Abstract
The aim of this study was to evaluate the toxicity of seven insecticides (g a.i./L): chlorpyrifos (2.25), cartap hydrochloride (1.66), pyriproxyfen (0.33), profenofos/lufenuron (1.33/0.13), fenpropathrin (0.40), deltamethrin/triazophos (0.02/0.70), and zeta-cypermethrin (0.05). These insecticides are commonly used on coffee crop in Brazil and they were sprayed, by using a Potter tower, on pupae and adults of the insect predator Chrysoperla externa. The time of development of the treated pupae were not affected during the testing period and ranged from 9.2 to 9.5 days, although their survival was lower when chlorpyrifos and deltamethrin/triazophos were used, at approximately 62.5 and 75%, respectively. The other compounds did not aid in reducing this specific biological trait, with averages ranging from 90% to 95%. Chlorpyrifos and fenpropathrin were highly toxic to adults of C. externa, causing 100% mortality. Adult insects treated with deltamethrin/triazophos and zeta-cypermethrin showed survival rates of 15% and 22.5%, respectively. The other products did not affect the mortality of directly treated adult insects. Deltamethrin/triazophos and chlorpyrifos were classified as slightly harmful (class 2), whereas the other were considered harmless to C. externa pupae. The insecticides cartap hydrochloride and pyriproxyfen were considered harmless to adults; profenofos/lufenuron was slightly harmful; and chlorpyrifos, fenpropathrin, deltamethrin/triazophos, and zeta-cypermethrin were considered harmful.
Introduction
The state of Minas Gerais is one of the major producers of coffee in Brazil, occupying an area of approximately 1 million hectares, accounting for 52.8% of the national coffee production. It is estimated that Brazilian coffee production in 2012 will reach approximately 50 million 60-kg bags of processed coffee (Conab 2012).
The coffee agroecosystem hosts a great diversity of arthropod pests such as the coffee leaf miner Leucoptera coffeella (Guérin-Meneville & Perrottet, 1842) (Lepidoptera: Lyonetiidae), coffee berry borer Hypothenemus hampei (Ferrari, 1867) (Coleoptera: Scolytidae), cicada Quesada gigas (Olivier, 1790) (Hemiptera: Cicadidae), red coffee mite Oligonychus ilicis (McGregor, 1919) (Acari: Tetranychidae), broad mite Polyphagotarsonemus latus (Bankx, 1904) (Acari: Tarsonemidae), false spider mite Brevipalpus phoenicis (Geijskes, 1939) (Acari: Tenuipalpidae), among other species (Silva et al. 2010).
Amid sporadic outbreaks, other important pests causing damage to coffee plants, both in the roots and the shoots, are the mealybugs (Hemiptera: Pseudococcidae), including Dysmicoccus texensis (Tinsley, 1900), Planococcus citri (Risso, 1813), Planococcus minor (Maskell), and Pseudococcus longispinus (Targioni Tozzetti, 1867) (Souza et al. 2008; SantaCecília et al. 2009).
Those pests are usually controlled through the application of highly toxic, broad-spectrum pesticides containing organophosphates, pyrethroids, and neonicotinoids (Okumura et al. 2003; Souza et al. 2007; Silva et al. 2010). These compounds can be used up to three sprays consecutively per target pest (Agrofit 2009). However, for greater sustainability, it is necessary to associate chemical and biological methods of control (Ripper et al. 1951; Rigitano and Carvalho 2001; Degrande et al. 2002; Foerster 2002).
The family Chrysopidae consists of a large number of species that stand out among predators because of its prevalence in coffee crops, with Chrysoperla externa (Hagen, 1861) (Neuroptera: Chrysopidae) being the most common (Carvalho and Souza 2000). Its larvae exhibit predatory behavior throughout the developmental phase, which consists of three instars (Fonseca et al. 2000). The green lacewings are generalists and can feed on eggs and small caterpillars of moths, aphids, mealybugs, whiteflies, and psyllids thriving in various crop species (Fonseca et al. 2000; GonçalvesGervásio and Santa-Cecília 2001; Silva et al. 2002; Barbosa et al. 2003; Auad et al. 2007).
On the basis of the significance of this natural enemy to coffee crops, the aim of this study was to evaluate the impact of insecticides on pupae and adults of the predator C. externa under laboratory conditions to generate information that could be employed in the improvement of integrated pest management programs in coffee crops.
Material and methods
The experiments were performed in the laboratory at 25 ± 2 °C, RH 70 ± 10%, and photoperiod of 12 h. The products used in the study are shown in Table 1. The control treatment consisted only of distilled water.
Trade name, active ingredient, chemical group, doses of insecticides and concentrations of the active ingredients of the compounds registered for the control of Leucoptera coffeella in coffee crop, tested on pupae and adults of Chrysoperla externa, under laboratory conditions (Agrofit 2009).
Maximum dose of the commercial product (c. p.) (L or kg/ha.).
Concentration of the active ingredient (g or mL/L of water).
Effects of insecticides on C. externa pupae
After approximately 24 h of pupal formation, in which a small black disk develops during the last instar exuvia, forty pupae were collected from laboratory cultures and distributed to 15 cm diameter Petri dishes. The insecticides were sprayed directly on the pupae using a Potter tower, with an application of 1.5 ± 0.5 mg/cmβ and pressure of 15 lb/polβ, and after spraying the pupae were individualized in glass tubes. The experimental study was a completely randomized design with 8 treatments and 10 replicates, with each plot consisting of four pupae. The length of the pupal stage and survival of treated pupae were then evaluated.
Effects of insecticides on adults of C. externa
One to two days old adults were obtained from rearing maintenance, separated by sex, anesthetized using CO₂ for 30 s, and then placed in Petri dishes lined with filter paper. The insects were then sprayed with insecticides similarly to the methodology used on the pupae assay. After spraying, two insects were then transferred to a cylindrical PVC cage (diameter, 10 cm; height, 10 cm) covered with filter paper, which served as the substrate for oviposition, and placed on a Petri dish lined with the same type of paper and sealed at the top with voile fabric. The adult insects were fed with brewer yeast and honey, and distilled water, supplied through a moistened cotton wool placed over the voile fabric that overlaid the cages, was provided every two days intervals. Assessments of mortality of adult insects were initiated approximately 24 h after spraying.
Effects of insecticides on the reproductive parameters of predators treated during pupal and adult phases
Six couples per treatment, obtained from treated pupae and adults, were kept in cylindrical PVC cages (diameter, 10 cm; height, 10 cm) and coated with white filter paper, which served as the substrate for oviposition. The top of each cage was sealed with voile fabric and the bottom laid on a Petri dish lined with filter paper. The insects were fed with brewer yeast and honey, using a ratio of 1 g of commercial product to 1 mL of honey, and adding distilled water to the mixture. This diet was coated on Parafilm® strips that were hung inside the cages. Distilled water was supplied by means of a moistened cotton wool placed on the voile fabric that shrouded the cages.
The number of treatments varied because the formation of couples depended on the toxicity of the applied insecticides. Egg viability was assessed every 3 days intervals up to 28 days after the pre-oviposition. At each evaluation, 96 eggs were collected at random, which were individually placed in ELISA microwell plates. One egg was placed in each chamber; the plates were sealed with laminated PVC film, and were kept in an acclimatized room similar to the previous experiments until larvae hatching.
Data analysis
Pupal survival, length of pupal phase, survival of treated adults, number of eggs laid, and egg viability were analyzed using ANOVA and the means were compared using the Scott and Knott test, at 5% significance (Scott and Knott 1974).
Classification of insecticides based on standards established by the International Organization for Biological Control (IOBC)
The total mortality of insects (from the stage tested to adult emergence) was corrected by using Abbott's formula (1925). The tested insecticides were classified according to its toxicity (Table 2) based on the reduction of biological characteristics of the insects using Vogt's formula (1992) as follows:
Toxicity classes of pesticides to Chrysoperla externa, according to the total effect (E), in laboratory tests.
Toxicity classes (Hassan and Degrande 1996).
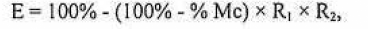
where:
E = total effect (%); Mc % = mortality corrected for control treatment (Abbott, 1925); R₁ = ratio of the average daily eggs laid per treated and untreated females; and R₂ = ratio of the average fertile eggs laid by treated and untreated females.
Results and discussion
Effects of insecticides on pupae of C. externa
Low survival rates were observed among pupae treated with chlorpyrifos and deltamethrin/triazophos, which were calculated as 62.5% and 75%, respectively (Table 3). These results are assigned to the fact that the products may have penetrated into the small holes present in the silk cocoon constructed by the insect. The cocoons of the pupae of C. externa features holes on its surface, with a diameter of about 6 µm that pass through the cocoon allowing the insect to breathe during the pupal stage (Cosme et al. 2009). Another important fact is that the chemical group of organophosphates has greater residual effect, which may have favored the death of adult in time of emergency, when contacted cocoons contaminated (Table 4). Silva et al. (2006) have also found that chlorpyrifos treatment (1.2 g a.i./L water) caused a 77.5% reduction in the pupal survival.
Length (days) and survival (%) (± SE) of pupae of Chrysoperla externa treated with seven insecticides. Temperature of 25 ± 2 °C, RH 70 ± 10% and photoperiod of 12 hours.
Means followed by the same letter in the column are not statistically different according to the Scott-Knott test (P < 0.05).
There was not statistically difference between treatments for duration of Chrysoperla externa (P > 0.05).
Number of couples, mortality (%), corrected mortality (%), average daily oviposition, egg viability (%), total effect (E%) and toxicity classes based on criteria established by the IOBC on pupae of Chrysoperla externa treated with the insecticides. Temperature of 25 ± 2 °C, RH 70 ± 10% and photoperiod of 12 hours.
Accumulated mortality (%) obtained during the development of the predator.
Corrected mortality (%) based in the control (Abbott 1925).
Number of eggs/day/female during the first 28 days after the pre-oviposition.
Viability (%) of eggs collected in the period of four consecutive weeks.
Total effect (%) of the products on the predator. E = 100% - (100% - M%) x R₁ x R₂.
Toxicity classes proposed by Hassan and Degrande (1996), as follows: 1 = harmless (E < 30%), 2 = slightly harmful (30 ≤ E ≤ 79%), 3 = moderately harmful (80 ≤ E ≤ 99%) and 4 = harmful (E > 99%).
No differences in pupal survival were observed for the other insecticides, which allowed survival rates ranging from 90% to 95%, suggesting that the morphology of the cocoon may have served as a barrier to the penetration of these compounds, thus protecting the developing insects (Table 3). These results are similar to those of Grafton-Cardwell and Hoy (1985) that showed green lacewings eggs and pupae to be more tolerant to pesticides. Godoy et al. (2004) also confirmed these results by showing that the insecticides (g a.i./L water) thiacloprid (0.036), deltamethrin (0.0125), lufenuron (0.0375), tebufenozide (0.12), fenbutatin oxide (0.4), and abamectin (0.0054) caused no negative effects on pupal survival. Although the compounds (g a.i./L water) endosulfan (1.050), esfenvalerate (0.075), fenpropathrin (0.090), trichlorfon (0.090), and triflumuron (0.0375) tested by Ulhôa et al. (2002) reduced the survival of C. externa pupae, their toxicities were classified as class 1, indicating that these reagents were safe, with average survival rates ranging from 70% to 86.7%.
The pesticides were also shown to impart no effects on the length of the pupal stage, which varied from 9.2 to 9.5 days (Table 3). The observed length of this stage was similar to that of previous reports on green lacewings fed with different types of chemically uncontaminated prey (Figueira et al. 2000; Costa et al. 2002; Maia et al. 2004; Macedo et al. 2010).
Chlorpyrifos and deltamethrin/triazophos were classified as class 2, where they are considered slightly harmful to pupae (30 ≤ E ≤ 79% mortality). The other insecticides were considered harmless; therefore, they were selective to pupae of C. externa. Hence, treatment during the pupal phase resulted in negligible negative effects (Table 4). Rocha (2008) examined the effects of insecticides (g a.i./L water), namely, thiamethoxam (0.50), imidacloprid (0.70), endosulfan (2.63), and dimethoate (0.48) and rated these as harmless to pupae of C. externa.
Effects of the insecticides on directly treated C. externa adults
Chlorpyrifos and fenpropathrin were shown to be highly toxic to adult C. externa, causing 100% mortality. The insects treated with deltamethrin/triazophos and zetacypermethrin were also affected, resulting in average mortality rates of 85% and 77.5%, respectively, indicating that the adults are more sensitive to certain compounds and insecticides, such as those classified as organophosphates and pyrethroids (Table 5).
Number of couples, mortality (%), corrected mortality (%), average daily oviposition, egg viability (%), total effect (E%) and toxicity classes based on criteria established by the IOBC on females of Chrysoperla externa treated with pesticides. Temperature of 25 ± 2 °C, RH 70 ± 10% and photoperiod of 12 hours.
Accumulated mortality (%) obtained during the development of the predator.
Corrected mortality (%) based in the control (Abbott 1925).
Number of eggs/day/female during the first 28 days after the pre-oviposition.
Viability (%) of eggs collected in the period of four consecutive weeks.
Total effect (%) of the products on the predator. E = 100% - (100% - M%) x R₁ x R₂.
Toxicity classes proposed by Hassan and Dagrande (1996), as follows: 1 = harmless (E < 30%), 2 = slightly harmful (30 ≤ E ≤ 79%), 3 = moderately harmful (80 ≤ E ≤ 99%) and 4 = harmful (E > 99%).
The sensitivity of adult insects was also examined by Godoy et al. (2010), who reported a high toxicity for thiamethoxan (0.05 g a.i./L water), resulting in low survival rates in C. externa and Ceraeochrysa cubana (Hagen, 1861) (Neuroptera: Chrysopidae), at 22.7% and 0%, respectively, approximately 4 days after application. Moura et al. (2010) showed that the application of the compounds (g a.i./L water) carbaryl (1.73), fenitrothion (0.75), and methidathion (0.4) resulted in 100% mortality of adult C. externa populations from two different localities (Bento Gonçalves and Vacaria - RS), thus classifying these pesticides as harmful (class 4). Bueno and Freitas (2001) classified imidacloprid as harmful to adult insects at doses of 14.0, 17.5, and 21.0 g p.c./100 L of water, whereas Castilhos et al. (2011) reported that the insecticides (g or mL of p.c./100 L water), malathion (300), phosmet (200), dimethoate (250), and fenitrothion (150) were harmful to adult insects. Rezaei et al. (2007) evaluated the impact of insecticides (L or kg of p.c./ha) imidacloprid (0.25), propargite (1.5), and pymetrozine (1.0), reporting mortality rates of 36.8%, 35.8%, and 37.1%, respectively, in Chrysoperla carnea (Stephens, 1836) (Neuroptera: Chrysopidae) adults. Studies conducted by Preetha et al. (2009) using the compounds (g or mL p.c./L) imidacloprid (0.56), methyl demeton (1.0), diafenthiuron (3.2), and monocrotophos (2.0) reported a mean mortality of 36.7%, 63.3%, 53.3%, and 60%, respectively, approximately 48 hours after application.
Pyriproxyfen was observed as a safe insecticide, at the concentration used and this may be attributable to the increased activity of growth regulators during the last larval or nymph instars (Faria 2009), or the ability of insect may have to rapidly excrete this compound from its body. Medina et al. (2002) showed that the topical application of 37.5 x 10-6 g a.i. pyriproxyfen per female of C. carnea resulted in 88% of the insects showing signs of penetration through the cuticle approximately one day after treatment; however, 90% of the insects showed a capacity for excretion approximately two days after treatment. Velloso et al. (1999) observed no changes on the survival of C. externa females treated with pyriproxyfen (0.100 g a.i./L of water), confirming the results obtained in the present study.
On the basis of the high mortality rates associated with the application of chlorpyrifos, fenpropathrin, deltamethrin/triazophos, and zeta-cypermethrin, these compounds were considered harmful (class 4) to C. externa adults. Although profenofos/lufenuron did not reduce the survival of treated adults, this insecticide affected the reproductive capacity of the insect, reducing the viability of eggs laid by treated females, thus allowing us to classify this product as slightly harmful (class 2). Cartap hydrochloride and pyriproxyfen were classified as class 1, indicating that these are harmless to adult insects and can be recommended for integration into pest management programs in coffee crops, as well as for the preservation of this green lacewing species (Table 5).
Other studies have demonstrated the toxicity of organophosphates and pyrethroids to adult lacewings, as work by Vilela et al. (2009) who classified as harmful insecticide fenpropathrin (0.3 g a.i./L) for adults of the same species of predator, as Carvalho et al. (2011) classified the insecticides triazophos (0.6 g a.i./L water) as harmful (class 4) to adult of C. cubana.
Conclusions
The examined pesticides showed no significant negative effects on C. externa pupae, as compared to the adult insects. Cartap hydrochloride and pyriproxyfen were classified as harmless. Profenofos/lufenuron was slightly harmful, whereas chlorpyrifos, fenpropathrin, deltamethrin/triazophos, and zeta-cypermethrin were categorized as harmful to C. externa adults. Further studies using field conditions should be done to confirm the toxicity levels of these compounds on this predator.
Footnotes
Acknowledgements
The authors express their gratitude to the Study funded by the Consórcio Brasileiro de Pesquisa e Desenvolvimento do Café - CBP&D/Café, Coordination for the Improvement of Higher Education Personnel-CAPES, and Foundation for Research Support of Minas Gerais, for their financial support for the implementation of the project and for providing the scholarships.
