Abstract

Introduction
In their book Good Pharma: the public-health model of the Mario Negri Institute, Donald Light and Antonio Maturo 1 documented the ethos, origins and development of the Istituto Farmacologiche ‘Mario Negri.’. One reviewer commented that the book combines ‘a devastating critique of the pervasive harms of patent-driven medical research by the pharmaceutical industry with a compelling account of an alternative patient-driven, no-patent research’. 2
In his Foreword in Good Pharma, Iain Chalmers 3 notes that the mission of the Mario Negri Institute since its inception in the 1960s has been ‘to do research of international quality to improve people’s health, based on independent, transparent science, openly used to educate doctors and patients about how best to address their health needs’.
The Mario Negri Institute has used a variety of strategies to promote its mission. One of these is to embed clinical research within publicly orientated healthcare. This requires collaboration with the doctors providing and the patients receiving care.
4
The philosophy underlying this element of the Institute’s mission has been elaborated as follows: Research is an expression of care. It cannot be separate, parallel or occasional (as it is for commercial testing) and yet still be clinically real. The greatest risk in clinical medicine is to dissociate care from research about how effective that care is. Yet this is what usually happens. Rather, research into treatments must be nested within practice.
5
Il Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico (GISSI)
Previous articles7,8 have reviewed the reasons for the interest generated by the first GISSI trial. 6 Together with the contemporaneous ISIS-2 trial,9,10 the first GISSI trial was considered to have made an innovative contribution to the evolution of trial methodology: it had bridged the usual gap between research and healthcare by embedding the former in the latter. In doing so, it addressed the difference between explanatory (efficacy) and pragmatic (effectiveness) trials, 11 as called for by Archie Cochrane. 12
The background for this innovation had been established a few years before, outside and beyond the lively debate about the different philosophies and purposes of explanatory versus pragmatic trials. 13 In particular, in mainly statistically oriented papers, Richard Peto 14 drew attention to the need for substantially larger trials in cancer15,16 and cardiovascular diseases. 17
A common goal of papers such as these was to stress that clinical trials were needed to compare interventions reliably in the type of patients (‘populations’) presenting in routine care settings, and they needed to be large enough to yield patient-important outcomes over periods of time that reflected the natural history of the conditions of interest. The first GISSI trial was the first to demonstrate that this approach was feasible. It was planned, implemented, concluded and published with dramatically innovative results over a period of only 2.5 years. More than 12,000 patients with definite or suspected heart attacks participated in the study, a sample size that was uniquely large for a treatment trial at that time.
The philosophy of this study design had been shown to fit well within usual healthcare: real-life data were collected within the routine and heterogeneous conditions of care; the data were collaboratively controlled and centrally supervised according to agreed pre-defined quality criteria and these were shown to be as informative as the rather artefactual information derived from strictly selected ‘samples’ of patients meeting well-defined diagnoses, but not representing real clinical populations.
The innovative aim of the principles followed and promoted by GISSI had been to provide clinical experiments with a natural epidemiological framework that bridged scientific rigour and basic needs: application of these principles in practice had been achieved by a nationwide network of non-academic NHS hospitals collaborating in strict alliance with an independent research institution. For the first time, a simple-to-use, inexpensive treatment had been shown convincingly to reduce premature death after myocardial infarction (heart attack). 6
Even before the results of the trial had been published, their importance was recognised in closing remarks at a meeting of the American Heart Association in November 1985 and in modifications of the US National Institutes of Health’s strategy on thrombolysis. Significantly, the drug regulatory authorities recognised that the available evidence supporting an old drug – streptokinase – was better than that supporting the technologically highly innovative drug tissue plasminogen activator (tPA). Although there was evidence favouring tPA in classical experimental settings, only surrogate laboratory outcomes were available.
Methodological, public health and cultural implications of GISSI’s research
Characteristics of the six studies organised by GISSI, 1984-2007.
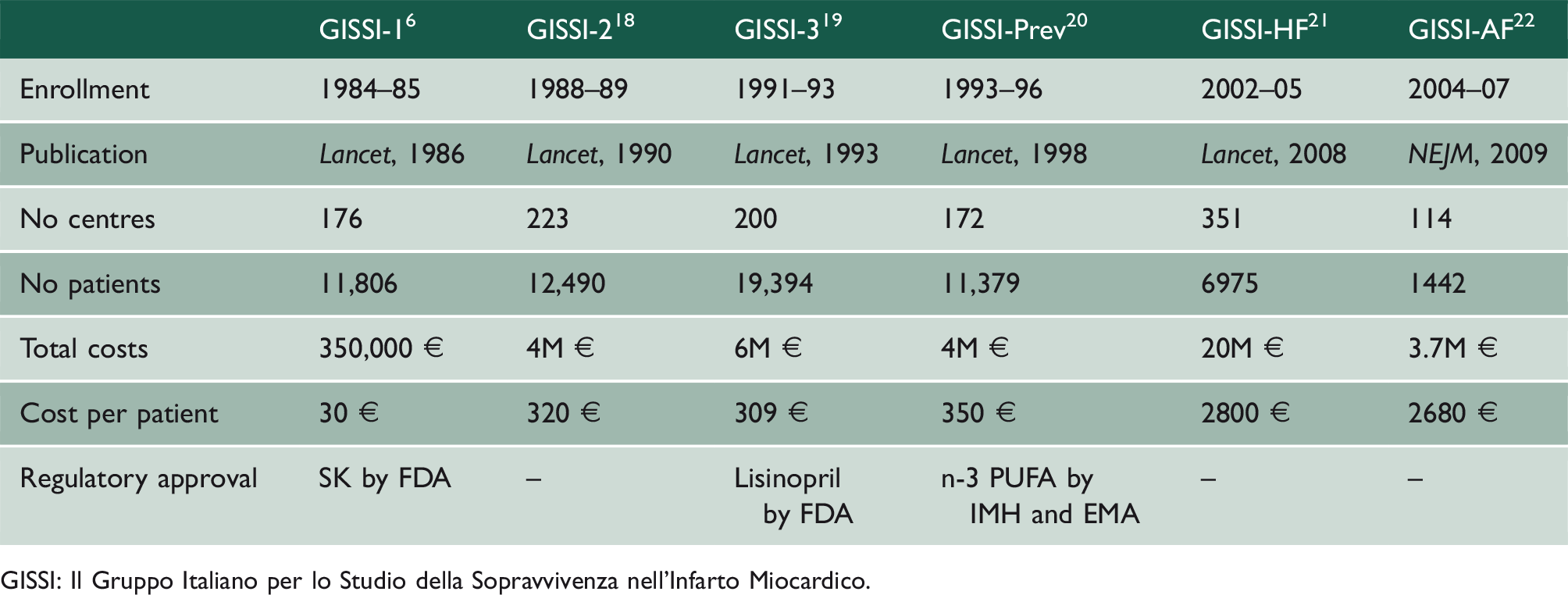
GISSI: Il Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico.
Which of the features of the GISSI trials might reasonably be accepted as fundamental to their success? And how applicable are they in the current regulatory framework for clinical trials?
Following intense and stimulating public discussions with key clinicians, the GISSI protocol was adopted as an expression of collective responsibility: it promised to produce original scientific knowledge, and to upgrade the methodological competence and the public health awareness of the cardiological community at a national level. Over a period of four months, in each of the 20 regions of Italy, the promoters of the trial were ‘on the road’ presenting, explaining and discussing what should be done to translate routine good practices into quality-controlled data collection. Participation by 90% of Italian Coronary Care Units and the completeness and quality of follow-up6 are testimony to the overall success of these initiatives.
GISSI has maintained a consistent and rigorous approach, even when faced with the new scenarios introduced with the Good Clinical Practice-International Committee on Harmonisation (GCP-ICH) rules. The support of pharmaceutical companies became necessary to comply with increasingly competitive and bureaucratically burdensome legislation and the increasing cost of trials in the trials ‘marketplace’. A clear and transparent pre-defined policy of exclusive GISSI data ownership ensured that only when the trial results had been accepted for publication would the trial datasets be made available for registration purposes.
Implications
The global events that ushered in a new era were already visible in the mid-1990s with the creation of the World Trade Organization and in the inverse hierarchy of values proposed by the philosophy and practice of epidemiology imposed by the Global Burden of Disease. 27 These made the visibility and the outcome of patients less relevant than the obsessive focus on the economic burden of diseases.
There appears recently to have been signs of a ‘re-discovery’ of the need to develop or to return to an approach that recognises that actively ‘learning health systems’ 28 are needed to assure ‘sustainability’. 29 Perhaps these can be seen as an indication that the scientific, methodological and cultural lessons from the era of GISSI-like experiences may yet be resuscitated.
