloidal dispersions in which the diameter of the dispersed phase particles is less than one-fourth the wavelength of visible light (Goering et al., 1983). To reduce of the high viscosity of vegetable oils, microemulsions with immiscible liquids such as methanol and ethanol and ionic or non-ionic amphiphiles have been studied (Billaud et al., 1995).
Liquid fuel and biodiesel from non-edible vegetable oils via pyrolysis
Conversion of vegetable oils using pyrolysis type reactions represents a promising option for the production of biodiesel fuel. Pyrolysis is the conversion of one substance into another by means of heat or by heat with the aid of a catalyst (Sonntag, 1979). Thermal decomposition of triglycerides produces the compounds of classes including alkanes, alkenes, alkadienes, aromatics, and carboxylic acids. Different types of vegetable oils produce large differences in the composition of the thermally decomposed oil (Srivastava and Prasad, 2000).
In recent years, many investigators have studied the pyrolysis of non-edible vegetable oils and their cakes such as Jatropha curcas (Manurung et al., 2009), Madhuca indica (Kalaiselvan et al., 2014), tung (Lima et al., 2004), and castor oil (Figueiredo et al., 2009) to obtain the suitable alternative fuel for CI engine. For instance, Figueiredo et al. (2009) described the production of pyrolysis oil from castor seeds via a low temperature conversion process and showed that the use of this oil in a pyrolysis oil–diesel blend is a good option.
Biodiesel via transesterification from non-edible vegetable oils
Transesterification is the chemical reaction between a triglyceride and an alcohol (mostly methanol or ethanol) in the presence of a catalyst and consists of a sequence three consecutive reversible reactions where triglycerides are converted to diglycerides and then diglycerides are converted to monoglycerides followed by the conversion of monoglycerides to glycerol. In each step, an ester is produced and thus three ester molecules are produced from one molecule of triglycerides (Sharma and Singh, 2008). Ethanol is a preferred alcohol in the transesterification process compared to methanol because it is derived from agricultural products and is renewable and biologically less objectionable in the environment (Demirbas, 2003). However, methanol is preferable because of its low cost and its physical and chemical advantages (polar and shortest chain alcohol). Triacylglycerols are esters of long-chain carboxylic acids combined with glycerol. Carboxylic acids {R– C( = O)–O–H} can be converted into methyl esters {R–C( = O)–O– CH3} by the action of a transesterification agent (Demirbas, 2009a). Figure 1 shows the transesterification reaction of triglycerides with alcohol.
Transesterification of triglycerides with alcohol.
Transesterification of vegetable oils can be carried out by two ways: (a) catalytic transesterification and (b) non-catalytic transesterification methods.
Catalytic transesterification methods
Vegetable oils can be transesterified by heating them with a large excess of anhydrous methanol and a catalyst. Transesterification reactions can be catalyzed by alkalis, acids, or enzymes.
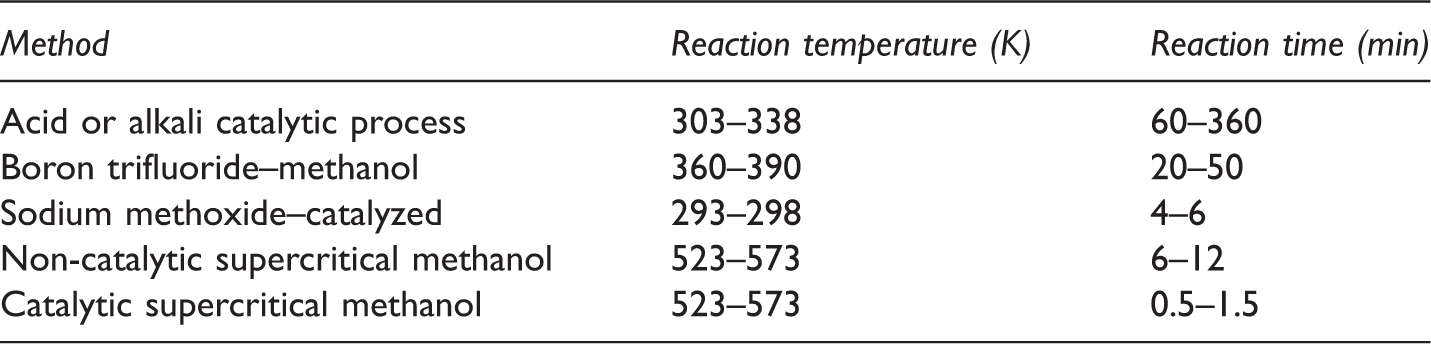
Comparison of various methanolic transesterification methods.
Transesterification can be catalyzed by alkaline catalysts, preferably by sodium or potassium methoxide (Muthu et al., 2010; Padhi and Singh, 2010; Prabaningrum, 2011; Thangaraj et al., 2014). Sodium methoxide (CH3ONa) is the most widely used biodiesel catalyst with over 60% of industrial plants using this catalyst (Huber et al., 2006). Alkaline catalysts have the advantages, e.g. short reaction time and relatively low temperature can be used with only a small amount for catalyst and with little or no darkening of color of the oil (Singh and Padhi, 2009). Alkaline metal alkoxides (as CH3ONa for the methanolysis) are the most active catalysts, since they give very high yields (>98%) in short reaction times (30 min) even if they are applied at low molar concentrations (0.5 mol%). However, they require the absence of water, which makes them inappropriate for typical industrial processes (Schuchardt et al., 1998). In the alkali catalytic methanol transesterification method, the catalyst is dissolved into methanol by vigorous stirring in a small reactor. The oil is transferred into a biodiesel reactor and then the catalyst/alcohol mixture is pumped into the oil. The final mixture is stirred vigorously for 2 h at 340 K in ambient pressure. A successful transesterification reaction produces two liquid phases: ester and crude glycerol. However, due to the high FFA content of non-edible oils, these oils cannot be transesterified in the presence of an alkaline base catalyst for it leads to catalyst deactivation and soap formation (Fadhil et al., 2012). For high FFA oils, two step acid-base catalyzed transesterification process is preferred. FFA content of oil could be reduced to less than 1% using an acid catalyst at the first stage. The second step is base catalyzed transesterification in which oil reacts with methanol in the presence of an alkaline catalyst to form ester and glycerol (Fadhil et al., 2012).
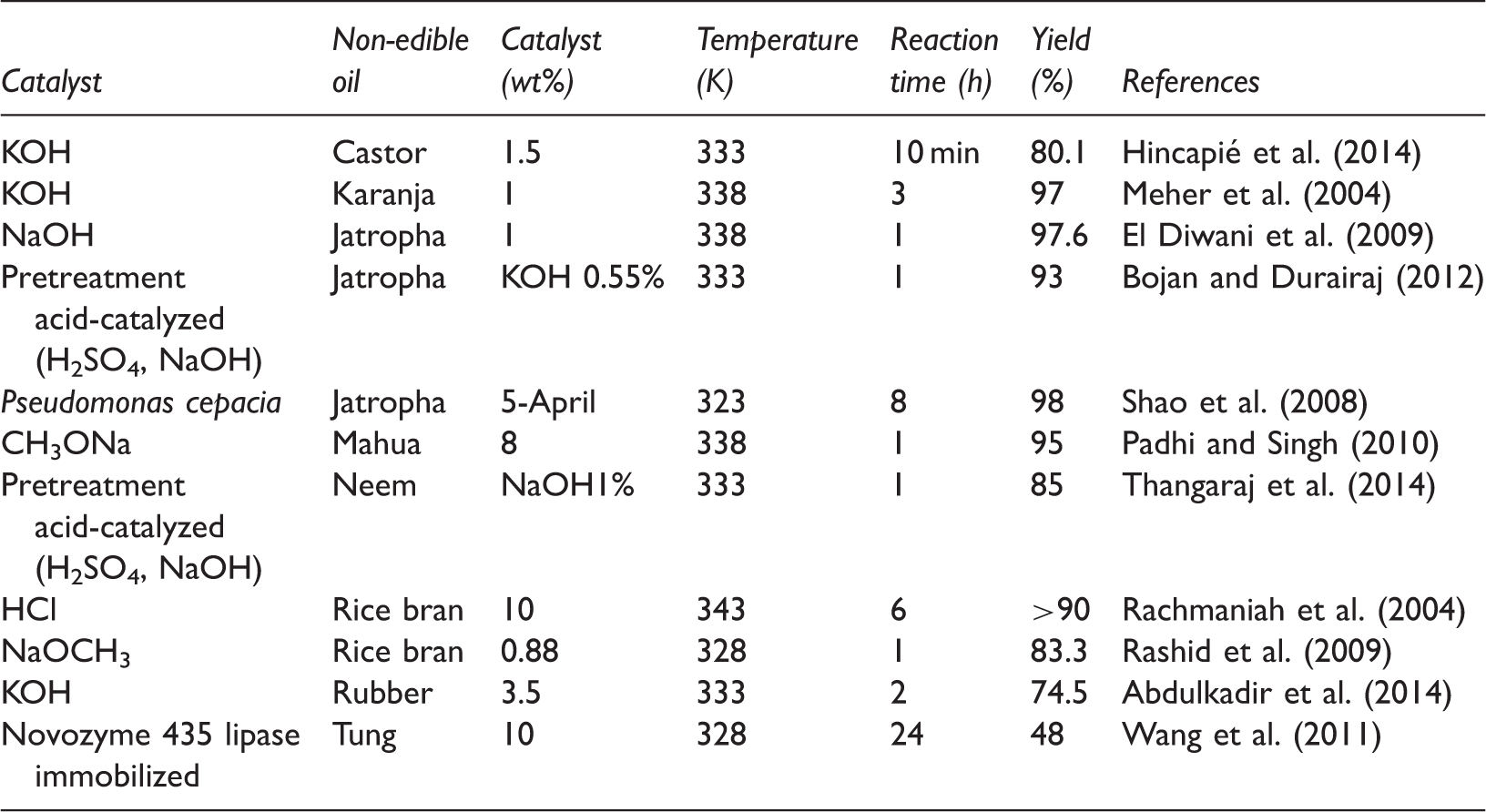
Summary of catalytic transesterification of some selected non-edible oils.
Non-catalytic transesterification methods
There are two basic routes to produce biodiesel by non-catalyzed transesterification. These are BIOX co-solvent process and supercritical alcohol process. BIOX co-solvent process uses either tetrahydrofuran (THF) or methyl tert-butyl ether as a co-solvent to generate a one-phase system. The result is a fast reaction, on the order of 5–0 min, and no catalyst residues in either the ester or the glycerol phase (Demirbas, 2009a). The THF co-solvent is chosen, in part, because it has a boiling point very close to that of methanol. This system requires a rather low operating temperature, 303 K (Helwani et al., 2009).
Comparisons between catalytic methanol (MeOH) method and SCM method for biodiesel from vegetable oils by transesterification.

SCM: supercritical methanol.
Effect of different reaction parameters on the biodiesel yield
The main factors affecting transesterification are alcohol/oil molar ratio, reaction time, reaction temperature and pressure, catalyst, water content, and FFA content. It is evident that at subcritical states of alcohol, the reaction rate is so low and gradually increased as either pressure or temperature rises (Madras et al., 2004). The most important variables affecting the methyl ester yield during transesterification reaction are alcohol/oil molar ratio and reaction temperature.
Effect of molar ratio and alcohol type
One of the most important variables affecting the yield of alkyl ester is the molar ratio of alcohol to oil. In the supercritical alcohol transesterification method, the yield of conversion raises 50%–95% for the first 10 min.
The stoichiometric ratio for transesterification reaction requires 3 mol of alcohol and 1 mol of triglyceride to yield 3 mol of fatty acid ester and 1 mol of glycerol. Higher alcohol/oil molar ratios are used to enhance the solubility and to increase the contact between the triglyceride and alcohol molecules (Noureddini et al., 1998). The alcohol/oil molar ratio of 6:1 or higher generally gives the maximum yield (higher than 98 wt%).
The vegetable oils are transesterified 1:6–1:40 oil/alcohol molar ratios in catalytic and supercritical alcohol conditions (Demirbas, 2002a). Transesterification of Jatropha curcas oil in supercritical methanol conditions was carried out at different oil/methanol molar ratios from 10 to 43 (Hawash et al., 2009). It was observed that increasing molar ratio had a favorable influence on methyl ester conversion.
Acid-catalyzed reactions require the use of high alcohol/oil molar ratios in order to obtain good product yields in practical reaction times. However, ester yields do not proportionally increase with molar ratio. Ramadhas et al. (2004) and Sahoo et al. (2007) have reported 6:1 alcohol/oil molar ratio during acid esterification and 9:1 alcohol/oil molar ratio during alkaline esterification to be the optimum amount for biodiesel production from high FFA rubber seed oil and polanga seed oil, respectively.
Another important variable affecting the yield of ester is the type of alcohol to triglyceride. In general, short chain alcohols such as methanol, ethanol, propanol, and butanol can be used in the transesterification reaction to obtain high ester yields.
Effect of reaction temperature and time
Transesterification reaction can occur at different temperatures depending on the type of oil employed. It was observed that increasing the reaction temperature, especially to supercritical conditions, had a favorable influence on the yield of ester conversion. In the alkali-catalytic transesterification reaction, the temperature maintained by the researchers during different steps range between 318 and 338 K. The boiling point of methanol is 337.9 K. Temperature higher than this will burn the alcohol and will result in much lesser yield. Leung and Guo (2006) showed that temperature higher than 323 K had a negative impact on the product yield for neat oil, but had a positive effect for waste oil with higher viscosities.
Encinar et al. (2002) studied the transesterification reaction, with ethanol, of Cynara cardunculus oils using NaOH and KOH as catalysts and reported 91.6% conversion at room temperature. Alkali-catalytic transesterification of Pongamia pinnata oil was carried out at five different temperatures with 1% KOH catalyst concentration, methanol/oil ratio 6:1, and 30 min of reaction time. The conversion was increased with increase in temperature and the optimum temperature was found to be 333 K beyond that point no change was observed with increase in temperature.
Effect of reaction temperature and the reaction time was investigated on the conversion of Jatropha curcas oil to biodiesel (Vyas et al., 2011). Maximum FAME yields 93.5% was observed at 323 K and 45 min of reaction time.
The methyl ester conversion rate increases with the reaction time. Different researchers have reported different reaction times for the transesterification process. Kim et al. (2004) studied the transesterification reaction, with methanol/oil ratio (6:1), of vegetable oils using NaOH and Na/NaOH/c-Al2O3 as catalysts at 333 K. They reached the maximum biodiesel yield within 1 h both for the case of homogeneous and heterogeneous catalyst systems.
Effect of water and FFA
The water and FFA content are key parameters for determining the viability of the vegetable oil transesterification process. In the transesterification process, the vegetable oil should have an acid value less than 1 and all materials should be substantially anhydrous. If acid value is greater than 1, more alkali (NaOH or KOH) is injected to neutralize the FFAs.
Water content is an important factor in the conventional catalytic transesterification of vegetable oil. Water can cause soap formation and frothing. The resulting soaps can induce an increase in viscosity, formation of gels and foams, and made the separation of glycerol difficult (Agra et al., 1992; Ghadge and Raheman, 2005). In the conventional transesterification of fats and vegetable oils for biodiesel production, FFAs and water always produce negative effects, since the presence of FFAs and water causes soap formation, consumes the catalyst, reduces its catalytic effectiveness which results a low conversion in biodiesel production. FFAs react with the alkaline catalyst to produce soaps that inhibit the separation of the ester, glycerin, and wash water (Canakcı and Van Gerpen, 1999). Ma et al. (1998) reported that water and FFA contents of beef tallow catalyzed by NaOH had to be maintained below 0.06 wt% and 0.5 wt%, respectively. Using NaOH catalyzed transesterification, methyl esters can generally be prepared in high yields for low FFA oils.
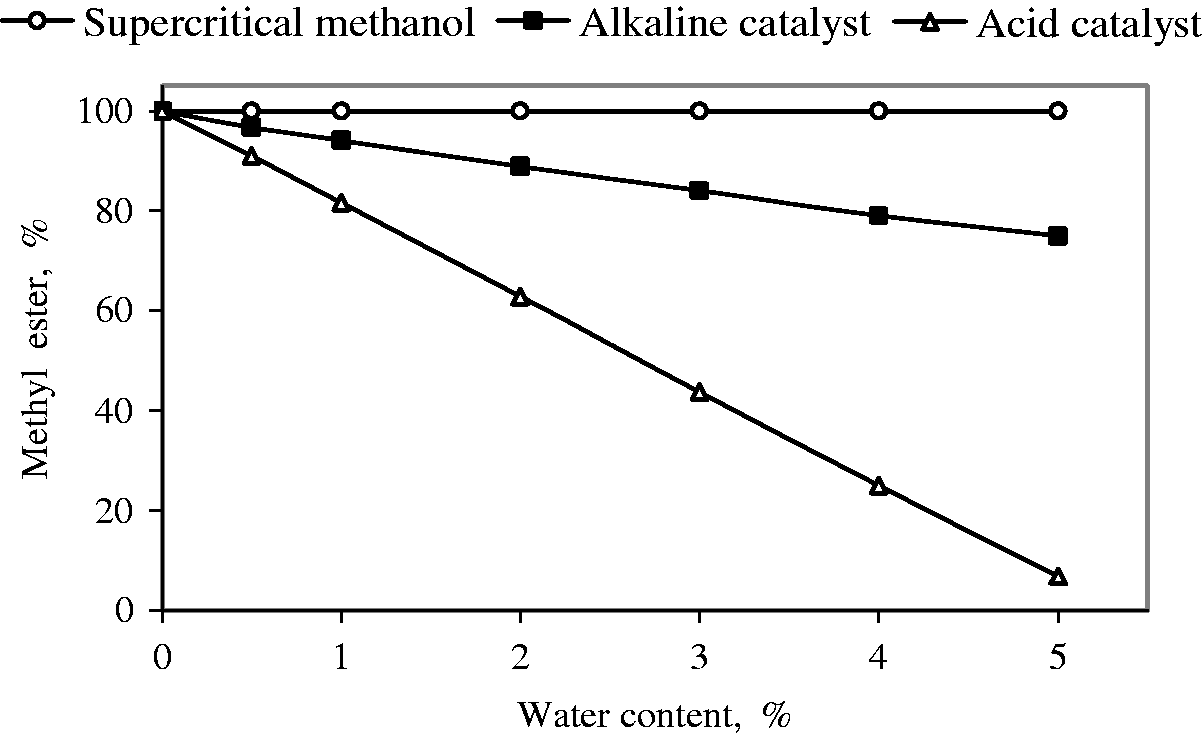
In catalyzed methods, the presence of water has negative effects on the yields of methyl esters. The presence of water had negligible effect on the conversion while using lipase as a catalyst (Madras et al., 2004). At the same time, the presence of water had a positive effect in the yield of methyl esters when methanol at room temperature was substituted by supercritical methanol (Demirbas, 2006). Kusdiana and Saka (2004) studied the effect of water and FFA contents on the yield of methyl esters by the supercritical methanol treatment (623 K, 43 MPa, 4 min of treatment with a methanol to oil molar ratio of 42:1) compared with those from alkaline- and acid-catalyzed method. Effects of water and FFA contents on the alkaline- and acid-catalyzed and supercritical transesterification of triglycerides are shown in Figures 2 and 3.
Yields of methyl esters as a function of water content in transesterification of triglycerides. Yields of methyl esters as a function of free fatty acid content in transesterification of triglycerides.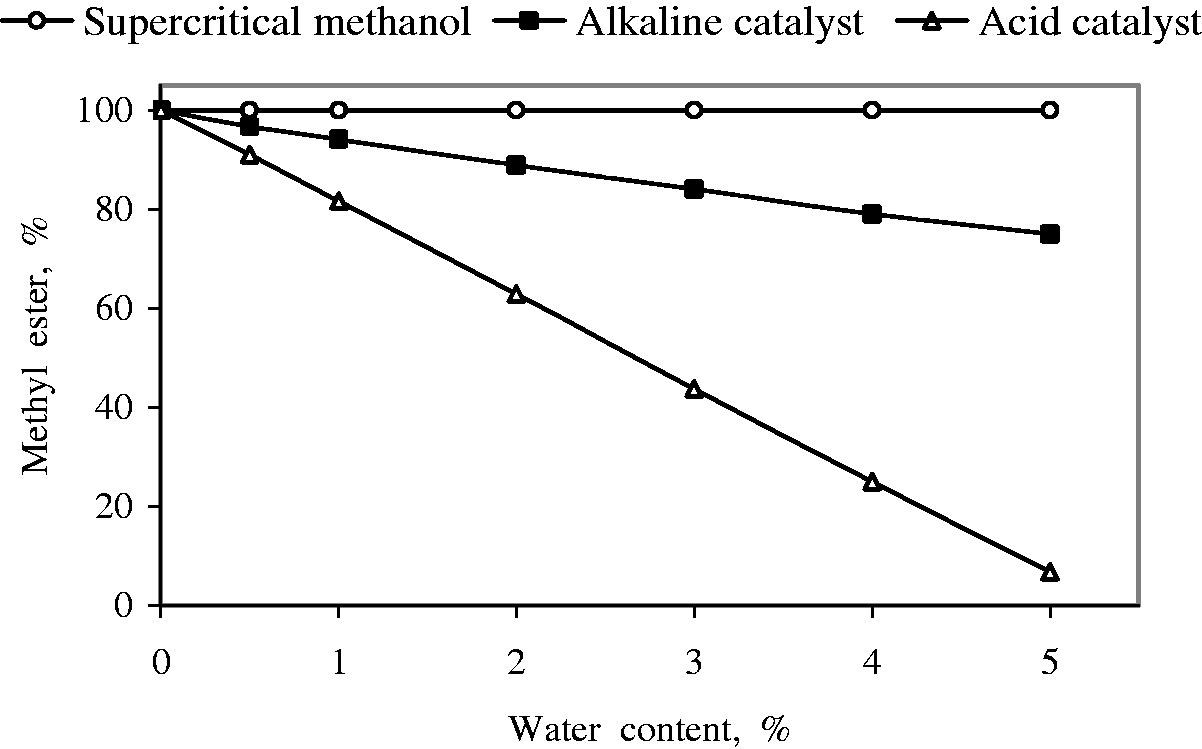
Effect of catalyst content and type
Catalysts used for the transesterification of triglycerides are classified as alkali, acid, enzyme. Several studies have examined the effects of catalyst content and type in transesterification of triglycerides (Ma et al., 1998; May, 2004; Meneghetti et al. (2006); Stavarache et al., 2005).
The effect of different catalyst concentrations on base-catalyzed transesterification during biodiesel production from vegetable oil by means of ultrasonic energy were investigated by Stavarache et al. (2005). The highest yields were obtained when the catalyst was used in small concentration, i.e. 0.5% wt/ of oil.
Hossain and Mazen (2010) investigated the effects of different homogeneous catalysts (NaOH, KOH) on the biodiesel yield. The experiments were carried out at room temperature with 0.5% catalyst concentration, methanol/oil ratio 1:1, and 2 h of reaction time. The results showed that NaOH (71.2%) gave the better yield, compared with KOH (68.9%). In addition, different concentrations of NaOH like 0.5, 1.0, and 1.5% were used. The highest yield was obtained with 1.0% of NaOH concentration. In another study, Meneghetti et al. (2006) reported that HCl is much more effective than NaOH at higher reaction temperatures.
Fuel properties of biodiesel from non-edible vegetable oils
ASTM standards of maximum allowed quantities in diesel and biodiesel (Lotero et al., 2006).
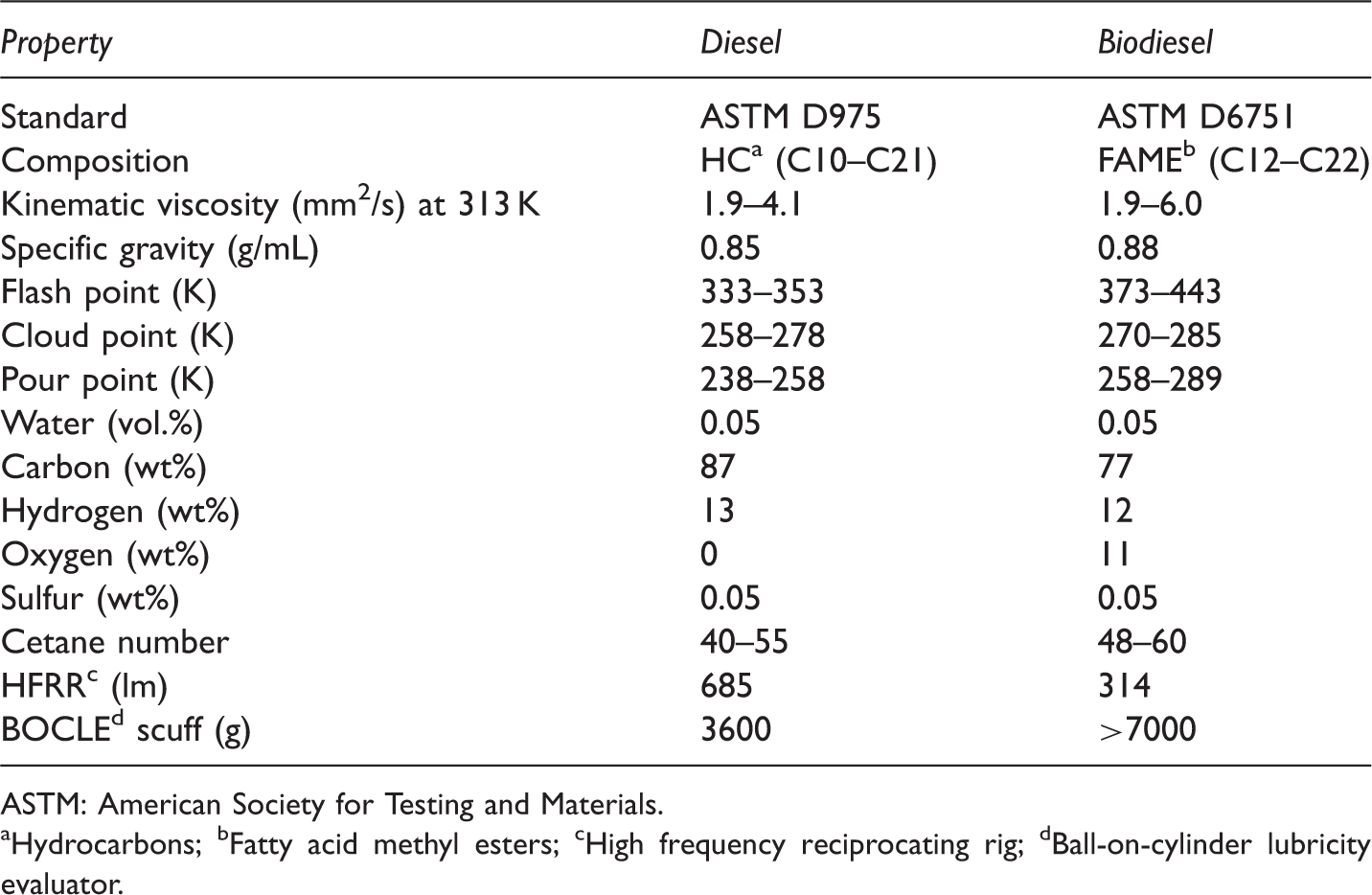
ASTM: American Society for Testing and Materials.
Hydrocarbons; bFatty acid methyl esters; cHigh frequency reciprocating rig; dBall-on-cylinder lubricity evaluator.
Fuel-related properties of biodiesel from non-edible oils.
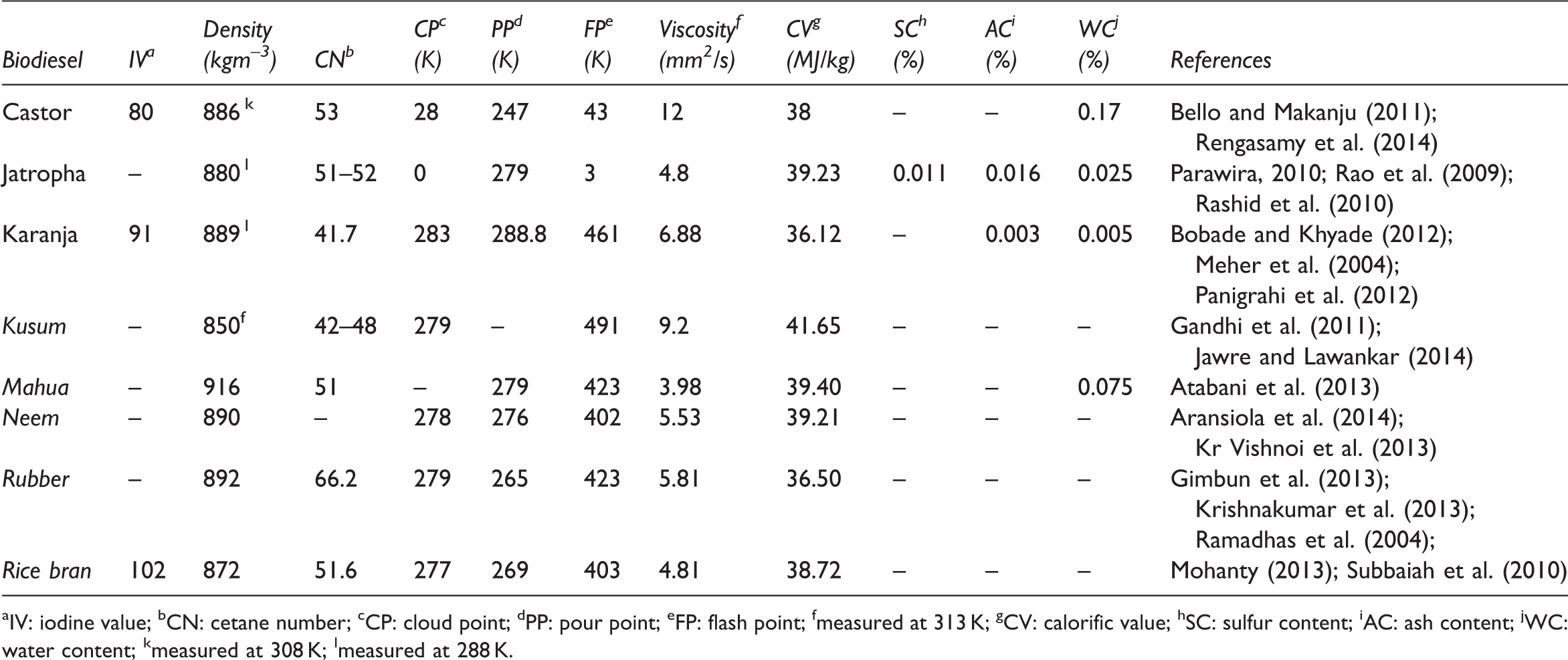
IV: iodine value; bCN: cetane number; cCP: cloud point; dPP: pour point; eFP: flash point; fmeasured at 313 K; gCV: calorific value; hSC: sulfur content; iAC: ash content; jWC: water content; kmeasured at 308 K; lmeasured at 288 K.
The injection and atomization characteristics of the vegetable oils are significantly different than those of petroleum-derived diesel fuels, mainly as the result of their high viscosities (Cigizoglu et al., 1997). High viscosity leads to poorer atomization of the fuel spray and less accurate operation of the fuel injectors. The lower the viscosity of the biodiesel, the easier it is to pump and atomize and achieve finer droplets (Islam and Beg, 2004). The viscosity values of vegetable oil methyl esters decrease sharply after transesterification. The viscosity values of vegetable oils are between 27.2 and 53.6 mm2/s, whereas those of vegetable oil methyl esters are between 3.6 and 4.6 mm2/s (Demirbas, 2006). The kinematic viscosity in biodiesel is determined using ASTM D6751 (1.9–6.0 mm2/s) (Table 8). Compared to ASTM D6751 standards for biodiesel fuel, some of non-edible vegetable oil methyl esters are highly viscous, such as Ricinus communis and Schleichera oleosa methyl esters (Table 9).
The cetane number is an important parameter for the determination of diesel fuel quality, especially the ignition quality. It measures the readiness of the fuel to auto-ignite when injected into the engine. Ignition quality is one of the properties of biodiesel that is determined by the structure of the FAME component (Bamgboye and Hansen, 2008). As shown in Table 9, cetane number of non-edible oil methyl esters is higher than that of petroleum; therefore they can replace diesel for the combustion engine.
Another important property of biodiesel fuel is its flash point. Flash point of a fuel is the lowest temperature at which it will spontaneously ignite without the presence of a flame or spark. Non-edible oil methyl esters have quite high flash points which make them less volatile and safer to transport or handle than diesel fuel.
Two important parameters for low-temperature applications of a fuel are cloud point and pour point. The cloud point is the temperature at which wax first becomes visible when the fuel is cooled. The pour point is the temperature at which the amount of wax from a solution is sufficient to gel the fuel; thus it is the lowest temperature at which the fuel can flow. Compared to ASTM D6751 standards for biodiesel fuel, cloud and pour points of non-edible oil methyl esters are similar biodiesel fuel. Biodiesel has a higher cloud and pour points compared to conventional diesel (Prakash, 1998). The vegetable oil methyl esters have cloud point and pour point that are 15–25 K higher than those of diesel fuels (Table 8). The cloud and pour points of non-edible methyl esters vary significantly with feedstock depending on fatty acid compositions (Atabani et al., 2013; Silitonga et al., 2013).
The density is an another important property of biodiesel fuel. It is the mass per unit volume of any liquid at a given temperature. Specific gravity is the ratio of the density of a liquid to the density of water. Specific gravity of biodiesels ranges between 0.87 and 0.89 (Demirbas, 2006). An increase in density from 860 to 885 kg/m3 for vegetable oil methyl esters increases the viscosity from 3.59 to 4.63 mm2/s and the increases are highly regular (Demirbas, 2005, 2009a). There is high regression between the density and viscosity values of vegetable oil methyl esters (Demirbas, 2009a). According to ASTM D6751 standards, density should be tested at the temperature reference of 288 K. The density of non-edible methyl esters is higher than that of common diesel fuel. This may be due to the presence of higher molecular weight triglycerides (Hotti and Hebbal, 2015).
HHV is an important parameter in the selection of a fuel. The HHVs of biodiesels (39–41 MJ/kg) are slightly lower than those of gasoline (46 MJ/kg), petrodiesel (43 MJ/kg), or petroleum (42 MJ/kg) (Demirbas, 2008b) because of their higher oxygen content. Ash and extractive content are two important parameters directly affecting the heating value. High ash content of a plant part makes it less desirable as fuel, whereas high extractive content adds to its desirability (Demirbas, 2002c).
Conclusion
Edible plant oil fuels presently do not compete economically with petroleum-based fuels because they are more expensive but due to the recent increase in petroleum prices and uncertainties concerning petroleum availability, there is renewed the interest in non-edible plant oil fuels for diesel engines.
Non-edible plant oils are unsuitable for human food because of the presence of some toxic components. These plants are easily available in developing countries and are very economical comparable to edible plant oils. Biodiesel produced from non-edible plant oils has good potential as an alternative diesel fuel. However, as a serious drawback, most non-edible oils contain a high content of FFAs, which increases the biodiesel production cost.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
