Abstract
Children with disruptive mood dysregulation disorder have difficulties with emotion regulation and recognition which impacts their social functioning. Due to its recent definition, there is limited information on the neural mechanisms of this disorder. This exploratory study examined the role of autonomic heart regulation in children with disruptive mood dysregulation disorder, motivated by suggestions of reduced autonomic regulation (measured by heart rate variability) associated with psychiatric disorders in adults and children. The cross-sectional design sampled two groups of children, one with disruptive mood dysregulation disorder (n = 15; 14 male) and the other with typical development (n = 15; 14 male). Heart rate variability, facial emotion recognition accuracy and speed, and prosody were measured. There were no significant differences in heart rate variability between the two groups before, or during, the tasks. However, children with disruptive mood dysregulation disorder experienced significantly greater difficulty recognising fear in faces compared to controls and confused other negatively valenced emotions. There was also a significant negative correlation between heart rate variability and prosody modulation (verbal expression of emotion) in the control group, a relationship that was absent in the disruptive mood dysregulation disorder group. These results suggest that atypical autonomic regulation during emotionally evocative situations and difficulty with facial emotion recognition may contribute to the challenges experienced by children with disruptive mood dysregulation disorder. This is valuable knowledge for psychologists, which can contribute to individualised, effective, and comprehensive care that addresses the child’s emotional, social, and relational well-being.
Keywords
Disruptive mood dysregulation disorder (DMDD) is a new diagnostic category in the Diagnostic and Statistical Manual of Mental Disorders, Text Revision (5th ed.; DSM-5-TR; American Psychiatric Association [APA], 2022). Symptoms include persistent, debilitating irritability and recurrent temper outbursts. Despite low global prevalence of DMDD (0.8%–3.3%), many risk factors for DMDD (i.e., adverse childhood experiences) are ubiquitous in the lives of South African children, which may increase their risk of developing this disorder (Copeland et al., 2013; Slopen et al., 2010). Empirical data on DMDD is limited due to its recent inclusion and focus on its predecessor, severe mood dysregulation (SMD). Available research focuses on the prevalence, comorbidity, and cognitive and behavioural correlates of DMDD (Althoff et al., 2016; Dougherty et al., 2016). Atypical social behaviour such as irritability, misinterpretation of social, emotional, and threat cues characterise SMD (Zepf et al., 2015) and was hypothesised to be present in children with DMDD. However, the extent of these difficulties and their neurophysiological correlates have not yet been investigated. Consequently, this study explored the relationship between factors that may contribute to difficulties in social engagement behaviour (viz., facial emotion recognition and prosodic features of speech), and neurophysiological processes (viz., vagal tone, which controls heart rate) in children with DMDD.
The investigation is grounded in Porges’ (2001, 2003, 2007) Polyvagal Theory, particularly focusing on the social engagement system (SES), which regulates attention and emotion through a neural connection between the face and heart with mechanisms for expressing and receiving social cues. This theory underscores the role of the parasympathetic nervous system, particularly the vagus nerve, in governing social and emotional behaviours. The vagus nerve links the heart with facial muscles, affecting facial expressions and speech. The theory posits that humans have evolved to regulate heart rate rapidly, either by increasing it for fight/flight responses or by slowing it to promote prosocial behaviours (calming/co-operation; Stewart et al., 2013). This regulation, known as ‘vagal tone’ (Porges, 1992), is indexed by heart rate variability (HRV). Reduced HRV has been associated with psychiatric disorders in both children and adults, suggesting links between attenuated HRV, emotion reactivity, and poor emotion regulation in children (Graziano & Derefinko, 2013).
The primary component of high-frequency HRV is respiratory sinus arrhythmia (RSA). The amplitude of RSA – the phenomenon where the heart rate (i.e., the number of heart beats per minute; Palatini et al., 2006) increases during inhalation and decreases during exhalation – is a selective, valid, non-invasive index of the parasympathetic nervous system’s contribution to cardiac regulation (i.e., vagal tone; Laborde et al., 2017). Moderate RSA indicates improved vagal tone regulation and is positively correlated with prosocial behaviours in neurotypical individuals (Miller et al., 2017).
Impairments in the SES (i.e., through atypical vagal tone) may account for the social difficulties experienced by children with DMDD (and its precursor, SMD). Limited evidence indicates that relative to healthy controls, children with DMDD often label ambiguous faces as angry rather than happy (Stoddard et al., 2016) and demonstrate an attention bias towards angry faces compared to happy faces, which correlates with increases in symptom severity (Hommer et al., 2014). Compared to healthy volunteers and those with bipolar disorder, children with SMD show over-reactivity to angry faces (Thomas et al., 2014). This signifies that children with DMDD may experience difficulties in emotion recognition relative to peers, an issue investigated in our study.
Children with SMD also experience difficulties in determining the correct emotional tone of verbal speech (Deveney et al., 2012) and difficulties with vocal emotion labelling (Brænden et al., 2022). No studies have investigated possible difficulties in expressive prosody in children with DMDD, a focus of the current study. Evidence suggests that flat affect and monotone voice during childhood are behavioural predictors of depressive disorders in adulthood, which children with DMDD are more likely to develop (Yang et al., 2013). Therefore, children with DMDD may exhibit reduced prosody relative to their peers.
An understanding of the diagnostic features of DMDD as emergent properties of deficits in the SES would contribute towards an integrated neurocognitive perspective of these symptoms. This could inform intervention and treatment strategies to ameliorate vagal system functioning. It may also identify a physiological variable that could be an early predictor of DMDD. Consequently, the study research questions asked whether children with DMDD (compared to controls) have:
atypical autonomic regulation, indicated by shorter heart period (HP, that is, the inverse of heart rate) and lower RSA before, during, and after all tasks;
difficulties accurately identifying facial emotion, that is, reduced accuracy and increased reaction time;
reduced prosody, indicated by reduced modulation depth, increased bandwidth of fundamental frequency (BW50), and reduced spectral tilt (see Method for detail).
Method
Study design and sample population
This quasi-experimental study compared children who met the DSM-5 criteria for DMDD to gender, ethnicity, and grade-matched typically developing controls. English-proficient participants were recruited via purposive sampling (Table 1). Participants in the DMDD group were recruited through psychiatrists and psychologists in Johannesburg. Comorbid disorders in this group included attention-deficit hyperactivity disorder (ADHD; n = 13), anxiety (n = 1), conduct disorder (n = 1), and unspecified mood disorder (n = 1). Exclusion criteria included compromised neurology (e.g., epilepsy) or serious illness (e.g., meningitis) in the last 3 months. Control participants were recruited from schools in Johannesburg that disseminated information letters, consent, and assent forms to parents and learners. Control participants were excluded if they met any of the DSM-5 criteria for DMDD or had first-degree relatives with psychiatric illness diagnoses (e.g., depression, anxiety). The sample comprised 30 children and adolescents (15 per group; MAgeControl = 10.13, SDAgeControl = 1.85; MAgeDMDD = 10.13, SDAgeDMDD = 1.73).
Demographic variables, by group.
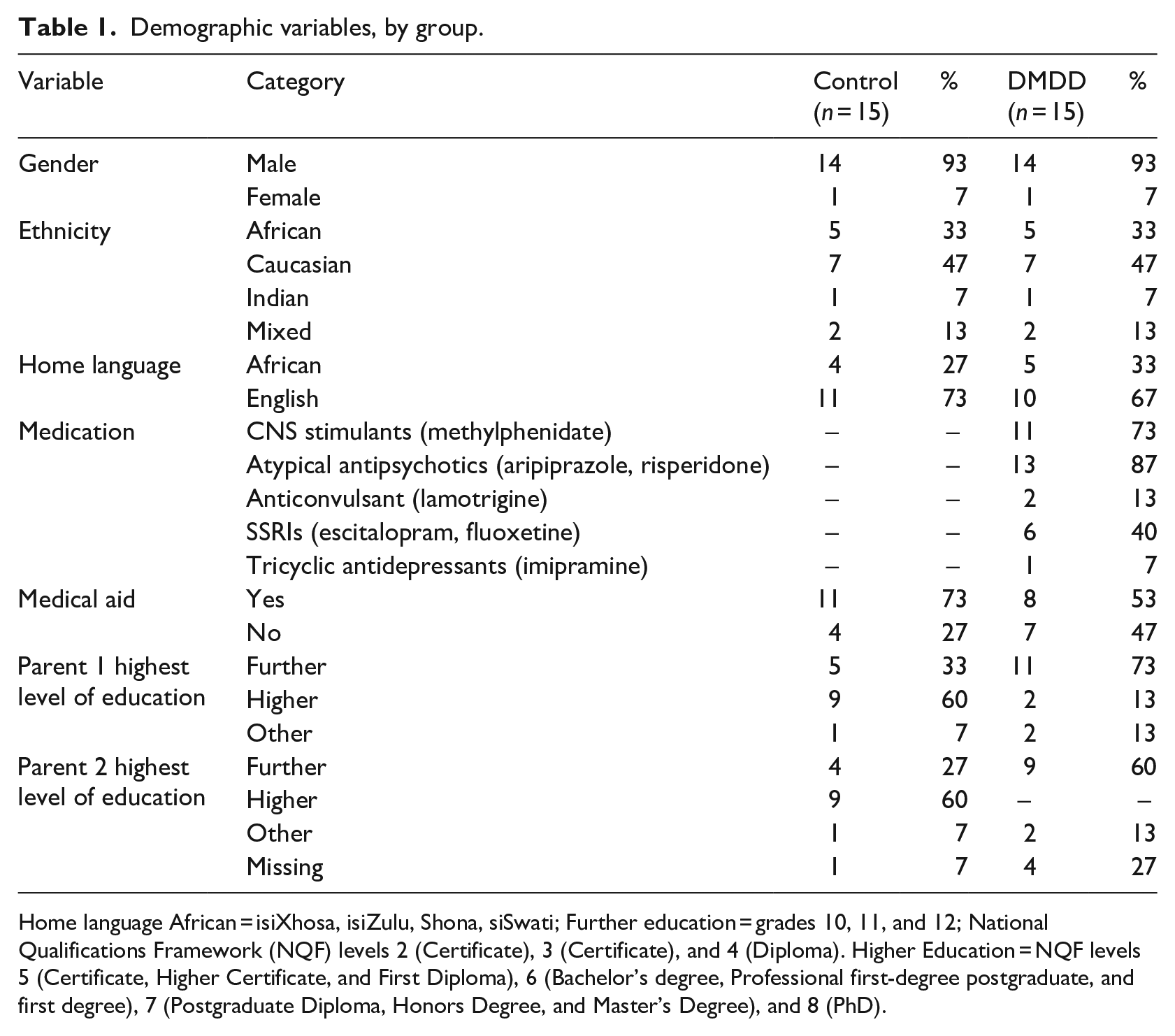
Home language African = isiXhosa, isiZulu, Shona, siSwati; Further education = grades 10, 11, and 12; National Qualifications Framework (NQF) levels 2 (Certificate), 3 (Certificate), and 4 (Diploma). Higher Education = NQF levels 5 (Certificate, Higher Certificate, and First Diploma), 6 (Bachelor’s degree, Professional first-degree postgraduate, and first degree), 7 (Postgraduate Diploma, Honors Degree, and Master’s Degree), and 8 (PhD).
Instruments
Physiological measures
Heart rate was recorded at 1024 Hz using a wireless single-channel electrocardiogram (ECG) recorder. The interval (IBI) series was extracted from HP with QRSTool (Allen et al., 2007). The IBI data were visually inspected for missed R-wave detections and artefacts and edited in CardioEdit (Brain-Body Centre, University of Illinois at Chicago). Less than 5% of each file required editing. CardioBatch (Brain-Body Center, University of Illinois at Chicago) was used to calculate RSA (Porges, 1985). A band-pass filter was applied to the detrended residual series to extract the variance in the HP pattern at the frequency of respiration for children (0.12–1.0 Hz).
Facial emotion recognition
The Dynamic Affect Recognition Evaluation (DARE; Porges et al., 2007) is a standardised measure of emotional facial expressions comprising a series of still images representing six basic emotions (anger, disgust, fear, happiness, sadness, and surprise) that morph into videos of a face starting with a neutral expression and transitioning into one of the six target emotions. Images include male and female college-aged students. The task consists of 4 sets of videos for each of the six emotions (24 videos) and has been used with US paediatric populations, but not in SA (Bal et al., 2010). The emotion displayed, the participant’s response, and the response latency (in milliseconds) were recorded.
Prosody
Prosody was recorded during a narrative task designed to evoke emotion through the recounting of everyday positive (e.g., ‘Tell me something good that happened in school’) and negative experiences (e.g., ‘Tell me about a time when you argued with your friend’). Audio files (20+ s each) were recorded at 44.1 kHz and saved in .m4a format. Prosodic variables included modulation depth (i.e., the variation in tone, volume, and rhythm), bandwidth of fundamental frequency (BW50; indicated an increased variability in pitch, with higher bandwidth denoting greater prosody), and spectral tilt (measures the energy distribution, or balance, between low and high vocal frequencies; Kakouros & Räsänen, 2016).
The study used a band-pass filter (0.2–6 kHz) and a 22.05 kHz sampling rate to analyse audio files for spectral tilt and fundamental frequency variability using the SHRP MATLAB function (Sun, 2002–2016). Files were separated into 40 ms windows, decomposed using Fast Fourier transformation. The mean peak frequency was computed.
Procedure
Individual assessments occurred at school between 08:00 and 10:00 a.m. as cardiac activity varies during the day. All participants were requested to avoid stimulants (e.g., caffeine or sugar) 2 hr before testing. The DMDD group was asked to delay medication on the day of testing until after the session to minimise cardioactive effects.
For both groups, 2 min of baseline HP and RSA data were collected at the start of the session while participants sat quietly, looking at a fixation cross on the laptop screen. Recording continued throughout the 15-min DARE task and the 40-s+ prosody task. After each task, a 2-min HP and RSA post-task response was recorded.
Participants completed the DARE first to avoid emotional carry-over from the prosody task. They had to identify the displayed emotion as quickly as possible and choose the correct answer from the six basic emotions.
On completion of the final post-DARE heart rate recording, participants completed the prosody task. Positive questions were posed first with time given to formulate a response, after which they were recorded while speaking. The same procedure was followed for the negative questions.
Ethical considerations
This study was approved by the Medical Research Ethics Committee (MREC) at the University of the Witwatersrand. Participants assented and their legal guardians gave written informed consent before participation, with appropriate opportunities for withdrawal without prejudice. Anonymity and confidentiality were assured. A psychologist was available during all sessions for debriefing, although none of the participants expressed distress. The MREC did not permit compensation for participation.
Data analysis
To determine group differences in performance on the DARE and prosody tasks, a repeated-measures mixed-effects model was employed with the outcome (i.e., RSA and HP; response time; accuracy; and prosodic features) as dependent variables, and age, group, test (DARE, prosody tasks), and the group–test interaction as independent variables (fixed effects; Ellis, 1999). ‘Participant’ was treated as a random repeated measure to capture the matched case–control nature of the data.
Normality of the model residuals was confirmed by inspecting model diagnostics. Post hoc comparisons used the Tukey–Kramer adjustment for multiple comparisons.
Results
RSA and HP
Tables 2 and 3 summarise the descriptive statistics for the groups’ RSA and HP measurements before, during, and after the DARE and prosody tasks.
Descriptive statistics for the dynamic affect recognition (DARE) physiological measurements by group.
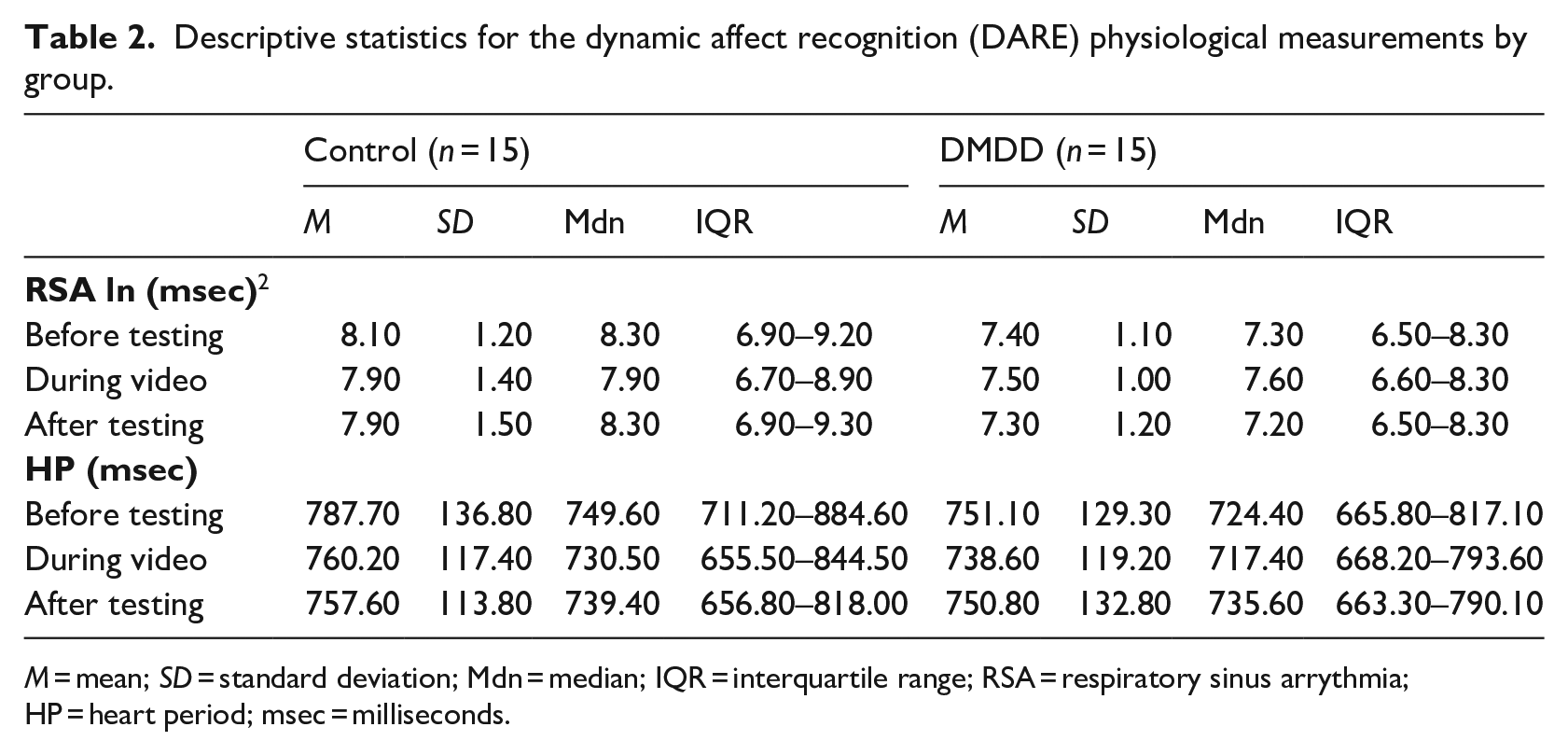
M = mean; SD = standard deviation; Mdn = median; IQR = interquartile range; RSA = respiratory sinus arrythmia; HP = heart period; msec = milliseconds.
Descriptive statistics for the prosody task physiological measurements, by group.
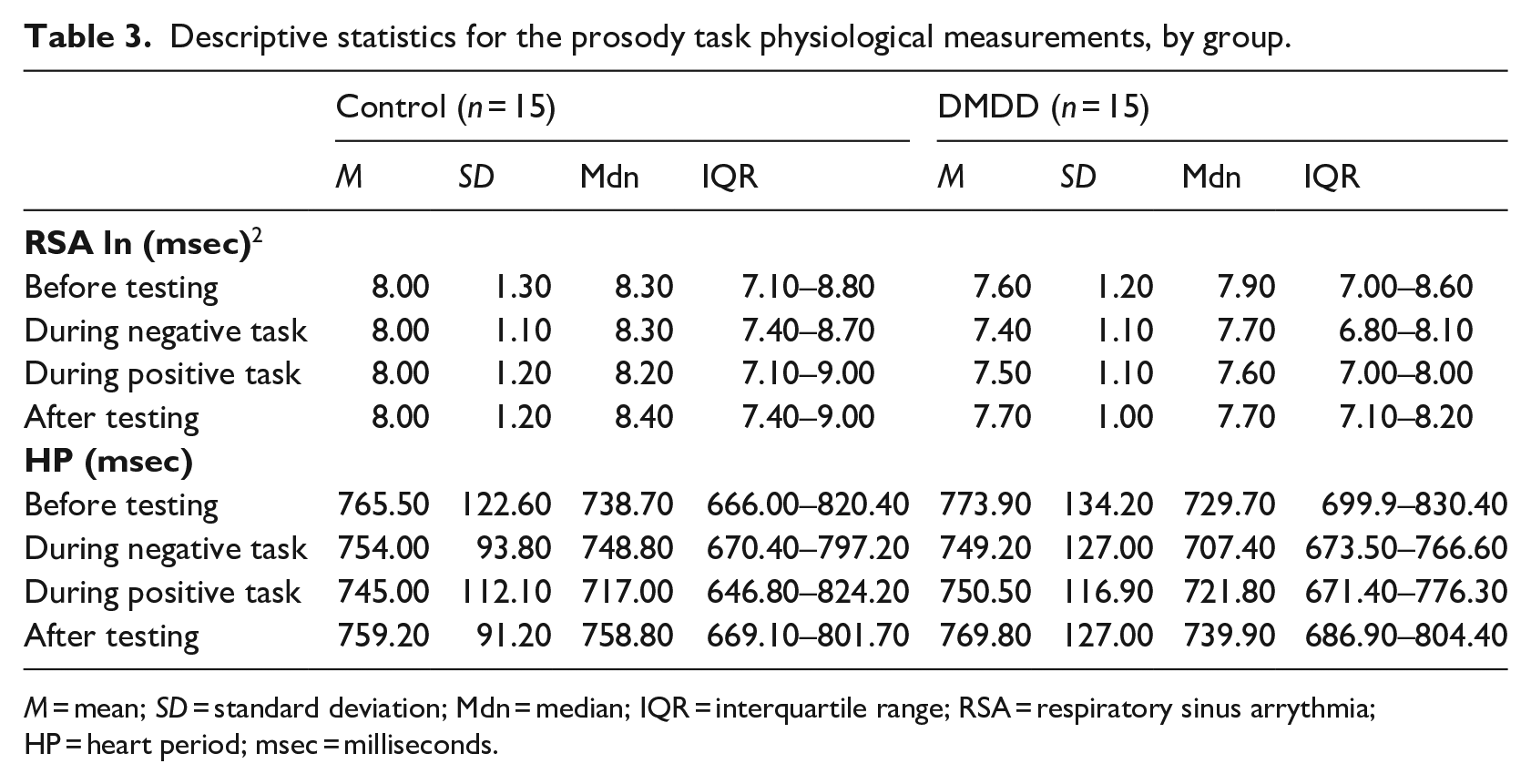
M = mean; SD = standard deviation; Mdn = median; IQR = interquartile range; RSA = respiratory sinus arrythmia; HP = heart period; msec = milliseconds.
The results of a mixed-effects model, controlling for age and physiological measurements (RSA and HP) before testing, showed that the DDMD and control groups had similar RSA during (F[1,15] = 1.60, p = .225) and after (F[1,16] = 0.78, p = .391) the DARE and prosody tasks (positive: F[1,23] = 0, p = .986; negative: F[1,11] = 1.94, p = .190; Table 2).
Similarly, HP did not differ between the groups during the DARE (F[1,14] = 0.92, p = .353), nor during and after the positive or negative prosody tasks (positive: F[1,24] = 0.02, p = .880; negative: (F[1,23] = 1.99, p = .172; after: F[1,12] = 0.02, p = .883). Heart period was significantly shorter in the control group after the completion of the DARE (F[1,14] = 5.13, p = .040, d = 0.36).
Affect recognition
Groups were compared on their ability to accurately and quickly recognise each emotion using the DARE (Table 4). They differed significantly in their accuracy for recognising fear, which was higher for the control group than the DMDD group (F[1,28] = 5.12, p = .032, d = 0.29). No significant differences were found for recognising anger (F[1,14] = 2.64, p = .127), disgust (F[1,28] = 1.85, p = .184), happiness (F[1,14] = 0.01, p = .928), sadness (F[1,14] = 0.01, p = .915), or surprise (F[1,14] = 1.56, p = .232). Control participants mistook fear for surprise (38%) and then happiness (26%), while DMDD participants typically misidentified fear as disgust (34%) and then happiness (29%).
Descriptive statistics for the dynamic affect recognition (DARE), by group.
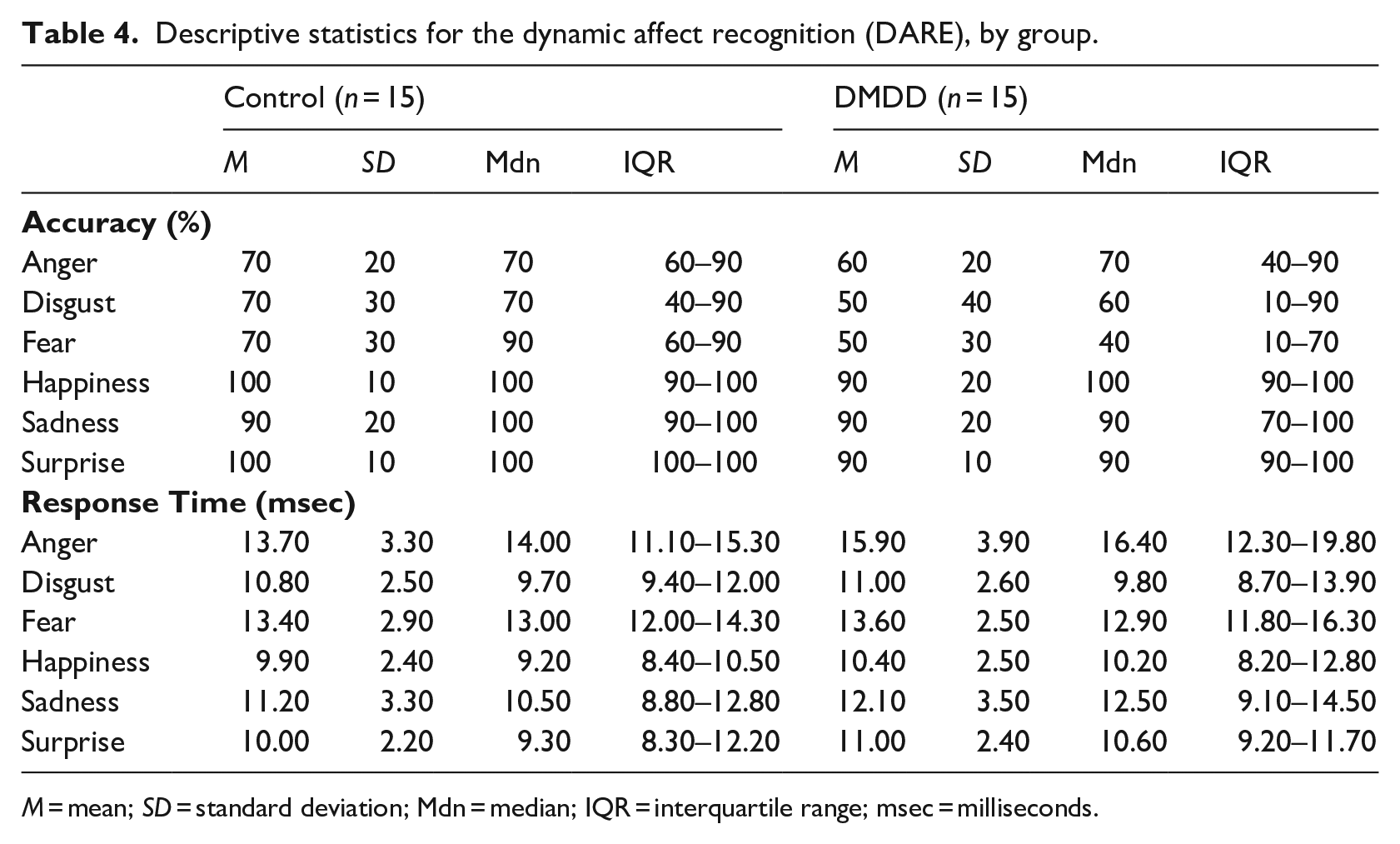
M = mean; SD = standard deviation; Mdn = median; IQR = interquartile range; msec = milliseconds.
Response latency did not differ significantly between the groups: anger (F[1,14] = 3.06, p = .102), disgust (F[1,14] = 0.05, p = .832), fear (F[1,27] = 0.79, p = .381), happiness (F[1,14] = 0.40, p = .539), sadness (F[1,14] = 0.47, p = .504), or surprise (F[1,28] = 1.42, p = .243).
Prosody
A main group effect was found for spectral tilt (F[1,25] = 4.79, p = .044, d = 0.45; Table 5) during the prosody tasks, meaning there was considerably more variation between the low and the high frequencies in the DMDD group’s speech compared to the control group. No main effects of group were found for BW50 (F[1,26] = 0.14, p = .714) or modulation depth (F[1,13] = 3.96, p = .068).
Descriptive statistics for prosody features, by group.
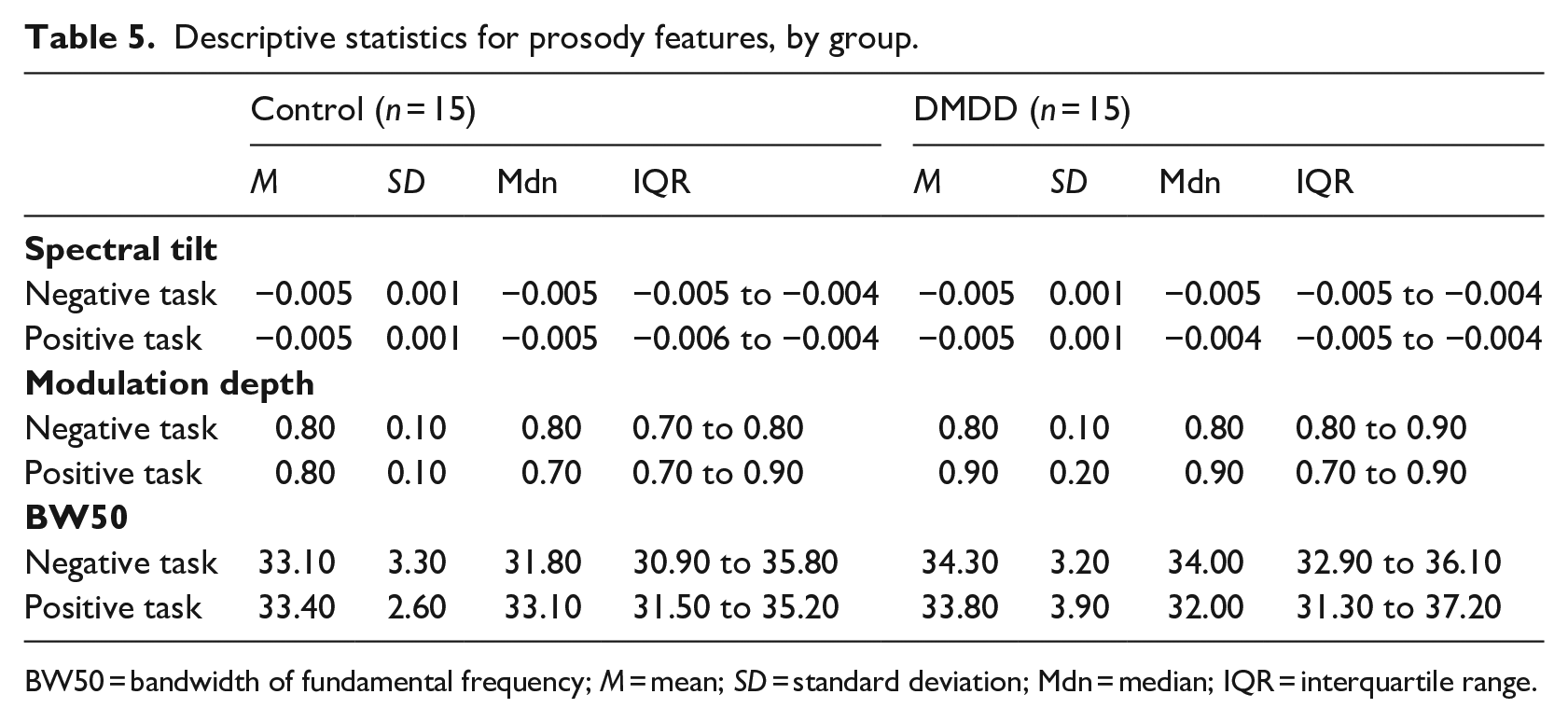
BW50 = bandwidth of fundamental frequency; M = mean; SD = standard deviation; Mdn = median; IQR = interquartile range.
Modulation depth differed significantly between the groups during the negative prosody task (F[1,25] = 5.26, p = .031, d = 0.39). The speech of the DMDD group was more modulated in frequency and time than that of the control group. There were no group differences on spectral tilt (F[1,13] = 0.61, p = .450) and BW50 (F[1,26] = 0.93, p = .344).
The relationship between modulation depth and RSA differed by group, both during the positive narrative task (Figure 1; F[1,14] = 8.24, p = .012, d = 0.39) and after the task (Figure 2; F[1,15] = 11.34, p = .004, d = 0.46). In both instances, the correlation between these variables was not significant for the DMDD group (Positive: rho = 0.319, p = .290; After: rho = 0.326, p = .280), but significant and negative for the control group (Positive: rho = −0.518, p = .048; After: rho = −0.608, p = .016).
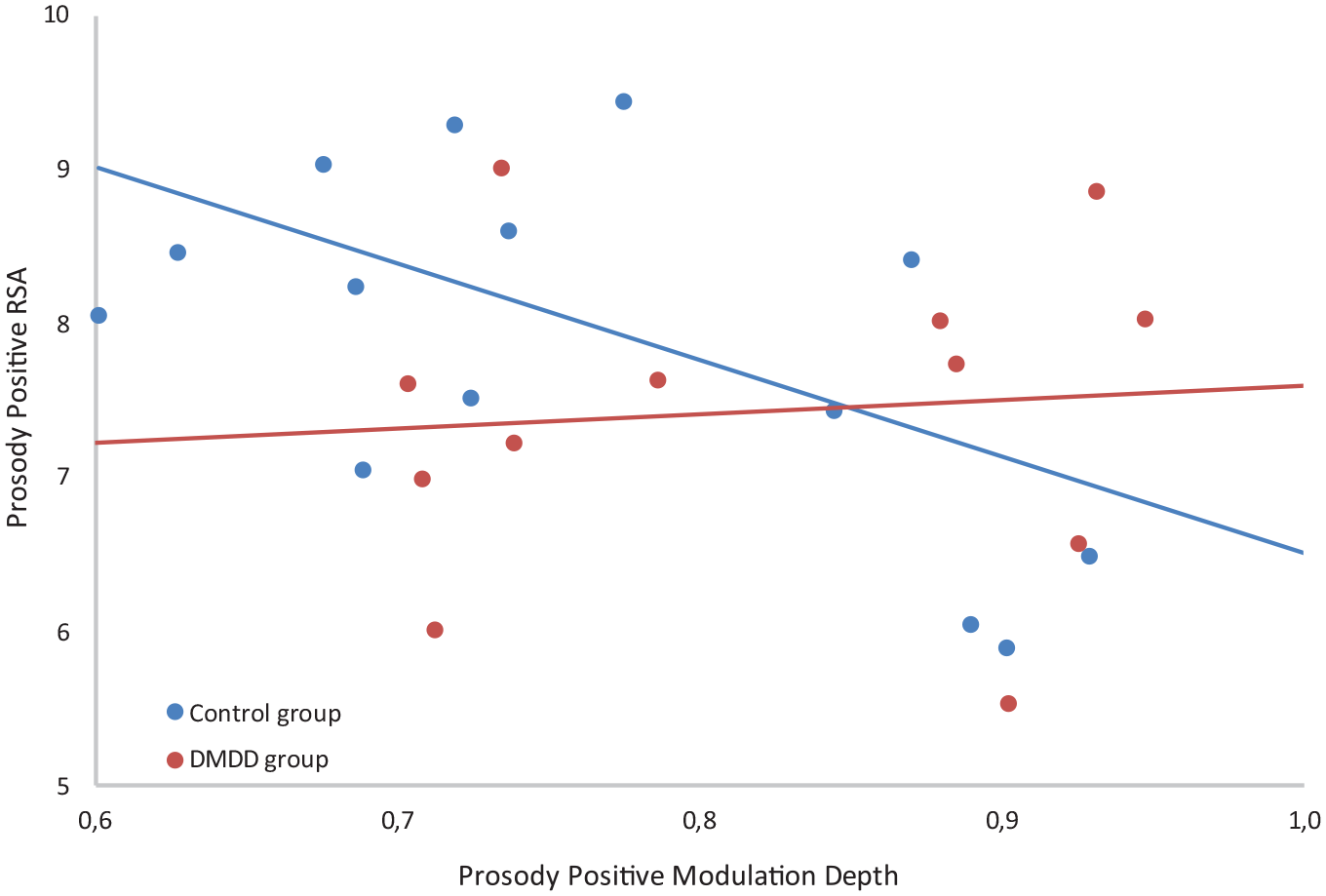
Relationship between modulation depth and RSA during the positive prosody task.
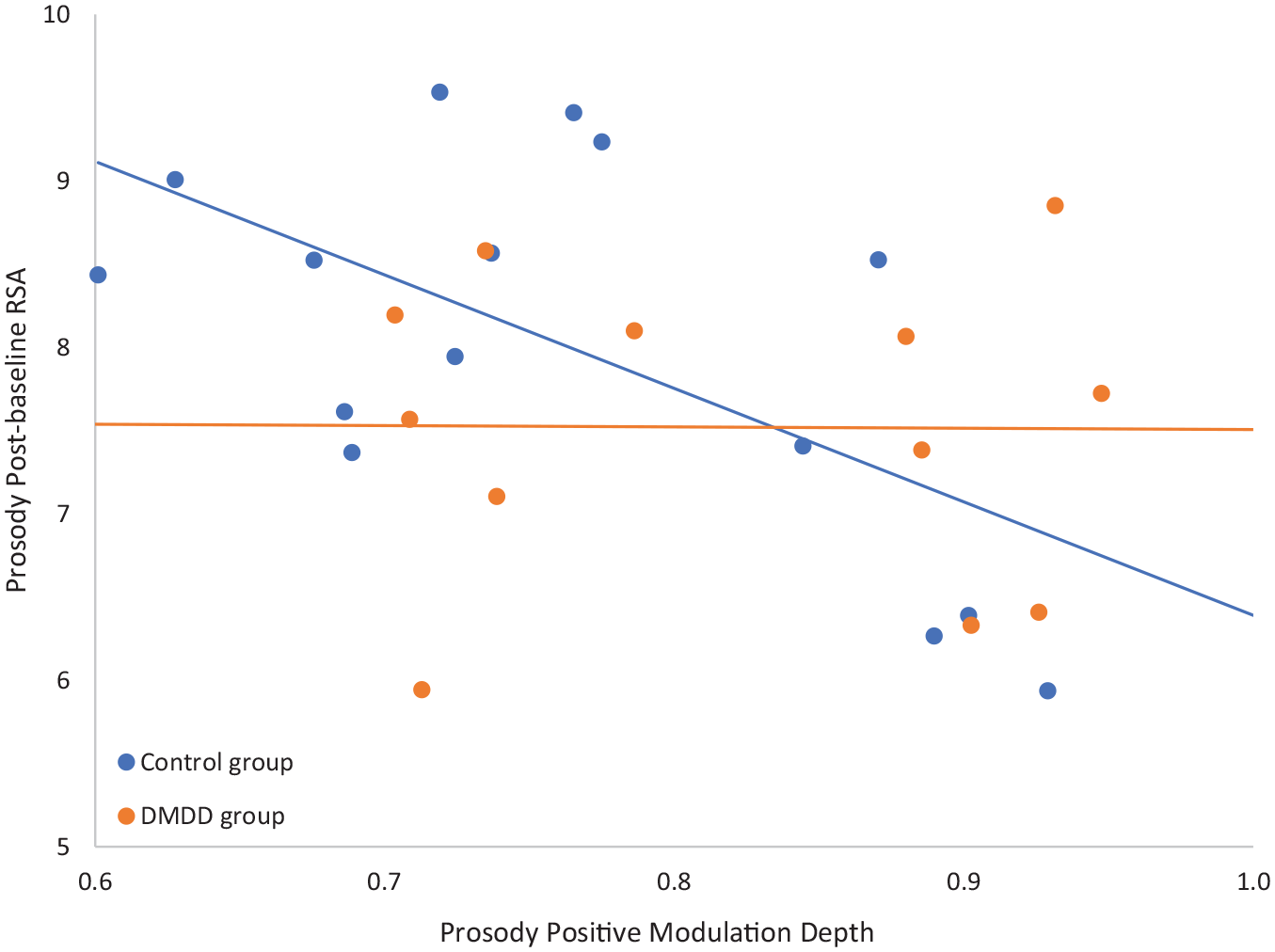
Relationship between modulation depth during the positive prosody condition and post-task respiratory sinus arrythmia (RSA).
Discussion
This study investigated whether atypical regulation of the SES within the Polyvagal Theory (Porges, 2001) is related to DMDD. This was operationalised by exploring the relationship between HRV (RSA and HP), emotion recognition, and expressive prosody in children with DMDD. To our knowledge this is the first study to explore these associations.
Unexpectedly, participants in the DMDD group did not have atypical autonomic regulation before, or during, any of the tasks compared to controls. Research on the link between RSA and emotional and behavioural dysregulation is equivocal (e.g., Beauchaine, 2015). This could be due to methodological heterogeneity across studies, inconsistent control of several transient and stable variables that affect RSA (e.g., weight, height, and waist-hip ratio), and quantification of RSA (e.g., domain vs frequency domain assessments; Beauchaine & Bell, 2020). Some of these variables could have influenced the results of our study, for example, cardioactive medication in the DMDD group, which would have medically regulated HRV. Although the control group had significantly faster heart rate after the DARE, shorter HP in the absence of significantly lower RSA amplitude does not necessarily implicate the vagus nerve in HRV (Porges, 2007) and so cannot be considered theoretically meaningful in terms of affect recognition.
The hypothesis that children with DMDD would have impaired emotion recognition abilities relative to controls was partially supported. While the groups’ response times in recognising facial emotions were similar, the DMDD group was significantly less accurate at recognising fear than the control group. The former also confused other negatively valenced emotions, such as anger and disgust, more than the control group. Such difficulties were anticipated in this group, as children with SMD have difficulty with correctly identifying emotions in facial expressions (Guyer et al., 2007). This aligns with the well-replicated finding that individuals who exhibit inappropriate social behaviour have problems in facial emotion recognition (Hubble et al., 2015). The finding suggests that children with DMDD either fail to perceive relevant emotional cues due to attentional difficulties or a physiological state that does not support the reception of social cues. It is also possible that they misinterpret these cues, particularly negative ones, which may result in a continuation of their disruptive or inappropriate behaviour.
In addition to the facial expression of emotions, we explored the verbal expression of emotion (via prosody). It was anticipated that the DMDD group would have reduced prosody, as low HRV is linked to reduced prosody (Stewart et al., 2013) and is also associated with emotion dysregulation (Beauchaine & Bell, 2020). The results showed that the children with DMDD had several prosody features that differed significantly from those of their peers. The former group displayed significantly increased modulation depth on the negative prosody task which might indicate animation, suggesting an inability to perceive and adapt to social situations appropriately when the demands on social behaviour are changed or unstated (e.g., using a more subdued voice in a formal situation). It is also possible that this strong modulation may indicate vocal instability (such as quavering). As this was not related to shifts in RSA, there may be a disconnect, or inefficiency, between RSA and intonation in children with DMDD. A relationship between RSA and modulation was found in the control group, albeit not as expected. Reduction in RSA is usually paralleled by reductions in intonation variations, yet the opposite occurred in the control group. It is possible that the control group interpreted the prosody task as stressful, and as RSA dropped, they were less able to smoothly regulate their voices. Research in this area is limited and restricted to infants, so the link between RSA and intonation in older children may be more complex, allowing for an optimal balance between arousal and vocal regulation that enables adaptive responding to, and engagement with, the environment (Kolacz et al., 2018). Our findings highlight expressive prosody as an area of potential difference in children with DMDD and a topic that warrants further research.
The study conclusions are limited by the small sample size resulting from difficulties locating children with a DDMD diagnosis. Its inclusion in the DSM-5-TR was met with substantial criticism relating to its reliability, stability, comorbidity, and the potential for pathologising normal childhood behaviour, which could have affected diagnoses locally (Bruno et al., 2019). Underscoring concerns regarding comorbidity, most children with DMDD have at least one comorbid disorder (Mürner-Lavanchy et al., 2021), as was the case in the current study, which complicates the interpretation of findings. As mentioned earlier, ADHD was most common, and future research should investigate how this combination (ADHD + DMDD) affect functioning. Alternatively, comparing children with only ADHD to those with ADHD and DMDD could provide further insight into the differences in SES regulation between these groups (e.g., Özyurt, 2023). Other limitations include an inability to measure all potential confounding variables that influence HRV (e.g., cardioactive medication 1 in the DMDD group), a lack of ethnic diversity in the DARE stimuli, and a predominantly male sample (the disorder is more prevalent in males; Spoelma, 2023).
Conclusion
While there were no marked physiological differences between the DDMD children and controls, they did differ significantly on some of the behavioural measures, particularly prosody features and negative emotion recognition, which could impact effective social engagement. These findings could inform targeted psychological intervention strategies for children with DMDD, such as facial emotion recognition training to improve social interactions (Hubble et al., 2015) and HRV biofeedback, which may improve SES regulation and could have downstream effects on emotion regulation and well-being (Dormal et al., 2021).
Footnotes
Acknowledgements
LifeMax (Pty) Ltd, South Africa, provided the Actiwave Cardio wireless single-channel ECG waveform recorder for data collection. We thank the participants who participated in the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The financial assistance of the National Research Foundation (NRF) towards this research is hereby acknowledged. Opinions expressed and conclusions arrived at are those of the authors and are not necessarily to be attributed to the NRF. This work is based on the research supported by the National Institute for the Humanities and Social Sciences (NIHSS).
