Abstract

This is the fourth in a series of articles on making evidence-based medicine work for you. The series is based on the book ‘Attenti Alle Bufale’ by Tom Jefferson (www.attentiallebufale.it).
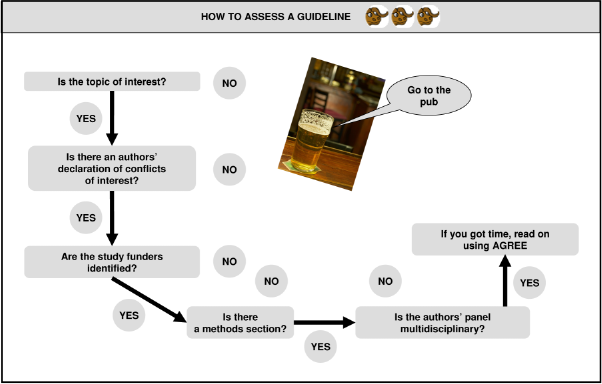
QUICK INSTRUMENT (Figure 1)
Beware of guidelines prepared by single scientific societies or groups. Bin guidelines with no methods chapter or conflict of interest statement, or those not drafted by a multidisciplinary group.
FULL INSTRUMENT
AGREE (Appraisal of Guidelines Research and Evaluation) is a complex but bufala-unfriendly instrument (www.agreecollaboration.org/instrument/). It is made up of six specific domains reflecting key aspects of a guideline, including:
Scope and purpose; Stakeholder involvement; Rigour of development; Clarity and presentation; Applicability to real-world situations; Independence of the editorial group that developed the guideline.
RATIONALE
Guidelines are a flourishing industry. Everyone writes them, but few will withstand the rigours of the quick instrument and even fewer of AGREE. Clinical guidelines are supposed to apply to real world situations, so the idea that they can be written by members of a single discipline is ridiculous. Bear in mind that even the best guideline has a heavy qualitative component and manipulations are still possible.
