Abstract
Family Health Conversations (FamHC) increase health and well-being, but knowledge about their cost-effectiveness, and how to best calculate this, is lacking. In this feasibility study we evaluated the cost-effectiveness of using FamHC with families in which a middle-aged family member had suffered stroke. Seven families participated in a FamHC intervention and seven families received ordinary care. Health-related quality of life (HRQoL) was estimated with SF-6D and EQ-5D over a six-month period. The cost-effectiveness of the intervention was calculated. Families receiving FamHC intervention had significantly increased HRQoL at follow up. Cost per quality adjusted life year differed depending on the instrument and analysis method used in the calculation. However, all calculations showed that FamHC were cost-effective. We conclude that FamHC significantly increase HRQoL and suggest that they are cost-effective. Both instruments seemed to be able to capture changes. Considering the participants’ experience of answering the two instruments, we advocate the use of EQ-5D.
Introduction
Family Health Conversations (FamHC) are a family system nursing intervention that has been suggested as a way to increase health and well-being in the whole family when a family member suffers long-lasting illness. Knowledge about the cost-effectiveness (CE) of FamHC and even how to best calculate this is lacking. Knowledge about the CE of various interventions makes a significant contribution to decisions about resource allocation and there is an urgent need to find ways to calculate CE in relation to family system nursing interventions.
Background
Family system nursing occurs in a collaborative, non-hierarchical relationship between a family and a nurse in which the nurse implements a non-judgmental approach and takes into consideration the legitimacy of multiple realities in a strengths-based orientation, moving health-promoting conversations toward family competences and strengths. 1
In a review study, 2 family system nursing interventions have been described as improving families’ communication and increasing their confidence and capability in relation to life with illness. Families were shown to become more caring and supportive toward each other and to coordinate activities in a better way. Feelings of closeness and affirmation within family relationships were enhanced, suffering eased, and well-being increased for both individual family members and the family as a whole.
The family system intervention, FamHC, has in Sweden been developed 3 by adapting and modifying the Calgary Family Assessment and Intervention Models 4 and the Illness Beliefs Model 5 to Swedish conditions.6–11 The FamHC takes a systemic approach simultaneously focusing on the individual and the family unit, and always on the interaction, reciprocity and circularity between the person with ill-health and other family members.3,10 The aim of FamHC is to create a context for support and/or change; to make beliefs conscious; to challenge, facilitate or create new beliefs, ideas and meanings about complex family problems that might be experienced when a family member has become ill. Several assumptions underpin the FamHC. For example, each family member’s view is to be acknowledged as equally valid, the importance of narratives and reflective thinking is to be emphasized, and beliefs are the heart of healing in families and for illness.3,5,10
Evaluations concerning the effects of FamHC are scarce and, to our knowledge, health economic evaluation is lacking. It is important to evaluate whether new interventions are worthwhile compared to other interventions with respect to costs in relation to effects. This can be done in different ways: one is to estimate the effects in terms of gains in quality adjusted life years (QALYs) and to compare the costs arising from the intervention. Quality adjusted life years are a proxy for health-related quality of life (HRQoL) over a certain period of time. 12 They are calculated from quality weightings which can be obtained from the use of standardized instruments that assess health-related quality of life, such as, for example, the EQ-5D 13 and the SF-6D. 14 The QALY weights are simply multiplied by the time period they are valid for (e.g. the QALY-weight 0.7, times one year is equal to 0.7 QALY).
FamHC are assumed to be suitable for use in families where one family member suffers from long-lasting illness, and in this study we evaluate FamHC in families in which one family member had suffered stroke. The whole family is significantly affected when a family member had suffered stroke. The family is important in the person’s recovery process but needs support. Family Health Conversations are suggested to be an intervention that improves family health; however, their cost-effectiveness has, to our knowledge, not yet been calculated. Further, we found no suggestions for instruments assessing HRQoL for QALY-calculations in families taking into consideration that family members are linked in a system, and most likely have different health statuses.
Aim
The aim of this feasibility study was to evaluate the cost-effectiveness of using FamHC with families in which a middle-aged family member had suffered stroke.
Research questions
In families in which one middle-aged family member has suffered from a stroke:
^ is HRQoL better in the families that participate in FamHC compared to that of families that do not? ^ are FamHC cost-effective? ^ does EQ-5D and/or SF-6D capture changes in HRQoL when seeing a family as a unit? ^ is EQ-5D and/or SF-6D suitable for assessing HRQoL?
Method
A feasibility study was done. It had a non-equivalent comparison group design with pre- and post-tests. The study took place in two counties in Sweden.
Procedures and participants
Eligible participants were families that included one member less than 65 years of age who had suffered a stroke (infarction or haemorrhage) and been admitted to a rehabilitation clinic. Exclusion criteria were family members who did not speak and read Swedish and stroke diagnosed as transient ischemic attack (TIA). For this study we decided to consecutively include 14 families (two groups of seven), a number we considered large enough to provide enough data to find indications for future studies of the feasibility of performing the intervention and its effects.
15
For practical reasons of geographical distances, the participants for the intervention group were to be recruited from one county and those for the comparison group from another county. However, it turned out that few families fulfilled the inclusion criteria in the first county in reasonable time so two families for the intervention group were recruited from the other county. For the intervention group, a total of 12 people who had suffered a stroke were consecutively
16
approached during their stay in a rehabilitation centre and invited to participate in the study. Six males and one female consented to participate. For the comparison group, 12 people diagnosed with stroke were consecutively asked to participate and four males and three females consented to participate (Figure 1). These participants were then asked to identify who they defined as belonging to their family, as described elsewhere.
4
In total, seven families consisting of 17 family members were included in the intervention group and seven families consisting of 21 family members were included in the comparison group. All participants signed an informed consent. After the families had given their consent to participate in the study, nurses approached them to plan appointments. Before and during the study, written and verbal information concerning the study aim, voluntary participation, and confidentiality was given to the participants.
Flow diagram of inclusion of participants and drop-outs.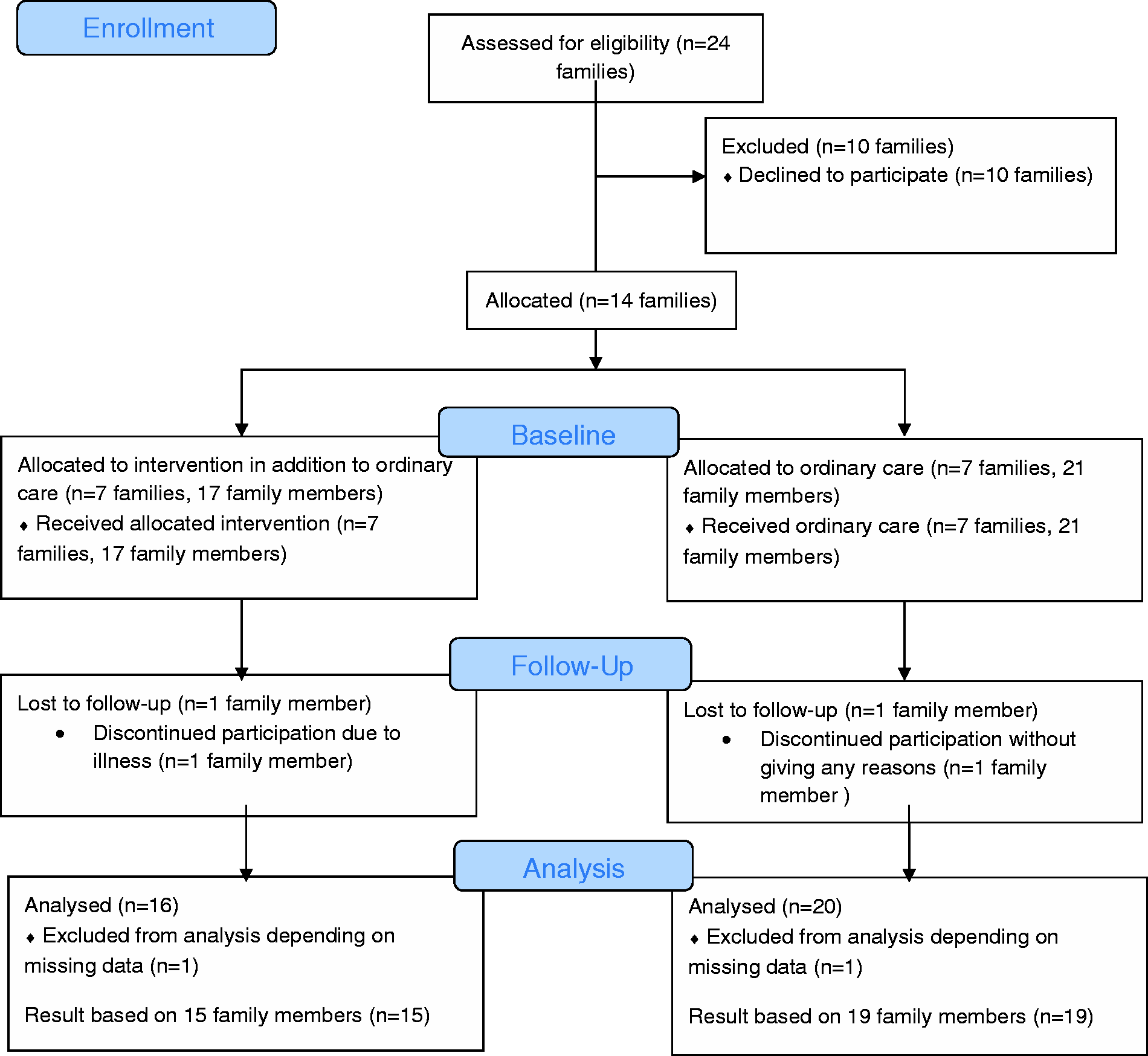
Intervention
The persons with stroke in both groups received rehabilitation at rehabilitation clinics. In addition, the intervention group participated in FamHC that took place in the families’ homes. Time between stroke diagnosis and the intervention start was 3–5 months for three, 7–8 months for two and 10–12 months for two of the families. The intervention consisted of a series of three conversations, each lasting about one hour, and held at an interval of two to three weeks. The conversations were led by two nurses following a structure identified from the theoretical assumptions underlying FamHC. 10 Core components of the intervention include: reflecting with the family on expectations of the conversations; exploring the family structure; jointly prioritizing which problem(s) to discuss; ensuring all family members have the opportunity to narrate their experience; exploring significant parts of the narratives shared by the family; inviting family members to reflect on each other’s narratives; using reflective questions and other questions to challenge family beliefs; giving commendations and acknowledging suffering; offering the family the chance to listen to nurses’ reflections; and, finally, during the last conversation, closing the conversations series by 1) summarizing verbally what had been experienced during the series and 2) offering written reflections in a ‘closing letter’ 17 sent to the family two weeks after the last conversation. During the conversations, one nurse took the major responsibility for the conversation while the other was a co-participant considering meta-communications and responsible for offering reflections on the conversation’s content. 3 Further details about the content and purpose of FamHC core components are given elsewhere. 10
Data collection
Data were collected between 2010 and 2012. In the intervention group, data were collected at baseline and six months after the FamHC were completed. For the comparison group, data were collected at baseline and at a time equivalent to the six-month follow up for the intervention group.
Measures
To measure HRQoL the following instruments were used: the EQ-5D classification system and SF-6D. The EQ-5D classification system consists of the EQ-5D index and the EQ VAS. The EQ-5D index has a self-rated health state description in five dimensions: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. 13 Each health description has a preference-based value derived from a British general population 18 with values between –0.59 (worst state) and 1.0 (best state). The health state in the present study was also evaluated with the EQ VAS, which is a 20-cm-long visual analogue scale ranging from 0 (worst imaginable health) to 100 (best imaginable health). The EQ-5D classification system has been found to have acceptable validity when assessing HRQoL after stroke. 19
The SF-6D is a six-dimensional health state classification 14 derived from SF-36. 20 The dimensions are: physical functioning, role limitations, social functioning, pain, mental health, and vitality. The SF-6D asks several questions for each dimension and the answers are combined to generate a score for that dimension. Preference-based values with 1 as the best health and 0 as death are reported. 14 Our study used the acute recall version (1-week recall). SF-6D is based on a selection of questions from SF-36. SF-36 has been found to be a valid and reliable assessment tool when used among stroke patients. 21
Both the EQ-5D classification system and the SF-36 are considered to be appropriate for use in a general population. 22 However, the SF-36 has been found to be more sensitive among individuals without health problems or with only minor health problems when compared to the EQ-5D classification system. 22 Further, patients with stroke have been found to provide complete data to a greater extent when using EQ-5D classification system than when using SF-36, probably due to the simplicity of EQ-5D. 23
The research group has license and approval to use both EQ-5D and SF-36. All family members responded to the questions by rating their own health and well-being.
Data analysis
One participant in each group (family members without stroke) withdrew from the study (n = 2); further, one participant in each group did not answer all questions (n = 2) (see Figure 1). Only those forms that were fully completed were analysed.
Treatment effect
SPSS version 19 was used to analyse the data. Paired sample t-test was used to calculate differences in HRQoL within groups between baseline and six-month follow up. Because the non-equivalent groups differed at baseline the analyses were done with a difference variable, i.e. the observed change between baseline and six-month follow up in each group. For between-group comparisons, independent sample t-test was used. A p-value of < 0.05 was considered statistically significant. All answers were weighted equal in the analysis regardless of whether they were answers from the person with stroke or from any family member.
In addition to calculating statistically-significant differences in HRQoL gains, we also calculated effect size to estimate clinical significance. As there is no preferable single approach when calculating effect size and the use of multiple strategies is recommended, 24 we used three approaches, whereas two have been used previously in relation to stroke 25 (standardized effect size and standardized response mean). Standardized effect size (ES) was calculated on data from the within-group analysis by dividing the difference between baseline mean value and mean value at follow up with the standard deviation from baseline score. Standardized response mean (SRM) was calculated on data from the within-group analysis by dividing the observed change by the standard deviation of the observed change. 26 Effect size was also calculated from data in the between-group analysis by dividing the mean difference by pooled standard deviation. 27 Regardless of the calculation method, the effect size is usually interpreted as follows: 0.2 is a small effect size, 0.5 is medium and 0.8 is a large effect size, using Cohen’s schema. 26
Cost-effectiveness
Quality adjusted life years were calculated as follows: change in QALY weights from baseline to six-month follow up was multiplied by 0.5, which was the length of the study in years. Since the QALY weights were assumed to have increased linearly over the six months, the sum from the multiplication was divided by 2 (change in QALY weights *0.5)/2 = QALY gained). Because QALY weights differed between groups at baseline, QALYs were calculated both from changes within the intervention group and from mean difference in the between-group analysis.
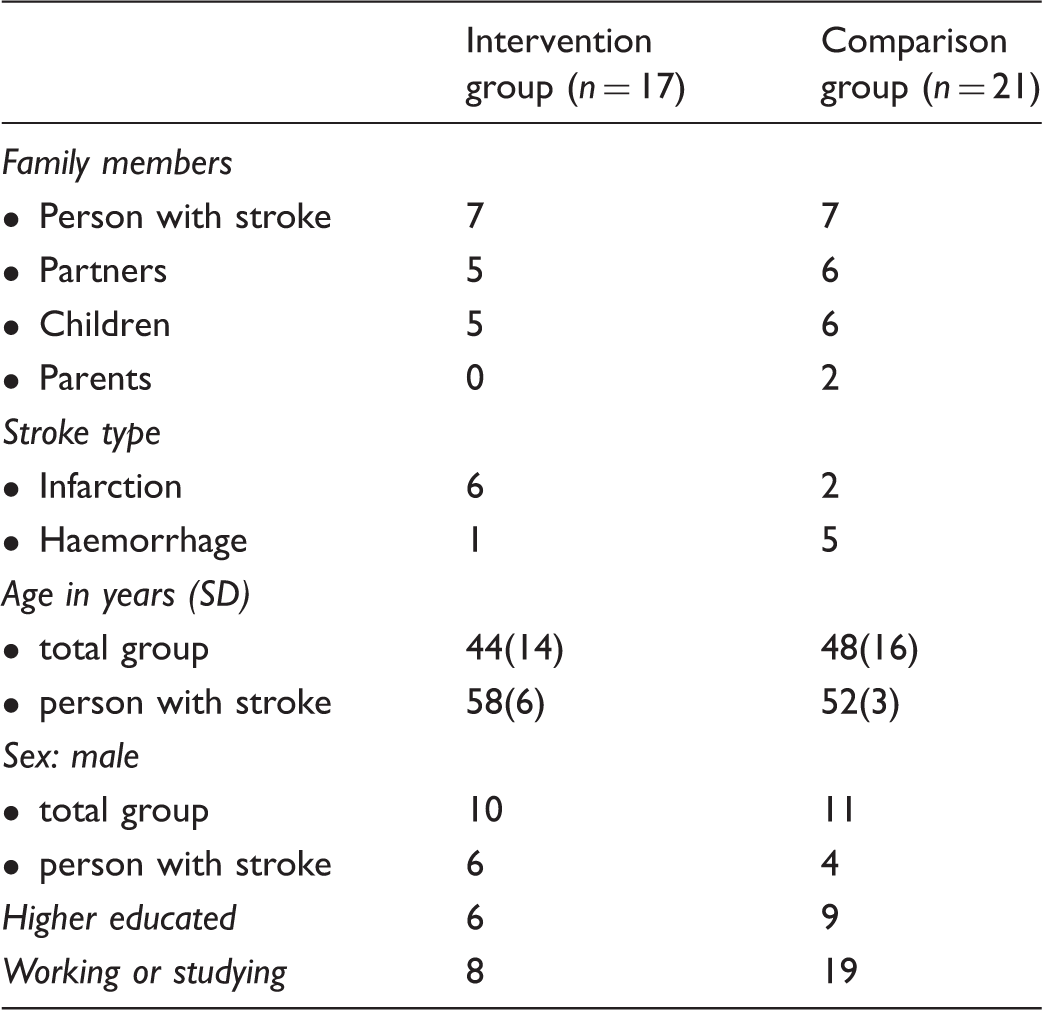
Demographics of participating families.
Results
The family members were between 16 and 72 years of age and the family members who had suffered stroke were between 48 and 64 years of age. The participants’ demographics can be seen in Table 1.
Treatment effects
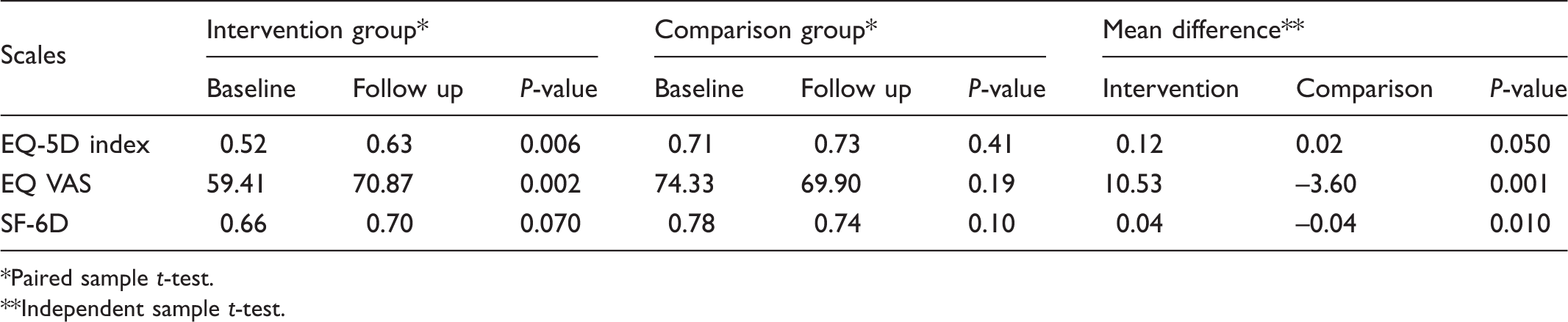
Results from within- and between-group comparisons.
Paired sample t-test.
Independent sample t-test.
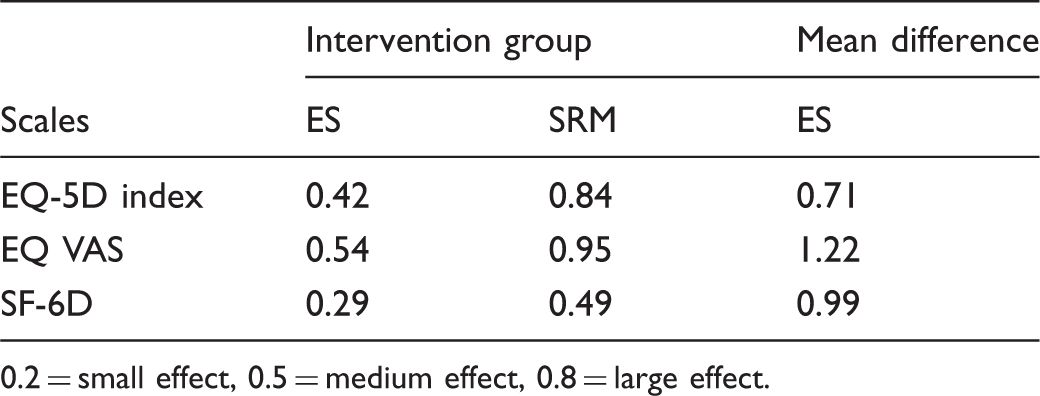
Effect size calculated in standardized effect size (ES) and standardized response mean (SRM) in the intervention group and in effect size on mean difference in the between-group analysis.
0.2 = small effect, 0.5 = medium effect, 0.8 = large effect.
Cost-effectiveness
Quality adjusted life years (QALYs) calculated on changes within intervention group and mean difference between groups.
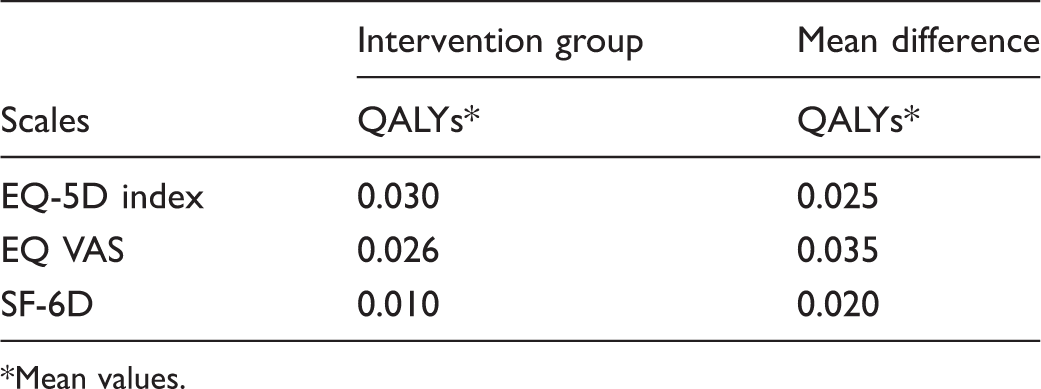
Mean values.
Costs for FamHC in Euro (€).

Cost-effectiveness of FamHC based on within-group and between-group analyses.
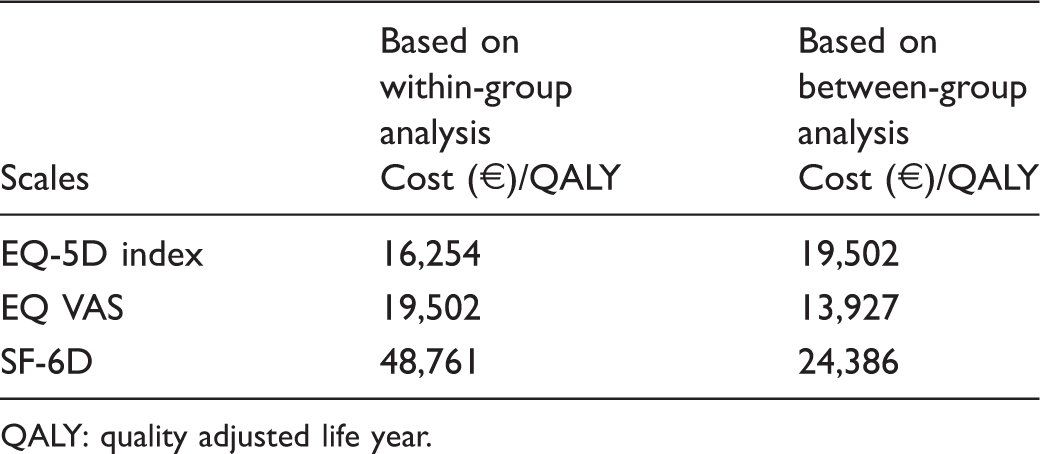
QALY: quality adjusted life year.
Discussion
The results show that in comparison with a group of families receiving standard care, families that additionally had taken part in FamHC had significantly increased HRQoL six months after the intervention. The effect size was shown to be medium to large in seven of nine estimations, which indicates that FamHC provide clinically important changes and are clinically relevant.
The cost-effectiveness of FamHC was between about €14,000/QALY and € 49,000/QALY, depending on the instrument used to estimate HRQoL. The finding is in line with previous studies that have shown that results from health economic evaluations vary according to the choice of instrument. 28 The threshold for cost-effectiveness usually used is €55,000/QALY as described elsewhere.29,30 This means that interventions that cost less than €55,000/QALY are considered to be cost-effective. Considering this threshold, FamHC are a cost-effective intervention.
Evaluation of FamHC and family system nursing interventions have previously been predominantly qualitative. 31 A review of family system nursing interventions revealed that only five empirical papers used quantitative measures to evaluate the interventions. 2 In another review 32 of nursing studies on patients and family members (not specifically with a systemic approach), 22 empirical studies had used quantitative methods. None of the studies included in either review reported having conducted health economic evaluations, which makes the present study unique.
Consequently, there were no previous studies to guide us in how to best evaluate cost-effectiveness in a family system nursing intervention. We decided to use two instruments to estimate HRQoL, which have both been used among persons with stroke and also among people in general. In our study, the EQ-5D index and the EQ VAS showed significant increases in HRQoL in the intervention group while no change was shown with the SF-6D from baseline to follow up. The comparison group showed no changes over time. Previously, the QALY gain in persons with stroke has been found to be larger when using the EQ-5D then when using the SF-6D, 25 which was partly consistent with our findings. In line with results from other studies,21,33 we experienced that the SF-36 was comprehensive and difficult for some participants to complete. Both instruments seemed to be able to capture changes even in this small group of participants. Considering the difficulties some participants experienced, we advocate the use of the EQ-5D to estimate HRQoL when evaluating FamHC in families where one family member has suffered stroke.
Clinical significance was calculated using both ES and SRM, which showed some different results. In the analysis of clinical significance in the intervention group, the great difference in the results of ES and SRM was likely due to the difference in standard deviations. It is supposed that SRM is less dependent on sample size 26 and it is therefore reasonable to assume that this calculation is preferable. Even if we take the lowest value of effect size, our results show clinically significant effects from taking part in FamHC.
In the estimation of cost and calculation of QALYs, we have maintained a cautious approach in order not to overestimate cost-effectiveness. Estimation of alternative costs for participants’ time in taking part in conversations was based on relatively generous wages irrespective of whether the participants were adults in employment, adults on sick leave or children. Further, staff costs were based on wages for a researcher, which are higher than wages for nursing staff. In addition, travel required was longer than travel would be if the intervention were part of ordinary health care. Taken together, these factors means lower costs are likely to occur when the intervention is implemented in clinical practice. Also, in the calculation of QALYs gained, the increased QALYs were assumed to not persist for more than six months (last follow up), which probably induces an underestimation. It is reasonable to assume that the effects, if not persisting, would to some extent persist for some time after last follow up. Altogether, it is reasonable to assume that FamHC are more cost-effective in clinical practice than shown in this study.
Limitations
This study should be interpreted as a primary feasibility study, and therefore cautiously. 15 In this initial test of the intervention we considered that the number of participants was large enough to provide an indication of measures for health economic evaluations that could guide future studies. Moreover, even if the FamHC are targeting the family as a system, HRQoL was measured at an individual level. Estimating effects of interventions at a family level is challenging; one approach can be to use individual measures. Individual family members are certainly on various health trajectories but their experiences do not exist in isolation from family; rather, mutual interactions influence their perceptions. 34
Furthermore, the intervention and comparison groups in this study were non-equivalent at the start. The comparison group rated higher in HRQoL than the intervention group at baseline which can probably be explained by there being more healthy family members in the comparison group than in the intervention group. Because of this non-equivalence, the QALY was calculated from mean difference in HRQoL between the intervention and comparison groups and from the observed change in HRQoL in the intervention group. There was a significant difference according to subtype of stroke (infarction or haemorrhage) at baseline where more people in the control group had haemorrhage. As patients with haemorrhage have been found to achieve greater functional outcome in the rehabilitation phase compared to patients with infarction,35,36 and consequently could have been expected to gain higher HRQoL, it is unlikely that this could be an explanation of the difference in change in favour of the intervention group at follow up.
Conclusions
Our results indicate that FamHC for families in which a member under 65 years of age suffered unhealthy conditions such as stroke result in clinically relevant increases in HRQoL and can be suggested to be cost-effective. We have shown that individual measures of HRQoL in families can capture improvements even if the families consist of both ‘healthy’ and ‘unhealthy’ members. We suggest the use of the EQ-5D when estimating HRQoL in studies that evaluate the effects of FamHC. This study provides guidance for future studies on the effects of participating in family system nursing interventions.
Footnotes
Ethical approval
The study has been approved by the Regional Ethical Review board in Umeå, Sweden (Dnr 2011-68-32M).
Acknowledgements
The authors would like to express their gratitude to the participating families and to the staff at the rehabilitation departments for assisting with the recruitment of the families. We would like to acknowledge Dr Lars Lindholm, Professor in Health Economics at Umeå University and Dr Marie Lindkvist, Umeå University for their valuable support during the analysis.
Funding
This research was supported by grants from the Strategic Research Program in Health Care—Bridging Research and Practice for Better Health (SFP-V), the Swedish STROKE Association, and the Faculty of Medicine, Umeå University.
Conflict of interest
The authors declare that there is no conflict of interest.
