Abstract
Objective
To present a case of probable autoimmune encephalitis initially misdiagnosed as a primary psychiatric disorder.
Method
A case of probable autoimmune encephalitis presenting with catatonia in a young woman 6 months post-partum is described in this article.
Results
The patient was admitted to an outside hospital, diagnosed with a primary psychiatric condition, and sent to psychiatric inpatient, where she was denied admission due to labile hypertension. After admission to the medical service (and ICU), she responded to a lorazepam challenge, and made a complete recovery within several weeks after combination treatment with IV methylprednisolone, IV immunoglobulin (IVIG), and rituximab.
Conclusion
Autoimmune encephalitis should be suspected in patients presenting with labile vital signs, family history of autoimmunity, and new psychosis without prior history of psychiatric problems. Other important diagnostic considerations include neuroleptic malignant syndrome, substance-induced psychosis, or catatonia secondary to post-infectious immune-mediated encephalitis. While a first psychotic break should always be considered, the presence of autonomic instability, catatonia, seizures, or dyskinesias in a young woman postpartum should prompt a thorough medical and neurological work-up.
Introduction
Autoimmune encephalitis (AE) is an uncommon manifestation of underlying autoimmunity that can present with acute psychiatric symptoms, thus representing an important cause of reversible non-infectious encephalitis. Some patients have made a complete recovery with steroids and immunomodulatory drugs, implicating autoimmune etiology even without confirmatory serology.1–3 Prodromal syndromes, orofacial dyskinesia, catatonia, and extrapyramidal signs suggest AE. 1 Early recognition may reduce mortality and morbidity. 4 Especially in young patients with no psychiatric history, autoimmune encephalitis should be considered in the differential diagnosis for acute psychotic or catatonic presentations.
Case Presentation
A 30-year-old female 6 months post-partum presented to our hospital for subacute altered mental status. Two weeks prior, she had developed a sudden increase in anxiety, coupled with echolalia, fearful grimaces, and possible visual hallucinations (Figure 2, Disease Timeline). An outside hospital (OSH) had diagnosed primary psychosis and recommended inpatient psychiatric care, but she was rejected per new-onset high blood pressure with systolic blood pressure in 220s millimeters of mercury; therefore, the patient was sent to our Emergency Department (ED) for further medical stabilization.
At outside ED, she was disoriented, nonverbal, and not following commands, with systolic blood pressure to 170s, tachycardia, and elevated C-reactive protein (CRP) at 2.4 mg/dL. Initial drug screen was negative. She received one droperidol dose (1.25 mg), two lorazepam doses (1 mg and 2 mg), and 2 liters lactated ringer’s fluids. She was transferred to inpatient psychiatry, rejected for labile hypertension, transferred back to initial hospital, and admitted for medical work-up. This work-up, while negative for infection, showed electrolyte derangement, ketonuria, thrombocytosis, and mildly elevated creatine kinase, d-dimer, ammonia, and cortisol. She received electrolytes, fluids, parenteral nutrition, two IV haloperidol doses (1 mg) over three days, two bedtime quetiapine doses (25-mg), and one ziprasidone dose (10-mg), without improvement in agitation or hallucinations.
She received 4 2-mg and 3 1-mg lorazepam doses, which significantly improved alertness and visual tracking. Tachycardia and hypertension persisted despite fluids, amlodipine, and metoprolol. She intermittently fevered to 39.4 C. OSH favored post-partum depression with psychosis but wanted to rule out autoimmunity. Patient was transferred to our institution for medical work-up.
Six months prior to presentation at our institution, she delivered a baby girl through uncomplicated spontaneous vaginal delivery and had just stopped breastfeeding the day before acute symptom onset. The infant was diagnosed with sickle cell anemia. The patient herself has sickle cell trait, and a history of anemia, with reportedly previously elevated CRP and erythrocyte sedimentation rate (ESR). Family history was also significant for lupus in three first degree relatives, one in whom lupus was fatal. Per family, the patient had begun developing slowly worsening anxiety and depressive behaviors since childbirth (Figure 2).
Several weeks prior to presentation, her husband reported she was becoming increasingly anxious about returning to work (Figure 2)
Labs
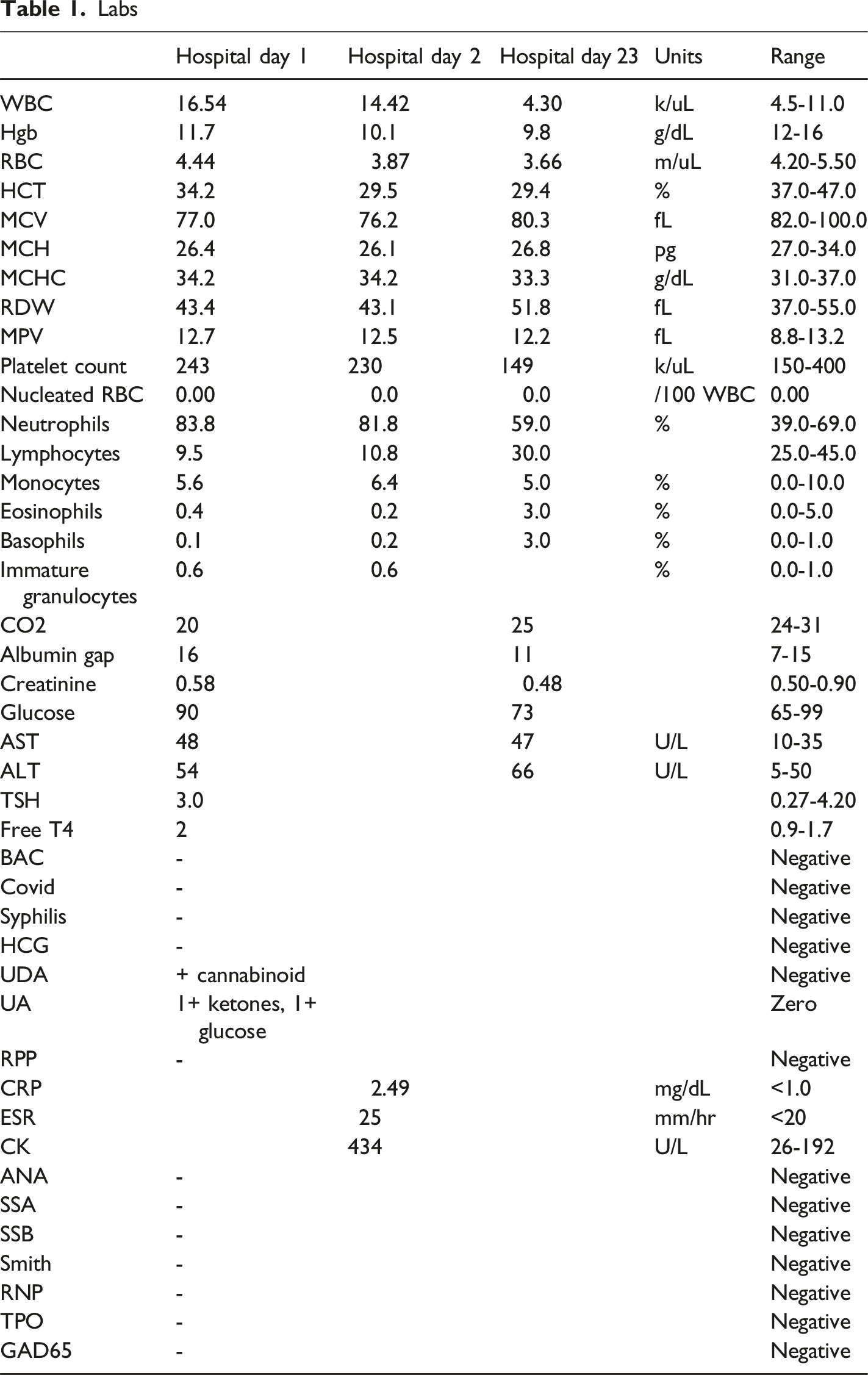
Additional lab work revealed elevated sedimentation rate (25 mm/hour) and C-reactive protein (2.49), with mildly elevated creatine kinase (434 U/L), but negative anti-nuclear autoantibodies (ANA), anti-Sjogren’s syndrome type A and B (SSA, SSB), anti-Smith, anti-ribonucleic acids (RNP), anti-thyroid peroxidase (TPO, and anti-glutamic acid decarboxylase 65-kilodalton isoform (GAD65) antibodies (Table 1). Infectious disease screening for syphilis and human immunodeficiency virus (HIV) was negative. Leukocytosis with neutrophilia suggested Systemic Inflammatory Response Syndrome (SIRS), so blood cultures were ordered, and ampicillin was added, along with dextrose 5% with thiamine. Drug screen was positive for cannabinoids, although at OSH she had had two negative drug screens. Husband reported smoking cannabis but said patient does not. The medical record revealed no history of drug use in the patient.
Psychiatry consultation-liaison service was consulted for management of psychiatric symptoms and catatonia. She was nonverbal, fearful at approach, and tried to move away from the interviewer. She was unable to follow commands or state her name, and was staring and blinking, with bilateral extremity and trunk rigidity, facial grimaces, arm tremor, and dynamic vital sign changes. During the interview, her heart rate rose to 160s (beats per minute), with blood pressure spiking to 180s millimeters of mercury systolic, despite no history of tachycardia or hypertension per family. Her Bush Francis Catatonia Rating Score (BFCRS) was 35. Administration of lorazepam showed improvement in catatonia. After initial lorazepam challenge showed improvement, the dose was scheduled to 2 mg twice daily.
CSF Results
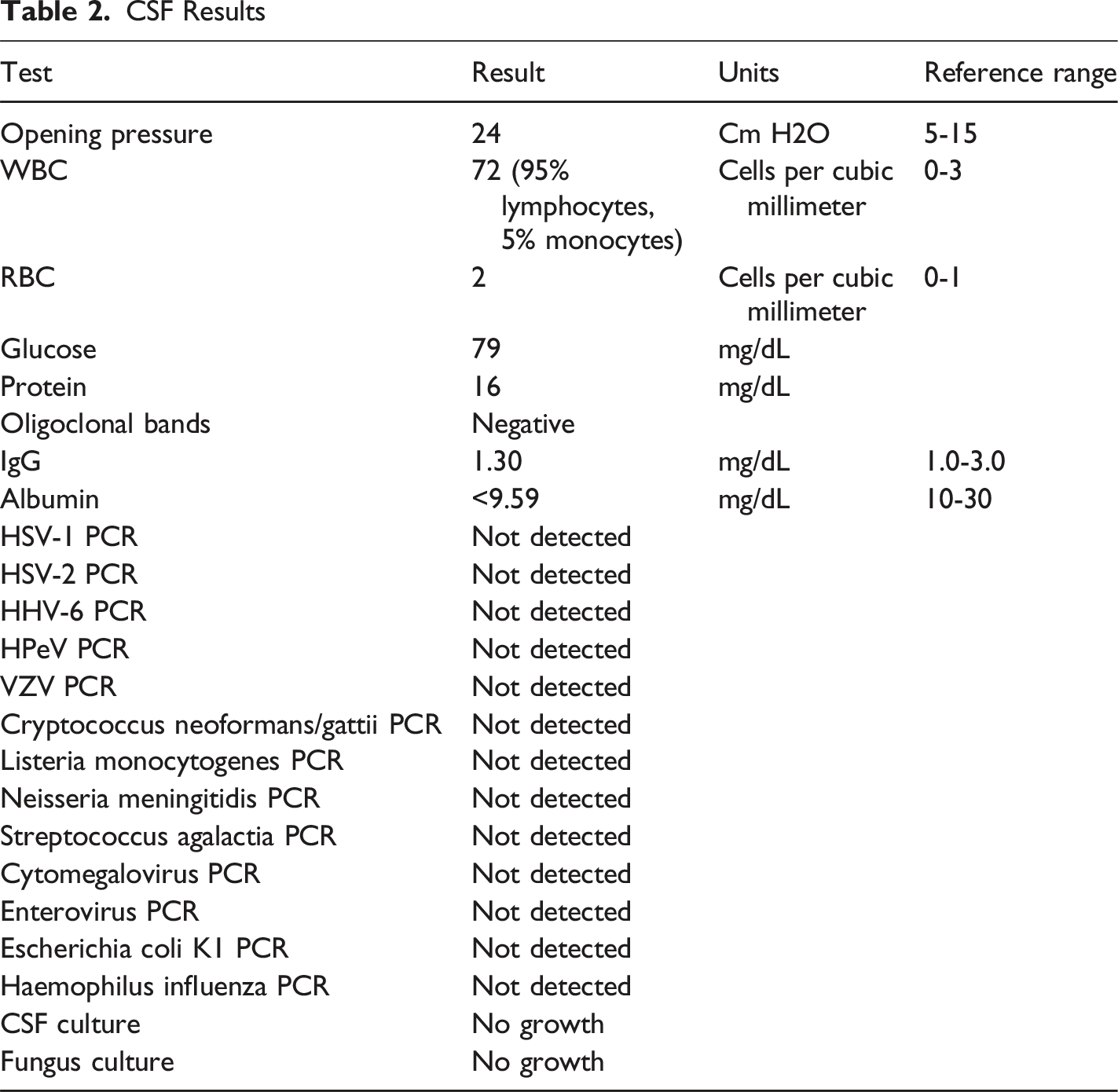
By hospital day 4, lorazepam dose was increased to 2 mg every 4 hours. The patient was transferred to the intensive medical unit per hospital’s procedures for closer monitoring on high doses of benzodiazepines. Over the next 2 days, her symptoms improved, with stereotypy, excitability, and fixed gaze disappearing. She became more alert. Additional medication options were discussed with patient’s husband, who shared that olanzapine given in outside hospital had helped control manic symptoms. Per current catatonic state, aforementioned dopaminergics like olanzapine were held, but IV divalproex sodium was initiated after discussion of risks, benefits, and alternatives as treatment for possible underlying manic condition in addition to adjunctive treatment for catatonia. She was also started on IV methylprednisolone, with a peripherally inserted central catheter (PICC) line placed for total parenteral nutrition on hospital day 6.
By hospital day 8, she had become temporarily verbal, but reverted to muteness, was stiff on exam, and continued to have autonomic instability. Lorazepam was increased to 2 mg every 2 hours. On hospital day 9, her BFCRS was down to 15 (from 35 at presentation). Autonomic instability persisted, so lorazepam dose was increased to 30 mg in 24 hours.
Electroconvulsive therapy (ECT) and rituximab were discussed as potential treatment options. However, the state of Texas rules that only the patient or a court-appointed legal guardian can consent to ECT. Patient remained altered and was unable to consent to ECT. The hospital’s legal team was contacted and advised next of kin did not count as legal guardian; unless an emergent legal guardian was court-appointed, family could not consent to ECT.
IV immunoglobulin (IVIG) was administered hospital days 8-11, with amantadine added day 9, 200 mg daily. She was maintained on 60 mg of oral prednisone daily.
After amantadine addition and 24 hours of high-dose lorazepam, her Bush Francis score dropped to 10; autonomic instability and stiffness began resolving. Lorazepam was decreased to 2 mg every 2 hours, with planned taper next day for 2 mg every 3 hours, then 2 mg every 4 hours on the subsequent day. Unfortunately, lowering lorazepam failed, with returning autonomic instability and worsening catatonia (BFCRS up to 26). ECT was still not an option per state of Texas laws, as she could not consent and would require guardianship proceedings or out of state transfer to fulfill ECT consenting in the state of Texas. Off-label amantadine use, in addition to FDA-approved dosage, was discussed with family; doses were increased to 200 mg twice daily.
By hospital day 17, she had improved, with less catatonia (BFCRS down to 15 from 26), able to shake her head yes or no, but still disorganized and confused. By hospital day 21, she could ambulate from bed to chair. She was alert, mouthing words, and able to write on a pad, though written statements did not match questions asked. She endorsed feeling sad, writing that she does not know where she is and thinks her family has dropped her off. She was given rituximab 1000 mg after a negative hepatitis and tuberculosis screen.
By hospital day 23, she was brushing her teeth and trying to communicate, while retaining some perseveration with actions and words. By hospital day 28, she was transferred to inpatient rehabilitation for physical and occupational therapy (PT/OT), where her mental status continued to improve. The psychiatry consultation-liaison service continued to follow her at inpatient rehabilitation. On interview on hospital day 31, patient appeared alert and responsive to questioning, able to follow commands, with minor residual grimacing. She reports amnesia regarding the experience and ringing in her ears. Patient’s husband reports that her overall state seemed to improve with amantadine and PT/OT. Her last BFCRS was down to a 5 (Figure 2). She was discharged home after 6 days of rehabilitation, on amantadine, divalproex, carvedilol, Vitamin D, lorazepam taper, prednisone taper, and prophylactic sulfamethoxazole-trimethoprim, with outpatient follow-up to psychiatry, physical medicine and rehabilitation, and neurology.
Discussion and Literature Review
The post-partum period is associated with instances of new-onset or worsening autoimmunity, as the immunosuppressive state of pregnancy can shift to a potentially labile immunologic period (Figure 1).
5
Systemic Lupus Erythematosus, Multiple Sclerosis, and inflammatory bowel diseases can manifest or flare in the post-partum stage.5-8 This immune-labile period has neuropsychological implications. Accumulating evidence points to an immune-mediated encephalitic mechanism underpinning at least some forms of post-partum psychosis.
9
Autoimmune Encephalitis and Associated Autoantibodies. The Post-partum Period May Unmask Autoimmunity and is Often a Presenting Period of New-Onset Autoimmune Disease. In Autoimmune Encephalitis, Antibodies Directed Against Neuronal Targets Such as the N-Methyl-D-Aspartic Acid (NMDA) Receptors, as Well as Myelin Oligodendrocyte Protein (MOG), although Sometimes the Neuronal Antigen is Not Identified. Figure created With Biorender.com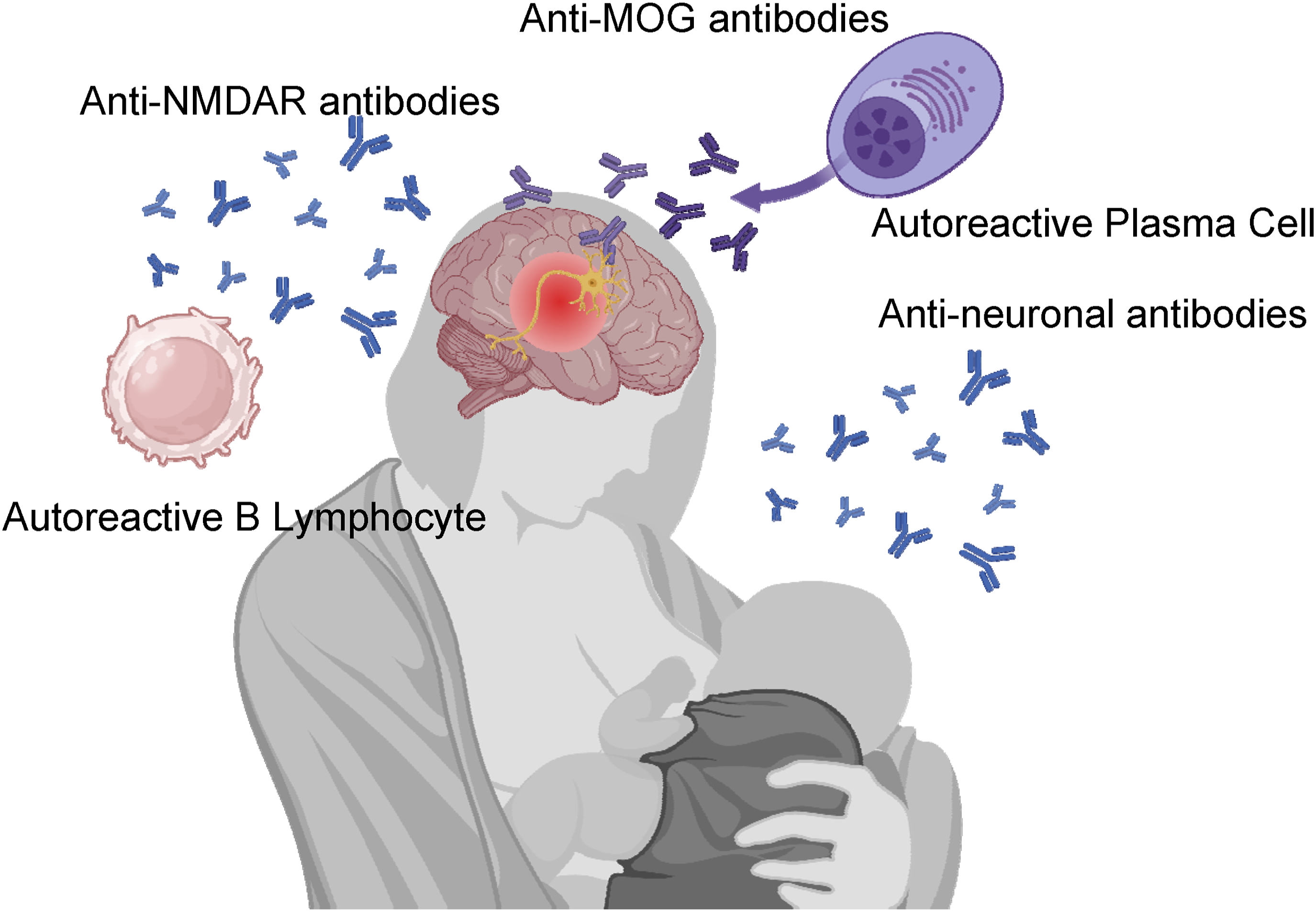
The DSM-5 does not uniquely recognize post-partum psychosis but uses the qualifier “with peripartum onset,” for cases arising during pregnancy or within four weeks post-partum. 10 Post-partum psychosis usually begins within several weeks of birth. However, the literature contains nearly 400 reports with psychosis onset more than 3 weeks after birth. 11 Some hypothesize menses resumption triggers later-onset post-partum psychosis. Our patient had weaned her infant just before psychosis onset. Her anxiety behaviors began at childbirth, without evaluation, and worsened overtime until acutely worsening at weaning.
Autoantibodies against anti-N-methyl-D-aspartic acid (NMDA) receptors or myelin oligodendrocyte glycoproteins (MOG) may be implicated in some cases diagnosed as post-partum psychosis.12-18 Sometimes, while antibodies against neuronal surfaces are noted to be present, the exact neuronal antigen cannot be found (Figure 1). 19 In some instances, autoantibodies may be negative. 1 Autoimmune thyroiditis and pre-eclampsia, both autoimmune in origin, predict higher rates of postpartum psychosis diagnoses. 20
Acute encephalitis is a neurological condition manifesting with rapidly progressive encephalopathy from brain inflammation. 21 While infectious causes of encephalitis have established diagnostic criteria and guidelines, immune-mediated encephalitis is an emergent, less-understood entity. 21 The autoimmune encephalitides represent a range of syndromes developing sub-acutely, involving impaired consciousness, behavior disturbances, psychiatric features, and movement disorders. 22 Antibody-associated immune-mediated neuropsychiatric disease falls in this class, as does antibody-negative, probable autoimmune encephalitis.
At least seven cases of fulminant post-partum encephalitis have been reported in the literature.12-18,23 Teratomas can trigger a paraneoplastic syndrome manifesting as encephalitis and should be excluded in the diagnostic work-up.18,23 Koksal and colleagues described a case of NMDAR encephalitis, initially misdiagnosed as postpartum psychosis, where a young woman three months post-partum presented with sudden delusions, hallucinations, and psychomotor excitement. 12 She was kept in psychiatric inpatient, failing risperidone, zodiazepine, aripiprazole, and two electroconvulsive therapy (ECT) sessions. A generalized seizure prompted her re-evaluation by neurology, who discovered anti-NMDAR antibodies in the CSF, despite their absence in the serum. An abdominal ultrasound and MRI showed an ovarian cystic teratoma. While she failed IV immunoglobulin (IVIG) and IV methylprednisolone, she made a sudden and nearly complete recovery after laparoscopic ovarian tumor removal. 12
Rhombencephalitis associated with MOG-IgG antibodies has occurred suddenly post-partum, successfully treated with plasmapheresis. 13 Shaaban and colleagues successfully treated a case of postpartum anti-NMDA-receptor encephalitis with rituximab. 14 Similarly, Lalanne and colleagues used corticosteroids and immunoglobulin, attaining full recovery in a post-partum woman with anti-NMDA receptor limbic encephalitis. 17 Steroid therapy and seven cycles of plasmapheresis were insufficient for one case, although the family members attained discharge against medical advice within the first two months of hospital treatment. 15
A prodromal period, sometimes after viral illness, may herald post-partum autoimmune encephalitis, often occurring in young women without a psychiatric history, who may have known autoimmunity or family risk factors.1,19
Differential Diagnosis
Differential Diagnoses
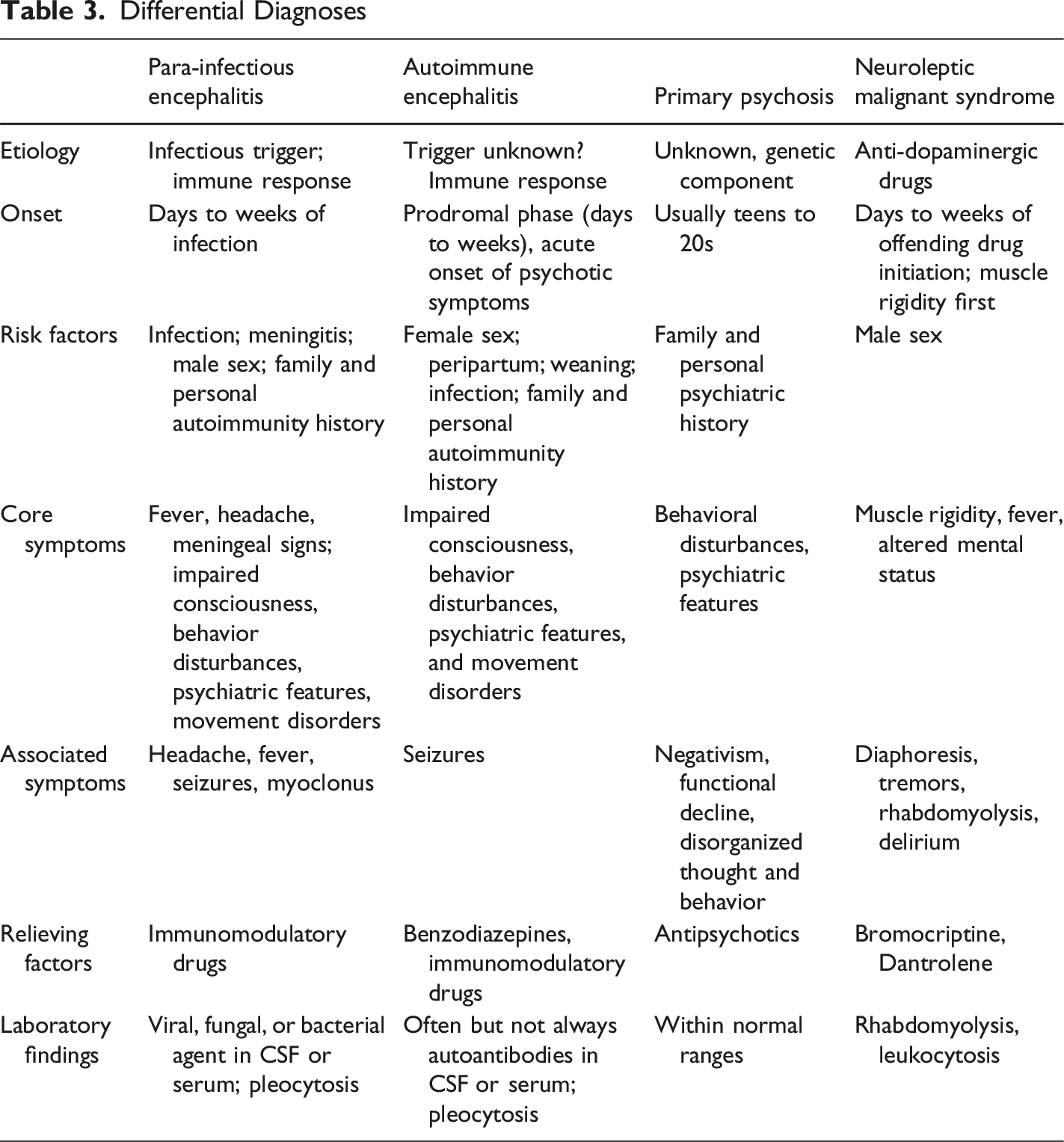
NMS arises from dopamine receptor-antagonist drugs, or their rapid withdrawal, especially haloperidol, which our patient received from OSH. 24 NMS typically occurs within hours or days of drug exposure, in a triad of fever, muscle rigidity, and altered mental status. Muscle rigidity usually begins first, then fever, then mental status changes, ranging from somnolence, agitation, or confusion to severe delirium. 24 Our patient certainly had muscle rigidity, intermittent low-grade fever, and altered mental status, with diaphoresis, tremor, mutism, tachycardia, labile blood pressure, and elevated creatine kinase. However, the temporal sequence was wrong; symptoms occurred prior to antidopaminergics (Table 3).
Consistent with immune-mediated encephalitis, she had prodromal worsening mood symptoms (days to weeks), then acute hallucinations and mania, with labile hypertension and tachycardia developing by outside presentation. Further, her family reported “rigidity,” strange movements, and facial grimaces, before pharmacotherapy. Her tremors, chorea, and muscle rigidity pre-dated antipsychotics (Figure 2). Disease Timeline. In This Timeline, We Note Our Patient’s Recent Childbirth Experience in the Fall, Along With the Psychosocial Stressors Faced From Fall to Spring, the Building Anxiety Behaviors, and the Sudden Onset of Bizarre Behaviors which Prompted Medical Evaluation. We Note her Bush Francis Catatonia Rating Scale (BFCRS) Scores as she Progressed to the Last Day of Hospitalization (Day 44 of Illness). Figured created With Biorender.com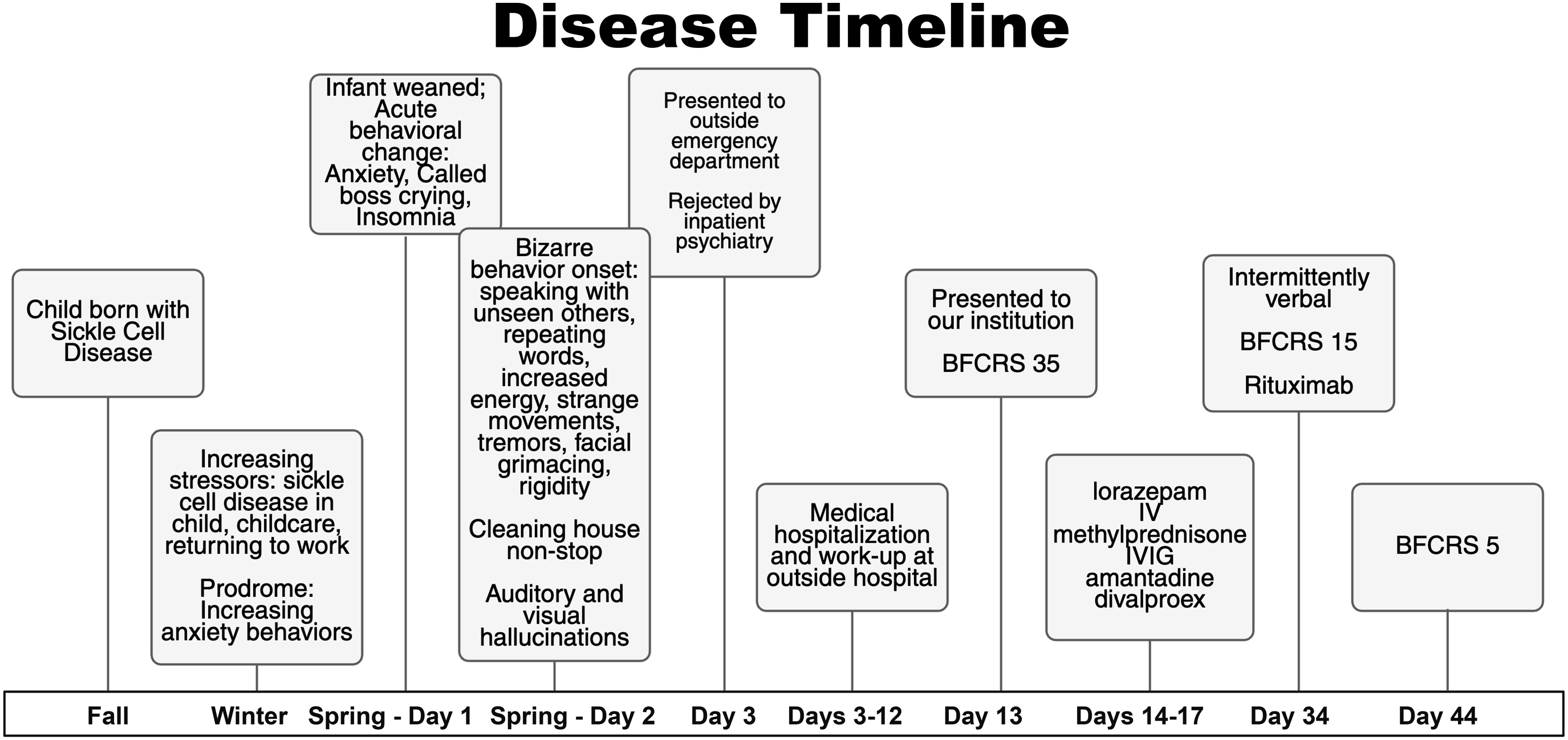
As our patient tested positive for THC, we considered cannabis-induced psychosis. Her negative initial OSH drug screen was positive at our institution, which could reflect varying sensitivities, substance use in between hospitalizations, or false-positives, like consumption of opiate-containing foods. Family may have tried cannabis to modulate psychotic symptoms. A Swedish cohort showed sampling cannabis by age 18 increased schizophrenia risk 2.4-fold, with a dose-response relationship later emerging.25-27 However, initial OSH negative THC screen lowers cannabis-psychosis suspicion; plus, intermittently negative THC screens suggest low total dose. Cannabis may have lowered her psychosis threshold, however.
Post-infectious immune-mediated encephalitis (IE) can cause catatonia; our patient had presumed infectious bronchiolitis treated with antibiotics, 28 and pleocytosis on CSF, suggesting an immune response. IE clinically resembles AE; the real difference is etiology. 28 Cytotoxic T cells and autoantibodies drive AE, whereas direct infectious insult and inflammation underpin IE. Fever, headache, and meningeal signs present saliently in IE, whereas movement disorders, memory deficits, and seizures commonly characterize AE. Elevated protein, causative culture agent, and prominent fever would be expected in an IE. Our patient’s anemia, recent childbirth and weaning, family lupus history, female sex, elevated inflammatory markers with intermittent fevers, strong positive response to immunosuppressants, and prominent movement disorder favor autoimmune over infectious. A multi-hit immunologic hypothesis could be proposed: autoimmunity predisposition, post-partum, recent weaning, cannabinoids, and infectious bronchiolitis.
Our case contains several unique aspects of autoimmune encephalitis presentation and represents important technical and legal constraints. Due to inadequate CSF collection, we were unable to obtain autoimmunity studies, which would have been helpful at confirming the diagnosis of autoimmune encephalitis. Also, additional imaging, like positron emission tomography (PET), could have further elucidated autoimmune encephalitis.
Our patient required remarkably high doses of lorazepam up to 30 mg per 24 hours, as attempts at lowering the doses resulted in rebounding catatonia and autonomic instability. Addition of IV divalproex sodium was not effective in managing her catatonia; consequently, amantadine was added. Only significantly higher doses of amantadine (400 mg per 24 hours) resulted in improvement of her catatonia and eventual lowering of lorazepam dosing.
Lorazepam, a benzodiazepine, is classically used to diagnose catatonia via the “lorazepam test.” BFCRS improvement following 1-2 mg of lorazepam is considered a positive test, supporting the diagnosis of catatonia. Benzodiazepines act on gamma-aminobutyric acid type A (GABA-A) receptors in the CNS, enhancing inhibitory effects, reducing neuronal hyperactivity implicated in encephalitis. 21 Multiple sclerosis studies suggest that lorazepam may indirectly modulate immune cell activity, lowing CNS inflammation. 29 Amantadine is an anti-viral drug that also non-competitively antagonizes NMDA receptors, excitatory glutamate receptors targeted by anti-neuronal antibodies in autoimmune encephalitis. 30 However, amantadine also has anti-inflammatory effects, lowering pro-inflammatory cytokines. 31
While ECT is an ideal treatment to break catatonia, the State of Texas requires direct patient consent or that of a legal guardian. Our patient was not able to grant consent and did not have a legal guardian. Administration of ECT would have required either out of state transfer or guardianship proceedings which would delay treatment for weeks; Family was not additionally ready to proceed with legal action to obtain emergent guardianship through the court.
Conclusion
In this case, malignant catatonia was the presenting sign of autoimmune encephalitis, which responded strongly to immunomodulatory therapy. Our patient had a strong family history of autoimmunity, no prior psychiatric history, and was in the first 6 months of the post-partum period. Rapid diagnosis and appropriate treatment permitted a complete neurological and physical recovery.
The combination of autonomic instability, catatonia, and dyskinesias make a primary psychiatric condition less likely. In primary care and consult psychiatry, patients may present with an outside diagnosis; women and minorities face an especially high risk of incorrect psychiatric diagnoses. 32 It is critical to avoid anchoring bias by creating a thorough medical and neurological differential. This case illustrates the first rule of Psychiatry: underlying medical causes of psychiatric presentations should always be thoroughly ruled out.
Footnotes
Author contributions
NG, DJ, RS: conceptualization, data acquisition, and interpretation. DJ, VS: data acquisition and interpretation. NG, VS, DJ: drafting the manuscript and interpreting data. NG, VS, DJ, RS: critical revision of manuscript. All authors gave final approval of the version to be published and agree to be accountable for accuracy and integrity of the manuscript, ensuring that questions related to accuracy or integrity are appropriately investigated and resolved.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
