Abstract
Objectives:
Emerging evidence suggests that glucagon-like peptide-1 (GLP-1) receptor agonists and sodium-glucose co-transporter 2 (SGLT2) inhibitors may exert neuroprotective effects. However, the comparative efficacy of individual agents remains unclear. This network meta-analysis (NMA) aimed to evaluate the differential impacts of these therapies on the incidence of mental status changes, specifically delirium, depression, dementia and coma.
Design:
A frequentist NMA was conducted using data from randomised controlled trials (RCTs) investigating GLP-1 receptor agonists or SGLT2 inhibitors. The robustness of the findings was verified through a Bayesian NMA framework.
Setting:
This study adopted a confirmatory framework focusing on pre-defined neuropsychiatric adverse outcomes in alignment with Cochrane recommendations.
Participants:
Included trials enrolled individuals without baseline cognitive or psychiatric disorders.
Main outcome measures:
The primary endpoint was the incidence of delirium, depression, dementia or coma during treatment. Secondary endpoints included changes in cognitive performance and drop-out rates.
Results:
A total of 62 RCTs comprising 200,068 participants were included. Among all treatments, only high-dose dapagliflozin (10 mg/day) significantly reduced the occurrence of delirium and depression, particularly in patients with type 2 diabetes. Dulaglutide and liraglutide were the only agents associated with cognitive improvement. No significant benefits were observed for dementia or coma across all agents.
Conclusions:
This analysis highlights agent-specific neuroprotective profiles: SGLT2 inhibitors, especially high-dose dapagliflozin, may mitigate the onset of delirium and depression, while GLP-1 receptor agonists, notably dulaglutide and liraglutide, may enhance cognitive function. These findings warrant consideration in selecting antihyperglycemic therapies for individuals at elevated neuropsychiatric risk.
Trial registration:
PROSPERO CRD42024601021.
The study protocol was approved by the Institutional Review Board of the Tri-Service General Hospital, National Defense Medical Center (TSGHIRB E202516007).
Introduction
Glucagon-like peptide-1 (GLP-1) receptor agonists and sodium–glucose co-transporter 2 (SGLT2) inhibitors represent a new generation of antihyperglycemic agents with mechanisms distinct from older glucose-lowering drugs. While their primary application has been glycemic control in type 2 diabetes, accumulating evidence suggests potential effects on neurological and psychiatric outcomes. In particular, Mehan et al. proposed neuroprotective roles of GLP-1 receptor modulators in neurodegenerative conditions, 1 while anti-inflammatory and immunomodulatory properties of SGLT2 inhibitors such as dapagliflozin may provide protective benefits in acute mental status alterations.2,3
Despite such promising preclinical data, clinical evidence remains fragmented. Randomised controlled trials (RCTs) have rarely prioritised neuropsychiatric endpoints, limiting the ability of individual studies to establish definitive effects. Meta-analyses, particularly network meta-analyses (NMA), offer a robust means to synthesise comparative data across interventions and address indirect treatment comparisons.
While prior analyses have examined GLP-1 receptor agonists and SGLT2 inhibitors in relation to neurodegenerative disease 4 and hearing loss, 5 few have comprehensively assessed their broader impact on mental status changes, including delirium, depression and coma. Given the clinical burden of such syndromes – especially in older adults or individuals with diabetes – understanding agent-specific neuropsychiatric outcomes is increasingly relevant.
This study aimed to compare the efficacy of individual GLP-1 receptor agonists and SGLT2 inhibitors in preventing mental status deterioration using both frequentist and Bayesian NMA approaches. We focused on four key clinical syndromes: delirium, depression, dementia and coma, as defined by Patti and Gupta. 6 Our hypothesis was that pharmacological and pharmacokinetic differences among agents would translate to differential effects on mental health outcomes.
Methods
Setting
This NMA employed a confirmatory analytic approach, emphasising predefined neuropsychiatric adverse effects as per Cochrane guidance. 7 The study adhered to the PRISMA extension (Supplementary Table 1) for network meta-analysis and was registered with PROSPERO (CRD42024601021). Ethics approval was obtained from the Tri-Service General Hospital IRB, Taipei, Taiwan (TSGHIRB E202516007).
Search strategy and study selection
Systematic searches were conducted across PubMed, Embase, ClinicalKey, Cochrane CENTRAL, ProQuest, ScienceDirect, Web of Science and ClinicalTrials.gov up to 14 October 2024 (Supplementary Table 2). Two reviewers independently screened titles, abstracts and full texts. Disagreements were resolved by consensus. Additional manual searches of reference lists were performed. There were no language restrictions.
Eligibility criteria
We included peer-reviewed RCTs that met the following PICOS criteria:
Population: Adults without known cognitive or psychiatric diagnoses.
Intervention: Any GLP-1 receptor agonist or SGLT2 inhibitor.
Comparison: Placebo or standard of care.
Outcomes: Incidence of delirium, depression, dementia, coma or changes in cognitive performance.
Study design: Parallel-group RCTs.
We recognised the fact that there were only few RCTs specifically choosing mental health outcomes as their primary outcomes. Therefore, in order to prevent risk of reporting bias, we only included RCTs with designation of systematic reporting adverse event. Exclusion criteria included animal studies, observational designs and trials lacking adverse event surveillance. For cognitive outcomes, broader inclusion was permitted given their presence as prespecified endpoints in select RCTs. If mental status events were not reported in manuscripts, data from trial registries (e.g. ClinicalTrials.gov) were retrieved.
Risk of bias and data extraction
Two reviewers assessed risk of bias using the Cochrane RoB Tool 1.0. Extracted data included demographics, intervention details and outcomes. Events linked to metabolic crises (e.g. hypoglycemic coma) or substance withdrawal (e.g. alcohol-induced delirium) were excluded. Further, to uniformly extract data, we extract the data of ‘end of study’.
Statistical analysis
A random-effects NMA was performed using the frequentist netmeta package via MetaInsight. Categorical variables were analysed using odds ratios (ORs) with its 95% confidence intervals (95%CIs). We recognised the fact that the rating scales for cognitive function might vary across the recruited RCTs, we choose to use standardised procedure (i.e. standardised mean differences (SMDs)) to pool them into estimates. Treatment hierarchies were established using surface under the cumulative ranking curve (SUCRA). Inconsistency was assessed via loop-specific, node-splitting and design-by-treatment interaction models. Between-study heterogeneity was quantified using tau estimates.
To ensure robustness, we conducted a Bayesian NMA using SUCRA heatmaps, Rank-O-Gram plots and deviation models.
Additional subgroup analyses were conducted by baseline diabetes status and treatment duration over 52 weeks, referencing dementia-risk stratification frameworks.8,9 Finally, we evaluated the overall certainty of evidence in our NMA with the GRADE. Potential publication bias was evaluated through visual inspection of comparison-adjusted funnel plots and Egger’s regression tests.
Results
Study characteristics
Figure 1 illustrates the flowchart summarising the literature search and screening process for this NMA. After excluding 135 articles for various reasons (Supplementary Table 3), 62 RCTs across 59 publications (n = 200,068 participants) met eligibility criteria. Mean age was 63.0 years (range: 44.9–71.9), and mean female proportion was 37.7%. Study duration averaged 129.2 weeks (range: 8–281; Supplementary Table 4). Among the included RCTs, about 93.5% and 8.1% RCTs exclusively included participants with diabetes or obesity, respectively. About 59.7% RCTs provided treatment over 52 weeks.
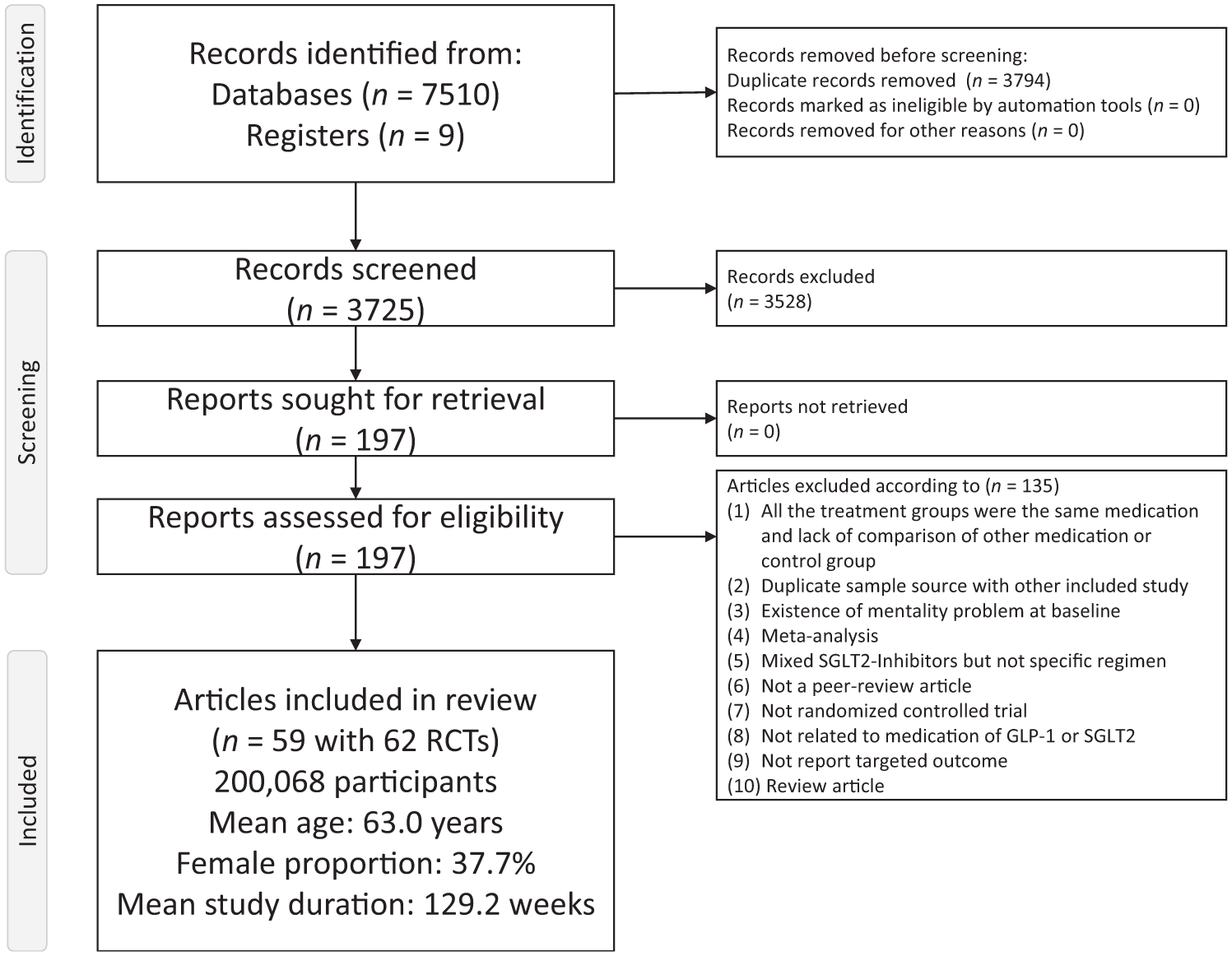
PRISMA2020 Flowchart of current network meta-analysis.
Primary outcomes
Only high-dose dapagliflozin (10 mg/day) was associated with significantly fewer composite mental status events relative to placebo (OR = 0.34, 95%CIs = 0.34–0.79; Figures 2 and 3, Supplementary Figures 3A and 4A and Table 1).
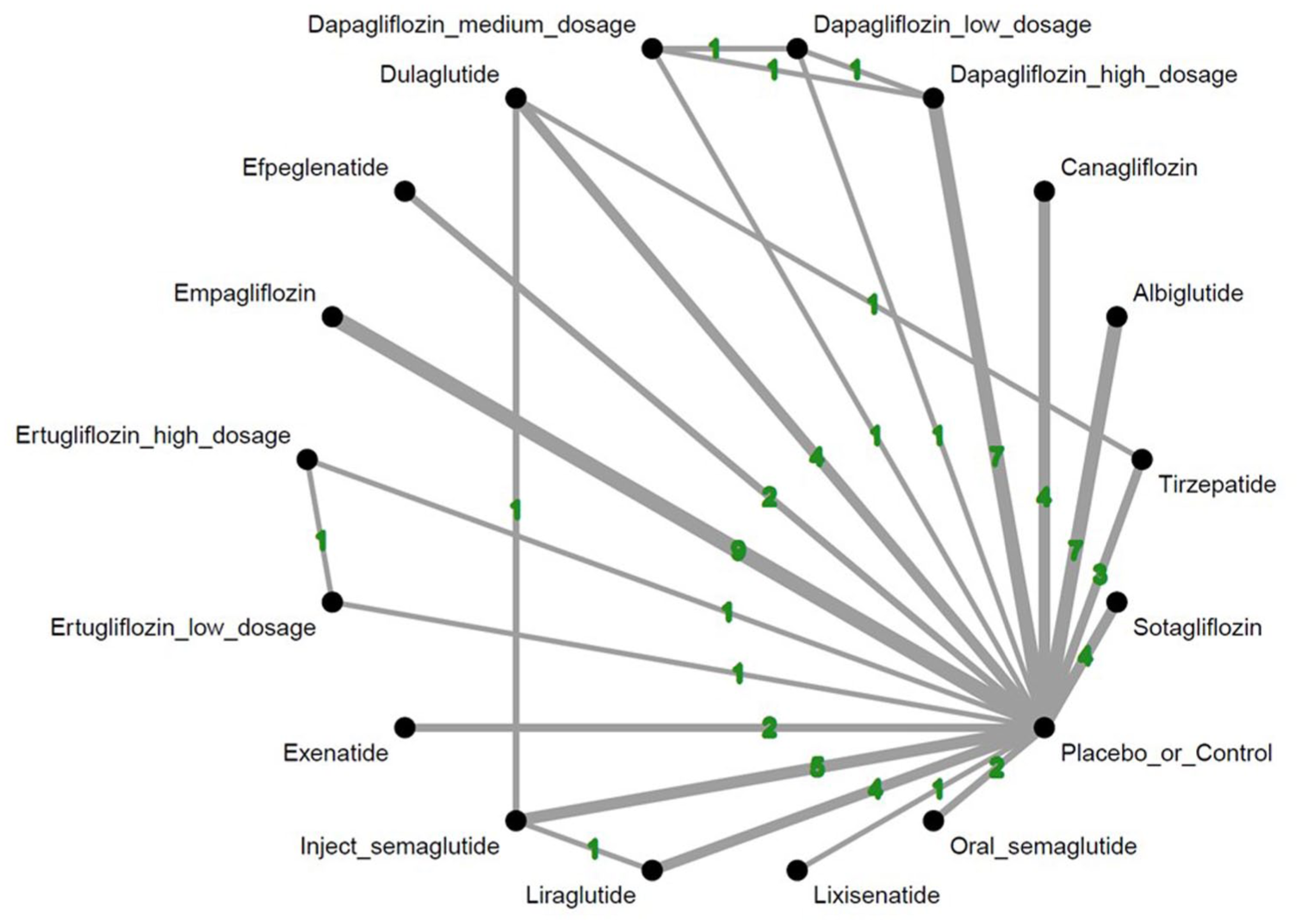
Network plot of the primary outcomes: overall events of mental status changes.
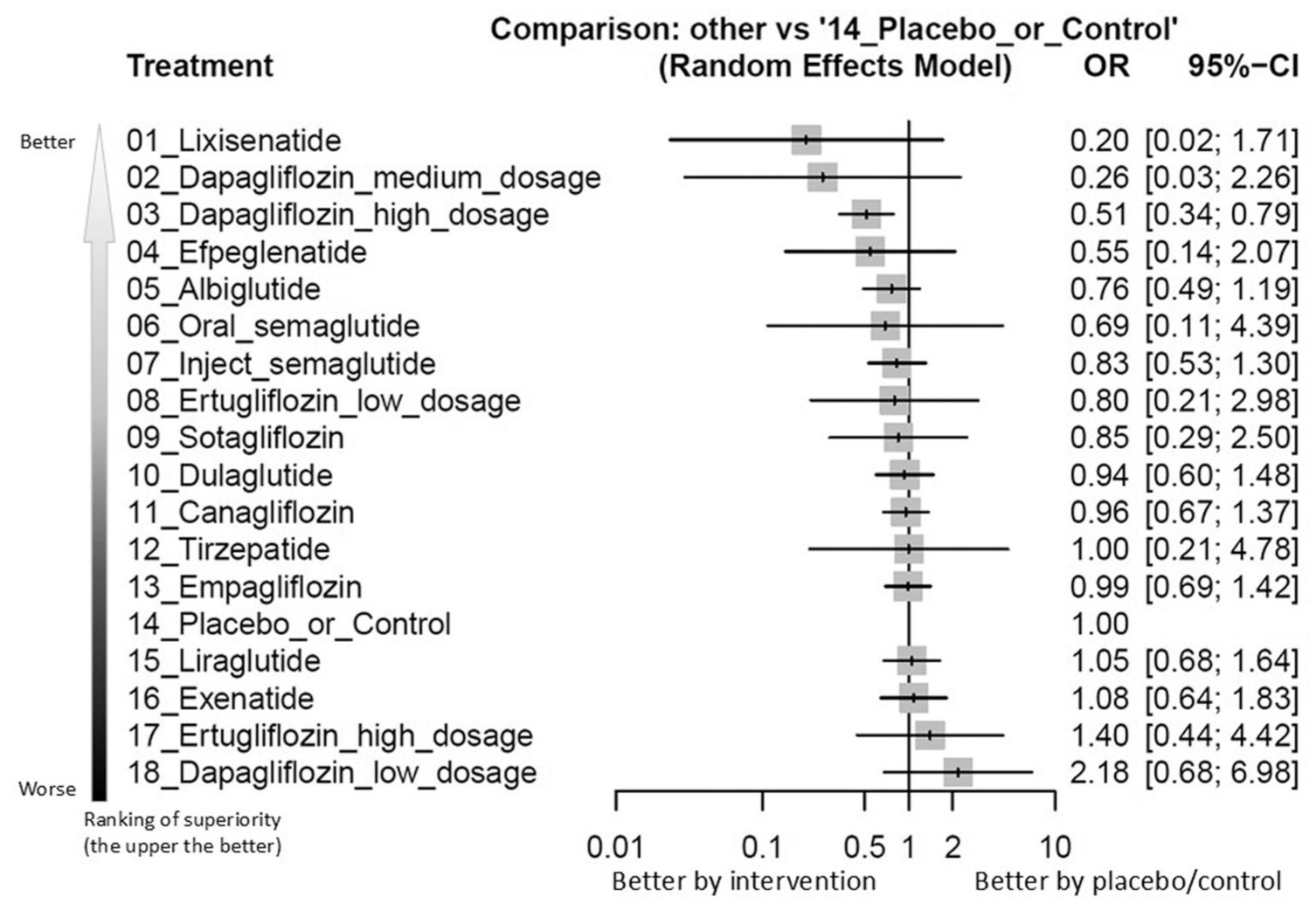
Forest plot of primary outcome: overall events of mentality status change.
League table of the primary outcome: overall events of mentality status change.
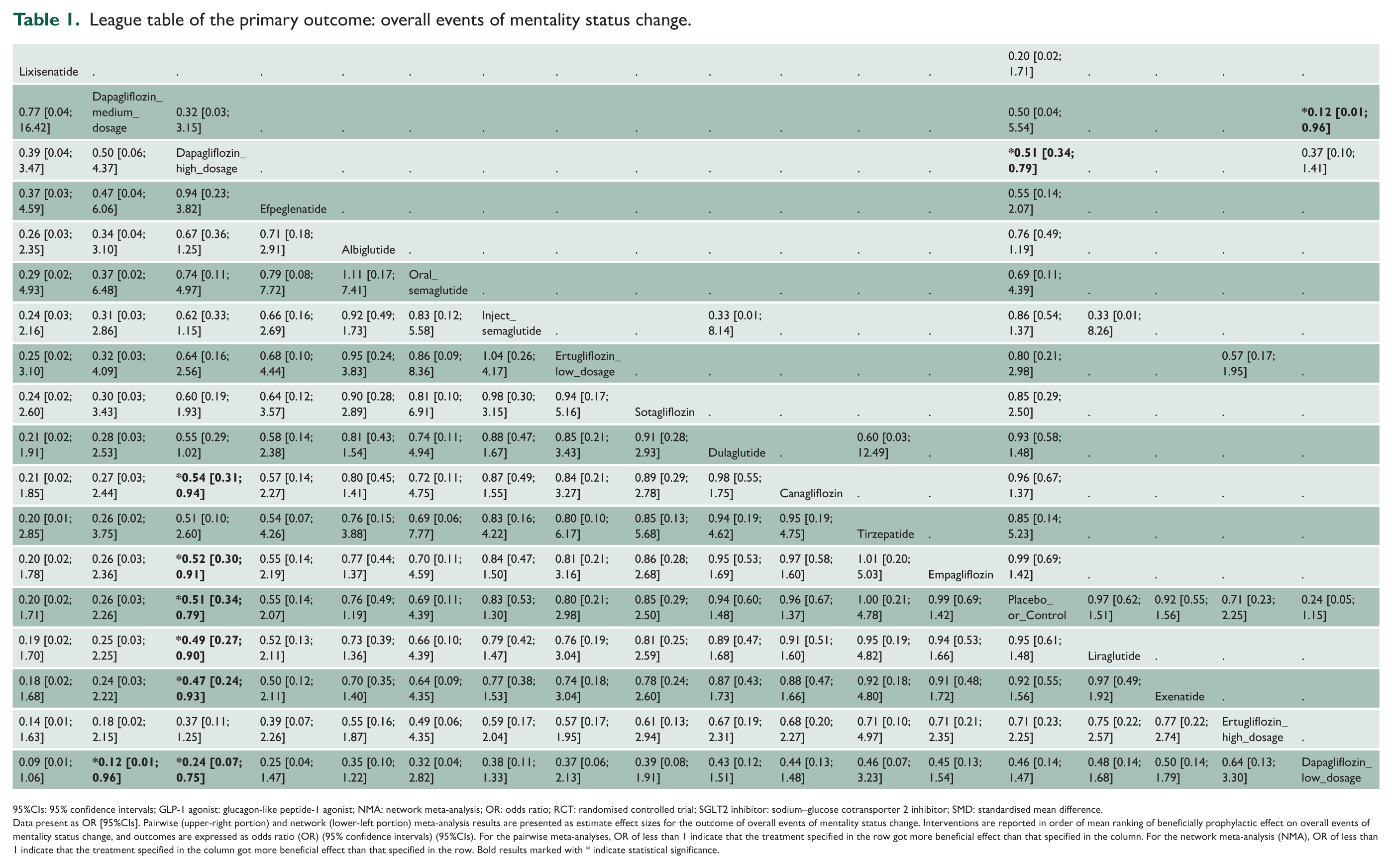
95%CIs: 95% confidence intervals; GLP-1 agonist: glucagon-like peptide-1 agonist; NMA: network meta-analysis; OR: odds ratio; RCT: randomised controlled trial; SGLT2 inhibitor: sodium–glucose cotransporter 2 inhibitor; SMD: standardised mean difference.
Data present as OR [95%CIs]. Pairwise (upper-right portion) and network (lower-left portion) meta-analysis results are presented as estimate effect sizes for the outcome of overall events of mentality status change. Interventions are reported in order of mean ranking of beneficially prophylactic effect on overall events of mentality status change, and outcomes are expressed as odds ratio (OR) (95% confidence intervals) (95%CIs). For the pairwise meta-analyses, OR of less than 1 indicate that the treatment specified in the row got more beneficial effect than that specified in the column. For the network meta-analysis (NMA), OR of less than 1 indicate that the treatment specified in the column got more beneficial effect than that specified in the row. Bold results marked with * indicate statistical significance.
Delirium subgroup
Dapagliflozin (10 mg/day) was associated with significantly less delirium incidence (OR = 0.21, 95%CIs = 0.05–0.83) (Supplementary Figure 1A, Supplementary Figure 2A, Supplementary Figure 3B, Supplementary Figure 4B and Supplementary Table 5A).
Depression subgroup
The same dapagliflozin dosage was associated with significantly reduced depression risk (OR = 0.43, 95%CIs = 0.20–0.91) (Supplementary Figure 1B, Supplementary Figure 2B, Supplementary Figure 3C, Supplementary Figure 4C and Supplementary Table 5B). Effects were most pronounced in patients with pre-existing diabetes.
Dementia and coma subgroup
No treatment was significantly superior to placebo in reducing dementia or coma incidence (Supplementary Figures 1C–1D, Supplementary Figures 2C–2D, Supplementary Figures 3D–3E, Supplementary Figures 4D–4E and Supplementary Tables 5C–5D).
Secondary outcome: change of cognitive function
The applied rating scale of cognitive function among the recruited RCTs included minimum mental state examination, Montreal cognitive assessment and memory part of standardised neuropsychological assessment tests. After converting the effect sizes of those rating scales into SMD, only dulaglutide (SMD = 1.46, 95% CIs = 0.85–2.08) and liraglutide (SMD = 0.70, 95%CIs = 0.24–1.16) was associated with significantly improved cognitive performance over controls (Supplementary Figure 1E, Supplementary Figure 2E, Supplementary Figure 3F, Supplementary Figure 4F and Supplementary Table 5E).
Acceptability: drop-out rate
Agents associated with significantly lower drop-out rates included tirzepatide (low, medium, high doses), canagliflozin (low, high), liraglutide and low-dose empagliflozin (Supplementary Figure 1F, Supplementary Figure 2F, Supplementary Figure 3G, Supplementary Figure 4G and Supplementary Table 5F).
Subgroup analyses of baseline diabetes or not
In trials recruiting patients with diabetes (total 58 RCTs in this subgroup), dapagliflozin (10 mg/day) retained significant preventive effects for composite mental status (OR = 0.49, 95% CIs = 0.29–0.81) and depression (OR = 0.32, 95% CIs = 0.14–0.74). Insufficient data precluded subgroup analysis among non-diabetic individuals (Supplementary Figures 1G–1K, Supplementary Figures 2G–2K and Supplementary Tables 5G–5K).
Subgroup analysis of treatment duration over 52 weeks
Among trials exceeding 1 year (total 37 RCTs in this subgroup), only high-dose dapagliflozin was linked to fewer mental status change events (OR = 0.48, 95%CIs = 0.31–0.76) (Supplementary Figure 1L, Supplementary Figure 2L and Supplementary Table 5L).
Bayesian sensitivity analyses
Bayesian NMA confirmed the primary findings. Rankograms and SUCRA plots consistently ranked dapagliflozin highest for depression/delirium prevention and liraglutide/dulaglutide for cognitive gains (Supplementary Figures 5A–5E, Supplementary Figures 6A–6J and Supplementary Tables 6A–6E). The deviation-model assessment did not demonstrate significant deviation among the current NMA (Supplementary Figures 7A–7O).
Risk of bias and inconsistency
No substantial publication bias was detected through visual inspection of the comparison-adjusted funnel plots or by Egger’s regression analysis (Supplementary Figures 8A–8B). Most studies exhibited low RoB (344/434 items). Common issues included inadequate allocation concealment and lack of outcome assessor blinding (Supplementary Figures 9A-9B). Inconsistency testing, including loop-specific analysis, node-splitting and design-by-treatment interaction models, did not reveal major violations of consistency assumptions (Supplementary Tables 7A–7H). Measures of between-study heterogeneity showed acceptable levels across most comparisons (Supplementary Table 8). The GRADE evaluation rated overall certainty of evidence as moderate to high (Supplementary Tables 9A–9G).
Discussion
Overall, both GLP-1 receptor agonists and SGLT2 inhibitors showed beneficial effects on mental status, though with specific strengths in different areas. GLP-1 receptor agonists, particularly dulaglutide and liraglutide, demonstrated a notable advantage in enhancing cognitive function, while SGLT2 inhibitors, especially high-dose dapagliflozin, were more effective in preventing depression, especially in subjects with baseline diabetes, and delirium. However, neither class of drugs had a significant impact on dementia or coma outcomes. To our knowledge, this NMA is the first to systematically compare the mental health benefits of GLP-1 receptor agonists and SGLT2 inhibitors, particularly in relation to delirium and depression prevention.
One key finding of this NMA was that SGLT2 inhibitors were more effective in reducing delirium and depression. Specifically, dapagliflozin contributed to lowering the incidence of these conditions. To date, there have been few reports discussing the preventive effects of SGLT2 inhibitors, especially dapagliflozin, on delirium and depression. A potential mechanism could involve dapagliflozin’s anti-inflammatory properties. Previous studies have suggested that delirium precipitating factors, such as infections and hypoglycemia, may trigger an overt inflammatory response in affected patients. 3 These factors could compromise the integrity of the blood–brain barrier, allowing inflammatory agents (e.g. cytokines) to enter the central nervous system. 3 Elevated cytokines, such as interleukin-1 beta (IL-1B), 10 and overexpression of toll-like receptors (TLRs) 11 in the brain can lead to changes in neural function, resulting in delirium symptoms. Similarly, dysregulated TLR expression is thought to play a crucial role in depression, 12 and higher levels of IL-1B have been observed in patients with depression. 13 Dapagliflozin has been shown to exert anti-inflammatory effects by inhibiting TLR overexpression in human endothelial cells and differentiated macrophages. 2 Further, dapagliflozin had demonstrated the benefits in neuroinflammation reduction and synaptic plasticity promotion in animal models, which served as an indicator of neuroprotection. 14 Additionally, in human studies, dapagliflozin has been reported to reduce systemic inflammation in patients with type 2 diabetes. For example, a study by Wang and colleagues found that plasma IL-1B levels decreased in patients treated with dapagliflozin for 12 months. 15 Further, in contrary with the other SGLT2 inhibitors, the dapagliflozin had an adequate range of volume of distribution in the view of pharmacokinetics (118 L). 16 An adequate volume of distribution could contribute to sufficient distance of penetration and local concentration of drugs. This theory could be supported by another SGLT2 inhibitor with adequate volume of distribution. Ertugliflozin, with volume of distribution 85.5 L, 17 had a trend to reduce risk of delirium although the statistics result did not achieve significant level (OR = 0.07, 95% CIs = 0.00 to 1.38 in comparison with controls). On the other hand, regimens with overt high volume of distribution would lead to insufficient local concentration and result in unsatisfactory protective effects, such as sotagliflozin. 18 In addition to mechanisms above, the neuroprotective effects of dapagliflozin might also come from its metabolic health enhancing effect. However, since dapagliflozin’s metabolic benefit was not the highest among this category, the neuroprotective effect from metabolic benefit by dapagliflozin might be less predominant.
Another important finding is the benefit of GLP-1 receptor agonists in improving cognitive function. In addition to showing the strongest effect on the primary outcome, with albiglutide ranking highest for overall mental status changes, only GLP-1 receptor agonists (specifically dulaglutide and liraglutide) were associated with significantly greater improvements in cognitive function compared with controls. GLP-1 receptor agonists were initially developed for diabetes management due to their ability to extend the biological activity of GLP-1, 19 which promotes insulin secretion, delays gastric emptying, inhibits glucagon release and subsequently reduces blood glucose levels. 20 Their relevance to neuropsychiatric applications arose from the discovery that GLP-1 receptors are expressed in several key areas of the central nervous system, including the hippocampus, neocortex, hypothalamus and cerebellum. 19 Studies in animal models have suggested that GLP-1 analogs offer neuroprotective effects 21 by regulating mitochondrial biogenesis and tau protein phosphorylation. 22 However, while these findings have been promising in animal studies, evidence for their efficacy in humans remains inconclusive. For instance, there is still uncertainty regarding the ability of GLP-1 receptor agonists to cross the blood–brain barrier. 19 Despite this, clinical observations have shown improvements in various cognitive domains in patients receiving GLP-1 receptor agonists, such as dulaglutide 23 and liraglutide. 24 The underlying physiological mechanisms may include anti-inflammatory effects, 25 inhibition of amyloid-beta aggregation, 26 promotion of neurogenesis, enhancement of synaptic plasticity and prevention of neuronal apoptosis. 27 The findings of this NMA provide further support for the association between GLP-1 receptor agonist treatment and improved cognitive function.
Strengths and limitations
This NMA provides a comprehensive comparison of neuropsychiatric outcomes among contemporary antidiabetic drugs. Strengths include RCT-only inclusion, stratified subgroup analysis and confirmatory Bayesian modeling.
Nonetheless, our findings are constrained by trial durations. While sufficient for short-term outcomes like delirium, the 129.2-week average may not capture late-onset syndromes such as dementia. Blinding and allocation bias also warrant caution, as does selective adverse event reporting. Pre-existing comorbidities (e.g. obesity) may have confounded results despite diabetes-based subgrouping. Data heterogeneity and absence of diagnostic standardisation remain challenges inherent to meta-analyses.
Implications
Targeted use of antidiabetic agents may yield mental health benefits beyond glycemic control. Consideration of neuroprotective profiles in pharmacologic decision-making is warranted, particularly in vulnerable populations.
Conclusion and interpretation
This NMA identifies distinct roles for SGLT2 inhibitors and GLP-1 receptor agonists in protecting mental health. High-dose dapagliflozin appears effective in mitigating depression and delirium, especially among diabetic patients. Dulaglutide and liraglutide may enhance cognition. In the absence of guidelines on psychiatric protection in diabetes management, our findings support preferential selection of neuroprotective agents – especially for patients at high risk for mental status changes. Future trials with dedicated neuropsychiatric endpoints are needed to guide personalised prescribing.
Supplemental Material
sj-docx-1-jrs-10.1177_01410768251395877 – Supplemental material for The concentration-dependent protective effects by new generation hypoglycemic agents on delirium, depression, dementia and coma: evidence from a network meta-analysis
Supplemental material, sj-docx-1-jrs-10.1177_01410768251395877 for The concentration-dependent protective effects by new generation hypoglycemic agents on delirium, depression, dementia and coma: evidence from a network meta-analysis by Ping-Tao Tseng, Bing-Yan Zeng, Chih-Wei Hsu, Mein-Woei Suen, Chao-Ming Hung, Andre F. Carvalho, Brendon Stubbs, Yen-Wen Chen, Tien-Yu Chen, Wei-Te Lei, Pao-Yen Lin, Jiann-Jy Chen, Kuan-Pin Su, Hung-Yu Wang, Bing-Syuan Zeng, Yow-Ling Shiue and Chih-Sung Liang in Journal of the Royal Society of Medicine
Footnotes
Acknowledgements
There is not any funding resource or sponsorship related to this work. This article presents independent research. The views expressed in this publication are those of the authors and not necessarily those of the acknowledged institutions.
Declarations
Data availability:
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request. All the data generated in this study came from HYPERLINK “https://nam12.safelinks.protection.outlook.com/?url=https%3A%2F%2Fclinicaltrials.gov%2F&data=05%7C02%7Cpeter.geraghty%40sagepub.co.uk%7Cf77ed17e9e704bff889808de2bc265ad%7C866b3abd7515461abdb412b4a1857f04%7C0%7C0%7C638996311114441113%7CUnknown%7CTWFpbGZsb3d8eyJFbXB0eU1hcGkiOnRydWUsIlYiOiIwLjAuMDAwMCIsIlAiOiJXaW4zMiIsIkFOIjoiTWFpbCIsIldUIjoyfQ%3D%3D%7C0%7C%7C%7C&sdata=BXbrlVFzPaVZQMrQL6Ygcd2j4WOrEW1iuJGxFdrkVzs%3D&reserved=0” ![]()
Supplemental Material:
Supplemental material for this article is available online.
Use of generative AI:
No generative AI was used during the preparation of this manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
