Abstract

Introduction
Stereotactic ablative radiotherapy
Stereotactic ablative radiotherapy is defined as the use of external beam radiotherapy to deliver high doses of radiation (usually ≥6 Gy) very precisely to an extracranial target within the body, as a single or small number of fractions. High doses of radiation result in larger biological effective doses and have unique radiobiological processes, 1 resulting in dramatic tumour responses thus termed ‘ablative’.
It was first developed in 1951 to treat brain tumours which were surgically inoperable. Over the next half century, significant improvements in radiotherapy techniques led to improvements in the precision and safety of radiotherapy thus allowing delivery of higher doses of ablative radiation to target lesions. This technique is also known as stereotactic body radiotherapy for extracranial lesions and stereotactic radiosurgery for intracranial lesions.
Technicalities of stereotactic ablative radiotherapy
To achieve ablative doses, the machines employed to deliver stereotactic ablative radiotherapy treatments utilise multiple, finely collimated radiation beams or arcs to produce highly reproducible and conformal treatment plans. Radiotherapy plans will require tight margins and steep dose gradients between tumours and surrounding normal tissues. Four-dimensional computed tomography simulation scans that acquire images throughout the respiratory cycle are often used to take into account organ movements due to free breathing.
An important aspect of the treatment is precise patient positioning and immobilisation using body fix or similar devices to ensure set-up accuracy. Accurate targeting of lesions using image guidance or fiducial matching is also important. During the treatment of lung and liver sites which are susceptible to intrafraction respiratory movements, multiple methods can be employed to reduce movement uncertainties such as respiratory gating, abdominal compression, deep inspiration breath-hold techniques or real-time tracking.
Given that high doses per fraction and tight margins are used, it is important to minimise any mistakes as this would result in significant target miss or serious overdoses, thus rigorous quality assurance programmes 2 must be in place and strictly maintained.
Stereotactic ablative radiotherapy in early non-small cell lung cancer
Historically, patients with inoperable early stage non-small cell lung cancer would be treated with conventionally fractionated radiotherapy (using <2–3 Gy per fraction over a period of 3–6 weeks). The results were often lower than surgical outcomes with five-year cancer-specific survival rates of between 12% and 39%. 3 Methods to improve on radiotherapy outcomes were needed.
Some of the earliest phase 1 study data in lung stereotactic body radiotherapy were published in 2003 by Timmerman et al. The same group subsequently reported in phase 2 trials excellent three-year local control rates of 90.6%. 4 This landmark trial now forms the basis for international lung stereotactic ablative radiotherapy practice.
Stereotactic ablative radiotherapy in early stage (T1-2aN0) inoperable non-small cell lung cancer has been established in several international guidelines (Figure 1). Many prospective trials using different dose regimens have published excellent 2–3-year local tumour control and 1–3-year overall survival rates: 84–98% and 43–72%, respectively.
5
These studies also reported very low rates of severe, grade 3–4 toxicities. The optimal dose and schedule for stereotactic ablative radiotherapy varies between centres and physicians. This is largely determined by tumour location, tumour size and normal organ constraints. In primary non-small cell lung cancer, ablative doses with a biological effective dose of >100 Gy at isocentre have been shown to be a significant factor in local control and survival outcome in primary lung cancer.
6
Case of stereotactic ablative radiotherapy to primary non-small cell lung cancer central lung tumour. (a) Pretreatment positron emission tomography-computed tomography imaging with solitary right upper lobe tumour; (b) stereotactic ablative radiotherapy planned to 54 Gy in three fractions, showing multiple beam arrangement, targeted at lesion and (c) three-month post-treatment computed tomography showed near complete resolution of lesion.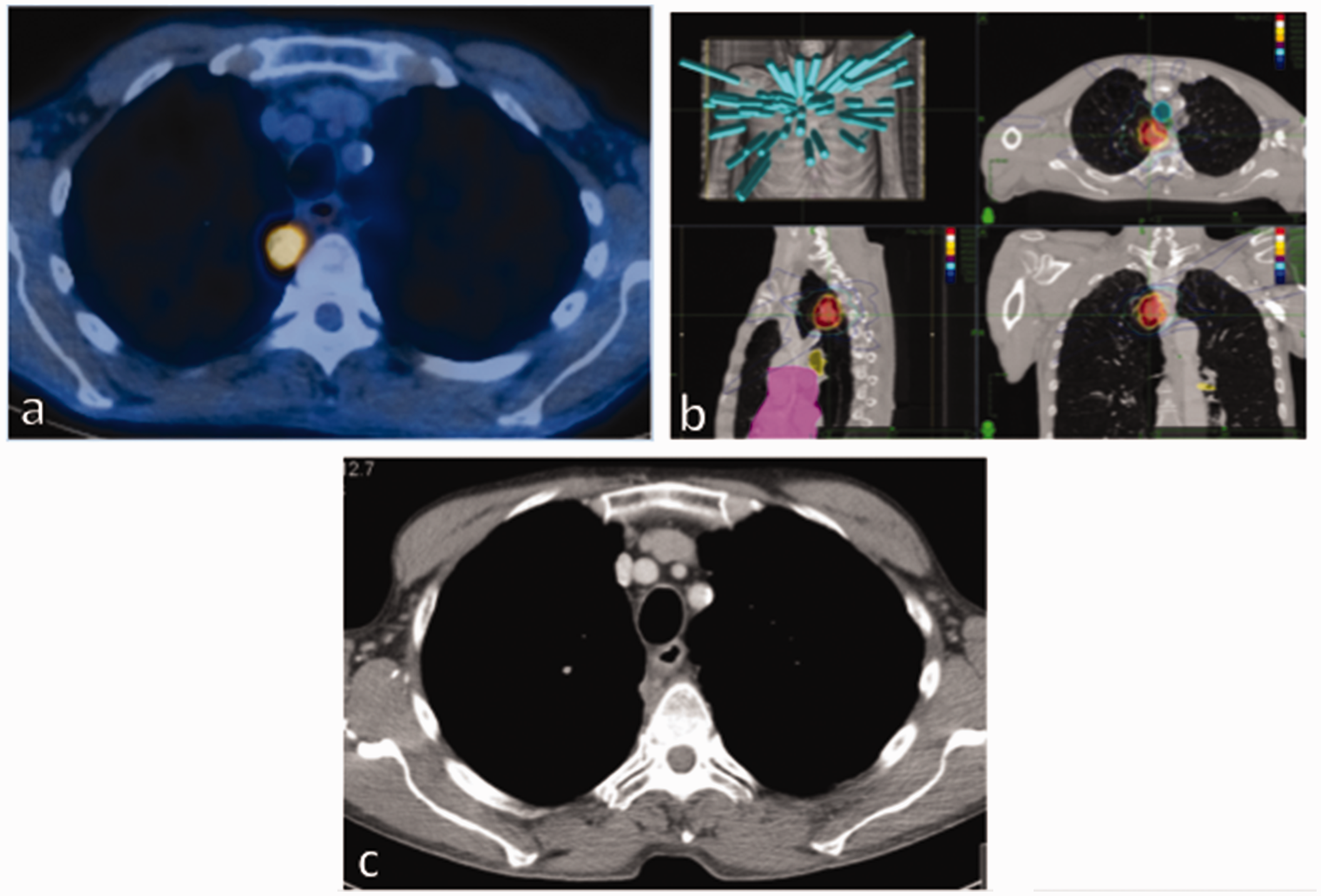
The role of stereotactic ablative radiotherapy in surgical candidates is less certain. Comparative data analysis for both stereotactic ablative radiotherapy and surgery was also conducted using the SEER database. 7 Analyses have shown that stereotactic ablative radiotherapy was associated with a lower risk of early mortality but long-term survival was inferior compared to the surgical group. This might, however, be partly explained by an elderly, less fit cohort in the inoperable stereotactic ablative radiotherapy group.
Recently, there has also been a pooled analysis 8 of two randomised trials called ROSEL (The Dutch Radiosurgery or Surgery for Operable Early stage non-small cell Lung cancer study) and STARS (Stereotactic Radiotherapy versus Surgery). Both trials set out to compare stereotactic ablative radiotherapy to surgery but were terminated early due to poor accrual. The pooled three-year overall survival was superior in the stereotactic ablative radiotherapy arm 95% versus 79% than in the surgical arm (hazard ratio 0.14 [95% confidence interval 0.017–1.190]). In the stereotactic ablative radiotherapy arm, three patients (10%) experienced a grade 3 toxicity and the three-year recurrence-free survival was 86%. The results are in keeping with other published institutional data, but due to the small numbers it is not feasible to claim superiority of one treatment option over another.
There are at least four ongoing prospective randomised trials that will provide additional guidance for surgical patients. In the UK, the SABRTooth trial has closed for accrual and results are awaited. Other internationally run trials are the joint Lung Cancer Trialist’s Coalition STABLE-MATES trial, POSTLIV from China and the Veterans Affairs lung cancer surgery or stereotactic radiotherapy VALOR study. In the meantime, management decisions in operable patients should be discussed as a multidisciplinary team with clear presentation of available evidence during physician–patient consultations. Nevertheless, stereotactic ablative radiotherapy remains a good option in high-risk surgical candidates.
Stereotactic ablative radiotherapy in oligometastatic non-small cell lung cancer
The oligometastatic paradigm in lung cancer has gained increasing acceptance. This select group of patients typically has up to three isolated metastases although some definitions allow as many as five. Traditionally, palliative systemic therapy remains the standard of care for stage IV non-small cell lung cancer patients. However, there is growing evidence both retrospectively and prospectively that offering select patients with few metastases a more aggressive approach to management can result in good outcomes. This is reflected by updates in international guidelines to now recommend more aggressive therapies in a select group of patients with oligometastatic disease.
In non-small cell lung cancer, recently implemented Eighth American Joint Committee on Cancer (AJCC) TNM staging system has redefined its classification for M1 stage disease by splitting solitary and multiple metastases on the basis that survival rates between these groups have been shown to be significantly different. 9 In a pooled analysis 10 of 2531 patients presenting with metastatic disease for treatment on consecutive SWOG protocols, those with a single metastatic lesion had significantly longer survival (8.7 months) compared to those with multiple metastases in a single organ (6.2 months) or those with multiple organs (5.1 months).
The incidence of oligometastatic disease is relatively common with a rate as high as 50% in stage IV non-small cell lung cancer patients. This reported incidence is likely to have been influenced using improved imaging techniques which have higher sensitivity for detecting more subtle sites of metastatic spread, e.g. positron emission tomography imaging and brain magnetic resonance imaging.
Notably, some oligometastatic patients do not progress to widespread disease on follow-up. 11 It would thus be reasonable to hypothesise that locally aggressive treatments could potentially ‘cure’ these patients or provide improved long-term disease control.
Traditionally this has often been done with surgery. However, due to existing co-morbidities or inoperability of the site of disease, this is often not an option. Non-invasive ablative methods such as stereotactic ablative radiotherapy, chemical ablation, radiofrequency ablation, cryoablation and high-intensity focused ultrasound become more attractive options (Figure 2). With the depth of experience, evidence and availability of intracranial stereotactic radiosurgery and primary lung stereotactic ablative radiotherapy treatments, stereotactic ablative radiotherapy in metastatic disease is quickly becoming a more established option and there is now a growing pool of literature supporting its use.
Several retrospective and a few single-arm prospective trials specific to lung cancer patients have reported their own experience in treating these select patients who have a limited number of metastatic lesions with stereotactic ablative radiotherapy. In general, these are single-arm studies that have reported improvements in the patient’s long-term disease-free survival and overall survival comparative to historical results.
Case of stereotactic ablative radiotherapy to adrenal gland oligometastases. (a) Pretreatment positron emission tomography-computed tomography imaging with solitary right adrenal metastases. (b) A fiducial seed (blue arrow) was inserted for radiotherapy treatment targeting. (c) Stereotactic ablative radiotherapy planned. Concentric coloured lines show steep dose fall off. (d) Ten-month post-stereotactic ablative radiotherapy positron emission tomography-computed tomography shows complete resolution of adrenal metastases.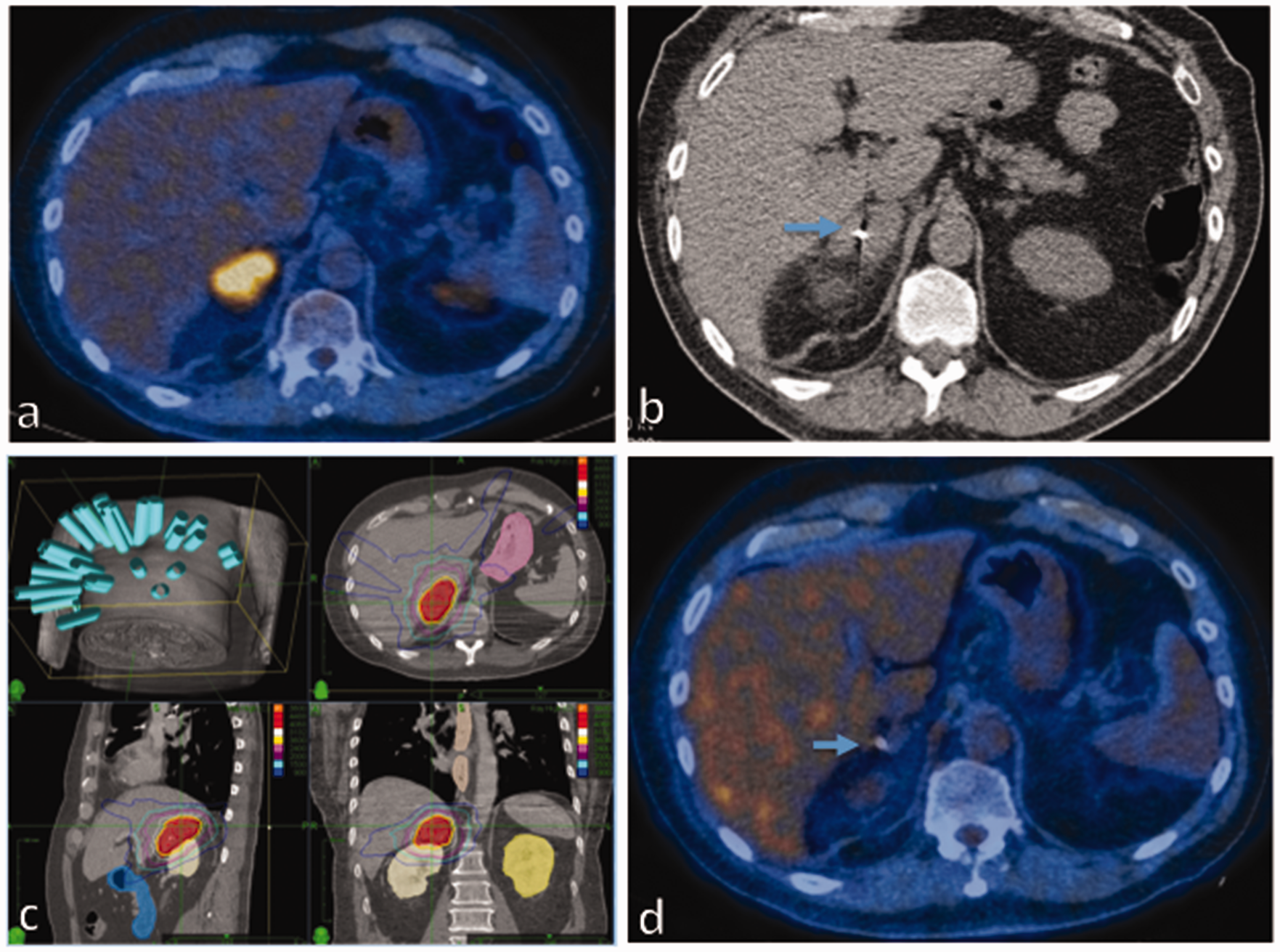
Ashworth et al. 12 reported a systematic review which included 757 patients internationally with treated non-small cell lung cancer oligometastases. All patients had the primary lung cancer treated aggressively as well as 1–5 oligometastases. A total of 37.7% of patients were treated with radiotherapy to their oligometastases. The pooled median overall survival was 26 months with a favourable overall survival of 29.4% at five years. Median progression-free survival was 11 months but at five years 13.1% of patients remained progression-free, suggesting benefit of local ablative technique. On multivariate analysis, nodal disease and synchronous metastases and non-adenocarcinoma histology had worse overall survival.
Two randomised trials have been reported so far. The first by Gomez et al. 13 was a multi-centre phase II trial which randomised non-small cell lung cancer patients with ≤ 3 metastases who did not progress after induction systemic treatment to either local consolidative therapy (surgery or radiotherapy to all metastases, with or without systemic therapy) or to systemic therapy alone. The study was closed early after 49 patients were enrolled as interim analysis found the median progression-free survival in the consolidative therapy arm to be 14.4 months compared to 3.9 months in the standard arm. Median overall survival was not reached. The second and recently published study was by Texas Southwestern Medical Centre. 14 They enrolled 29 patients with similar enrolment criteria, but their study design allowed for ≤6 sites of metastatic disease. The study was stopped early as interim analysis showed a significant progression-free survival advantage in the stereotactic ablative radiotherapy arm (9.7 versus 3.5 months). Stereotactic ablative radiotherapy resulted in no in-field failure versus seven in the maintenance only arm. No additional toxic effect was noted in the stereotactic ablative radiotherapy arm and median overall surival was also not reached.
In oligometastatic lesions, a dose–control relationship is also observed 15 although the optimal dose required has not yet been defined. The trials reported to date are heterogenous and have treated patients with metastatic lesions at many different organ sites with different doses. Thus, it is reasonable to give as high a biological effective dose as possible providing it does not compromise the radiation tolerance of local normal tissues.
Several studies have also looked at the role of ablative treatment to specific sites of metastatic spread in non-small cell lung cancer. The brain is a common site of spread with significant risk of morbidity; good control is thus essential.
Stereotactic radiosurgery to brain oligometastases
The role of aggressive management of non-small cell lung cancer with limited brain metastases was established by Patchell et al. 16 In it, patients who had surgery in addition to whole brain radiotherapy had better local control (52% versus 20%) and overall survival (40 versus 15 weeks).
Much of the evidence for stereotactic radiosurgery comes from its use in conjunction with whole brain radiotherapy. The RTOG 9508 trial
17
compared whole brain radiotherapy alone to whole brain radiotherapy with stereotactic radiosurgery boost to patients with oligometastatic brain lesions. Stereotactic radiosurgery boost allowed improved control. Importantly, there was an overall survival advantage in patients with better prognosis as well as an improvement in functional preservation. Other trials comparing the role of adjunct whole brain radiotherapy to stereotactic radiosurgery alone
18
showed that the addition of whole brain radiotherapy did not result in an overall survival advantage despite better local control. This could potentially spare patients the neurotoxicities of whole brain radiotherapy while improving the quality of life (Figure 3).
Case of stereotactic ablative radiotherapy to solitary non-small cell lung cancer brain metastases. (a) Pretreatment T1-weighted magnetic resonance imaging+contrast showing solitary right temporal brain lesion; (b) stereotactic ablative radiotherapy planned to 24 Gy in three fractions; (c) four-month post-stereotactic ablative radiotherapy magnetic resonance imaging showed good response and (d) unfortunately, 18-month follow-up magnetic resonance imaging showed disease progression of the treated lesion.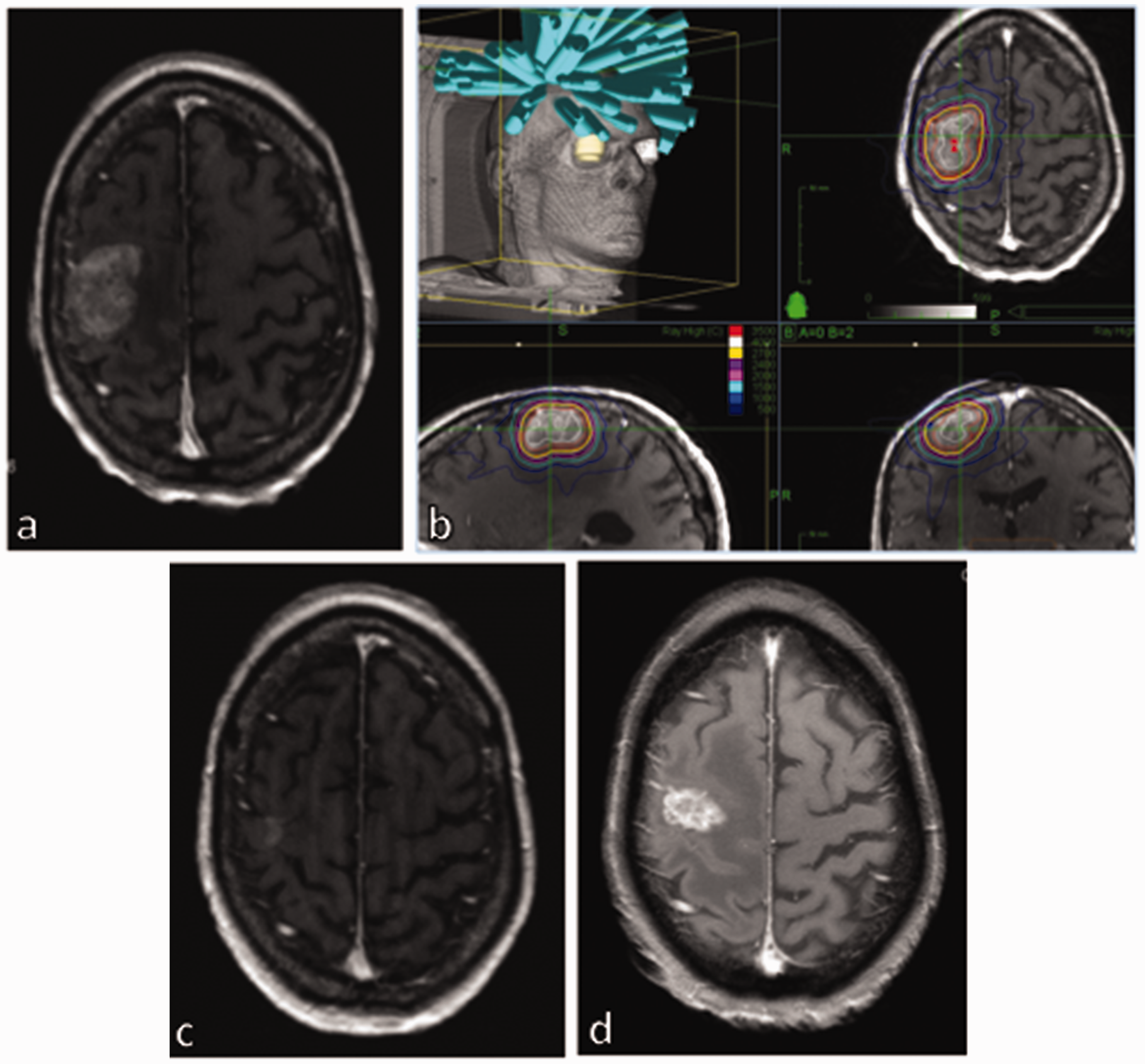
There is also evidence for the use of stereotactic radiosurgery to the surgical cavity postoperatively 19 to improve local control (72% versus 43% at 12 months) with no adverse events reported.
Stereotactic radiosurgery can be given in 1–5 fractions depending on size of tumour, location to critical structures (e.g. brain stem, optic chiasm) as well as prior brain radiation. The criteria for most trials limit stereotactic radiosurgery to four brain lesions each <3 cm; however, some centres treat even greater number of lesions. A common complication of stereotactic radiosurgery is tumour swelling that can result in compressive symptoms and delayed brain radiation necrosis in about 10% of cases, approximately half of which may be asymptomatic.
Although there are no comparative trials for stereotactic radiosurgery versus surgery in brain oligometastases, the evidence suggests that stereotactic radiosurgery is a very suitable option, especially in those patients with small inoperable lesions and treatment options should be individualised.
Conclusion and future directions
The optimal management of early and oligometastatic non-small cell lung cancer is likely to change over time as more clinical trial data become available. For inoperable early stage non-small cell lung cancer, stereotactic ablative radiotherapy is already an important non-invasive method of achieving good local control and survival outcomes with low rates of treatment toxicity. Trials looking at optimising the dose and fractionation schedules of central (RTOG 0813) and peripheral (NCT00843726, Roswell Park Cancer Institute) tumours are underway and will help guide physicians better. Continued improvements in radiation technology and planning systems are likely to increase our ability to deliver higher doses of radiation to tumours with improved accuracy and reduced normal tissue toxicity. Particle beam therapy has a different physical beam profile to that of photons and could allow significant improvement in dose distributions and potential treatment outcomes, although head-to-head trials would need to be conducted to validate this.
For operable, early stage non-small cell lung cancer patients, stereotactic ablative radiotherapy remains an important treatment modality particularly for those patients who are considered high-risk operative candidates. Future randomised trial results will hopefully help guide treatment decisions for these patients better.
In oligometastatic disease, novel biomarkers are currently being investigated to help define patients who are truly oligometastatic from those that are polymetastatic. Recently, microRNA 20 expression profiles have demonstrated promising results and have been found to have prognostic value. These studies though are limited by small sample sizes and therefore further prospective studies are needed to help develop and validate this method to help guide future patient selection.
Due to the lack of randomised prospective data, level 1 evidence to support the use of stereotactic ablative radiotherapy in oligometastatic non-small cell lung cancer patients is lacking. The timing and appropriate integration of stereotactic ablative radiotherapy into the treatment regimen of patients is still uncertain. Several ongoing trials will hopefully help clarify this matter. The NCT01185639, Wake Forest University and ATOM (NCT01941654) trials are looking at the role of consolidation radiotherapy after systemic treatment and epidermal growth factor receptor targeted therapies, respectively. In the UK, the SARON trial (NCT02417662) is a multicentre randomised phase III trial of stereotactic ablative radiotherapy with standard chemotherapy versus standard chemotherapy alone in synchronous oligometastatic non-small cell lung cancer.
The optimal dose fractionation when treating multiple oligometastases in various organ sites is still unknown and most published literature includes stereotactic ablative radiotherapy for solitary lesions. The NRG BR001 (NCT02206334) trial will attempt to clarify the safety and tolerability of stereotactic ablative radiotherapy to multiple metastases.
In the evolving field of metastatic non-small cell lung cancer treatments, targeted molecular agents such as epidermal growth factor receptor and anaplastic lymphoma kinase tyrosine kinase inhibitors are commonly used. Interaction with these agents is uncertain. NCT01796288, which is a multicentre randomised phase 2 trial comparing stereotactic ablative radiotherapy with erlotinib to erlotinib alone, will attempt to provide a measure of the benefit of stereotactic ablative radiotherapy as local ablative therapy of oligometastatic disease in oncogene-driven non-small cell lung cancer.
Of late, immunotherapy has been a hot topic in the management of non-small cell lung cancer. There is growing interest in combining stereotactic ablative radiotherapy, which is thought to be immunogenic, with novel immune checkpoint inhibitors such as anti-CTLA-4 and anti-PD-1 or PD-L1 antibodies to potentiate the antitumour immune response. Preclinical in vivo mouse models and case reports of combined treatments have suggested potential synergistic effects and are now being investigated in prospective trials such as the Dutch PEMBRO-RT study (NCT02492568).
While we await further prospective data, it is important to consider the evidence so far; stereotactic ablative radiotherapy for oligometastatic non-small cell lung cancer is promising but its current use should be weighed against the risks involved, be it a multidisciplinary team-based decision and if possible, conducted within the context of a well-designed prospective randomised trial. Stereotactic ablative radiotherapy for early non-small cell lung cancer, on the other hand, is well established and remains an important curative treatment option, particularly for those patients unsuitable for radical surgery.
