Abstract
Objective
Despite the well-established increased risk of cardiovascular mortality in individuals with bipolar disorder (BD), prevention and treatment of cardiovascular risk factors and diseases have been largely overlooked in this population. This manuscript reviews the pathophysiological basis of the connection between BD and cardiovascular diseases, highlighting their shared mechanisms, reciprocal interactions, and relevant prevention and treatment strategies.
Methods
For this narrative review, a search was carried out on PubMed using the keywords bipolar disorder, cardiovascular diseases, and cardiovascular risk factors.
Results
The increased frequency of cardiovascular morbidity in BD can be attributed to overlapping biological and psychosocial mechanisms. These mechanisms are complex and involve both direct effects of BD and indirect effects mediated by lifestyle and pharmacological factors. Cardiovascular diseases also significantly exacerbate the clinical course of BD and increase morbidity and healthcare costs; thus, their effective management can improve psychiatric outcomes and vice versa. However, patients with BD frequently encounter healthcare barriers.
Conclusion
Awareness initiatives and research on integrated care are needed to determine the best strategies for improving cardiovascular and psychiatric outcomes in individuals with BD.
Introduction
Bipolar disorders (BD) are a spectrum of conditions (bipolar I, bipolar II, and cyclothymic disorder) characterized by episodes of emotional states affecting a person’s mood, energy level, and ability to function. These mood episodes, lasting from days to weeks, are categorized as manic/hypomanic when the predominant mood is markedly elevated or irritable, or depressive episodes, when there is an intensely sad mood or the ability to experience joy or pleasure disappears. 1 These mood episodes can affect daily routines and social interactions, leading to disruption in relationships and difficulties at school or work. The prevalence of BD is around 2.4% and the condition is among the most disabling worldwide.1,2 In addition, life expectancy is significantly reduced in these patients. This fact is the result of the combination of multiple factors, including increased risk of suicide and death rate from cardiovascular diseases (CVD).1,2
The American Heart Association recognizes BD as a condition that increases the risk of early CVD and recommends earlier screening and tighter control of cardiovascular risk factors. 3 However, there has been limited attention to prevention and treatment of CVD comorbidities in BD. Furthermore, the link between BD and cardiovascular diseases (CVD) is complex, bidirectional and not completely understood.
The objective of this review is to investigate the connection between CVD and BD, describing their shared pathophysiological pathways, the role played by pharmacological treatments, and pitfalls in management strategies. For this review, a search was carried out on PubMed using the keywords bipolar disorder, cardiovascular diseases, and cardiovascular risk factors. Clinical trials, meta-analyses, retrospective/prospective cohort studies and current guidelines published until October 2024 were retrieved and discussed below.
Cardiovascular Diseases in People with Bipolar Disorder: An Overview
Premature death in patients with severe mental illness (SMI), especially schizophrenia and BD, has historically been attributed to suicide. 1 However, in the last 2 decades, studies have shown that CVD has more impact on reducing the life span of these patients than suicide. CVD encompasses a broad range of conditions, such as myocardial infarction, heart failure, ventricular arrhythmias that are potentially life threatening. People with BD have an increased incidence of CVD and have been observed to die twice as often as the general population.1,2
Recent studies have investigated vascular and cardiac dysfunction in patients with BD using different imaging modalities. For instance, a Taiwanese group observed that BD patients had significantly higher rates of left ventricular (LV) hypertrophy (63% vs 42%) and hyperdynamic heart (32% vs 2%) associated with diastolic dysfunction, and higher mean values of LV mass index and ejection fraction compared to healthy controls. Interestingly, there was a positive association between inflammatory markers (interleukin 8, IL-8; and soluble tumor necrosis factor receptor 1, sTNF-R1) and echocardiography parameters, suggesting a potential role of low-grade inflammation as a contributor mechanism to LV hypertrophy and cardiac dysfunction in BD patients. 4 Non-invasive ultrasound measures of carotid structure (ie, carotid intima media thickness) and function (ie, flow-mediated dilation) were associated with traditional vascular risk factors and predicted CVD among adolescents with BD. 5
A pivotal study conducted by Osby et al 6 from 1973 to 1995 involving 15 386 patients with BD in Sweden showed a higher risk of death with a standardized mortality ratio (SMR) of 2.6, with cardiovascular and cerebrovascular diseases contributing to a significant portion of excess deaths in this population. A more recent systematic review of the literature on CVD-related mortality in patients with BD showed a hazard ratio of 1.65 (CI: 1.10-2.47). 7 Another Swedish study found that BD patients died 9 years earlier than expected, even after adjusting for sociodemographic factors. 8 Longer duration of mood episodes (both depression and mania) has been linked to higher vascular mortality. 9 In summary, there is unequivocal evidence showing increased CVD-related mortality in BD. While this finding has been observed worldwide, the majority of the studies were carried out in developed countries. 10 Accordingly, further studies from diverse countries are warranted.
As expected, given the increased CVD-related mortality, BD is associated with increased CVD morbidity probably secondary to accelerated atherosclerosis. A group of mostly male individuals with BD receiving clinical care through the Veterans Administration (VA) had high rates of various cardiovascular conditions at a younger age compared to a group without mood disorders. 9 Data from the National Epidemiologic Survey on Alcohol and Related Conditions (NESARC) confirmed that individuals with BD I were significantly more likely to experience hypertension and CVD such as coronary arteriosclerosis, angina, and myocardial infarction with onset of illnesses occurring much earlier than in controls. 11 The same trend has been described for stroke. A systematic review and meta-analysis of studies evaluating stroke in BD showed that people with BD had a significantly increased risk of both stroke incidence (HR, 1.43; 95% CI, 1.24-1.66; P < 0.001) and stroke mortality (HR, 1.54; 95% CI, 1.09-2.18; P = 0.013) compared to participants without BD. 12 Importantly, stroke is among the leading causes of excess cardiovascular mortality in BD, secondary only to coronary artery disease.13,14
While the increased prevalence of CVD is also observed in other mental or psychiatric disorders, such as major depressive disorder, BD seems to have a particularly high frequency of comorbid CVD. 10 People with BD also show lower rates of screening, prescription, and treatment adherence targeting cardiovascular risk factors. 10 This seems to contribute to excess cardiovascular mortality and undertreatment at multiple levels, even in countries with universal healthcare systems. 10 Shifting the focus of CVD prevention and treatment earlier for those with BD could help address these issues. In this context, organizations like the American Heart Association have recognized BD as a condition that increases the risk of early CVD and recommend earlier screening and tighter control of cardiovascular risk factors. 3 Research programs like PRIMROSE have also shown the effectiveness of modified risk prediction models for people with SMI over traditional models (like UK NICE and Framingham scores) based solely on cardiovascular risk factors. 15 In addition to traditional factors, PRIMROSE models (PRIMROSE Lipid and BMI models) include specific variables for psychiatric diagnosis, psychotropic medication at baseline and harmful use of alcohol, anti-depressants and social deprivation score. 15
Potential Mechanisms Underlying the Connection Between Bipolar Disorder and Cardiovascular Diseases
The elevated frequency of cardiovascular morbidity and mortality in BD can be attributed to several overlapping biological, psychosocial, and pharmacological mechanisms. These mechanisms are complex and interrelated, involving both direct effects of BD and indirect effects mediated by lifestyle and treatment factors.
Shared Risk Factors
Obesity, dyslipidemia, diabetes, smoking are shared between CVD and BD.16-18 While these conditions are seen as traditional risk factors for CVD, they have also been implicated in the pathophysiology of BD. Actually, their presence usually indicates a more severe BD phenotype. For instance, in a study involving patients with BD I, obesity was associated with a faster recurrence of mood episodes and a lower chance of remission with lithium or valproate treatment. Moreover, higher BMI in individuals with BD has been linked to unique brain changes, including greater volume loss in specific areas such as anterior cingulate gyrus and orbitofrontal cortex.17-19 Dysglycemia, or abnormal blood sugar levels, in BD adults has also been associated with lower volumes in the hippocampus and insular cortex. 20 Altogether, these findings highlight the complex relationship among BMI, blood sugar levels, and brain structure in individuals with BD. 19
Among adolescents with BD, obesity is highly prevalent and related to proxies for illness severity, including suicide attempt, self-injurious behaviors, and oppositional defiant disorder.21,22 BD among youth is considered a tier II moderate-risk condition that predisposes to accelerated atherosclerosis and early CVD and, therefore, should be managed in accordance with the same guidelines made for other moderate-risk traditional CVD risk factors. 3 In the opposite age range, nearly two-thirds of obese older adults with BD have had a stroke or silent cerebral infarction, with a higher prevalence of silent cerebral infarctions compared to non-BD elderly individuals. 14 The number of recurrent mood episodes also seems to increase the risk of stroke in older adults with BD. 14 It is worth noticing the paradoxical relation between CVD and sex/gender in adults with BD. While men have higher frequency of CVD in the general population, women with BD have higher CVD than men with BD. 23
Shared Pathophysiology
The interaction between SMI and CVD is thought to be influenced by several biological processes. An important mechanism is the dysregulation of the hypothalamic-pituitary-adrenal (HPA) axis that can be aggravated by ongoing stressors. 24 Indeed, BD is frequently associated with elevated cortisol levels and impaired cortisol stress reactivity. The migration of circulating leukocytes to artery walls, imbalance between pro- and anti-inflammatory cytokines, and malfunction of endothelial cells can all be consequences of persistently elevated cortisol levels. 24 Each 1 of them can contribute to the development of atherosclerotic plaques. Cortisol also interferes with glucose metabolism, which can lead to hyperinsulinemia, insulin resistance, and ultimately diabetes. Individuals with BD also display blunted heart rate variability, indicating dysregulation of autonomic nervous system that can be influenced by chronic stress. 25 The resulting protracted hyperadrenergic state can stimulate oxidative stress and vascular inflammation, further contributing to the development of atherosclerosis and hypertension. 26
Mood disorders and CVD are individually linked to increased levels of inflammatory biomarkers, such as interleukin (IL)-1β, IL-6, and C-reactive protein 27-29. In addition, inflammation is linked to traditional cardiovascular risk factors, such as obesity and diabetes, indicating a pivotal role for inflammation on the comorbidity between CVD and BD.
28
Both peripheral and central nervous system inflammation have been described in BD
30
(Figure 1). In BD, inflammation may contribute to glucocorticoid resistance, endothelial dysfunction, blood-brain barrier disruption, altered neurotransmitter metabolism, and neuronal damage. Although the exact mechanisms underlying BD-related inflammation remain uncertain, gut dysbiosis, ie, reduced gut microbial diversity and related changes in metabolites, has been consistently reported in BD and implicated as 1 the probable mechanisms.31,32 Besides potentially contributing to systemic inflammation in BD, gut dysbiosis has also been implicated in metabolic, especially lipid disruption that heightens the risk of CVD in this population.
33
Pathophysiology and shared risk factors of bipolar disorders in cardiovascular diseases.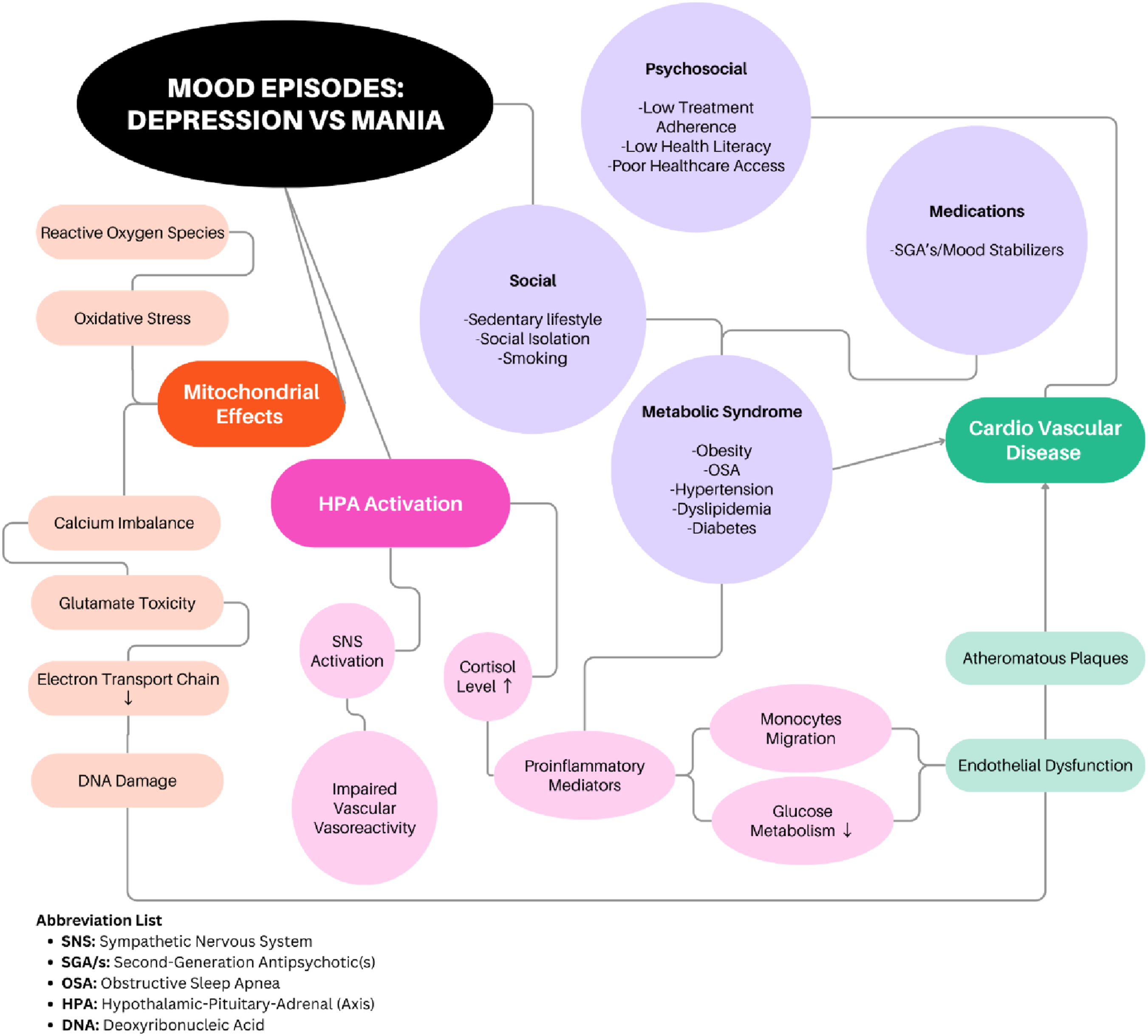
BD is often characterized by impaired energy production and increased cellular damage due to reactive oxygen species (ROS) build-up, indicating that problems on how the brain handles energetic metabolism, especially how it utilizes glucose within mitochondria, could play a significant role in the development of the illness. 34 Indeed, individuals with BD often exhibit abnormalities in glucose metabolism, including altered rates of glucose uptake and utilization in the brain, potentially leading to mitochondrial dysfunction. When mitochondria are not functioning optimally, they produce ROS, leading to oxidative stress, which damages cellular components like proteins, lipids, and DNA. 35 In addition, fluctuations in energy production due to mitochondrial dysfunction could contribute to the mood swings experienced by BD patients, with manic phases being associated with increased energy production while depressive phases with reduced energy. Oxidative stress secondary to mitochondrial dysfunction could also influence cognitive functioning in BD.36,37 Researchers have been investigating strategies that could improve mitochondrial function by enhancing energy production, regulating calcium levels, and/or reducing oxidative stress. For instance, supplementation with antioxidants could potentially mitigate the effects of increased ROS in BD. 38 While research is promising, more studies are needed to fully understand the mechanisms involved and to develop effective treatments targeting mitochondrial dysfunction in BD.
Finally, it is worth mentioning recent studies that have identified shared genetic factors between BD and CVD, including genes involved in MAPK signaling, lipid and glutathione metabolism pathways.39-41
Effect of Psychiatric Medications on Cardiovascular Health
Pharmacological treatments for BD, particularly second-generation antipsychotics (SGAs) and mood stabilizers, are associated with significant cardiovascular risk factors, including weight gain, dyslipidemia, and insulin resistance. 42 These metabolic side effects, coupled with the inherent biological vulnerability of BD, significantly increase the risk of CVD. 42 The risk of metabolic syndrome is nearly 2 times higher in patients taking SGAs than in those not on these medications. 42 Among SGAs, clozapine and olanzapine are known to significantly induce weight gain, while ziprasidone, lurasidone, aripiprazole and cariprazine have lower risk. 42 Regarding mood stabilizers, lithium can induce weight gain, but less so compared to valproate or SGAs. Lamotrigine and carbamazepine do not significantly affect weight.43,44
Mood stabilizers, particularly lithium and carbamazepine, can also have direct cardiovascular effects, mainly dysrhythmias. Lithium is known to cause QT interval prolongation, sinus node dysfunction, first degree AV block, myocarditis, chronic heart failure and orthostatic hypotension. Sinus tachycardia is the most frequently observed cardiac side effect of carbamazepine, while sinus bradycardia, atrioventricular block, premature ventricular contractions, ventricular tachycardia and junctional escape rhythms have been reported in patients with carbamazepine toxicity.42,43
Selective serotonin reuptake inhibitors (SSRIs) are considered safe for managing psychiatric disorders (eg, depressive and anxiety disorders) in patients with CVDs, although they are not usually indicated for BD. SSRIs can cause orthostatic hypotension, mild bradycardia, and QT interval prolongation, although these are generally less severe compared to mood stabilizers and tricyclic antidepressants.45,46
Lifestyle Factors
People with BD frequently have unhealthy dietary habits, a sedentary lifestyle, and diminished exercise capacity.47,48 Conversely, engaging in physical activity has been shown to reduce depressive symptoms, improve quality of life and overall function. Exercise may also promote neurogenesis in individuals with BD.48,49
A lifestyle intervention program tailored specifically for BD, known as the Nutrition, Exercise, and Wellness intervention (NEWTx), combined nutrition, diet, and exercise strategies. 50 NEWTx was a 20-week individual cognitive behavioral therapy-based treatment comprising 3 modules: (i) nutrition that teaches appropriate serving sizes and balanced diet; (ii) exercise that emphasizes increasing weekly physical activity; (iii) wellness focused on skills for healthy decision-making. Body weight, cholesterol and triglycerides levels decreased over the intervention as well as the number of daily calories and sugar intake. Weekly exercise duration more than tripled while depressive symptoms and overall functioning improved. 50
The episodic nature of BD, with alternating periods of mania and depression, often exacerbates unhealthy lifestyle behaviors. For example, depressive episodes are linked to increased sedentary behavior, while manic episodes can lead to impulsive behaviors, including overeating and excessive alcohol and other drugs use.51,52 Smoking and substance abuse are more prevalent among individuals with BD and contribute substantially to cardiovascular risk. 53
Sleep disorders are frequent in individuals with BD and are linked to weight gain, more severe mood symptoms, and poorer treatment response.54,55 For instance, a large cohort study reported that 54% of patients with BD were at risk for obstructive sleep apnea, a condition also implicated in CVD. 56 Sleep fragmentation and/or oxygen desaturation during sleep could induce and/or aggravate mood symptoms. 57 Accordingly, addressing sleep disorders should be an important intervention to reduce both illness severity and cardiovascular risk in patients with BD. 58 Pharmacotherapy such as melatonin or melatonin agonists, as well as psychosocial interventions, and continuous positive air pressure for obstructive sleep apnea may be of therapeutic value.59,60
Impact of Cardiovascular Diseases in Bipolar Disorder and Related Management
CVD significantly exacerbates clinical course of BD, leading to increased morbidity, mortality, and healthcare costs.7,62 Individuals with BD also experience cardiovascular events at a younger age than the general population, with the average age of onset being up to 10 years earlier.61-63 Despite this disturbing picture, patients with BD and comorbid CVD face several challenges, as described below.
Adherence and Polypharmacy
One of the major challenges in managing both BD and CVD is treatment adherence. Polypharmacy, common in BD management, increases the risk of drug-drug interactions and adverse effects, making adherence more difficult. Patients with BD may be prescribed with antipsychotics, mood stabilizers and cardiovascular medications, each of which carries its own risk of side effects that can be increased with combination of drugs. Nonadherence is a well-documented issue, often resulting in poor cardiovascular and psychiatric outcomes. 64
Impact of Cardiovascular Medications on Mental Health
Optimal management of CVD in patients with BD requires an integrated, multidisciplinary approach. Regular screening for metabolic syndrome, hypertension, and dyslipidemia is essential, particularly for patients receiving SGAs. Lifestyle interventions, including smoking cessation, dietary modifications, and exercise promotion, are critical components of managing cardiovascular risk in this population.7,62
Regarding more specific measures, several RCTs assessed the efficacy of metformin for antipsychotic-induced weight gain. Metformin treatment resulted in significantly better anthropometric and metabolic parameters than placebo with a mean difference in weight of about −3.2 kg with a significant reduction in BMI and insulin resistance index, contributing to weight loss up to 5% in about half of the patients. In most studies, the dose of metformin was 1000 mg a day for a period of at least 16 weeks. 65 By enhancing insulin sensitivity through the activation of AMP-activated protein kinase (AMPK) and reducing hepatic gluconeogenesis, metformin mitigates metabolic adverse effects, potentially supporting adherence to SGAs and reducing patient dropout due to weight gain or metabolic dysfunction.66,67 Furthermore, metformin can help regulate lipid profile by lowering triglycerides and LDL cholesterol, while modestly increasing HDL, thus countering dyslipidemia often associated with SGA. Its anti-inflammatory properties may reduce the chronic low-grade inflammation linked to metabolic syndrome, potentially lowering the risk of CVD in patients on long-term SGA treatment. Its impact on the gut-brain axis may influence appetite-regulating hormones, further aiding in metabolic control. 68
The use of cardiovascular drugs must be carefully considered to avoid exacerbation of psychiatric symptoms. Captopril and lisinopril, ACE inhibitors, have been noted to display mood-elevating or antidepressive effect in some cases, possibly through their action on the central angiotensin system that can influence serotonin and dopamine pathways in the brain. This effect is not uniformly observed across all ACE inhibitors, partly dependent on their ability to cross the blood-brain barrier. 69 Angiotensin receptor blockers (ARBs), such as losartan, have fewer documented psychiatric side effects compared to other antihypertensives, but instances of psychosis and depression have been reported, particularly in older patients. On the positive side, losartan has shown potential cognitive benefits, which could be advantageous in populations at risk for cognitive decline. 70
Calcium channel blockers, especially verapamil, have been studied for their effects on mood stabilization in BD. These drugs prevent calcium influx, which may stabilize neuronal excitability and affect mood by modulating neurotransmitter release. Some studies have also suggested that verapamil may have antimanic effects, but the evidence is mixed. 70
Although statins are generally safe, patients may experience neuropsychiatric side effects such as irritability, mood fluctuation, and memory deficits. By lowering cholesterol, an essential component of cell membranes, statins may indirectly affect neurotransmitter transport and receptors density. Atorvastatin and simvastatin have been linked to increased risks of depression and cognitive complaints, although these associations are rare. 70
Combining β-blockers with antipsychotics can lead to adverse cardiac outcomes, such as bradycardia, hypotension, and even arrhythmias. Risperidone or perphenazine combined with metoprolol can cause bradycardia and hypotension, while metoprolol combined with sertindole or ziprasidone can lead to ventricular arrhythmias. Lipophilic β-blockers like metoprolol can cross the blood-brain barrier, decreasing norepinephrine activity, which impacts CNS pathways involved in stress and mood regulation. Therefore, in addition to cardiac risks, β-blockers may also lead to fatigue and depression. 70
Diuretics, although less frequently associated with direct neuropsychiatric effects, can contribute to electrolyte imbalances, potentially exacerbating symptoms of confusion or fatigue. Electrolyte changes, particularly low sodium and potassium can disrupt neuronal communication, especially in older adults. Thiazide diuretics are known to interfere with lithium excretion increasing its plasma concentration when both are co-administered. Induction of lithium toxicity by thiazides can result in multiple neuropsychiatric symptoms including confusion, and tremors. 71
Different strategies like non-steroidal anti-inflammatory drugs, aspirin, polyunsaturated acids, N-acetylcysteine, anti-cytokines, and probiotics have provided promising results in terms of antidepressant efficacy in patients with BD.72,73 Some of them also have well-established (aspirin) or potential (polyunsaturated acids, N-acetylcysteine) preventive effects for CVD.74,75
The bidirectional interaction between the brain and the gut, named ‘brain-gut-microbiome axis' or ‘gut-brain axis', plays a key role in the link between nutrition and mood disorders. A ‘healthy' diet, generally characterized by a higher intake of fruits, vegetables, legumes, nuts, whole grains, and good quality sources of protein (ie, fish and/or seafood), is associated with a decreased risk of mood disorders which in addition has an independent positive impact on maintaining cardiovascular health. 76 Also, time-restricted eating along with caloric restriction have a positive impact on metabolic profile, symptoms of mood, and weight loss in obese women. 77 A ketogenic diet may have a positive impact on mood, reducing anxiety and improving sleep in BD patients by modifying neurotransmitter levels of glutamate and GABA.78,79 In addition, it has potential benefits on cardiovascular health, causing weight loss, lowered blood sugar and triglycerides and increased HDL levels. As a word of caution, under a ketogenic diet, certain individuals may experience a rise in LDL cholesterol levels, which could increase cardiovascular risk.78,79
Impact of Social Support on Management
Social isolation can lead to depression and aggravate BD course. People with BD may experience isolation for different reasons, including shame or guilt about their condition, fear of burdening others, which can further accentuate social withdrawal. 80 Stereotype endorsement, social withdrawal and discrimination experience explained most of the self-stigma in patients with mood disorders. 80 An early assessment of self-stigma and perception of control over illness (self-efficacy) could assist patients in enhancing their real situation, ultimately reducing social withdrawal. Because of its connection with clinical outcomes (eg, quality of life, adherence to treatment, hospitalization, and suicide risk), addressing social isolation and related issues can positively impact the clinical care of individuals with BD. 80
The role of family and social support in recovery cannot be overemphasized. Family, friends, and community support are essential in patients’ recovery and overall well-being. The support system offers not only emotional comfort, but also a range of benefits. It significantly influences the psychological health of the patient, thereby affecting their recovery and quality of life. Family members and friends may play a crucial role in providing daily support, which includes assisting with medication management, scheduling doctor’s appointments, as well as offering support during difficult periods. The presence of others can help alleviate feelings of isolation and loneliness, which are frequently experienced by patients with BD undergoing treatment for chronic illnesses like CVD. 81
Access to health services is a major issue for people with BD, including poor access to general and cardiovascular hospital cares. The integration of family members and/or friends in the therapeutic process of individuals with BD should be incentivized, tailored by the unique challenges and demands of both patients and families. Family intervention can help reduce the risk of recurrence and functional impairment of patients, improve health and wellbeing of family members, and optimize socioeconomic and health resources.82,83
Discussion
Individuals with BD have a greater risk of cardiovascular mortality due to a combination of factors, including higher prevalence of cardiovascular risk factors and CVD, unhealthy lifestyle, and medication side effects. Addressing these risk factors through lifestyle changes and appropriate medical management (eg, use of metformin for SGA-induced weight gain) is essential in reducing cardiovascular mortality in this population.
Patients with BD frequently encounter obstacles in obtaining cardiovascular treatment, with a major contributing factor being the prevalent stigma and prejudice against their mental health condition. These unfavorable perceptions may cause healthcare professionals to either ignore or underestimate physical symptoms in individuals with BD. Individuals with BD may also experience subjective symptoms that overshadow the concurrent physical ailment. In this context, cardiovascular risk factors can be overlooked or neglected, potentially leading to negative cardiovascular consequences. For instance, the majority of patients with SMI have a lower likelihood of undergoing invasive coronary procedures. Another important treatment gap is within post coronary angioplasty care, in which patients with BD and myocardial infarction are less likely to receive a combination of aspirin, P2Y12 inhibitors, β-blockers, statins and ACEIs/ARBs compared with the general population. 84
Effective management of physical illnesses can lead to better psychiatric outcomes, and vice versa. In order to enhance patient outcomes, it is essential that healthcare professionals further undergo thorough training in the intersection of mental health and cardiovascular care. An integrated approach to medical and psychiatric care, even at primary healthcare level, may be more beneficial than separate services. Further implementation research and awareness initiatives are needed to determine the best strategies for improving cardiovascular and psychiatric outcomes in individuals with BD.
Study Limitations
Given its non-systematic nature, although we attempted to provide a broad perspective on the topic, it is not possible to ensure that all relevant literature has been duly considered. We have also emphasized targetable shared mechanisms (eg, inflammation, metabolic dysfunction) in detriment of emerging areas of uncertain potential for clinical translation. In this regard, it is worth mentioning the emergent literature investigating common genetic background of psychiatric disorders, including BD, and CVD.
Conclusions
Individuals with BD face a significantly elevated risk of CVD, driven by both the biological underpinnings of BD and the side effects of pharmacological treatments. Multidisciplinary care models bridging the gap between mental health, primary care, and cardiology can contribute to more comprehensive and effective management of the excess cardiovascular risk in patients with BD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
