Abstract
Multidrug-resistant organisms (MDROs) represent a growing global threat due to resistance to multiple antibiotics, limited treatment options, longer hospitalizations, higher costs, and increased mortality. Although the clinical impact of MDROs is well recognized, their effect on patients’ functional recovery remains understudied. This study aimed to determine the prevalence of MDRO infections across hospital settings and examine their association with changes in functional status. A retrospective cohort study analyzed 30 428 adult hospitalizations from 2020 to 2023 in a 294-bed hospital in northern Italy. Demographic data, hospitalization characteristics, MDRO status, and Barthel Index scores at admission and discharge were collected. A multilevel linear regression model examined the effect of MDRO infection on Barthel variation, adjusting for age, sex, discharge area, and length of stay. MDRO infections occurred in 2.4% of hospitalizations (n = 732), mainly in medical and surgical wards, with the highest prevalence in medical and rehabilitation areas. The most common pathogens were ESBL-producing Enterobacteriaceae and vancomycin-resistant enterococci. Patients without MDRO infection showed greater improvement in Barthel scores than those with infection (+3.081; P < .001). Functional improvements were highest in rehabilitation and neurology, whereas medical and surgical wards showed declines. Each additional hospital day was associated with a modest increase in functional independence. Age and sex were not significantly related to functional change. MDRO infections are independently associated with reduced functional recovery, underscoring their clinical and rehabilitative burden and highlighting the need for integrated infection control and early rehabilitation strategies.
Keywords
Introduction
Multidrug-resistant organisms (MDROs) represent a significant and growing challenge in global public health due to their role in antibiotic resistance.1,2 MDROs exhibit a high capacity to withstand multiple classes of antibiotics and antimicrobial agents, severely limiting therapeutic options.1,3,4 Evidence shows that MDROs could highly impact patients’ outcomes, leading to prolonged hospital stays, increased healthcare costs, and higher mortality rates. 5 In Italy alone, up to 1 in every 205 people could lose a year of a healthy life due to antimicrobial resistance. 6
Prevalence of MDRO infections is steadily increasing, despite varying over time, across regions, and among healthcare settings, posing an escalating global threat.3 -5 In the United States, 5 more than 2 million people developed an MDRO-related infection each year. Furthermore, it was estimated that approximately 23 000 deaths per year were directly attributable to this type of infection. Worldwide, the number of deaths attributable to MDRO infections was estimated to be around 5 million in 2019 and is expected to reach approximately 10 million by 2050. 1 In addition, HAIs (Hospital-Acquired Infections) are also on the rise. 7 In the European Economic Area, 4.3 million patients in hospitals are affected by a HAI each year. 8 In Europe, 37 000 subjects die either directly from HAIs or as a result of increasing MDRO prevalence. 9
MDRO infections significantly increase mortality, hospital readmission rates, and length of stay, while worsening health outcomes in vulnerable patients.4,5,10,11 For example, in neurological patients, HAIs worsen the prognosis by increasing mortality and extending hospital stays. In acute stroke patients, HAIs reduce the likelihood of returning home and increase the need for intensive rehabilitation. 9 In addition, one of the most significant consequences of prolonged hospitalization in older adults is the increased risk of functional decline and long-term disability. This condition, known as hospital-associated disability, is characterized by the loss of independence in activities of daily living following hospitalization. 12 Functional decline and loss of independence require posthospitalization care, including protected discharge planning, integrated home care services, or transfer to specialized facilities. Furthermore, research indicates that extended hospital stays themselves predispose patients to the development of MDRO infections.9,11,13 This cyclical relationship further underscores the complexity of challenges posed by these infections, exacerbating the economic and social burden associated with MDRO infections.3 -5
Despite growing evidence on the adverse effects of MDRO infections, only 1 study has specifically examined their association with patient functional decline. Bartolo et al 9 investigated patients with severe acquired brain injury undergoing intensive neurorehabilitation and found that infections, often due to MDROs (55.5%), were significantly associated with reduced functional gains across multiple validated scales (Glasgow Coma Scale, Rancho Los Amigos Scale, Disability Rating Scale, and modified Barthel Index). However, this study focused on a specific population, highlighting the need to explore MDRO impact on functional outcomes across broader populations and settings.
Therefore, the present study aims to investigate the prevalence of MDROs across different hospital settings and examine their association with patient functional independence. This dual focus addresses a significant gap in the existing literature, as no prior studies have directly explored the relationship between MDRO prevalence and patients’ functional independence in hospital environments. Despite the growing concern about antibiotic resistance and its impact on patient care, the potential link between MDRO infections and changes in patient functional outcomes during hospitalization remains unexplored. By first establishing the prevalence of MDROs and then assessing its relationship with patient autonomy, this research seeks to provide a comprehensive understanding of this critical issue. The findings could inform improvements in hospitalization management, discharge planning, and the development of proactive, targeted interventions.
Material and Methods
Study Design and Setting
We conducted a retrospective single-center cohort study, reported in accordance with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines 14 (Appendix 1). This study took place over a 4-year period, from January 1, 2020, to December 31, 2023, at a 294-bed hospital located in a mountain district in northern Italy. The center provides essential medical and surgical care to a community of 20 625 inhabitants.
Participants
All consecutive hospitalizations between January 1, 2020, and December 31, 2023, were screened for eligibility. Inclusion criteria were: (a) age ≥ 18 years; (b) complete Barthel Index assessment at both admission and discharge. Exclusion criteria were: (a) pediatric population (age < 18 years), due to distinct clinical characteristics requiring separate analysis; (b) obstetric admissions for childbirth only, as these differ substantially from general medical-surgical hospitalizations and may introduce systematic bias; (c) incomplete Barthel Index data at admission or discharge, preventing calculation of functional change; (d) in-hospital death, as discharge functional status could not be assessed. This study included all hospitalizations meeting the inclusion criteria during the 4-year study period. No a priori sample size calculation was performed, as this was an exploratory observational study analyzing the complete available dataset rather than a hypothesis-testing study with predetermined effect size targets.
Variables
The following variables were extracted as part of routine data collection across the hospital settings: patient characteristics (ie, age, gender); length of stay (days); presence of MDRO infections (yes/no); type of infection (bacterial or fungal); microorganism type and gram classification; discharge areas, classified according the distribution of patients across the wards (critical care, surgical, neurological, rehabilitation and medical areas); and Barthel Index at admission and discharge.
For the purpose of this study, length of stay was included as a covariate for the differential care exposure and hospitalization experience across varying durations, and as a proxy for clinical severity in the absence of standardized severity scores.
In this study, MDRO infection was operationally defined as the detection of a multidrug-resistant organism from a clinical diagnostic specimen (eg, blood, urine, respiratory, wound, or other sterile sites) obtained as part of routine medical care for suspected infection. All cases met clinical criteria for infection (eg, fever, elevated inflammatory markers, clinical signs and symptoms) and required antimicrobial treatment. MDRO infections were detected using the Allplex™ Entero-DR Assay (Seegene, Seoul, Republic of Korea), a multiplex real-time PCR platform that simultaneously identifies 8 genes associated with carbapenem resistance, vancomycin resistance, and extended-spectrum β-lactamases. The assay provides qualitative results (positive/negative) for each resistance gene based on real-time PCR amplification. Each hospitalization was classified as MDRO-positive if at least 1 resistant organism was detected during that admission. MDRO categories included ESBL-producing Enterobacteriaceae, vancomycin-resistant Enterococcus (VRE), meticillin-resistant Staphylococcus aureus (MRSA), carbapenem-resistant Enterobacteriaceae (CRE), third-generation cephalosporin-resistant Enterobacteriaceae, non-fermenting Gram-negative bacilli (eg, Pseudomonas, Burkholderia, Stenotrophomonas), toxin-producing Clostridioides difficile, and Aspergillus in immunocompromised patients. Each hospitalization was classified once as MDRO-positive and attributed to a single predominant pathogen category based on clinical significance or the first organism detected.
The Barthel Index 15 is a widely adopted questionnaire to assess functional independence in the activities of daily living. It evaluates 10 essential activities, assigning scores ranging from 0 (complete dependence) to 100 (full autonomy). In the context of this study, nurses routinely administer the Italian validated version of the Barthel Index 16 as part of standard patient care at both admission and discharge, recording the scores in each patient’s medical record.
Data Sources
All patient data were recorded in the Datawarehouse RW10 management system, the hospital’s dedicated software for activity reporting and data analysis. The patients’ electronic records were examined by the center’s data manager, and the aforementioned variables were extracted into a custom-built database. The hospital’s data manager sent an Excel® file to the first author, an experienced nurse and master’s student. For privacy reasons, the file was anonymized by the hospital’s data manager.
Data Analysis
Data are presented as median (IQR, interquartile range) or proportion (%) according to the variable distribution. Multilevel linear regression models were used to examine the dependency of Barthel Index variation (as discharge score minus admission score). Coefficients (β) for each predictor variable and confidence intervals (CI) were reported for each predictor. The model included acquired MDRO (yes/no), age, sex, discharge area, and length of hospital stay as covariates. A random effect for the patient was included to account for multiple admissions. All analyses were conducted using R Core Team 2022.
Ethical Statement
The study was conducted in compliance with the principles of the Declaration of Helsinki. Our practices concerning data collection, processing, and storage adhere to the General Data Protection Regulation (GDPR Regulation [EU] 2016/679) core principles of legality, fairness, transparency, purpose limitation, data minimization, accuracy, storage limitation, integrity, and confidentiality. Participant privacy was rigorously protected. Due to the study’s retrospective design, informed consent from participants was not required. The ethical committee of the Veneto region (Italy) approved the research project (Prot. 0011472/25, internal code CET ANV 202-27).
Results
Characteristics of Hospitalizations
A total of 45 718 hospitalizations were recorded, of which 30 428 hospitalizations, accounting for 21 844 patients, met the inclusion criteria and were included in the study (Figure 1). Among these, 14 152 hospitalizations involved patients with multiple admissions in the study period, while 16 276 hospitalizations involved patients with a single admission. Consequently, multiple admissions by the same individual within the study period were counted separately.
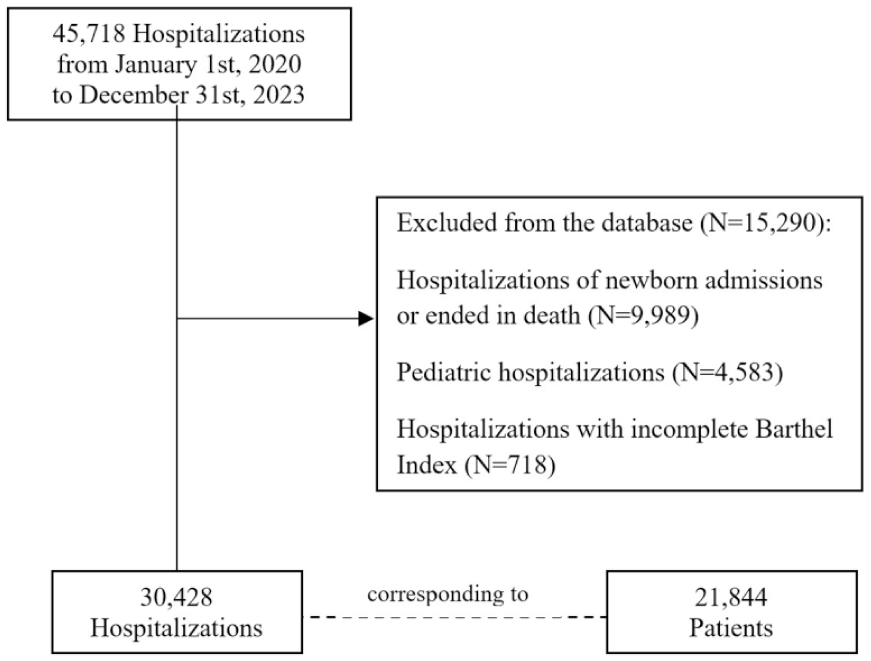
Flowchart of the study sample selection.
Regarding patient characteristics for the 30 428 hospitalizations, the median age was 69 years (IQR: 51-81; Table 1), and the majority were male (22 293, 73%).
Characteristics of Hospitalizations.
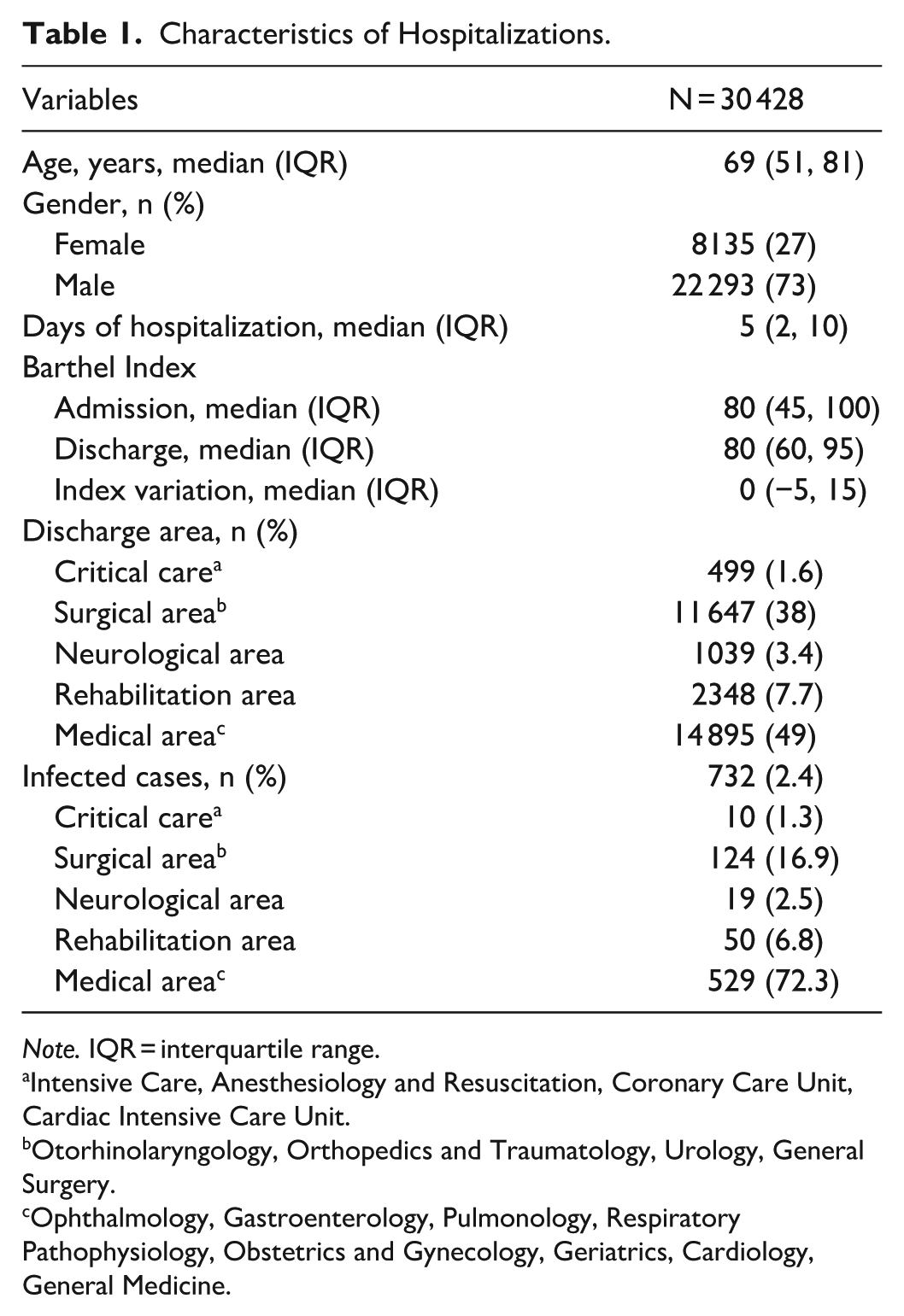
Note. IQR = interquartile range.
Intensive Care, Anesthesiology and Resuscitation, Coronary Care Unit, Cardiac Intensive Care Unit.
Otorhinolaryngology, Orthopedics and Traumatology, Urology, General Surgery.
Ophthalmology, Gastroenterology, Pulmonology, Respiratory Pathophysiology, Obstetrics and Gynecology, Geriatrics, Cardiology, General Medicine.
Most patients were discharged from the medical area (49%), followed by the surgical area (38%), with fewer discharges from the rehabilitation area (7.7%), neurological (3.4%), and critical care (1.6%) areas. The median length of hospital stay was 5 days (IQR 2-10). The median variation in the Barthel Index during hospitalization was 0 (IQR −5 to 15).
Among the 30 428 hospitalizations, 2.4% of cases (n = 732) were classified as MDRO infections.
Characteristics of MDRO Infection
Out of 732 infected cases, the majority occurred in the medical area (529 cases, 72.3%) and in the surgical area (124, 16.9%; Table 2). The majority of MDRO infections were bacterial, accounting for 99.7% of all cases (730/732), with fungal infections representing 0.3% (2/732). Among bacterial infections, Gram-negative organisms predominated (402 cases, 54.9%), while Gram-positive organisms accounted for 328 cases (44.8%).
Characteristics of MDRO Infection Cases.

Among the identified infections, extended-spectrum β-lactamase (ESBL)-producing Enterobacteriaceae were the most prevalent (249, 34.0%), followed by vancomycin-resistant Enterococcus faecalis and Enterococcus faecium (148, 20.2%), meticillin-resistant Staphylococcus aureus (137, 18.7%), and Enterobacteriaceae resistant to third-generation cephalosporins (125 cases, 17.1%).
Impact of MDRO Infection on the Barthel Index
The presence of an MDRO infection was associated with worse functional independence (Table 3). Patients without infection experienced a greater improvement in the Barthel Index compared to infected patients (β coefficient = 3.081; P < .001), translating to a 2.81% increase in variation of functional independence.
Log-linear Regression on Barthel Index Variation: Absolute (Barthel Points) and Relative (%) Effects on Barthel Change.

Note. Absolute effects are from the linear mixed model with untransformed Barthel change; relative effects are from the log-model back-transformation [(exp(β) − 1) × 100]. Same covariate structure in both models. Analytic sample: n = 30 428 complete cases.
CI = confidence interval.
Regarding the other variables in the model, age was not significantly associated with Barthel Index variation (β coefficient = .004; P < .570). Similarly, being male showed a non-significant decrease compared to females (β coefficient = .046; P < .877).
Length of stay showed a small but statistically significant positive association with functional improvement, with each additional day of hospitalization corresponding to a 0.20% increase in Barthel Index (β coefficient = .282; P < .001).
Discharge destination was significantly associated with variation in Barthel Index scores. Compared to patients discharged from critical care, those discharged from the rehabilitation area showed the greatest functional improvement (β coefficient = 6.859; P < .001), followed by those from the neurological area (β coefficient = 4.843; P < .001). In contrast, discharges from surgical (β coefficient = −16.600; P < .001) and medical areas (β coefficient = −7.249; P < .001) was associated with reduced functional recovery, corresponding to mean decreases of −13.31% and −4.40% in Barthel Index variation, respectively.
Discussion
This study investigated the occurrence of MDRO infections and their association with patient functional independence at discharge, as measured by the Barthel Index, across clinical settings in a small mountain hospital. Variation in functional recovery varied significantly depending on the discharge destination, likely reflecting clinical and organizational factors that influence both discharge planning and rehabilitation potential. MDRO infections were present in 2.4% of hospitalizations, predominantly among medical wards.
The prevalence rate in our study is comparatively lower than in several recent international studies. Cornistein et al 17 documented a prevalence of 15.1% in adult intensive care units across Argentina, while Alsaab et al 18 reported a rate of 37.3% among hospitalized patients in Saudi Arabia. Similarly, Patel et al, 19 in a longitudinal study at an Indian tertiary care hospital, recorded an increasing MDRO incidence density rate, rising from 4.2 to 8.77 per 1000 patient-days over a 10-year period. However, our study was conducted in a spoke mountain hospital with limited intensive care capacity and specialized surgical services—settings typically associated with higher antimicrobial selection pressures and greater patient acuity that may contribute to increased MDRO prevalence. In addition, most studies were conducted outside the European region, where local epidemiology, antibiotic stewardship programs, and infection control infrastructure may differ substantially, thereby affecting both the burden and detection of MDROs. Nevertheless, our findings align with those available in suggesting that MDRO infections are not confined to high-acuity wards.
Indeed, the distribution of MDRO infections in our study was highest in medical wards, suggesting a potential area of focus for infection control efforts, followed by surgical and rehabilitation areas. These findings align with Karakosta et al, 20 who reported substantial MDRO prevalence in non-ICU wards. Notably, the presence of MDROs in rehabilitation units in our study suggests that even sub-acute or recovery-focused settings are not exempt from colonization and transmission risks, potentially due to patient transfers between wards and less stringent infection control practices. Moreover, our findings may contribute to a better understanding of how MDRO distribution patterns differ between local spoke hospitals and highly specialized hub centers, emphasizing the need for infection control protocols tailored to local contexts.
Regarding pathogen distribution, the microbiological profile in our cohort was dominated by ESBL-producing Enterobacteriaceae [34.0%], followed by vancomycin-resistant Enterococcus faecalis and faecium [20.2%], and meticillin-resistant Staphylococcus aureus [18.7%]. A comparison of these data with existing studies reveals both differences and similarities. For example, Patel et al 19 identified Carbapenem-Resistant Enterobacteriaceae as the most common MDRO in a tertiary care teaching hospital in India, a pattern also observed by Alsaab et al 18 across different hospitals in Saudi Arabia. Similarly, Fan et al, 21 in a Chinese hospital-based cohort, reported a broader dominance of Gram-negative organisms in bloodstream infections, while Cornistein et al 17 identified Acinetobacter baumannii as both the most frequent and most lethal in their intensive care-based study in Argentina. Additionally, Gavi et al, 22 studying an Italian urological center, observed high rates of ESBL-producing Escherichia coli and moderate occurrence of Klebsiella pneumoniae, Enterococcus faecalis, and Enterococcus faecium.
Our finding that Gram-negative organisms predominated (54.9%), with ESBL-producing Enterobacteriaceae as the leading pathogen (34.0%), is consistent with several studies showing the growing burden of multidrug-resistant Gram-negative bacteria. However, variations in microbial profiles across studies likely reflects the clinical context, patient complexity, care settings, antibiotic prescribing patterns, and surveillance practices. While Gram-negative organisms were most prevalent in our cohort, the substantial proportion of Gram-positive infections (44.8%), particularly vancomycin-resistant enterococci and meticillin-resistant Staphylococcus aureus, reinforces the continued importance of closely monitoring both Gram-negative and Gram-positive organisms and boosting management strategies, particularly within internal medicine and rehabilitation departments.
Regarding the outcomes of MDRO infections, our study offers novel insights by investigating their association with post-acute functional recovery, a dimension rarely addressed in epidemiological research on antimicrobial resistance. The data revealed that MDRO infection independently and negatively affects patient functionality, with non-infected individuals exhibiting 3.081 points greater improvement in their Barthel scores compared to those with infection. These findings build upon prior studies, extending the documented association between infection and functional decline to a broader inpatient population. 9 While prior studies, such as those by Cornistein et al 17 and Patel et al, 19 focused on mortality and healthcare utilization, our analysis demonstrates that MDRO infections also substantially hinder rehabilitation and functional recovery, even in non-critical care populations. This observation expands the conceptual framework for assessing the burden of MDROs and underscores the need to integrate functional metrics into antimicrobial resistance surveillance and quality assessment programs.
Other noteworthy observations were the independent effect of longer hospital stays and discharge wards on functional independence, while age and sex did not show significant associations. Specifically, each additional inpatient day was linked to a 0.20% increase in Barthel Index score, consistent with previous research suggesting that extended hospital stay may provide greater opportunities for structured rehabilitation. 23 However, it must be balanced against the increased risks of nosocomial infection that accompany prolonged admissions, especially in high-risk units.5,13 In addition, neither age nor sex demonstrated a statistically significant association with functional outcomes, indicating that while demographic factors remain relevant to overall health risk, they may have limited predictive value for rehabilitation success during acute hospitalization. Instead, clinical factors, including the presence of complications such as MDRO infections, appear to exert a more direct influence on functional recovery.
Discharge ward emerged as another critical determinant of functional change. Patients discharged from rehabilitation and neurology wards achieved the greatest substantial improvements (+6.85 and +4.84, respectively), likely reflecting the intensity and specialized focus of therapeutic interventions in these settings. In contrast, patients discharged from surgical (−16.60) and general medical (−7.24) wards experienced declines in functional independence. These findings further underscore the need to consider the interaction between hospitalization ward and MDRO infection as a critical determinant of patients’ functional independence.
Study Limitations
Due to its retrospective nature, this study has several limitations. First, it was conducted in a single-spoke hospital without highly specialized services or a high-complexity intensive care unit, which may limit the generalizability of the findings and introduce selection bias through referral of more critical cases to larger hub centers. Second, patients who died during hospitalization were excluded due to the absence of Barthel Index scores, potentially underrepresenting the most severe cases and underestimating the true impact of MDRO infections. Third, this study period included the COVID-19 pandemic, during which hospital operations, patient management protocols, and infection control measures underwent substantial changes. These pandemic-related factors may have influenced both the prevalence of MDROs and patterns of functional recovery in ways that do not entirely reflect standard care conditions. Fourth, our dataset lacked information on infection timing (admission vs hospital-acquired) and infection site distribution (urinary, respiratory, bloodstream, surgical), preventing more granular analyses. Fifth, the absence of standardized comorbidity indices and clinical severity scores limits our ability to fully control for confounding. Patients with MDRO infections may have been systematically sicker at baseline in ways not captured by available covariates. However, our large sample size (30 428 hospitalizations), multilevel modeling, and consistent findings across settings partially mitigate this concern. Nonetheless, residual confounding remains possible, and findings should be interpreted as associations rather than causal effects. Additionally, including length of stay as a covariate may have introduced collider bias, as it is influenced by both MDRO status and other clinical factors. Sixth, while our outcome measure (Barthel variation) accounts for individual baseline functional status through within-person change, we could not explore whether the association between MDRO infection and functional recovery varies across different baseline functional levels. Finally, limitations in data availability restricted the ability to conduct subgroup analyses by diagnosis or care trajectory. For privacy reasons, only the initial admission and final discharge wards were accessible, precluding insight into ward transitions that could have affected both infection risk and rehabilitation outcomes. Moreover, data collection practices varied across wards with heterogeneous documentation methods (electronic and paper-based), and much clinical information resides in daily notes not systematically extracted into the centralized database. Our analysis was therefore restricted to standardized variables uniformly collected hospital-wide, ensuring consistency but limiting clinical granularity. Lastly, our analysis was limited by the institutional database’s predefined categorization of pathogens, preventing species-specific analyses that could have provided more granular epidemiological insights.
Implications for Research and Practice
The findings of this study carry important implications for clinical governance, infection control, and health system organization, particularly given the observed association between MDRO infections and impaired functional recovery. Although the overall infection prevalence was moderate (2.4%), the highest rates occurred in medical settings. The significantly reduced improvement in Barthel Index scores among infected patients highlights the broader impact of MDROs—extending beyond mortality. This underscores the necessity of incorporating functional metrics into antimicrobial resistance surveillance and patient management strategies.
This evidence suggests that infection prevention efforts should not be limited to intensive care or acute treatment settings but must be systematically extended to post-acute and rehabilitation pathways, especially within less specialized hospitals. In addition, advanced infection management technologies and protocols—such as ultraviolet disinfection, antimicrobial surfaces, targeted decolonization, staff screening, and rapid polymerase chain reaction diagnostics on admission—are often underused in medical and rehabilitation areas.24,25 These settings are perceived as lower risk and, as a result, receive comparatively fewer resources. 25
This calls for the need to reinforce, care models in which infection prevention and functional rehabilitation strategies are not siloed but are complementary priorities. In medical and rehabilitation settings, especially, coordinated care pathways should ensure that patients with MDRO infections receive not only appropriate bundled infection control strategies 25 but also uninterrupted access to rehabilitative interventions, which may otherwise be limited during infection-related isolation. This integration requires fostering the competencies of healthcare professionals within antimicrobial stewardship programs, since they play a pivotal role in medication administration, patient education, and the early identification of adverse effects or resistance patterns.26,27
Furthermore, the heterogeneity of patient presentations—ranging from acute to chronic and from high-functioning to functionally dependent states—calls for more stratified analytical approaches in future studies. The synergic adoption of early MDRO screening protocols 25 and of subgroups most susceptible to functional decline could enable targeted rehabilitation plans and inform the prioritization of infection control resources. In addition, transitions between wards should also be further investigated in terms of impact on both and MDRO infection risk and functional outcomes.
Moreover, traditional infection surveillance systems must also evolve to incorporate broader outcome metrics. Beyond mortality and readmission rates, functional status metrics, such as changes in the Barthel Index, should be considered as core indicators of recovery, particularly in older or comorbid populations where autonomy is a primary therapeutic objective. This functional perspective not only aligns with patient-centered priorities but also enables a more comprehensive evaluation of the consequences of MDRO infections across care settings. In parallel, enhancing the clinical MDRO dataset with clinical variables such as immune biomarkers, nutritional status, and multi-morbidity indices could provide a deeper understanding of how host factors modulate recovery trajectories in the context of microbial resistance.
Lastly, future research should extend beyond hospitalization to incorporate longitudinal follow-up and assess whether infection-related functional impairments persist, resolve, or worsen over time. Understanding how intra-hospital mobility, ward transitions, and structural factors contribute to infection risk and recovery disparities will also be critical in refining institutional policies.
Conclusions
This study shows that infections caused by multidrug-resistant organisms, even when relatively uncommon in a secondary care hospital, are linked to reduced recovery of functional independence at discharge. These infections were most frequent in medical wards and were mainly due to extended-spectrum beta-lactamase-producing Enterobacteriaceae, vancomycin-resistant enterococci, and meticillin-resistant Staphylococcus aureus. The results highlight the importance of strengthening infection prevention measures beyond intensive care units, with particular attention to general medical and post-acute care settings. Functional outcomes should be systematically integrated in monitoring systems. Additionally, early detection protocols, continuous staff training, and coordinated care strategies should be prioritized. Future studies should follow patients after discharge and explore how intra-hospital transfers and differences in care environments influence both infection risk and functional recovery.
Footnotes
Appendix
STROBE Statement—Checklist of Items That Should Be Included in Reports of Observational Studies.

| Item no. | Recommendation | Page | |
|---|---|---|---|
| Title and abstract | 1 | (a) Indicate the study’s design with a commonly used term in the title or the abstract | 1 |
| (b) Provide in the abstract an informative and balanced summary of what was done and what was found | 1 | ||
| Introduction | |||
| Background/rationale | 2 | Explain the scientific background and rationale for the investigation being reported | 2-3 |
| Objectives | 3 | State specific objectives, including any prespecified hypotheses | 3 |
| Methods | |||
| Study design | 4 | Present key elements of study design early in the paper | 3 |
| Setting | 5 | Describe the setting, locations, and relevant dates, including periods of recruitment, exposure, follow-up, and data collection | 3-4 |
| Participants | 6 | (a) Cohort study—Give the eligibility criteria, and the sources and methods of selection of participants. Describe methods of follow-up | 3-4 |
| Case-control study—Give the eligibility criteria, and the sources and methods of case ascertainment and control selection. Give the rationale for the choice of cases and controls | |||
| Cross-sectional study—Give the eligibility criteria, and the sources and methods of selection of participants | |||
| (b) Cohort study—For matched studies, give matching criteria and number of exposed and unexposed | |||
| Case-control study—For matched studies, give matching criteria and the number of controls per case | |||
| Variables | 7 | Clearly define all outcomes, exposures, predictors, potential confounders, and effect modifiers. Give diagnostic criteria, if applicable | 3-4 |
| Data sources/measurement | 8* | For each variable of interest, give sources of data and details of methods of assessment (measurement). Describe comparability of assessment methods if there is more than one group | 4 |
| Bias | 9 | Describe any efforts to address potential sources of bias | 4 |
| Study size | 10 | Explain how the study size was arrived at | 3-4 |
| Quantitative variables | 11 | Explain how quantitative variables were handled in the analyses. If applicable, describe which groupings were chosen and why | 3-4 |
| Statistical methods | 12 | (a) Describe all statistical methods, including those used to control for confounding | 4 |
| (b) Describe any methods used to examine subgroups and interactions | 4 | ||
| (c) Explain how missing data were addressed | |||
| (d) Cohort study—If applicable, explain how loss to follow-up was addressed | |||
| Case-control study—If applicable, explain how matching of cases and controls was addressed | |||
| Cross-sectional study—If applicable, describe analytical methods taking account of sampling strategy | |||
| ( |
|||
| Results | |||
| Participants | 13* | (a) Report numbers of individuals at each stage of study—eg numbers potentially eligible, examined for eligibility, confirmed eligible, included in the study, completing follow-up, and analysed | 5 |
| (b) Give reasons for non-participation at each stage | |||
| (c) Consider use of a flow diagram | Figure 1 | ||
| Descriptive data | 14* | (a) Give characteristics of study participants (eg demographic, clinical, social) and information on exposures and potential confounders | 5 |
| (b) Indicate number of participants with missing data for each variable of interest | |||
| (c) Cohort study—Summarise follow-up time (eg, average and total amount) | |||
| Outcome data | 15* | Cohort study—Report numbers of outcome events or summary measures over time | |
| Case-control study—Report numbers in each exposure category, or summary measures of exposure | |||
| Cross-sectional study—Report numbers of outcome events or summary measures | |||
| Main results | 16 | (a) Give unadjusted estimates and, if applicable, confounder-adjusted estimates and their precision (eg, 95% confidence interval). Make clear which confounders were adjusted for and why they were included | 5-6 |
| (b) Report category boundaries when continuous variables were categorized | |||
| (c) If relevant, consider translating estimates of relative risk into absolute risk for a meaningful time period | |||
| Other analyses | 17 | Report other analyses done—eg, analyses of subgroups and interactions, and sensitivity analyses | |
| Discussion | |||
| Key results | 18 | Summarise key results with reference to study objectives | 6-7-8 |
| Limitations | 19 | Discuss limitations of the study, taking into account sources of potential bias or imprecision. Discuss both direction and magnitude of any potential bias | 8 |
| Interpretation | 20 | Give a cautious overall interpretation of results considering objectives, limitations, multiplicity of analyses, results from similar studies, and other relevant evidence | 8-9 |
| Generalisability | 21 | Discuss the generalisability (external validity) of the study results | 8-9-10 |
| Other information | |||
| Funding | 22 | Give the source of funding and the role of the funders for the present study and, if applicable, for the original study on which the present article is based | Title page |
Acknowledgements
None.
Ethical Considerations
The ethical committee of the Veneto (Italy) region approved the research project (Prot. 0011472/25, internal code CET ANV 202-27).
Consent to Participate
Due to the study’s retrospective design, informed consent from participants was not required.
Consent for Publication
The author consents to the publication of this manuscript in this journal.
Author Contributions
SG: conceptualization, project administration, validation, methodology, and writing–review and editing. LV: conceptualization, project administration, data analysis, investigation, methodology, visualization, validation, and writing–original draft, writing–review and editing. AS: data curation, investigation, writing–review and editing. SM: data curation, investigation, writing–review and editing. JL: Supervision, data analysis, writing–review and editing. MD: conceptualization, project administration, validation, methodology, and writing–review and editing. All the authors have read and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The dataset that supports the findings of this study is available from the corresponding author upon reasonable request.
