Abstract
Over the last thirty years, the management of Malignant Ureteric Obstruction (MUO) has evolved from a single disciplinary decision to a multi-disciplinary approach. Careful consideration must be given to the risks and benefits of decompression of hydronephrosis for an individual patient. There is a lack of consensus of opinion as well as strong evidence to support the decision process. Outcomes that were identified amongst patients undergoing treatment for MUO included prognosis, quality of life (QOL), complications, morbidity and prognostication tools. A total of 63 papers were included. Median survival was 6.4 months in the 53 papers that stated this outcome. Significant predictors to poor outcomes included low serum albumin, hyponatremia, the number of malignancy related events, and performance status of 2 or worse on the European cooperative cancer group. We propose a multi-centre review of outcomes to enable evidence-based consultations for patients and their families.
Keywords
Introduction
Malignant ureteric obstruction is a condition that affects patients with advanced stages of cancer. An obstructed single system can significantly reduce patients’ quality of life especially if infection ensues; however, bilateral obstruction will lead to a certain death. In fact, upper urinary tract obstruction is a prognostic indicator of morbidity for many cancers.1–3
Over the last 30 years, the management has evolved from a single disciplinary decision to a multi-disciplinary approach involving urologists, oncologists, palliative care physicians, general medicine physicians and interventional radiologists. This is mainly due to the fact that advanced stages of cancer is now treated with this multi-disciplinary approach; in addition, the surgical approach to malignant ureteric obstruction has evolved from predominantly highly morbid open surgical procedures 4 to minimally invasive techniques. 5 Brin et al. described their ‘disappointing’ experiences of open palliative procedures with patients suffering ‘an inexorable downhill course’. 6 Interestingly, oncologists are more likely to push for decompression in asymptomatic patients with a poor prognosis than urologists. 7
Individualised consideration must be given to the risks and benefits of decompression.6,8–12 Although there are recommendations within cancer-specific guidelines, both the European Association of Urology and the American Urological Association guidelines recommend decompressing the urinary systems,13,14 there is a lack of consensus of opinion as well as strong evidence to support the decision process.2,4,16,17 The National Institute for Health and Care Excellence guidelines concluded that patients should be offered decompression, but that the option of ‘no intervention should also be discussed’. They noted that there was insufficient low-grade evidence in this arena.16,17 None of these recommendations take into consideration the implications of quality of life.
To this end, we aimed to conduct a review of the literature to be able to inform the decision-making process of managing patients with malignant ureteric obstruction. Specifically, we aim to distil the relevant evidence in this paper to help facilitate an evidence-based consultation with patients and their families on prognostic outcomes of decompression in the setting of malignant ureteric obstruction. 16
Methods
Search strategy
The review was conducted using Cochrane and PRISMA guidelines.17–19 The search strategy included the following databases: the US National Library of Medicine’s life science database (MEDLINE) (1975–September 2017), EMBASE (1975–September 2017), Cochrane Central Register of Controlled Trials – CENTRAL (in The Cochrane Library – 2017), CINAHL (1975–September 2017), Clinicaltrials.gov, Google Scholar and individual urological journals.
Search terms used included: ‘malignant ureteric obstruction’; ‘percutaneous nephrostomy’; ‘stent’; ‘quality of life’; and ‘prognosis’.
Medical Subject Headings (MeSH) phrases included:
((“Stents”[Mesh]) AND “Neoplasms”[Mesh]) AND “Quality of Life”[Mesh] (((“Stents”[Mesh]) AND “Ureter”[Mesh]) AND “Neoplasms”[Mesh]) AND “Quality of Life”[Mesh] (((“Stents”[Mesh]) AND “Ureteral Obstruction”[Mesh]) AND “Neoplasms”[Mesh]) AND “Quality of Life”[Mesh] (((“Stents”[Mesh]) AND “Ureteral Obstruction”[Mesh]) AND “Neoplasms”[Mesh]) AND “Prognosis”[Mesh] ((“Nephrostomy, Percutaneous”[Mesh]) AND “Neoplasms”[Mesh]) AND “Quality of Life”[Mesh] (((“Nephrostomy, Percutaneous”[Mesh]) AND “Ureteral Obstruction”[Mesh]) AND “Neoplasms”[Mesh]) AND “Prognosis”[Mesh] (((“Nephrostomy, Percutaneous”[Mesh]) AND “Ureteral Obstruction”[Mesh]) AND “Neoplasms”[Mesh]) AND “Quality of Life”[Mesh]
Study selection
Three authors (JP, TA and OA) independently completed the review of literature independently and followed predefined inclusion criteria. Disagreement between the authors in study inclusion was resolved by consensus.
Inclusion criteria
All types of publications were included. Manuscripts involving adult patients (18 years old and above) with malignant ureteric obstruction in the English language were included. If only abstracts were available, these were included if sufficient data were extractable. We included papers reporting on benign disease if the data could be extracted separately.
Our outcome measures were:
Prognosis in patients diagnosed with malignant ureteric obstruction (across all tumour groups) who received decompression via percutaneous nephrostomy or ureteric stenting; Quality of life associated with the above; Major and minor complications; Morbidity defined as hospitalisation post intervention; Effect of decompression on renal function; Prognostication tools in use to predict poor outcomes from intervention.
Data extraction
Data of each included study were independently extracted initially by two authors (JP and TA) after which a senior author (OA) extracted the data independently and cross-checked data extraction to ensure quality assurance of data. Data were tabulated using Microsoft Excel and inbuilt formulae utilised.
The following variables were extracted from each study: number of patients; gender; intervention; age; primary diagnosis; median survival; complications; amount of time spent in hospital; proportion of lifetime spent in hospital; proportion of patients not discharged; mortality, prognostication (where available); and quality of life.
Results
The initial review yielded 169 papers (see Figure 1). Of these, 54 were excluded after abstract screening and 47 were later excluded after full manuscript review. Of the 47 papers excluded, 18 papers included benign causes, 16 had no survival data, four used surgical diversion techniques and one paper excluded patients with poor outlook. Four were not available in the English language. Three authors were contacted to obtain manuscripts but did not respond. In total, 63 papers were included in the review.
PRISMA flow chart.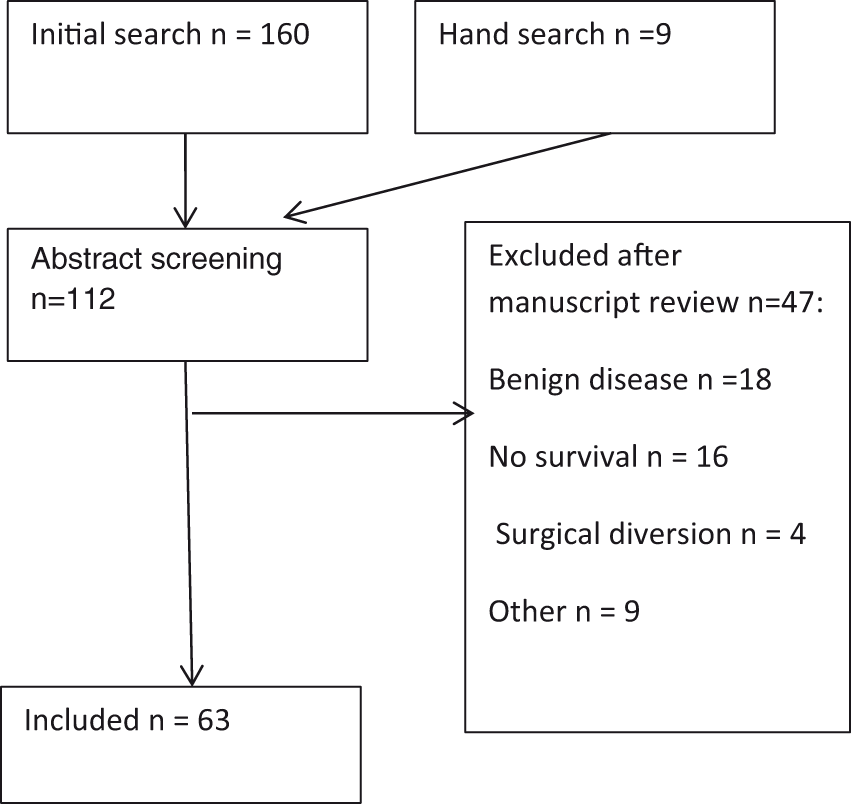
Characteristics of included studies
Seventeen studies were from United States of America, 14 from the United Kingdom, seven from Japan, five from Brazil, three from Germany, two from Greece, two from Korea. There was one paper authored from Serbia, the Philippines, Singapore, China, Pakistan, Jordan, Turkey, Israel, Sweden, New Zealand, Australia and Austria.
Only nine studies were prospective in nature; of these, one was a prospective cohort study. There were no randomised controlled trials. The follow-up period ranged from six months to eight years.
Demographics
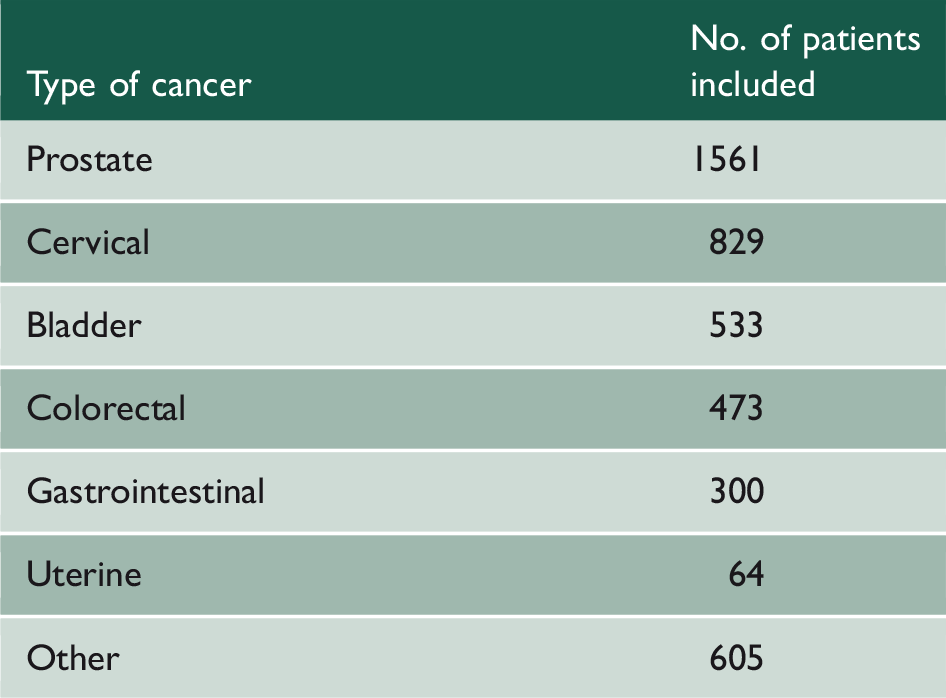
Distribution of cancers.
Prognosis
Fifty papers included prognosis as an outcome measure with a total of 2790 patients included. This ranged from 21 h to 140 months with a median survival of 6.4 months.1,3,8–11,16,20–44,44–53,55,57–60,62–65
Eight papers provided a mean one-year survival; the aggregate mean of the percentage of patients surviving one year was 23%.3,9,10,24,25,29,33,39,45
Quality of life
Twenty papers assessed quality of life with a total of 824 patients.9,11,12,20–23,25,28,31,33,45,46,49,50,61,62,66–68 Measures included time spent in hospital, pain assessment and qualitative interviews.
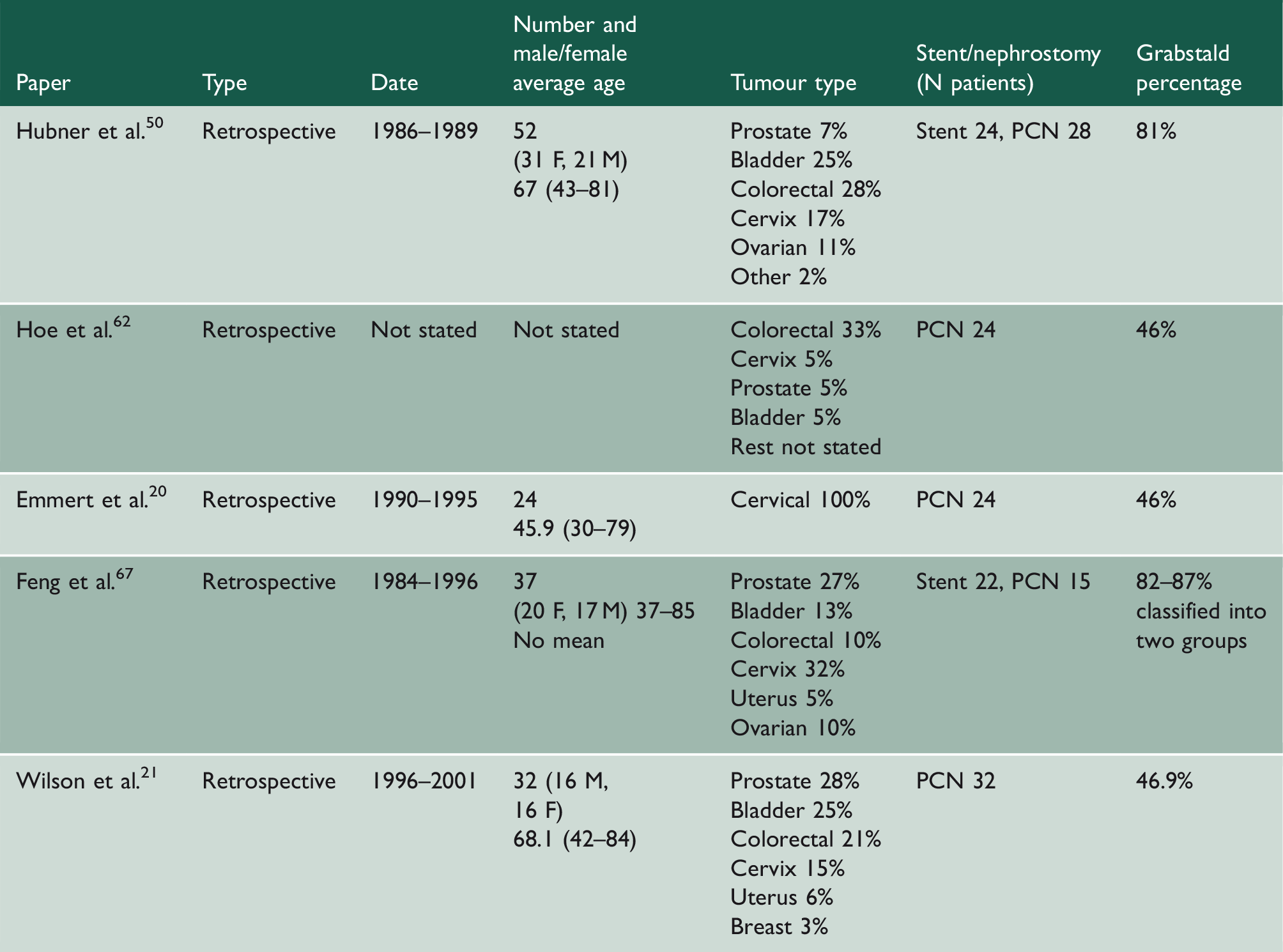
Papers using Grabstald ‘useful life measure’.
Quality of life.
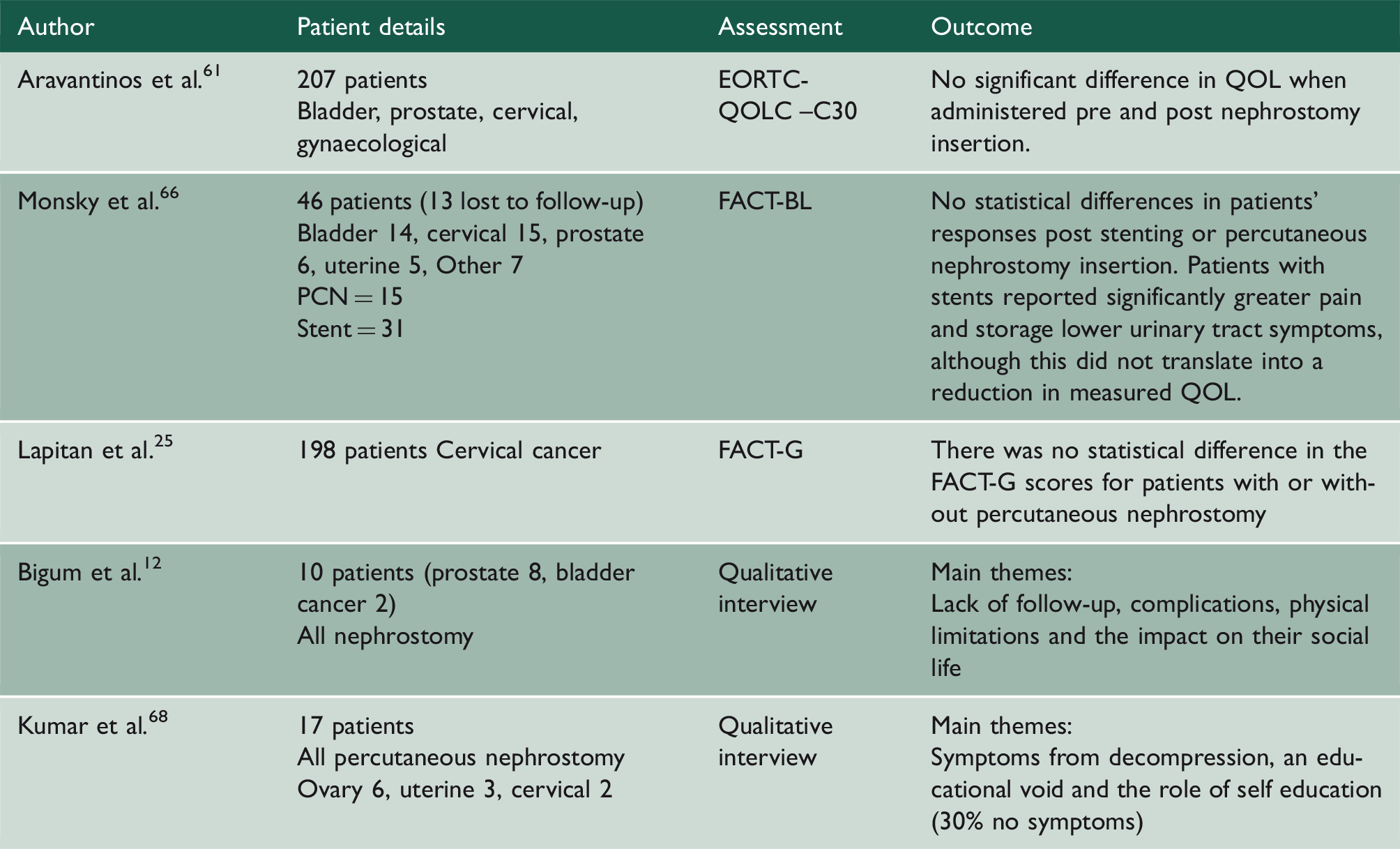
PCN: percutaneous nephrostomy; QOL: quality of life.
Complications
Twenty-four of the papers commented on the frequency of complications with a total of 1891 patients. The overall complication rate was 41%.10,11,16,22,23,26,32,33,36,39,45,51,59
Twenty-six per cent (439/1658) of patients with nephrostomies developed urinary infection, while 14% (26/180) of patients with stents placed developed infections. Ten per cent (173/1658) of patients experiencing dislodged nephrostomies, while 7% (113/1658) of patients developed blocked nephrostomies. Stent migration/dislodgement was reported in 6% (10/180). Haematuria rate was 8% (15/180) in patients stented compared to 3% (49/1658) in patients with nephrostomies. Nephrectomy rate was 0.2% (4/1658) following percutaneous nephrostomy placement; two for perinephric abscess (the indication for the other two patients was not stated).32,33 Mortality rate was 0.2% (4/1658): three from haemorrhage and one from sepsis. In the three papers that reported mortality, the overall rate was 5% (4/82).22,52,62
Twelve papers (628 patients) calculated the proportion of patients who never left hospital post decompression,9,11,21–23,28,31,33,45,55,62,64 with the pooled mean for this being 26% (range: 5–69%). Patients spent 20% of their remaining lifetime in hospital.8,23,28,33,37,44,45,48,49,53,62,64 Twelve papers included renal function pre and post procedure (a total of 1135 patients). Pre-nephrostomy, the average creatinine was 624 mmol/L and post procedure, the creatinine improved to 212 mmol/L on average.9,10,21,23,31,43,44,49,54,60,61,70
Prognostication tools
Summary of literature on prognostication.
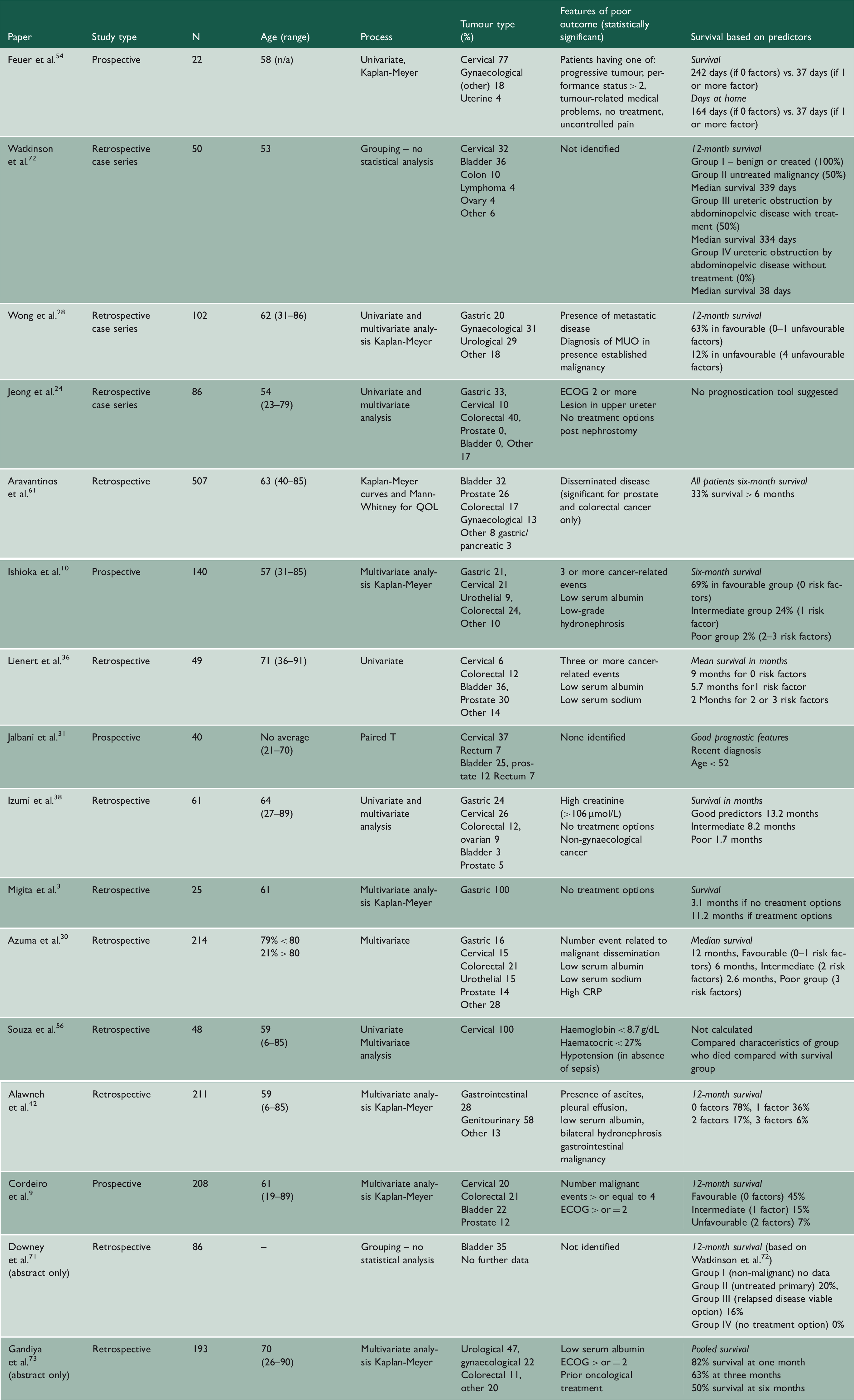
ECOG: European Cooperative Cancer Group; MUO: malignant ureteric obstruction; QOL: quality of life.
Proposed prognostication tool (PALLIATE).

Most commonly occurring statistical significance included low serum albumin,10,30,36,42,73 no further treatment options,3,24,38,54 hyponatraemia,30,36 number of malignancy-related events (pleural effusion, metastatic disease, ascites),10,30,36,42 the presence of metastatic disease,28,61 performance status of 2 or worse on the European Cooperative Cancer Group.9,24,54,73
Patients with a malignancy of unknown primary or gastrointestinal origin were identified as having poorer outcomes,28,42 whereas gynaecological malignancies had a better outcome.38 Other variables included patients with upper ureteric obstruction, 24 moderate–severe hydronephrosis, 25 bilateral hydronephrosis,42 elevated creatinine,25,38 anaemia56 and patients with an elevated C-reactive protein. 30
Discussion
This review of 63 papers gives a broad survival range for patients with malignant ureteric obstruction between 21 hours and 140 months.1,3,8–11,16,20–44,44–53,55,57–60,62–64 The median survival was 6.4 months and the percentage of patients alive at one year was 23%.3,9,10,24,25,29,33,39,45 Reasons for this variation in survival include the heterogeneous patient and cancer groups involved. Additionally, the data are limited by the fact that researchers in some instances may have included patients with retroperitoneal fibrosis secondary to treatment (such as radiotherapy) rather than ongoing or recurrent disease.74,75 The benign nature of this aetiology for obstruction would skew results towards improved outcomes. The majority of the papers reported on patients who underwent decompression, thereby not capturing a proportion of patients who were not decompressed.
Two pertinent questions are always presented: what are the preferred options for relieving malignant ureteric obstruction? And what is the expected prognosis? 5 In terms of methods of decompression, a previous comparative study discovered no relative superiority of retrograde stenting to percutaneous nephrostomy in the setting of infected obstructed uropathy caused by stones.76 Two recent review articles concluded that there were no data on the superiority of stent vs. percutaneous nephrostomy ± subsequent antegrade stent when considering malignant and benign ureteric obstruction.5,77
Clearly, the aim of relieving the obstruction depends on patient factors but would include improving renal function to enable further oncological treatment, to correct the symptoms of renal failure and to improve pain. 7 This must be balanced against a patient’s expectation of quantity and quality of life. This is, of course, a challenging consultation, particularly in the acute setting when such patients often present.71 Despite extensive retrospective publications and several review articles, there is a paucity of data assessing the important issues of quality of life and prognostication in this cohort of patients. 5
European Association of Urology guidelines on pain management recommend that for pelvic malignancies ‘it is good practice to drain symptomatic hydronephrosis at once, and to drain only one kidney (the less dilated and better appearing kidney or the one with the better function, if known) in asymptomatic patients’. 14 They conclude that a nephrostomy tube is superior to a double-J stent for drainage for pelvic malignancies but advocate either stenting or nephrostomies in other tumour groups. 14 Neither of these recommendations reference the literature nor do they mention implications of quality of life.
In the context of locally advanced non-metastatic bladder cancer with hydronephrosis, American Urological Association guidelines suggest placement of a ureteral stent. 13
Complications
Another frequently neglected statistic for patients before them undergoing decompression is the proportion of time spent in hospital and the risk of complications. The proportion of patients who had complications was 41%, with 26% of patients never leaving hospital. Those who had an intervention spent 20% of their resultant lifetime in hospital. One paper stated that 69% of their patients never left hospital. Removing this apparent outlier from the pooled mean resulted in the figure of 17.8%. 22 Despite improving renal function, creatinine did not return to baseline for patients, potentially avoiding an emergency situation but not reversing the damage caused by hydronephrosis.
Quality of life
Quality of life can be challenging to measure; early papers measured quality of life using the ‘useful life’ measure.50,67 Feng et al. and Hubner et al
Three studies utilised patient-reported outcome measures demonstrating no statistically significant improvements in quality of life pre and post decompression.25,61,66 However, despite improvement in the use of patient-reported outcome measures, there were several limitations with the studies. Limitations of the Monskey et al
Two papers looked at qualitative interviews. Qualitative analysis is helpful to develop themes. However, a small sample size, the challenges of confounders, single tumour group inclusion and the exclusion of patients without nephrostomies may limit its wider application.12,68
Prognostication
One group performed a prospective cohort study of patients with cervical cancer. Lapitan et al. followed up a cohort of patients who had malignant ureteric obstruction and assessed the outcomes of two groups: those who were decompressed and those who were not. 25 At the outset, there appears to be a survival benefit with 38% vs. 28% survival at six months for those who underwent decompression vs. those who did not. By 12 months, however, both groups had the same survival of 16%. 25
The most frequently found statistically significant indicators of poor prognosis among the literature were low serum albumin, no further treatment options, number of malignancy-related events (pleural effusion, metastatic disease, ascites), performance status of 2 or worse on the European Cooperative Cancer Group, the presence of metastatic disease and hyponatremia (Table 4). Two papers divided patients into groups depending on treatment options available; those with no treatment options had 0% 12-month survival and a median survival of 38 days. Combining these parameters with a larger patient group may help develop a prognostication tool for clinicians to aid decision-making (See Table 5).
Comparison of prognostication tools demonstrates that patients with none or one risk factor have more favourable outcomes with 12-month survival ranging from 20% to 78%28,42,71,72 and a median survival ranging between 9 and 13 months.30,36,38 In those patients with ‘intermediate’ risk factors (see Table 3), median survival ranged from 5.7 to 8.2 months.30,36,38 For those patients with two or more risk factors, median survival ranged from 1.7 to 2.6 months30,38 and 12-month survival ranged from 0% to 12%.9,28,42,71,72
Limitations
The reviewed data have significant heterogeneity, making conclusions regarding specific tumour types challenging. Sountoulides et al
There are only 25 papers published in the last 10 years; in this time, however, there have been significant advances in oncological treatment options. Therefore, outcomes may well be influenced by more conservative historic data.
Another limitation of this analysis is the retrospective nature of the data; there were only nine papers that were prospective in nature.9,10,25,31,43,48,54 Data on survival are lacking on those patients conservatively managed with only one paper including untreated patients in its survival data, which may lead to overstating median survival. 25 There are clear worldwide variations in practice regarding discharge home. In one series, 69% of patients did not leave hospital after decompression; 28 this contrasts with an average of 17% among other papers.
Limitations when reviewing prognostic tools include the fact that the majority of studies were retrospective in nature. The use of statistical analyses included univariate, paired-t as well as multivariate analysis, thus limiting transferability and utility on an individual patient basis.
Conclusion
In this post Montgomery era with the concept of the ‘reasonable patient’, can we continue to justify discussing decompression without stating to patients the evidence-based risks and benefits from the emergent body of literature?82 An overall complication risk of 41% and up to one-quarter of patients' remaining lifetime spent in hospital with a median survival of 6.4 months may encourage clinicians and patients to rethink the appropriateness of such interventions. We propose a contemporary multicentre prospective review of outcomes of this cohort of patients to enable evidence-based consultations for patients and their families. Further work in the domain of prognostication is needed to help best identify those patients who may benefit the most from decompression.
Footnotes
Declarations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
