Abstract
Acute diarrhoeal illness remains a common medical problem in children with nearly 1.7 billion cases globally every year. We report five infants who, following severe diarrhoea, developed methaemoglobinemia. This is an altered state of haemoglobin presenting with cyanosis and can pose a diagnostic dilemma. It should be suspected in young infants without cyanotic heart disease presenting with severe diarrhoea, sepsis and cyanosis disproportionate to their clinical status. Its outcome depends on prompt treatment, the severity of underlying sepsis and co-morbidity.
Introduction
Methaemoglobinemia may be congenital or acquired.1,2 The latter may be due to endogenous conditions, such as diarrhoea, sepsis and acidosis or exogenous, due to drugs or toxins.2–5 An association of diarrhoea with methaemoglobinemia can produce dangerously high methaemoglobin levels especially in young infants <6 months old.2–5
Case series
Over 13 months (December 2021 to January 2023), five infants were admitted with diarrhoea, vomiting and severe dehydration. All were from different suburbs of our city. There was no history of consanguinity in any infant. There was no history of exposure to drugs prior to presentation. Except for infants 4 and 5 who were exclusively breastfed, the remainder fed on diluted cow milk. Their demographic profile and relevant clinical and laboratory characteristics are shown in Table 1. On admission, all infants presented with lethargy tachypnoea, tachycardia, central cyanosis, acidotic breathing, severe dehydration and shock. No heart murmurs were found. Normal air entry and clear breath sounds were found at auscultation in all.
Clinical presentation and laboratory investigations.
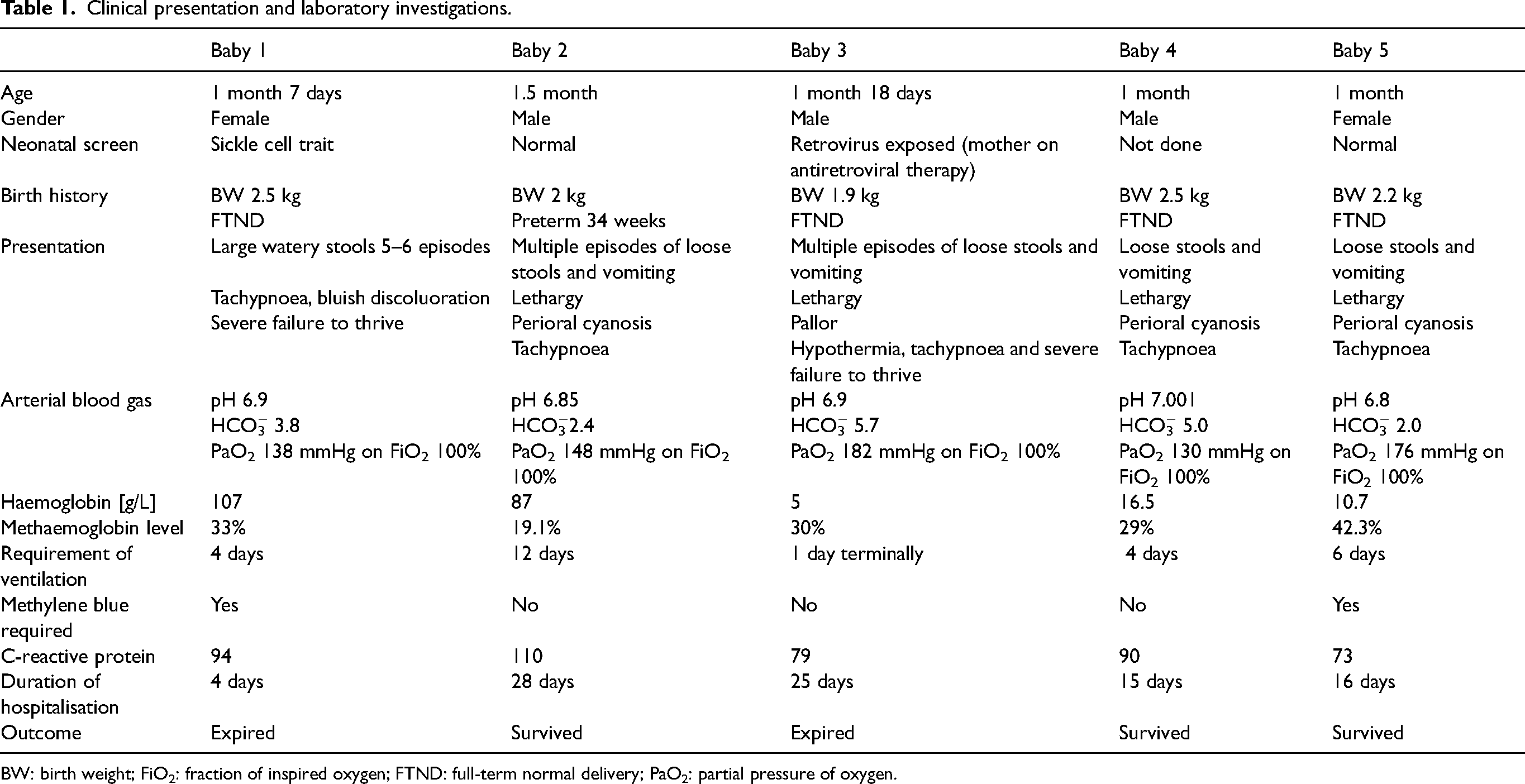
BW: birth weight; FiO2: fraction of inspired oxygen; FTND: full-term normal delivery; PaO2: partial pressure of oxygen.
After initial resuscitation, oxygen saturation remained below 90%, and so all infants were mechanically ventilated. Saturation did not, however, improve even with 100% FiO2. Arterial blood gas analysis produced blood was darker than normal, immediately suggesting methaemoglobinemia. This was confirmed on co-oximetry. There was a leucocytosis in all infants, but glucose-6-phosphate dehydrogenase, blood and urine culture were normal in all. The remaining investigations are shown in Table 1. All infants received intravenous fluids for severe dehydration, antibiotics (intravenous ceftriaxone and amikacin), inotropes and other supportive care as indicated. Infants 2, 3 and 4 showed a spontaneous gradual decline in their methaemoglobin levels within 24 h with fluid resuscitation alone. Infants 1 and 5 had persistent elevated levels hence received methylene blue, following which a decline in methaemoglobin levels was noted within 72 h. However, owing to disease severity, baby 1 died and because of an immune-compromised state and fungal sepsis, baby 3 succumbed. No workup for congenital methaemoglobinemia was performed owing to financial constraints.
Discussion
Clinicians are usually aware of the common complications of diarrhoea, viz. dehydration, oliguria and electrolyte imbalance. Although rare, transient methaemoglobinemia can occur, being first reported in 1954. 6 Methaemoglobin is an altered state of haemoglobin in which the ferrous iron of haeme is oxidised to the ferric state. This cannot bind oxygen, causing impaired tissue delivery and cyanosis. The normal methaemoglobin level is <1% and is reduced primarily by cytochrome b5 and nicotinamide adenine dinucleotide phosphate cytochrome b5 reductase. 1 Other mechanisms of reduction are glutathione, superoxide dismutase and glutathione peroxidase, which protect the body from oxidative stress. In infectious diarrhoea, gram-negative bacteria (viz. Enteropathic Escherichia coli, Shigella, Salmonella and Campylobacter) cause gut inflammation, and altered bacterial flora convert nitrates to nitrites, which oxidises haemoglobin. 4
Other predisposing factors are acidosis, failure to thrive, urinary tract infection, formula feeding and cow milk intolerance. 3 Staphylococcus aureus sepsis is another cause. Young infants <4 months are particularly vulnerable because of immature enzyme systems with low levels of cytochrome b reductase and higher levels of foetal haemoglobin. 7
Clinically, cyanosis occurs when methaemoglobin levels exceed 10%. Manifestations vary with rising levels: 10–30% cyanosis; 30–50% tachypnoea, headache and irritability; 50–70% stupor, acidosis and seizures; and >70% coma and death. 3 Healthy infants may have few symptoms when methaemoglobin levels are <10%. However, patients with cardiorespiratory co-morbidity may experience moderate-to-severe symptoms at much lower levels (5–8%), 3 and result in poor response to treatment. Important diagnostic clues include cyanosis not improving with administration of supplemental oxygen, blood that appears darker than normal, low haemoglobin oxygen saturation in pulse oximetry and a normal blood gas PaO2.3,4,7,8
Intravenous methylene blue is the first-line antidote.3,4,7,8 After initial stabilisation with fluids and antibiotics, the need for methylene blue should be guided by the symptoms and the decline in methaemoglobin levels. 9 Patients with mild asymptomatic methaemoglobinemia without any co-morbidity should simply be observed and treated symptomatically to avoid complications, especially where there is glucose-6-phosphate dehydrogenase (G6PD) deficiency. 9 The recommended intravenous dose of methylene blue is 1 to 2 mg/kg as an infusion over 5 to 10 min. The total dose should not exceed 7 mg/kg, as methylene blue acts as an oxidant at higher doses and can induce haemolysis, especially in children with G6PD deficieny. Methylene blue helps to accelerate the enzymatic reduction of methaemoglobinemia by nicotinamide adenine dinucleotide phosphate methaemoglobin reductase and reduces leuco-methylene blue formation resulting in non-enzymatic reduction. Hyperbaric oxygen exchange transfusion and vitamin C supplementation may be used in patients resistant or where methylene blue is contra-indicated.3,8
Footnotes
Acknowledgement
We thank Dr Mohan Joshi, dean of our institution for permitting us to publish this manuscript, and Dr Radha Ghildiyal, professor and head of Department of Paediatrics for her guidance.
Author contribution
SM conceived and designed the study, collected data, analysed the study, and prepared and edited the manuscript. JV helped in the management of patients and collection and compilation of the data. SAZ edited the article and revised it critically. PP helped in the management of patients and collection of the data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
