Abstract
The development of Horner’s syndrome during routine neuraxial anaesthesia suggests anatomic, technical or physiologic variance. Even more importantly, it warrants immediate cessation of the anaesthetic intervention.
Case report
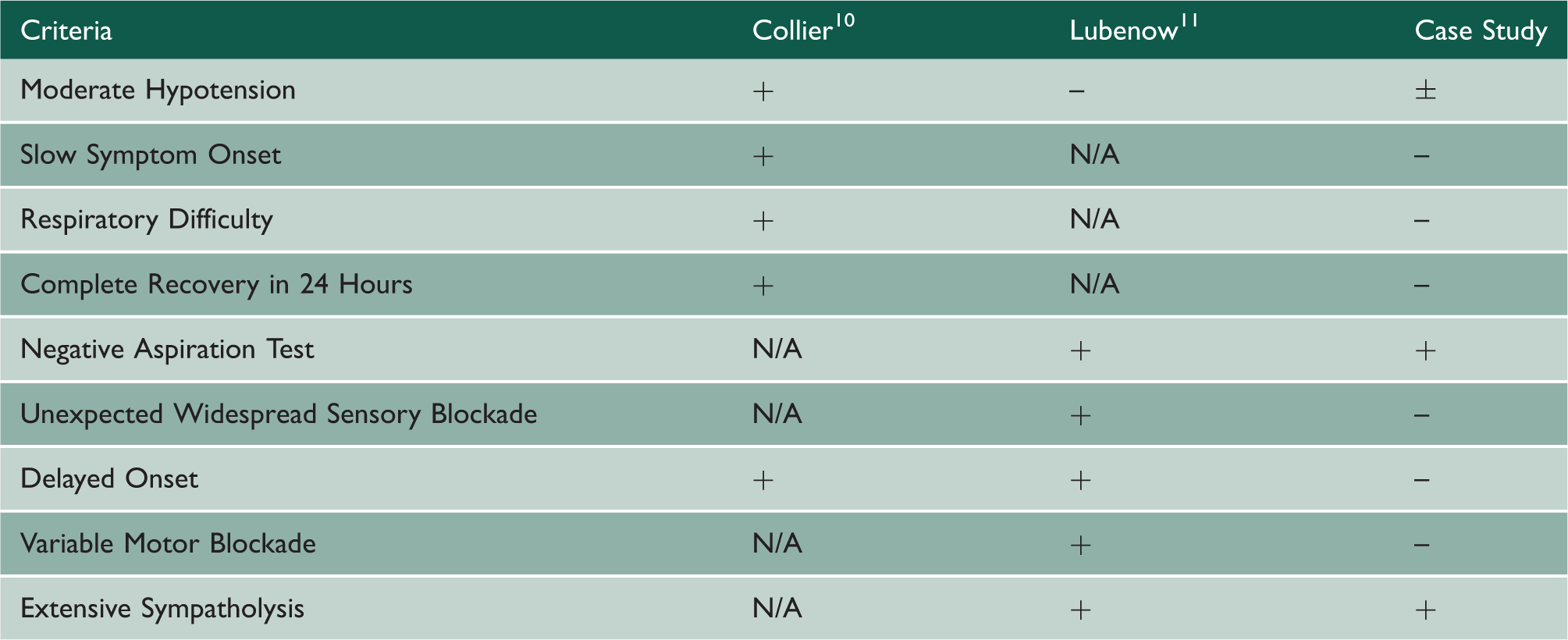
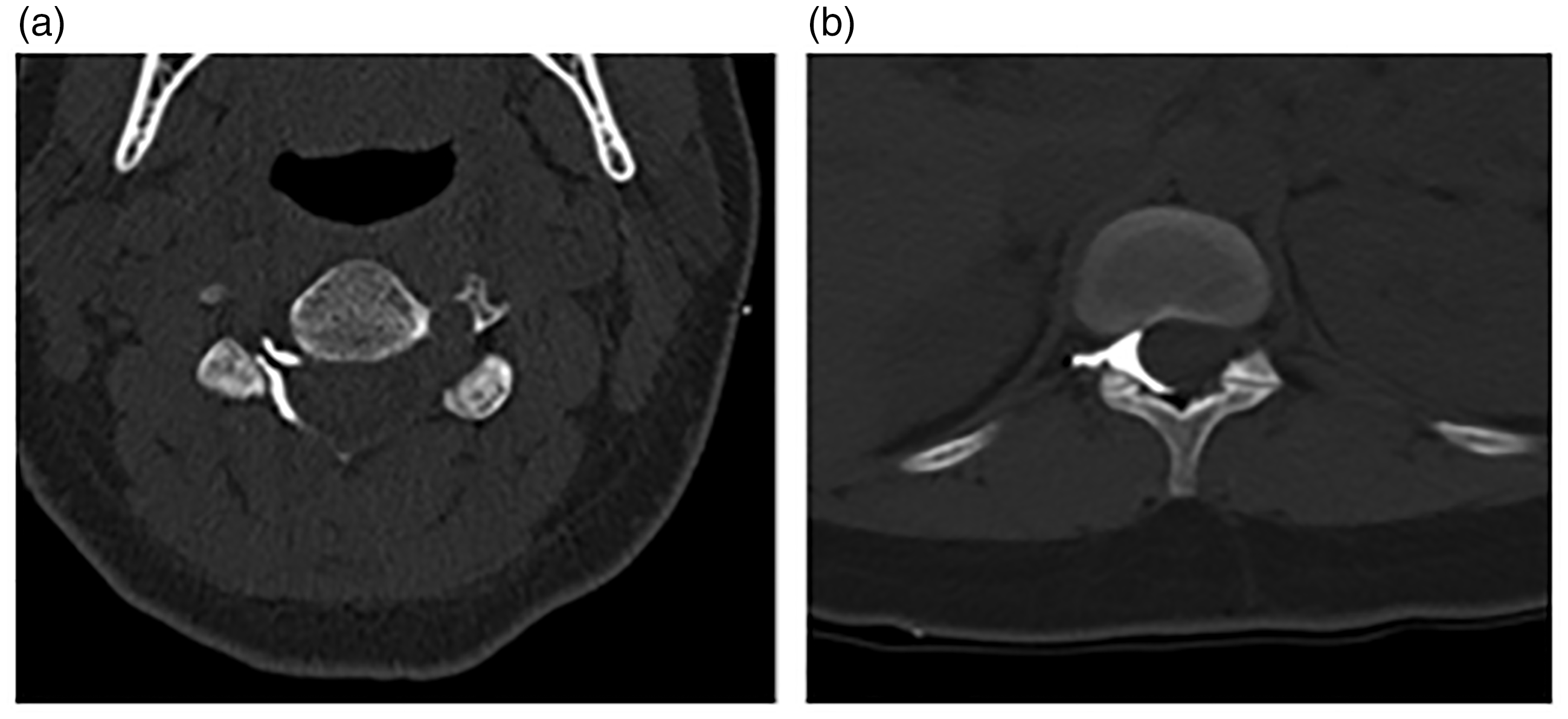
The patient is 28 years old, Gravida 5, Para 2, 75 kg, parturient at 39 weeks’ gestation who presented for induction of labour for suspected fetal macrosomia. Her medical history includes celiac disease and anaemia. Her surgical history includes a low transverse, primary C-section for failure to progress. She previously had one epidural placement that she described as ‘somewhat’ successful, with her first pregnancy. Per the patient, she had undergone general anaesthesia for Caesarean section because of the unilateral distribution of that epidural. No records from the previous delivery at a different hospital were available at that time, nor have they have been received to date. She was admitted to the obstetric ward and an oxytocin infusion was begun. At 5 cm cervical dilation, the patient requested an epidural for pain control. Syringe aspiration during the first epidural attempt, in the midline between the third and fourth lumbar levels, was positive for blood. A second epidural catheter placement attempt was made in the midline between the third and fourth lumbar levels. When the epidural catheter was inserted, there was a temporary paresthesia to the patient’s right hip, which resolved. The epidural catheter was threaded to 4 cm in the epidural space. Syringe aspiration was negative for cerebrospinal fluid or blood, and an epidural test dose injection (3 mL 1.5% lidocaine with 1:200,000 epinephrine) was negative for any haemodynamic or neurodynamic change. The patient received an epidural bolus of 0.25% bupivacaine, in two 4-mL injections and 100 µg fentanyl. An epidural infusion consisting of a 0.0625% bupivacaine and 2 mcg/mL fentanyl solution was begun at 14 mL/h. Shortly thereafter, she complained of shortness of breath, became hypotensive with a blood pressure of 77/39 and tachycardia with a heart rate of 120. She had one watery emesis, developed right eye ptosis and reported right-arm paresthesias. Concurrently, variable decelerations were noted on fetal tracings. A fluid bolus and phenylephrine were administered and the epidural infusion was stopped. Her vital signs stabilised and she remained comfortable despite the cessation of the epidural infusion for the rest of her labour. She delivered a male infant via spontaneous vaginal delivery without additional anaesthetic intervention. A computed tomography epidurogram was obtained the evening following delivery with 13 mL of Omnipaque-300. The study revealed continuous dye spread from the fifth lumbar level to the second cervical level and was seen almost exclusively on the right side of the epidural space. There were no complaints of discomfort or other symptoms during or following the contrast injection. The epidural catheter was removed at the end of the epidurogram with the tip intact. The official computed tomography imaging report stated contrast predominantly on the right side of the spinal canal from L5 to C1 with some distribution to the left side at T10-L2 (Figure 1a and b). The report further stated that contrast surrounded the right nerve roots and foramina (Figure 2a and b). Radiologists at our institution determined that the computed tomography images were consistent with epidural radiopaque dye spread.
(a) Computed tomography coronal view of the vertebrae after contrast and (b) lateral view of the neck after contrast. (a) Computed tomography transverse view of cervical vertebrae after contrast and (b) computed tomography transverse view of lumbar vertebrae after contrast.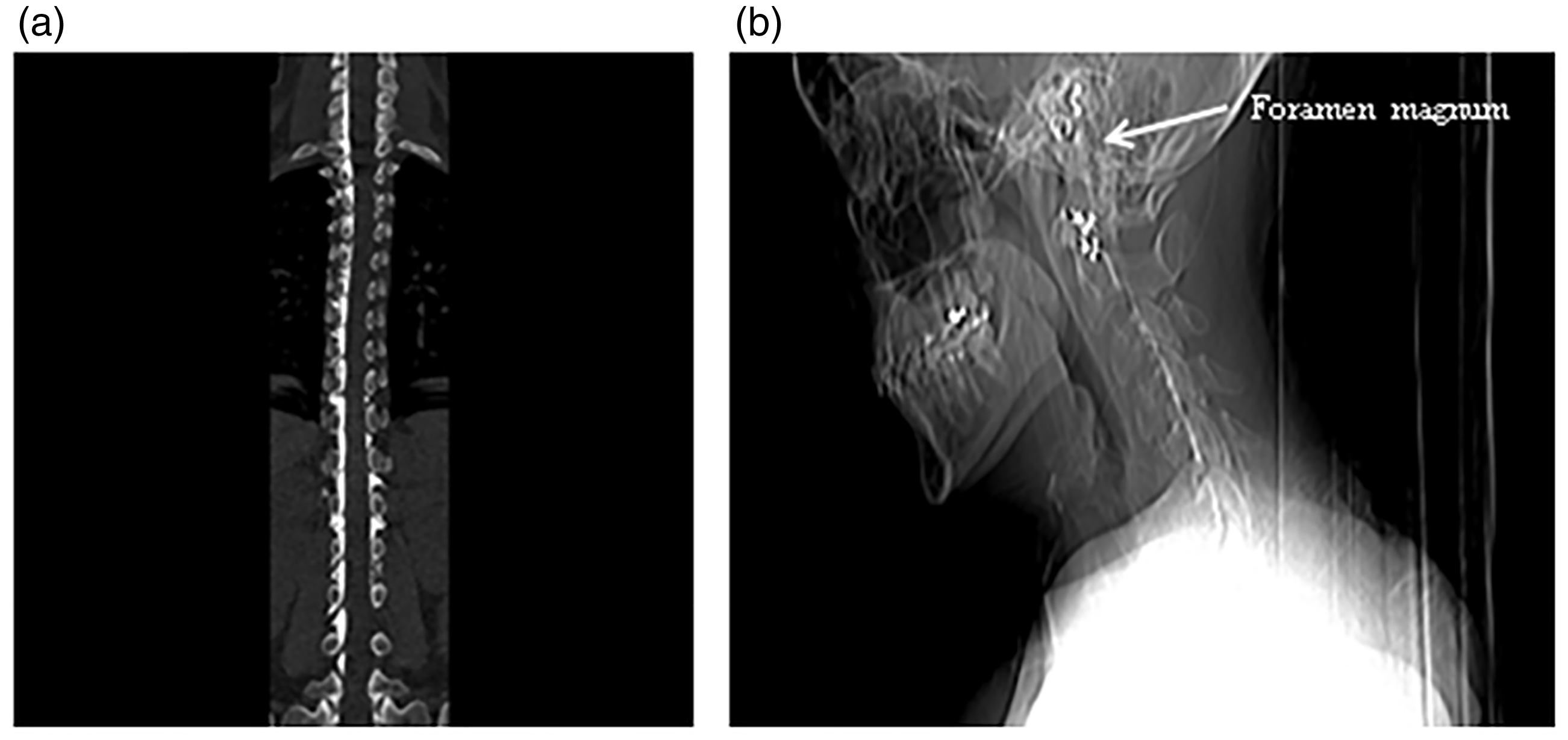
The patient’s symptoms resolved completely by the next morning and she was discharged home that afternoon with her healthy baby.
Discussion
Horner’s syndrome secondary to neuraxial anaesthesia is typically an uncommon and benign process. It has been reported in both obstetric and non-obstetric populations. 1 When unpredicted events occur following induction of neuraxial anaesthesia, the differential diagnosis should be broad and include patient variation in anatomy as well as accidental subdural placement. It has been postulated that pregnant patients tend to develop Horner’s syndrome because of changes in anatomy associated with pregnancy, with the epidural space becoming narrowed and less compliant from the vascular engorgement and increased intra-abdominal pressure from the gravid uterus. Hogan 2 demonstrated that cerebrospinal fluid volume variations among individuals and increased intra-abdominal pressure from obesity and pregnancy-reduced cerebrospinal fluid volume may produce extensive neural blockade. Unilateral cephalad spread of local anaesthetic has been attributed to septation in the epidural cavity 3 and, indeed, the existence of epidural septae has been documented. 4 Hogan et al. 5 showed in their cadaveric study that the lumbar epidural space is made up of discrete segments, determined that an epidural injection is distributed as rivulets through separate channels rather than a uniform one, 6 and concluded that patients have a wide variation in epidural anatomy. Thus, it is not unexpected that the response to a local anaesthetic injection through an epidural catheter varies among patients. These studies suggest that it is possible to achieve a higher sensory level than predicted with a commonly used volume of local anaesthetic, with or without the development of Horner’s syndrome.
The unique characteristics of our case were, first, that this procedure was lumbar in origin. Most descriptions of subdural spread of local anaesthetic into the intracranial space involved cervical epidural injections.8,9 In the clinical setting of a cervical approach, it is logical to assume that the proximity to the foramen magnum would predispose the operators to have a higher degree of caution with respect to the possibility of an intracranial event. Second, the unexpected (in spite of the vague patient history which was compounded by the absence of previous medical records) unilaterality of the injectable space (whether dural or subdural) roughly halved the available volume of the epidural space and led to enhanced cephalad spread. This brought us dangerously close to what may have incited a potentially lethal event had we proceeded with usual, clinically relevant dosing protocols.
The most important aspect of this case, however, is the fact that the epidural block intervention was halted as soon as the Horner’s syndrome was recognised. The recognition of the syndrome is of note because its discovery dictated appropriate treatment. Coverage of obstetric anaesthesia varies at many institutions. It may be attending-based, resident-based with attending coverage from a local site or a remote site, or the coverage may be by a nurse anaesthetist with or without attending anaesthesiologist supervision. In any event, the critical component is the clinical vigilance and the acumen of the individual providing the coverage. No title or level of training can automatically preclude overlooking a clinical change. Given the widely variable level of activity during a shift on a labour and delivery ward, subtle clinical findings such as a Horner’s sign may go unnoticed. This may be further masked by a more noticeable subjective reporting by the patient of pain stemming from uterine contractions. Thus, the emphasis must be upon appropriate teaching of obstetric caregivers in all disciplines to notice important changes in clinical status of these patients however subtle.
In addition to the above, other techniques for establishing and confirming proper placement of the epidural catheter in a normal epidural space are needed. We emphasise “normal” here since anatomic variability within the space exists with unknown frequency. The ability to recognise these variants by direct, real-time means rather than via sole reliance upon indirect techniques such as test dosing with local anaesthetics and pressors is not only desirable but necessary. Absent these developments we iterate vigilance and knowledge-based recognition followed by immediate action (in this case cessation of the treatment).
Conclusion
Instruction in regional anaesthesia, especially when guiding clinicians in training, poses a multitude of challenges for both the instructor and the trainee. Once the technique of the procedure has been operationalised, there remains the need for feedback (either positive of negative) which can help guide the management of the intervention. In certain circumstances, such as the one we identify in this case report, the epidural test dose that is typically used may not provide enough evidence that it is safe to proceed with the usual dosing protocols. The need for more definitive ways to determine ideal placement of indwelling neural blockade catheters of all types and especially those in the neuraxis still exists.