Abstract

This article is in our series on thoracic surgery.
Introduction
Lung ablation can be achieved using a variety of techniques. Most use thermal energy to destroy a small volume of lung tissue. This can be done by cooling (cryoablation) or heating (usually with radiofrequency or microwave energy).
There is growing evidence to support the use of image-guided ablation in oligometastatic pulmonary disease. The evidence for use in primary non-small cell lung cancer is less convincing.
A radiofrequency electrode or microwave antenna is passed through the patient’s skin under image guidance into the tumour. A generator powers the device which discharges energy into the tissues adjacent to its tip. Much of the available literature is based on outcomes after radiofrequency ablation, but there is a growing body of evidence to support microwave ablation.1–4 Generally, image-guided ablation is safe, repeatable and well tolerated.
Methods
An online literature review was performed. Personal opinions expressed in this commentary are based on the authors' 10-year experience of lung ablation.
Technique
Radiofrequency ablation and microwave ablation both produce focal heating. Microwave ablation times are typically much shorter (2–5 min as opposed to 12–15 min for a similar size treatment area). There are several theoretical advantages of microwave ablation, although these are yet to be validated. For example, charring, which can effectively impede the current in radiofrequency ablation by causing a high tissue resistance, is not an issue. There is also some suggestion that recurrence, which can occur because of the cooling effect of adjacent vessels (the so-called ‘heat sink’ effect), is reduced, although there are few rigorous data to support this. Recent guidance published in the United Kingdom by the National Institute for Health and Care Excellence (NICE) has summarised the case study data for microwave ablation in the chest and was unable to draw any firm conclusions on its safety or efficacy, but commented that because its mechanism of action is very similar to radiofrequency ablation, similar outcomes are expected. 5
Ablation can be performed under general anaesthetic or deep conscious sedation. There are advantages and disadvantages to both and ultimately this will depend upon local preferences. No difference in outcome was reported by Hoffman et al. when comparing procedures performed under general anaesthesia or sedation; however, several of the treatments could not be completed under sedation and a subsequent general anaesthesia was needed. 6
The patient is positioned on the computed tomography couch to allow a safe percutaneous access route. Computed tomography fluoroscopy with single shot axial image acquisition is used routinely. Full computed tomography helical imaging is used to confirm the position of the electrode with regard to both the lesion and any adjacent structures. Needle positioning is planned such that the predicted ablation zone encompasses the mass with an adequate margin (Figures 1 and 2). A pneumothorax or dextrose buffer can be introduced to protect the chest wall or other thoracic structures and traction can be applied to the electrode to move lesions into more favourable positions for treatment (Figure 3).
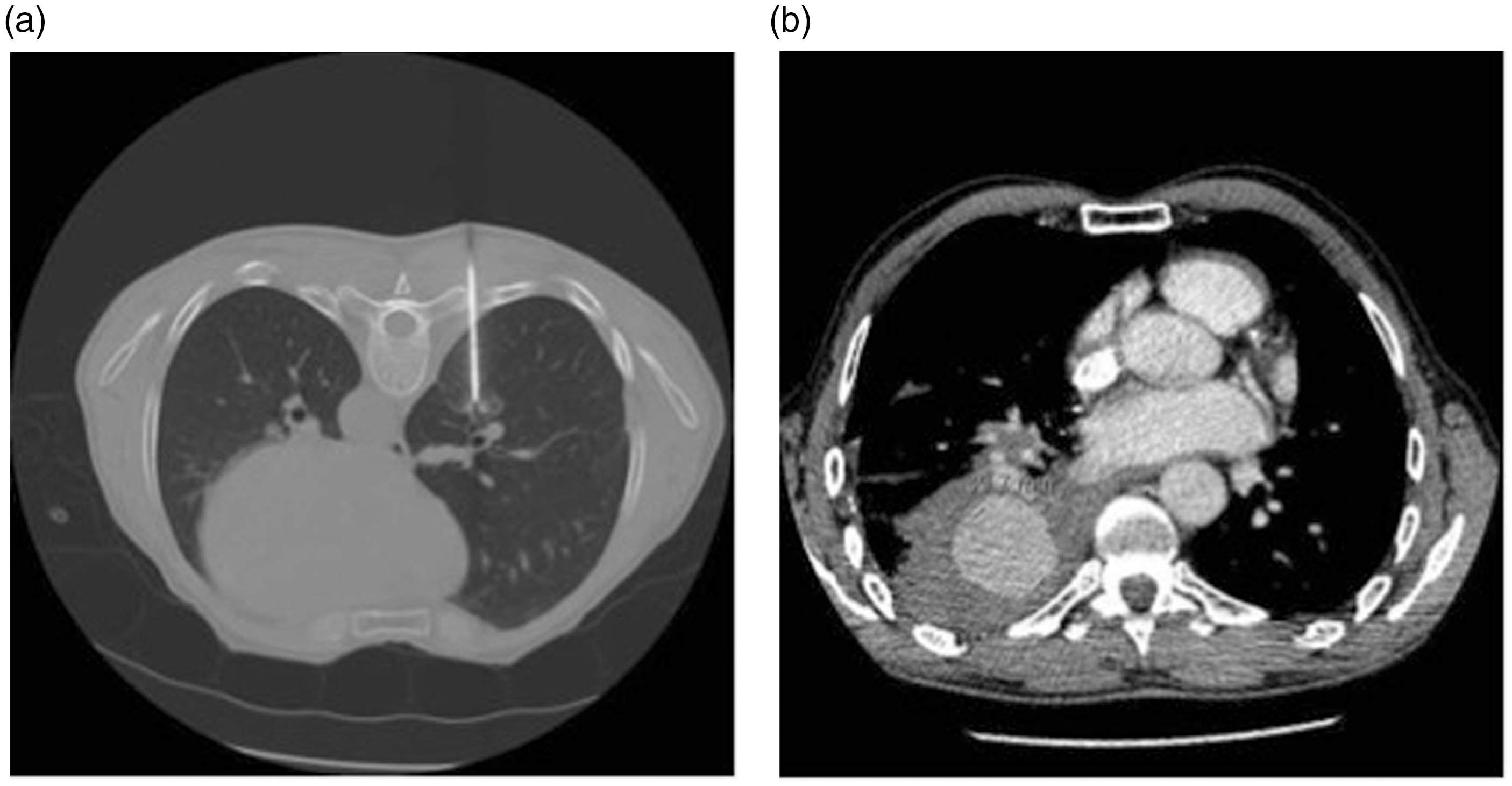
Computed tomography showing radiofrequency ablation electrode with expandable tines deployed in right lung to treat 5 mm lung mass. (a) Computed tomography showing radiofrequency electrode placed in small right lower lobe biopsy proven non-small cell lung cancer. (b) Computed tomography at one month after treatment showing cavity encasing tumour. Patient remained disease free and this area remained positron emission tomography negative until his death from unrelated causes seven years later. (a) Computed tomography showing radiofrequency electrode deployed to treat small right lower lobe sarcoma metastasis. Treatment proceeded without incident. (b) Computed tomography performed 24 h after (a) when patient's condition deteriorated. Computed tomography shows large enhancing mass at site of ablation, consistent with a large false aneurysm.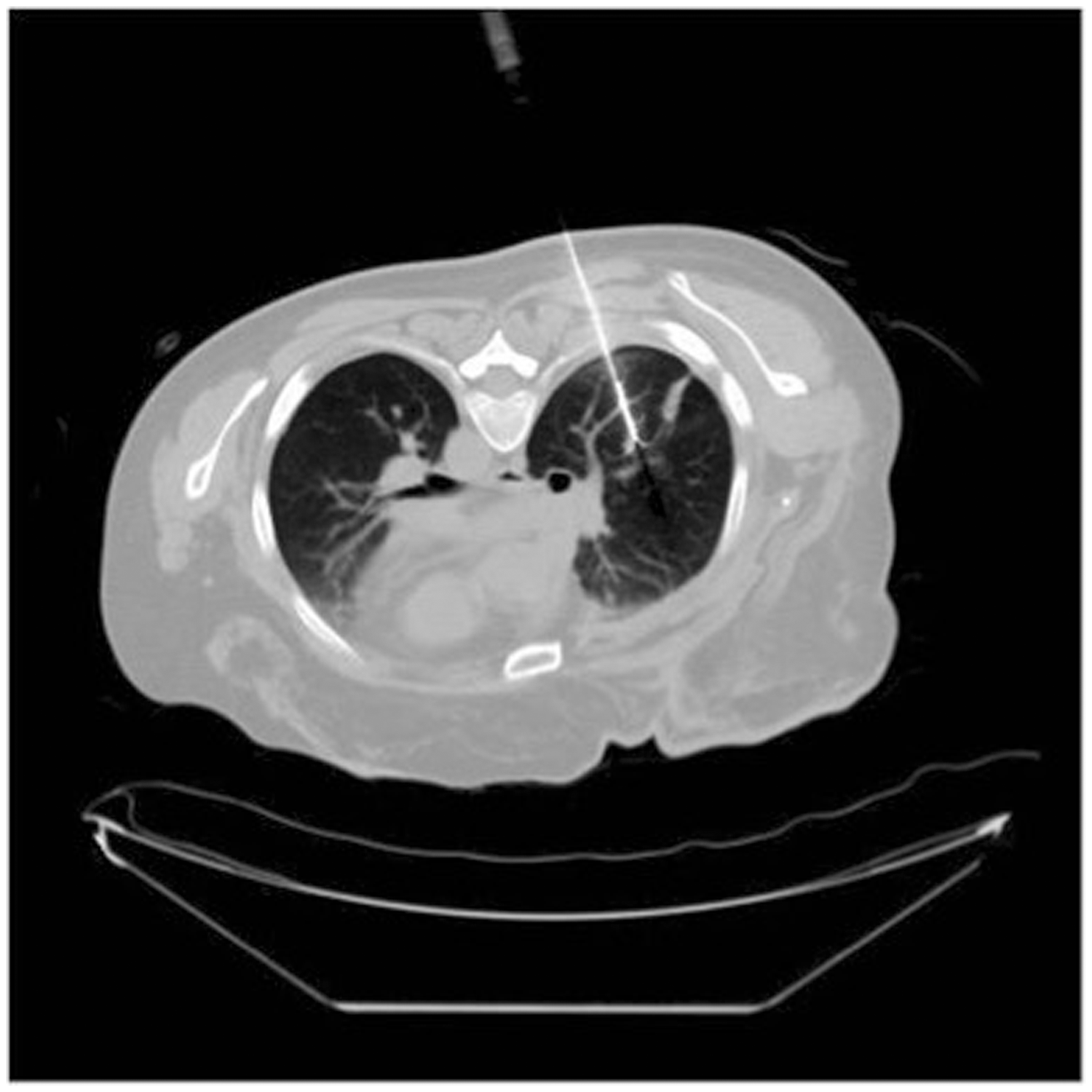
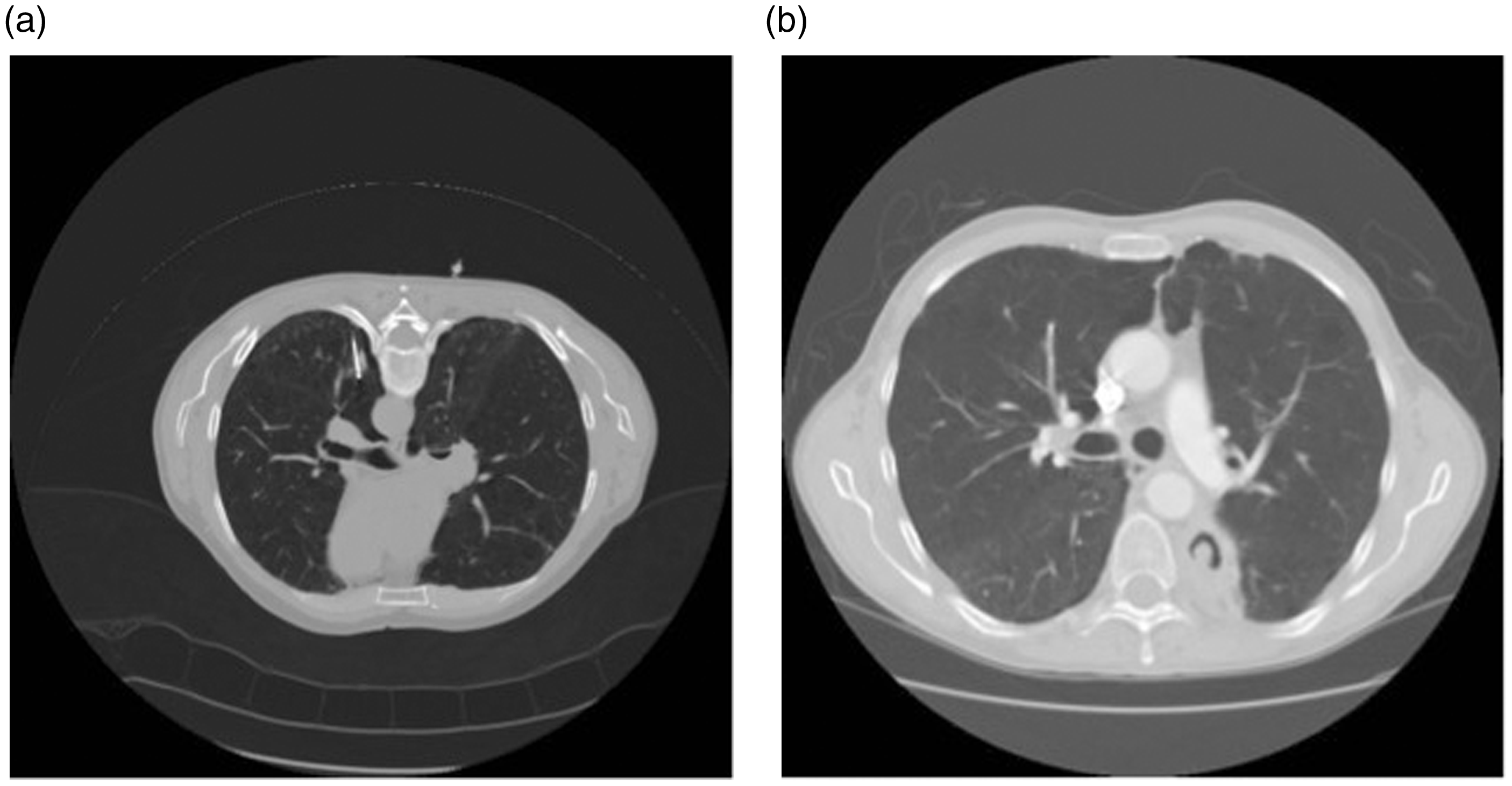
Multiple lesions can be treated at one sitting, although most practitioners will not treat bilateral disease at the same attendance because of the risk of bilateral pneumothoraces or severe haemorrhage. Procedure duration will vary, but a single lesion can be easily treated in 30–60 min.
Length of stay will depend upon local facilities and practice, but many centres now offer lung ablation as a day-case procedure. In rare instances, the opinion of a thoracic surgery centre may be needed in the management of complications but on site thoracic surgery is not mandatory.
Given its minimally invasive nature, ablation can be repeated if there is failure of local disease control or if new tumours appear. This is an obvious advantage over surgery.
Indications
Lung thermal ablation can be used to treat both primary and secondary thoracic malignancies. Treatment criteria vary but typically patients with primary tumours of 3 cm or less in diameter are considered suitable. Preferably lesions should be surrounded by aerated lung, but the suitability of more central or peripheral masses is made on a case-by-case basis.
Evidence to support the use of thermal ablation, particularly for metastases from colonic primary tumours, is now strong, with survival data in selected groups of patients, approaching that seen after surgery.7,8
Primary lung tumours
The use of ablation in primary lung tumours is driven by the fact that approximately one-third of non-small cell lung cancers are unsuitable for curative surgery. The RAPTURE multicentre prospective trial, which reported in 2008 and included patients with both primary and secondary tumours, showed a 99% technical success rate and a two-year overall survival rate of 48% in patients with non-small cell lung cancer and 66% in patients with pulmonary metastases from a colorectal primary. 7 A recent retrospective analysis in a group of 64 patients showed similar outcomes when thermal ablation and sublobar resection were compared and other studies suggest a particular advantage of local ablation in medically unfit patients.9,10 As yet, there are no data available directly comparing ablation with high-dose targeted radiotherapy (stereotactic body radiotherapy). However, in a recent review of the literature, there is clear support for stereotactic body radiotherapy in the treatment of early non-small cell lung cancer. This review concluded that stereotactic ablative therapy offered a five-year local control rate of 83–89.5% as opposed to 58–68% with radiofrequency ablation, which had a short follow-up of only 18 months. Both overall survival and cancer-specific survival were also better with stereotactic body radiotherapy with a three-year overall survival in the range of 38–84.7% and a cancer-specific survival of 64–88%, whereas overall survival data were only available for two radiofrequency studies and reported as 47–74%. Furthermore, the post procedural morbidity was higher for radiofrequency ablation at 33–100%, mainly due to the incidence of pneumothorax. Radiation pneumonitis and rib fracture were the most frequently seen post-radiotherapy complications in 3–38% and 1.4–6%, respectively. 11
A more recent review of radiofrequency ablation in non-small cell lung cancer reached a similar conclusion. Radiofrequency ablation was shown to have a higher local failure rate, with 31–42% of patients showing local progression and overall survival rate lower than stereotactic body radiotherapy. The authors of this study conclude that radiofrequency ablation may be best reserved for early stage non-small cell lung cancer patients who are unfit for surgery or sublobar lung resection. 12
It is worth noting that the small studies of image-guided ablation which have included primary non-small cell lung cancer are very heterogeneous in composition. Lesions with a wide range of volumes in patients with varying co-morbidities have been reported. There may be a role for ablation as a first treatment modality in small primary lung tumours <15 mm in size, where the ability to obtain a good treatment margin may rival that seen in conventional surgery, although at present, this remains speculative. Computed tomography screen and incidentally detected pulmonary nodules may lend themselves to such intervention.
Overall survival is a poor indicator of the efficacy of treatment as the literature is predominantly based on cohort studies where survival is highly influenced by patient selection. For this reason, a better marker of success is probably local disease control. It is also noteworthy that there are no randomised controlled trials comparing ablation with lung resection or stereotactic body radiotherapy. Furthermore, it is important to understand that for the most part, the published evidence focuses on efficacy rather than clinical effectiveness, for which there is a paucity of evidence. The absolute lack of randomised clinical trials also means there is no evidence to support or refute the role of non-curative or ‘debulking’ ablation. Early reports of debulking hepatic metastases from breast carcinoma suggest that the technique is feasible, but it is difficult to draw any firm conclusions with regard to survival advantages. 13 There are case reports of symptom relief after debulking of thoracic malignancy, but this remains an area where further investigation is required. 14
There may be a role for biopsy immediately followed by ablation, particularly in those patients unfit for other treatment, although there is little current evidence to support this. This approach does have potential advantages, namely the ability to make an accurate diagnosis and potential curative treatment with a single clinical episode. Many of the existing studies suffer from a lack of pre-treatment histology and such an approach would also allow a better degree of prognostication.
Local control for all tumour types does seem to be influenced by the size of the treated lesions, with lesions in excess of 3 cm at the time of treatment having a much higher incidence of local progression. 15 An early report by Lee et al. 16 described success rates of only 38% for primary tumours over 50 mm.
Post procedure
Post procedure imaging appearances are variable and can range from a rounded area of relatively dense scarring to a barely perceptible region of atelectasis or fibrosis. Pleural effusions are common as are small pneumothoraces. These have usually resolved by three to six months.
Generally, lung ablation has no measurable long-term effect on pulmonary function. No changes in formal pulmonary function tests were found one month after radiofrequency ablation in a study by de Baere et al. 17 There is no defined cut-off with respect to forced expiratory volume below which ablation is considered unsafe. Treatments are reported in patients with an forced expiratory volume of less than 0.8 l.
Complications
Mortality
Ablation has a reported specific mortality rate in the range of 0.4–2.6%. 18 Patients with a single lung have been treated safely. In a retrospective analysis published in 2007, four deaths occurred in 153 patients as a consequence of radiofrequency ablation and two of these four were single lung patients.
Morbidity
The rate of major complication (as defined by the Society of Interventional Radiology as one requiring remedial action or where the patient experiences significant morbidity) is 9.8–17.1%.18–20
Pneumothorax
The rate of traumatic pneumothorax developing during a procedure can be as high as 50% with a chest drain needed in up to 50% of these pneumothoraces.21,22 The threshold for placing a chest drain varies. 22 Pre-existing emphysema is also an independent risk factor for pneumothorax requiring pleurodesis. 18 There may also be an increased risk in the context of previous thoracic surgery. 23 Persistent air leak as a consequence of broncho-pleural fistula is reported but is very rare. 24 Surgical emphysema is common but is usually self-limiting.
Pleural fluid
The incidence of an aseptic pleuritis is reported as 2.3%. 18 The number of pleural punctures and previous systemic chemotherapy are significant risk factors. 18 Large pleural effusions are relatively uncommon and can simply be reactive. If patients are symptomatic or have abnormal inflammatory markers, aspiration is advised. Haemothorax is also rare.
Pulmonary abscess
Pneumonia occurs in 1.8% of treated patients with an increased risk in patients who have received prior radiotherapy. Age also seems to be an independent risk factor for pneumonia. 18 Lung abscess is rare, with a reported incidence of 1.6% patients. 18 Pre or post procedure antibiotics are not recommended routinely, but some centres do advocate their use in high-risk patients.
Vascular complications
Pulmonary haemorrhage
Mild pulmonary parenchymal haemorrhage is a near universal finding on the immediate computed tomography. This usually settles and significant haemoptysis is uncommon. Large parenchymal haemorrhage occurs in 7–8%21,25 but is often asymptomatic. Tumour size and platelet count have been identified as independent risk factors for haemorrhage. 18 Using the Common Terminology Criteria for Adverse Events system, Grade 4 haemorrhage (defined as life-threatening) is rare and reported at 0.3% in a large series of ablations. 18 Such massive haemorrhage is usually as a result of bleeding from a bronchial artery operating at higher pressure than the pulmonary circulation.
There are several case reports of non-fatal large air embolism occurring as a consequence of electrode placement, rather than the ablation itself.26,27
Psuedoaneurysm
Vascular injury resulting in pseudoaneurysm is uncommon, occurring in 0.2% of patients in a series of 538 pulmonary ablations (Figure 4). The risk is higher when the treated lesion lies close to a large vessel. Most pseudoaneurysms are small and are often asymptomatic. Spontaneous resolution is reported, but rupture and massive haemorrhage can occur.
28
Rupture has been reported between 3 and 17 days post treatment.
28
Radiofrequency ablation electrode has been passed into a right para-mediastinal tumour (white arrow). A 21G needle (black arrow) has been inserted into the pleural space and 200 ml of air injected to produce a pneumothorax. Traction applied to the electrode to strip the tumour away from the mediastinum and superior vena cava (*) to prevent thermal injury.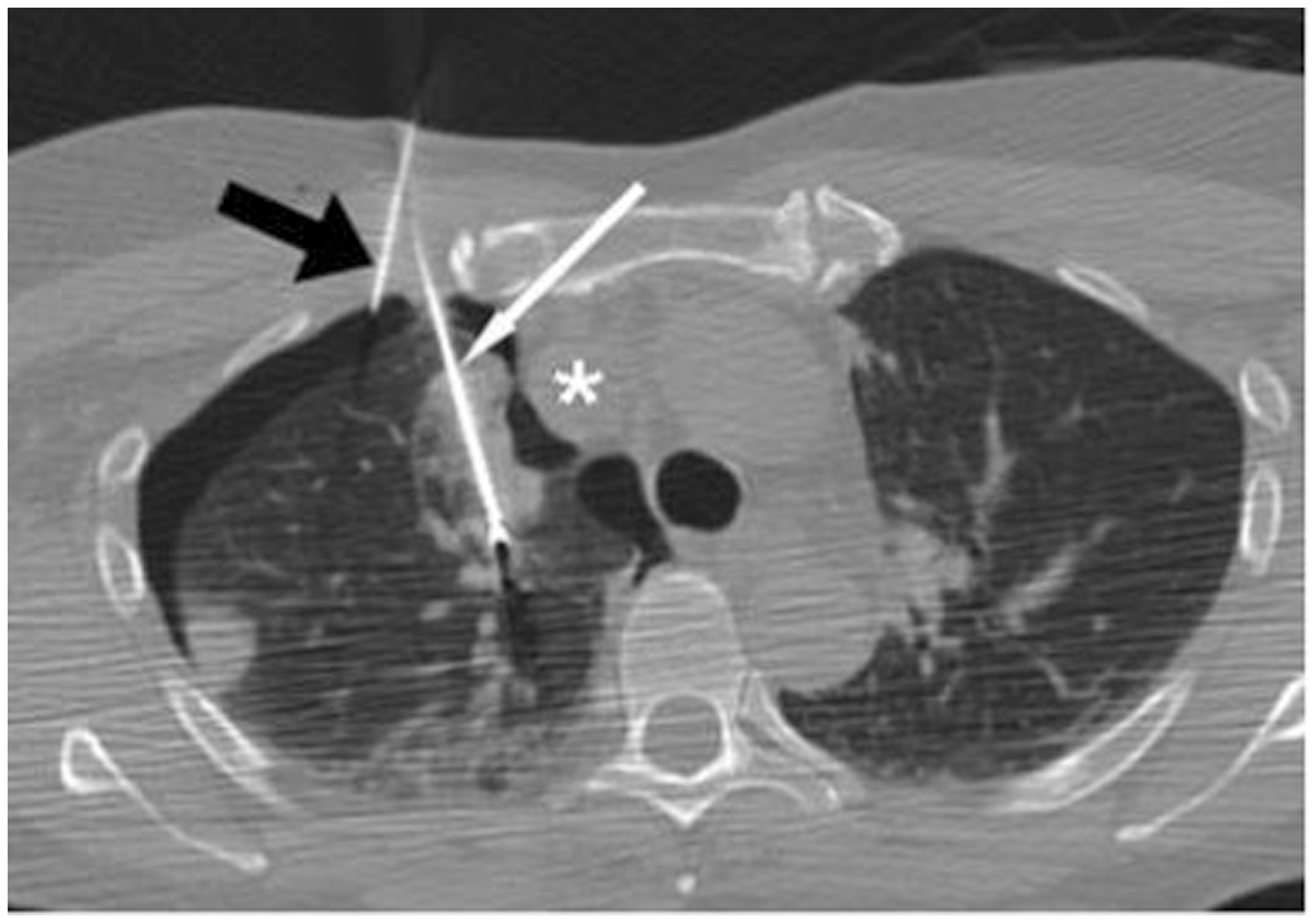
Lung inflammation
Acute interstitial pneumonitis has been reported, although its aetiology is uncertain. In a retrospective study of 327 ablations in 130 patients, the incidence of interstitial pneumonitis was 0.6%. The risk was greater in patients with tumours of 2 cm or more in diameter and in those individuals who had previously received external bean radiotherapy. The typical computed tomography findings are of dense ground glass change, interstitial thickening and frank consolidation. The disease can be fulminant and has a 50% reported mortality. 29
Thoracic wall injury
With lesions near to the pleura, there is a risk of pleural and chest wall injury. 30 Damage to intercostal nerves can lead to persistent neuralgia and these symptoms can be difficult to manage. However, the pain is almost always self-limiting. Superficial skin burns can occur, but should be avoided with careful multi-planar pre-treatment measurement of the electrode tip distance from the chest wall. The incidence of rib fractures related to thoracic ablation is reported to be as high as 13.5%, although the exact mechanism is uncertain. 31 Damage to the diaphragm is also reported, with an incidence of 0.1%. 32 This is usually self-limiting, but frank perforation requiring surgical repair is described. 32
Tumour recurrence
Local recurrence is the commonest pattern of relapse after treatment and is more commonly seen in primary tumours than with metastases, perhaps reflecting differences in tumour biology. It has been reported to be as high as 28% at two years, in a series of primary lung tumours.33,34 Risk factors include size of the tumour and the stage of disease at presentation. Masses close to blood vessels > 3 mm in diameter have a higher rate of relapse due to under treatment because of the so-called ‘heat sink’ effect where tissue is cooled by flowing blood
35
(Figure 5). Modalities other than computed tomography have been used to follow-up ablated tumours. Several studies exploring the role of positron emission tomography have been published and these suggest a characteristic set of findings in the successfully ablated tumour including a large decrease in the standardised uptake value in the ablated area. Positron emission tomography-computed tomography may detect recurrence earlier that conventional computed tomography, but there is limited evidence to support this. In a study of 68 patients, a standardised uptake value of less than eight was found to be a predictor of improved disease-free survival. After treatment reduced recurrence-free survival correlated with an unfavourable FDG uptake pattern, the absolute value of the post radiofrequency ablation standardised uptake value and an increase in standardised uptake value over time after ablation.
36
(a) Image showing radiofrequency electrode in position close to a small metastasis which lies immediately adjacent to the lower lobe pulmonary artery. (b) at one month after treatment, shows the metastasis is smaller, indicating a satisfactory response. (c) Computerised tomography at four months in the same patient showing an increase in the size of the mass (arrow) indicating recurrence. This is due to ‘heat sink’ effect of the adjacent large vessel which cools the tumour margin and prevents adequate ablation.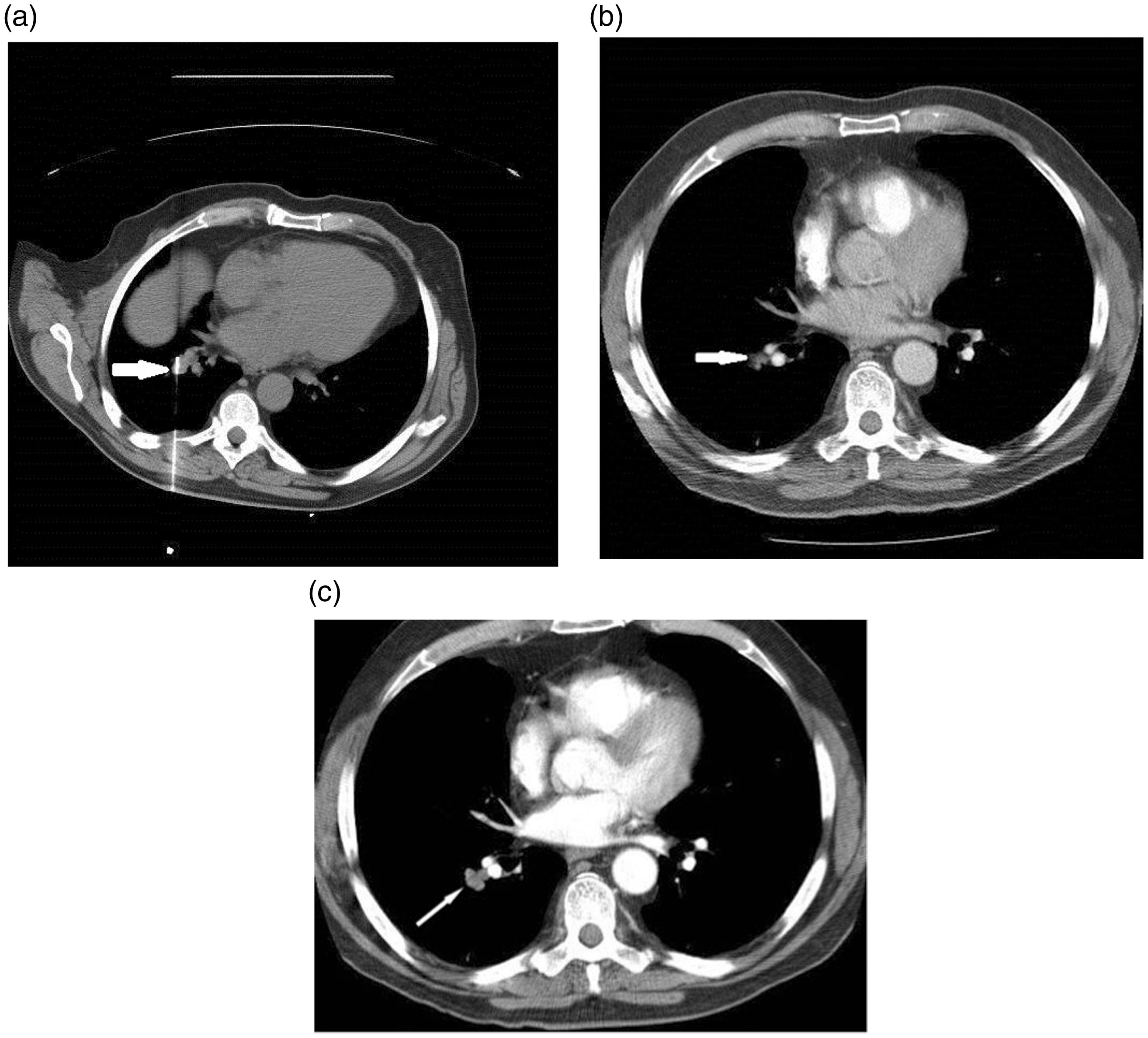
The use of diffusion-weighted magnetic resonance and magnetic resonance and computed tomography perfusion has also been reported, although again their effectiveness is undefined.
Future directions
With the advent of modern computed tomography, the incidence of incidentally detected lung nodules has increased. The possible introduction of lung cancer screening will also present a significant challenge with regard to the management of early lung cancer. Current British Thoracic Society guidelines suggest a role for image-guided ablation in those patients unfit for other treatment options. 37 With the projected increase in the number of very small early primary lung tumours, it is possible that ablation will have a bigger role, but there is a dearth of good quality trial data to support the clinical effectiveness of the technique.
Conclusion
Most published evidence for lung ablation is based upon radiofrequency ablation, but these results are translatable to microwave ablation. Microwave ablation does offer some theoretical advantages to radiofrequency ablation, including a shorter treatment time, higher end temperature and less 'heat sink effect'. Pulmonary thermal ablation is now an accepted treatment for lung metastases and some lung primary tumours.
Treatment has a high technical success rate, low mortality, low morbidity and low incidence of local recurrence which is comparable to surgery. Compared to surgery ablation offers a shorter hospital stay, with day case treatment feasible. It is also less expensive and potentially easier to repeat if there is failure of local control or if new tumours appear. There is growing evidence of its efficacy, particularly in those patients with oligometastaic disease and overall survival, local progression and disease free survival are similar when compared to sub-lobar resection for pulmonary metastatic disease.
The key to successful treatment depends upon patient selection. Those patients with true oligometastatic disease and a long disease free interval between the treatment of the primary tumour and the development of metastases have better outcomes. Lesions less than 3cm in diameter, situated away from large vessels are ideally suited for ablative therapy and have a low risk of local recurrence.
The evidence for using ablation in primary lung cancer is less compelling and SABR/surgery seems to offer advantages in this patient group. However there may be a role for small primary lung non-small cell cancer, particularly when other treatment options are limited.
The literature suffers from an absence of randomised control trials and there are many unresolved issues including the role of ablation in the treatment of small primary lung tumours, its efficacy when combined with other treatments particularly with small incidentally or screen detected tumours and its potential for tumour debulking, particularly as a prelude to systemic therapy.
