Abstract
Mixing pediatric medications with food is a common caregiver strategy to overcome challenges such as poor palatability, swallowing difficulties, and child refusal. This study aimed to assess maternal knowledge, perceptions, and practices concerning the preparation and mixing of pediatric medications with food in Palestine. A cross-sectional study was conducted between February and December 2025 in the West Bank, Palestine. Mothers were recruited from primary healthcare centers, antenatal clinics, and governmental hospitals across Nablus, Jenin, Tulkarem, Qalqilya, and Tubas. Data were collected using a semi-structured questionnaire. A total of 385 mothers participated. The majority of mothers reported not mixing medications with food (n = 267, 69.4%). Among those who did mix, the most common motivations were child refusal (n = 118, 30.6%) and unpleasant taste (n = 69, 17.9%). Urban mothers were significantly less likely to have been counseled about the safety of mixing compared with rural mothers (14.3% vs 16.1%, P = .021), and less likely to perceive mixing as safe (14.0% vs 15.8%, P = .022). Maternal age was positively associated with awareness that milk or juice may reduce drug efficacy (median 34.0 years [IQR 28.0-40.0] vs 32.0 years [IQR 26.0-37.0], P = .035), as was the age of the youngest child (median 4.0 years [IQR 1.5-7.0] vs 2.0 years [IQR 1.0-5.0], P = .021), suggesting that experiential factors influence knowledge acquisition. Nearly all mothers expressed a desire for additional information and educational support (n = 371, 96.4%), underscoring a critical unmet need. This study provides novel evidence on maternal knowledge and practices regarding medication-food mixing in Palestine. Findings highlight geographic disparities and age-related differences in awareness, alongside persistent gaps in counseling. Addressing these through culturally tailored education, strengthened healthcare provider involvement, and policy-level support could improve medication safety, adherence, and child health outcomes.
Keywords
Introduction
Mixing medications with food is a common caregiver strategy to facilitate drug administration in children.1 -4 Pediatric patients often resist medicines due to unpleasant taste, texture, fear, or swallowing difficulties, making administration particularly challenging for mothers, who are typically the primary caregivers.2,5,6 To overcome these barriers, many mothers mix medications with food or beverages to improve palatability and adherence.7 -9 However, this seemingly convenient practice carries important drug safety risks as food-drug interactions can compromise chemical stability, reduce bioavailability, and alter therapeutic efficacy, potentially resulting in treatment failure, toxicity, or dosing errors.10 -12 In some cases, mixing may confer benefits. For example, iron supplements with vitamin C-rich juices can enhance absorption, underscoring the dual potential for harm and benefit inherent in this practice. 10 Maternal knowledge, attitudes, and health literacy are central to safe pediatric medication practices.13,14 Research show that mothers’ knowledge and health literacy significantly predict their practices, with health literacy mediating much of the relationship between awareness and behavior. 13 This indicates that knowledge alone is insufficient. Therefore, broader factors such as confidence, attitudes, and access to reliable information determine whether safe practices are consistently applied.
Despite its clinical importance, maternal knowledge and practices surrounding medication-food mixing remain poorly characterized, particularly in resource-limited healthcare systems such as Palestine.8,14 In these contexts, mothers often administer pediatric medications with minimal counseling from healthcare providers, increasing the likelihood of unsafe practices and preventable adverse outcomes. Clarifying the extent of maternal awareness, the motivations behind mixing, and the contextual factors that shape these behaviors is therefore critical for designing effective educational interventions and safeguarding pediatric drug safety.
Structured counseling from healthcare providers is essential to ensure safe and effective practices in medication-food mixing. In the absence of professional guidance, many mothers rely on informal sources such as family, friends, or social media, which can perpetuate misconceptions and unsafe behaviors.4,8,9 Disseminating accurate information through physicians, pharmacists, and written medication leaflets can empower caregivers to make informed decisions, minimize harmful food-drug interactions, and support adherence to pediatric treatment regimens. Evidence shows that educational interventions, including workshops, printed guidelines, and audiovisual materials, enhance caregiver confidence and promote safer practices.4,5
The consequences of inadequate knowledge are substantial. When mothers are unaware of the risks of mixing medications with food, therapeutic outcomes may be compromised, resulting in treatment failure, adverse drug reactions, or dosing errors.7,8,11,12,14 In contrast, informed caregivers are better equipped to select appropriate food vehicles, avoid harmful interactions, and ensure accurate dosing. Evidence consistently shows that caregiver education improves medication adherence, reduces administration errors, and enhances child health outcomes.1,14
In Palestine, where mothers are the primary caregivers and often administer medications with minimal counseling, evidence on maternal awareness, motivations for mixing, and the sociodemographic determinants of these practices remains scarce. This gap limits the ability of healthcare providers and policymakers to design targeted interventions that address knowledge deficits, promote safe practices, and reduce preventable adverse outcomes in pediatric drug administration. Against this background, the present study aimed to comprehensively assess the knowledge, perceptions, and practices of Palestinian mothers regarding the mixing of pediatric medications with food, and to identify the sociodemographic, educational, and contextual factors associated with these behaviors. Specifically, the study evaluated maternal knowledge of food-drug interactions and safety guidelines, examined actual practices including motivations, food selection criteria, preparation methods, and reported adverse effects, and explored associations with key maternal characteristics such as age, education, occupation, socioeconomic status, area of residence, and sources of information. In addition, the study sought to identify educational needs and information sources that influence maternal confidence in safe medication administration, thereby providing evidence to guide future counseling strategies and policy initiatives.
Methods
Study Design and Settings
This study was conducted in adherence to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. Adherence to the STROBE checklist is shown in Supplemental Table S1. We employed a cross-sectional design to investigate maternal knowledge, perceptions, and practices regarding the preparation and mixing of pediatric medications with food. Data collection was carried out between February 2025 and December 2025 in the West Bank, Palestine.
The study was conducted across multiple healthcare settings to ensure representativeness of the target population. These included primary healthcare centers, particularly during vaccination days when mothers commonly bring their children for immunization; antenatal clinics; and governmental hospitals with pediatric departments. Recruitment sites spanned 5 major cities in the northern West Bank, Nablus, Jenin, Tulkarem, Qalqilya, and Tubas, as well as surrounding villages and refugee camps. Primary healthcare centers were selected as key venues because they serve as the first point of contact for pediatric care and provide access to a large proportion of mothers responsible for medication administration. Governmental hospitals were included to capture mothers of children with more complex medical needs, thereby broadening the scope of the study population. By integrating diverse healthcare settings and geographic areas, the study design ensured inclusion of mothers from varied sociodemographic backgrounds, thereby enhancing the generalizability of findings to the wider Palestinian population.
Study Population, Inclusion and Exclusion Criteria
The study population comprised Palestinian mothers residing in the West Bank who were directly responsible for administering medications to their children. Inclusion criteria were defined as mothers aged 18 years or older, serving as the primary caregiver for at least 1 child, and currently responsible for administering oral medications (liquid or solid dosage forms) to their child for any medical condition. Mothers were required to provide informed consent and to be able to complete a self-administered questionnaire.
Exclusion criteria included fathers, siblings, or non-biological caregivers; mothers younger than 18 years; mothers with significant cognitive impairments or illiteracy that would preclude completion of the questionnaire; and mothers of children who were fed exclusively via feeding tubes. This approach ensured that the study population represented mothers actively engaged in pediatric medication administration across diverse sociodemographic and healthcare settings, thereby enhancing the external validity and generalizability of the findings.
Sample Size and Sampling Technique
The sample size was calculated using an online Raosoft sample size calculator (www.raosoft.com). Assuming a population size of approximately 20 000 mothers in the northern West Bank with children receiving medications, a 95% confidence level, a 5% margin of error, and a 50% response distribution, the minimum required sample size was estimated to be 377 participants. The population size was conservatively set at 20 000 to approximate the number of mothers in the northern West Bank administering pediatric medications, given the absence of precise figures. A response distribution of 50% was selected following Raosoft recommendations, as this value maximizes the required sample size by assuming the highest variability. These choices ensured a sufficiently large and statistically robust sample size to capture diverse maternal knowledge and practices.
To achieve this target, we employed a convenience sampling method, recruiting mothers who met the inclusion criteria during routine visits to primary healthcare centers, antenatal clinics, and governmental hospitals with pediatric departments. This approach was chosen to maximize feasibility and ensure access to a representative cross-section of mothers actively engaged in pediatric medication administration.
Research Tools, Validity, Reliability, and Variables
Development of the Questionnaire
Data collection was based on a semi-structured questionnaire that was developed after a thorough review of the relevant literature on pediatric medication administration and food-drug mixing.1,2,7 -9 The instrument was adapted from previously validated questionnaires used in France, the Netherlands, the UK, Iran, and Palestine, and was carefully tailored to the Palestinian context to ensure cultural appropriateness and clarity for respondents.
Structure of the Questionnaire and Data Collection
The questionnaire consisted of 4 main components. The first part collected sociodemographic and household information, including maternal age, number of children, age of the youngest child, marital status, area of residence, perceived economic status, educational attainment, and employment status. The second part focused on child health and medication profile, asking mothers about the presence of chronic illness in their child, difficulty swallowing medications, and concurrent use of multiple medications. The third part assessed maternal knowledge of medication-food mixing, including awareness of guidelines or best practices, counseling received about safety of mixing medications with food, perception of safety, awareness that certain medications should not be crushed or opened, and awareness that milk or juice may reduce drug efficacy. Mothers were also asked about factors they believed influenced drug efficacy when mixed with food, such as type of food, temperature, timing of meals, dosage form, or uncertainty. The fourth part examined maternal practices in medication preparation and mixing, including reasons for not mixing, and motivations for mixing. Additional questions explored criteria for food selection, preparation methods, types of medications mixed, and types of food used. Finally, the questionnaire included items on information sources and educational needs, asking mothers about their reliance on physicians, pharmacists, medication leaflets, family or friends, internet, and social media. It also assessed frequency of reading instructions prior to administration, desire for additional information, factors that increase confidence, and willingness to attend workshops. The questionnaire is provided in Supplemental Table S2.
In this study, maternal knowledge was assessed through items that captured awareness of general guidelines/recommendations, counseling received, perception of safety, recognition that certain medications should not be crushed or opened, and awareness that milk or juice may reduce drug efficacy. Knowledge was therefore measured in relation to specific domains of awareness rather than by testing mothers on individual medications that can or cannot be mixed with food. Maternal practices were similarly assessed through self-reported behaviors, including motivations for mixing, reasons for not mixing, food selection criteria, preparation methods, and types of medications and foods used. This approach allowed for a descriptive and domain-specific evaluation of maternal knowledge and practices, consistent with the study’s exploratory objectives.
Data were collected using a self-administered semi-structured questionnaire. Mothers who met the inclusion criteria were approached by the field researchers (AH, AT, DT, and DY) during routine visits to primary healthcare centers, antenatal clinics, and governmental hospitals. After providing informed consent, participants were invited to complete the questionnaire on site. Field researchers were available to clarify items and ensure completeness, but mothers filled out the questionnaire independently to minimize interviewer bias. Completed questionnaires were collected immediately and checked for consistency before data entry.
Validity and Reliability
To ensure validity, the questionnaire was reviewed by a panel of experts in pharmacology (n = 3), pediatrics (n = 2), and public health (n = 3). The panelists were asked to rate each item for suitability. Item-level content validity index (I-CVI) and scale-level content validity index (S-CVI) were calculated, with values exceeding recommended thresholds (I-CVI ≥ 0.78, S-CVI ≥ 0.80). Content Validity Ratios (CVR) were also computed following Lawshe’s method, confirming that all items were essential. A pilot study was conducted with 35 mothers across included hospitals to assess clarity, cultural appropriateness, and content validity. Feedback from the pilot study further supported clarity and cultural appropriateness. Reliability was evaluated using both test-retest methods and internal consistency measures. The test-retest reliability demonstrated strong stability, with Pearson’s r = .91, indicating excellent reproducibility of responses over time. Internal consistency across knowledge and practice domains was acceptable, with a Cronbach’s alpha of .76, confirming that the items were sufficiently interrelated to measure the intended constructs.
Variables
Independent variables included maternal age, education level, occupation, socioeconomic status, area of residence, number of children, child’s age, child’s ability to swallow medications, presence of chronic illness, and type/frequency of medication use. Dependent variables comprised maternal knowledge (awareness of guidelines, counseling received, perception of safety, awareness of food-drug interactions) and maternal practices (motivations, food selection criteria, preparation methods, types of medications, and foods used). Information sources and educational needs were also analyzed as contextual variables.
Statistical Analysis
All data were entered and analyzed using the IBM Statistical Package for the Social Sciences (IBM SPSS), version 25.0. Prior to analysis, data were checked for completeness and consistency. Continuous variables were summarized using medians and interquartile ranges (IQRs), while categorical variables were presented as frequencies (n) and percentages (%). The distribution of continuous variables, such as maternal age, number of children, and age of the youngest child, was assessed using the Kolmogorov-Smirnov test. This step ensured appropriate selection of statistical tests for group comparisons. To evaluate differences in continuous variables across groups, non-parametric tests were employed. Specifically, the Mann-Whitney U test was used for comparisons between 2 groups, while the Kruskal-Wallis test was applied for comparisons across more than 2 groups. For categorical variables, chi-square tests were conducted to examine associations between maternal knowledge of medication-food mixing and sociodemographic or contextual factors. All statistical tests were two-tailed, and a P-value < .05 was considered statistically significant.
Results
Characteristics of the Participants
Of the 446 mothers invited to participate, 385 completed the study, yielding a response rate of 86.3%. The participant flow diagram is presented in Supplemental Figure S1. Among the respondents, 150 (39.0%) were recruited from healthcare centers in Nablus, 85 (22.1%) from Qalqilya, 65 (16.9%) from Tubas, 46 (11.9%) from Jenin, and 39 (10.1%) from Tulkarm. The median age of participating mothers was 33.0 years [28.0, 40.0], with a median of 3.0 children [2.0, 4.0] (Table 1). The youngest child had a median age of 3.0 years [1.0, 6.0]. The vast majority of mothers were currently married (n = 367, 95.3%), while only a small proportion were divorced or widowed (n = 18, 4.7%). More than half resided in urban areas (n = 215, 55.8%), whereas 170 (44.2%) lived in rural villages or refugee camps. Economic status was almost evenly distributed, with 187 (48.6%) reporting high status and 198 (51.4%) reporting low status. Educational attainment was notably high, as 286 (74.3%) had completed university education, compared to 99 (25.7%) with only some schooling. Employment status varied, with more than half being housewives (n = 202, 52.5%), followed by employment in the private sector (n = 74, 19.2%), the education sector (n = 61, 15.8%), and healthcare professions (n = 48, 12.5%).
Maternal Sociodemographic Characteristics and Child Health Profile (n = 385).

Q1: 1st quartile (25th percentile), Q3: 3rd quartile (75th percentile).
Regarding child health, only 13 (3.4%) of mothers reported that their child had a chronic illness (Table 1). However, swallowing difficulties were relatively common, affecting 78 (20.3%) of children. Additionally, 51 (13.2%) of mothers reported that their child was concurrently using multiple medications.
Maternal Knowledge of Medication-Food Mixing
More than half of mothers (n = 236, 61.3%) reported that they were aware of guidelines or best practices, yet only 117 (30.4%) had ever been counseled on the safety of mixing medications with food (Table 2). A minority of the mothers (n = 115, 29.9%) perceived the practice as safe. Importantly, a large proportion demonstrated awareness of specific risks as 314 mothers (81.6%) recognized that certain medications should not be crushed or opened, and 332 (86.2%) were aware that milk or juice may reduce drug efficacy. When asked about factors influencing drug efficacy, 204 (53.0%) identified the type of food, 127 (33.0%) mentioned temperature, 80 (20.8%) cited timing of meals, and 79 (20.5%) noted dosage form. However, 102 (26.5%) remained unsure, highlighting a persistent knowledge gap (Figure 1).
Maternal Knowledge of Medication-Food Mixing (n = 385).


Maternal perceptions of factors influencing drug efficacy when medications are mixed with food.
Association Between the Variables of the Mothers and Maternal Knowledge of Medication-Food Mixing
Univariate analysis showed that mothers residing in urban areas were less likely to have been counseled about the safety of mixing compared with those in rural areas (14.3% vs 16.1%, P = .021; Supplemental Table S3). Similarly, mothers residing in urban areas were less likely to perceive mixing as safe compared with those in rural areas (14.0% vs 15.8%; P = .022; Supplemental Table S4).
Univariate analysis showed that maternal age and the age of the youngest child were significantly associated with awareness that milk or juice may reduce drug efficacy. Mothers who reported awareness that milk or juice may reduce drug efficacy had a higher median age (34.0 years [28.0, 40.0]) compared with those who did not (32.0 years [26.0, 37.0], P = .035; Supplemental Table S5). Similarly, the youngest child’s age was higher among mothers who were aware of the risk (4.0 years [1.5, 7.0]) compared with those who were not (2.0 years [1.0, 5.0], P = .021; Supplemental Table S5).
Maternal Practices in Medication Preparation and Mixing
In practice, motivations for mixing included child refusal (n = 118, 30.6%), unpleasant taste (n = 69, 17.9%), ensuring full dose intake (n = 40, 10.4%), and swallowing difficulty (n = 30, 7.8%; Figure 2A). Conversely, the most common reasons for not mixing were uncertainty about safety (n = 103, 26.8%) and lack of perceived need (n = 98, 25.5%; Figure 2B).

Maternal reasons for not mixing (Panel A) and motivations for mixing (Panel B) pediatric medications with food.
Food selection was primarily guided by child preference (n = 83, 21.6%) or taste masking (n = 69, 17.9%), while only 37 (9.6%) relied on medical advice (Figure 3A). In terms of preparation methods, liquid mixing was most common (n = 141, 36.6%), followed by tablet crushing (n = 36, 9.4%) and capsule opening (n = 18, 4.7%; Figure 3B).

Criteria for selecting food vehicles (Panel A) and methods of medication preparation (Panel B).
Analgesics were the most commonly mixed medications (n = 281, 73.0%), followed by antibiotics (n = 198, 51.4%), cough medications (n = 136, 35.3%), vitamins/supplements (n = 100, 26.0%), and antihistamines (n = 36, 9.4%; Figure 4A). Juices (n = 100, 26.0%) and mashed fruit (n = 67, 17.4%) were the predominant food vehicles, while milk (n = 38, 9.9%) and yogurt (n = 27, 7.0%) were less frequently used (Figure 4B).
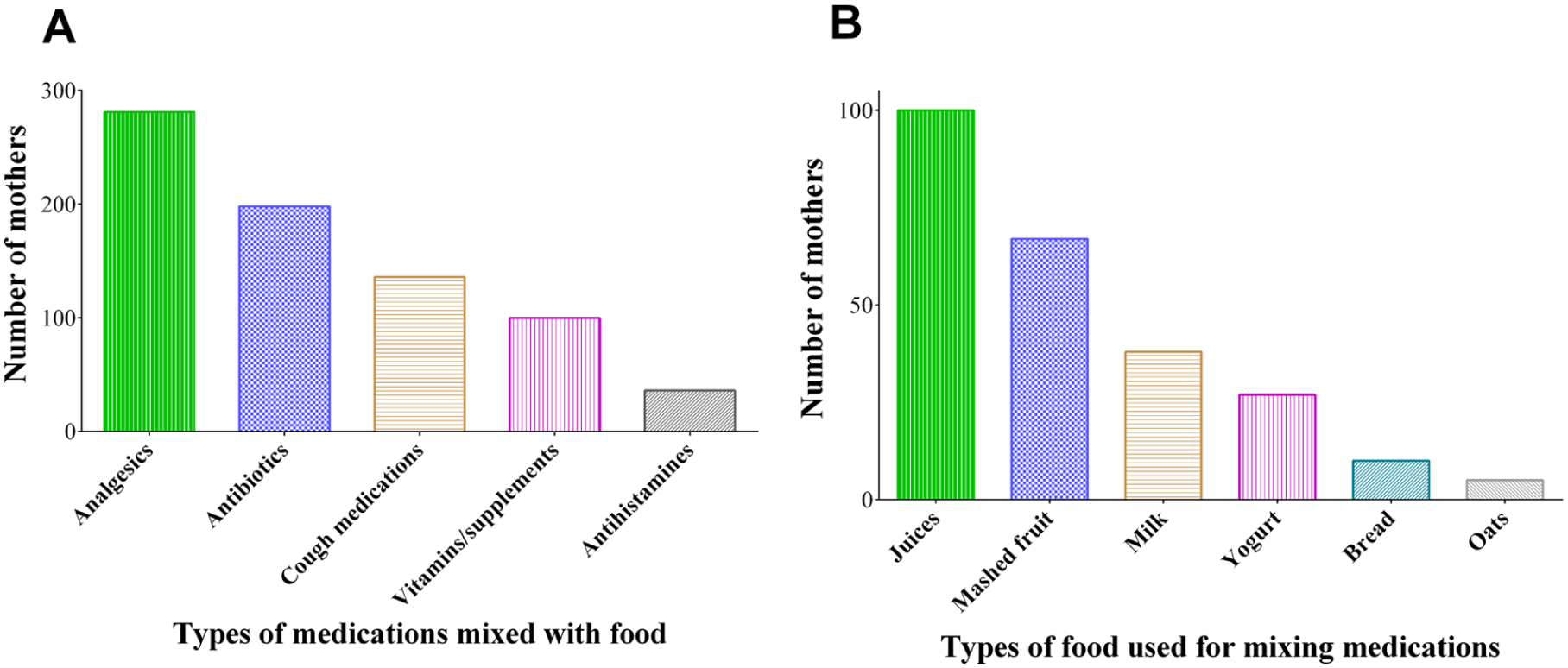
Types of medications mixed with food (Panel A) and types of food vehicles used for mixing medications (Panel B).
Maternal Information Sources, Safety Concerns, and Educational Needs
Physicians were the predominant source of information regarding pediatric medication administration, cited by 284 (73.8%) mothers, followed by pharmacists (160, 41.6%; Table 3). Other sources were less frequently consulted, including medication leaflets (85, 22.1%), family or friends (55, 14.3%), the internet (53, 13.8%), and social media (36, 9.4%). Reading instructions prior to administration was common, with 311 (80.8%) mothers reporting they always did so, while 22 (5.7%) read them sometimes and 52 (13.5%) never did.
Maternal Information Sources, Safety Concerns, and Educational Needs (n = 385).

Nearly all mothers expressed a desire for additional information on safe medication-food mixing (371, 96.4%), underscoring a critical educational need (Table 3). Confidence in mixing medications was most enhanced by clear instructions (239, 62.1%) and provider counseling (144, 37.4%), with fewer mothers citing printed guidelines (90, 23.4%) or educational videos (52, 13.5%). More than half of the participants (208, 54.0%) indicated willingness to attend educational workshops, while 124 (32.2%) were uncertain and 53 (13.8%) were unwilling.
Discussion
Understanding maternal perceptions and practices around mixing pediatric medications with food is critical for safeguarding drug safety in children.2,4,7,8,14 Mothers, as primary caregivers, often confront barriers such as poor palatability, swallowing difficulties, and child refusal, which drive the adoption of mixing strategies. This study demonstrated that although many mothers were aware of general principles, substantial gaps persisted in counseling, perceptions of safety, and practical decision-making. Mixing behaviors were strongly influenced by child-related challenges and reliance on informal information sources, while nearly all mothers expressed a need for structured guidance. These findings highlight priority areas for pediatricians, pharmacists, and primary healthcare providers, who play a central role in caregiver education. They also carry implications for policymakers seeking to strengthen support systems in resource-limited settings and for researchers designing culturally tailored interventions to promote safe medication administration.
Our findings align with international literature on caregiver and healthcare professional practices in pediatric medication administration. A recent multicenter study from Palestine reported that nurses frequently crushed tablets or opened capsules without adequate knowledge of the consequences, highlighting widespread gaps in awareness of food–drug interactions. 8 Similarly, our study showed that mothers often relied on informal sources and personal judgment when mixing medications with food, reflecting a comparable absence of structured guidance. This parallel indicates that knowledge deficits are not limited to caregivers but permeate healthcare systems, underscoring the need for comprehensive education and training at multiple levels to safeguard pediatric drug safety.
In high-income countries, pediatric medication administration is often supported by more systematic approaches. For instance, a study reported that structured counseling and clear instructions in Spain significantly improved adherence and reduced unsafe practices. 15 By contrast, our study found that Palestinian mothers received limited counseling and relied heavily on informal networks, revealing disparities in access to professional guidance. This contrast highlights the urgent need to strengthen healthcare provider involvement in caregiver education within resource-constrained settings to ensure safer medication practices. Similar patterns have been noted in biopharmaceutical reviews of pediatric oral administration strategies, which emphasize that food mixing is frequently driven by palatability and swallowing difficulties. 1 Our study confirmed that child refusal, unpleasant taste, and swallowing problems were the primary motivators for mothers to mix medications. These consistent findings across diverse contexts indicate that child-related challenges are universal drivers of caregiver behavior, transcending differences in healthcare systems and underscoring the need for child-friendly formulations to reduce unsafe mixing practices.
Taken together, the evidence highlights several priority directions for action. Caregiver counseling must be strengthened, with physicians and pharmacists providing routine, structured advice on safe medication-food mixing during vaccination visits and pediatric consultations. In parallel, culturally tailored educational materials—such as printed guidelines, audiovisual resources, and workshops—should be developed to address local practices and knowledge gaps, ensuring equitable access for mothers in both urban and rural areas. Healthcare providers should also promote safe alternatives to mixing, including child-friendly formulations such as flavored syrups or dispersible tablets, which can reduce reliance on potentially unsafe practices. Primary healthcare centers, already central to maternal and child health services, should be leveraged as venues for systematic dissemination of safe medication practices, embedding caregiver education into routine care. At the policy level, public health authorities in Palestine should prioritize caregiver education programs and establish clear regulations on which medications can safely be mixed with food. Situating these recommendations within the broader international literature underscores that while motivations for mixing are universal, the absence of structured counseling in Palestine represents a critical gap. Addressing this gap through targeted interventions has the potential to strengthen medication safety, improve adherence, and safeguard child health outcomes.
The associations observed in this study highlight how maternal knowledge and perceptions are shaped by both structural and experiential factors. The finding that urban mothers were less likely to have been counseled and less likely to perceive mixing as safe compared with rural mothers suggests that access to healthcare facilities does not automatically ensure effective communication. Busy urban clinics may prioritize efficiency over counseling, leaving caregivers without the guidance needed to make safe decisions.8,16 -18 This underscores the importance of embedding structured counseling into routine pediatric encounters, particularly in high-volume urban settings. Age-related associations further indicate that awareness of food–drug interactions, such as the impact of milk or juice on drug efficacy, increases with maternal age and with the age of the youngest child. This pattern suggests that knowledge is often acquired informally through experience rather than through systematic education.8,16 -21 Knowledge variables showed nuanced associations. Mothers who were aware of food-drug interactions, particularly the potential impact of milk or juice on drug efficacy, were paradoxically more likely to mix medications. This suggests that awareness alone does not necessarily translate into safer practices, and that knowledge must be paired with clear, actionable guidance from healthcare providers.2,4,7,8,14,17,18 Relying on experiential learning alone risks leaving younger mothers and those caring for younger children at a disadvantage, reinforcing the need for early, standardized counseling interventions. Taken together, these findings imply that interventions must be tailored to both geographic and demographic contexts, ensuring equitable dissemination of reliable information across urban and rural settings and across different caregiver age groups. Such strategies could help close existing gaps and promote safer medication practices in resource-limited healthcare systems.
Our findings resonate with regional and international studies examining maternal knowledge and practices in pediatric medication administration. In Palestine, previous work has shown that nurses frequently crushed tablets or opened capsules without adequate awareness of food-drug interactions, underscoring systemic gaps in knowledge across caregivers and healthcare professionals alike. 8 Another cross-sectional study from Palestine found that 91.9% of parents used medications without a doctor’s prescription, with frequent reliance on antipyretics, cough syrups, and antibiotics; importantly, 21.5% mixed medications with juice, 5.2% with food, and 4.7% with milk, highlighting widespread unsafe practices in home administration. 14 Similar deficits have been reported in Iran, where mothers relied heavily on informal sources and demonstrated limited understanding of safe administration practices. 14 Globally, studies from Europe highlight the importance of structured counseling. For instance, a study in Spain suggested that clear instructions significantly improved adherence and reduced unsafe practices, 15 while other studies in the Netherlands and the UK emphasized that caregiver uncertainty often leads to inappropriate mixing behaviors.7,9,18 A recent study from Sri Lanka highlighted that although most primary caregivers were aware of drug overdose risks and understood variations in syrup strengths, unsafe practices remained prevalent, with 37.7% administering medications without clinician consultation and 25.5% attempting to induce vomiting following poisoning. 22 Taken together, these findings demonstrate that maternal knowledge gaps in pediatric medication administration are widespread across diverse contexts. They reinforce the universal need for systematic caregiver education, culturally tailored interventions, and stronger healthcare provider involvement to ensure safe and effective practices.
Based on these findings, several specific and actionable recommendations can be made. First, pharmacies should be leveraged as accessible points of caregiver education, with pharmacists providing routine counseling on safe medication-food mixing during prescription dispensing. Second, guidance booklets and illustrated leaflets tailored to the local context should be developed and distributed through primary healthcare centers, vaccination clinics, and antenatal visits. Third, audiovisual materials and short instructional videos could be disseminated via social media platforms commonly used by mothers, ensuring wider reach. Finally, structured workshops integrated into maternal and child health programs would provide opportunities for interactive learning and confidence-building. These targeted strategies, implemented in collaboration with physicians, pharmacists, and public health authorities, could substantially improve maternal knowledge, reduce unsafe practices, and enhance pediatric medication safety in Palestine.
Strengths and Limitations
This study has a number of strengths. First, the study adhered to the STROBE guidelines, ensuring methodological rigor and transparent reporting across all stages of design, data collection, and analysis. This strengthens the credibility and reproducibility of the findings. Second, the study was conducted across multiple healthcare settings, including primary healthcare centers, antenatal clinics, and governmental hospitals, spanning both urban and rural areas. This diversity of recruitment sites enhanced the representativeness of the sample and allowed for meaningful comparisons across sociodemographic contexts. Third, the sample size was robust, exceeding the minimum required by formal calculation. With 385 mothers included, the study achieved sufficient statistical power to detect associations between maternal knowledge, practices, and sociodemographic variables. Fourth, the questionnaire was carefully developed and validated, drawing on international instruments and tailored to the Palestinian context. Reliability testing demonstrated strong reproducibility and acceptable internal consistency, ensuring that the tool effectively captured the intended constructs. Fifth, the study explored a comprehensive set of variables, ranging from sociodemographic characteristics and child health profiles to maternal knowledge, practices, information sources, and educational needs. This breadth allowed for a nuanced understanding of the determinants of medication-food mixing and provided actionable insights for healthcare providers and policymakers.
On the other hand, this study has also some limitations. First, the use of a cross-sectional design limits causal inference. While associations were identified between maternal knowledge, perceptions, and practices, these relationships cannot be interpreted as causal. For example, higher maternal age was associated with greater awareness of food-drug interactions, but this does not establish that age directly improves knowledge. Longitudinal studies are needed to track changes in maternal knowledge and practices over time, while interventional studies could evaluate the impact of structured counseling, pharmacy-based education, or guidance booklets on improving safe medication administration. Such designs would provide stronger evidence on causality and the effectiveness of targeted interventions. Second, responses may be subject to recall bias and social desirability bias. Because data were collected through self-reported questionnaires, mothers may have overreported awareness of safe practices or underreported unsafe behaviors to present themselves more favorably. This could lead to an overestimation of knowledge and an underestimation of risky practices. To mitigate these limitations, future studies should incorporate triangulation with observational methods, such as direct observation of medication administration or structured caregiver interviews, to validate self-reported data and provide a more accurate understanding of maternal practices. Third, the use of convenience sampling may introduce selection bias. Mothers recruited from healthcare facilities during routine visits may differ systematically from those who do not attend such facilities, for example in terms of health literacy, socioeconomic status, or access to professional counseling. As a result, the findings may not fully represent the broader population of mothers in the West Bank, particularly those with limited healthcare access. This limitation should be considered when interpreting the results, and future studies should employ probability-based sampling strategies to enhance representativeness and generalizability. Fourth, although the questionnaire was validated, the study relied on quantitative measures only, without qualitative interviews or focus groups. This restricted the ability to explore deeper contextual factors, such as cultural beliefs or emotional drivers, that may influence maternal practices. Fifth, the questionnaire focused on selected domains of maternal knowledge and practices, and did not capture all possible dimensions of caregiver behavior, which may restrict the comprehensiveness of the findings. Sixth, the study was conducted in the northern West Bank only, which may not fully capture variations across other regions of Palestine. Differences in healthcare infrastructure, cultural practices, and access to information in other areas could yield different results. Together, these strengths and limitations highlight the value of the study in providing novel, context-specific evidence while also pointing to areas for future research. Longitudinal designs, mixed-method approaches, and broader geographic coverage would further strengthen understanding and inform interventions to improve pediatric medication safety.
Conclusion
This study provides novel evidence on the knowledge, perceptions, and practices of Palestinian mothers regarding the mixing of pediatric medications with food. While many mothers demonstrated awareness of specific risks, persistent gaps in counseling and safe practices remain evident. The associations observed suggest that geographic disparities and age-related experience shape maternal awareness, yet informal and fragmented learning cannot substitute for structured guidance. The near-universal demand for more information underscores an urgent need for systematic interventions. Situating these findings within international evidence highlights that, although the motivations for mixing are universal, the absence of consistent counseling in Palestine represents a critical gap. Addressing this gap through culturally tailored education, strengthened involvement of healthcare providers, and policy-level support could substantially improve medication safety, adherence, and ultimately child health outcomes.
Supplemental Material
sj-docx-1-inq-10.1177_00469580261436352 – Supplemental material for Maternal Practices and Knowledge on Mixing Pediatric Medications with Food: Evidence of Counseling Gaps and Educational Needs in Primary Care
Supplemental material, sj-docx-1-inq-10.1177_00469580261436352 for Maternal Practices and Knowledge on Mixing Pediatric Medications with Food: Evidence of Counseling Gaps and Educational Needs in Primary Care by Ayman Hassan, Ali Titi, Dema Altouri, Dalya Yasin, Taysir Alsadder, Mohammad Jaber and Ramzi Shawahna in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-docx-2-inq-10.1177_00469580261436352 – Supplemental material for Maternal Practices and Knowledge on Mixing Pediatric Medications with Food: Evidence of Counseling Gaps and Educational Needs in Primary Care
Supplemental material, sj-docx-2-inq-10.1177_00469580261436352 for Maternal Practices and Knowledge on Mixing Pediatric Medications with Food: Evidence of Counseling Gaps and Educational Needs in Primary Care by Ayman Hassan, Ali Titi, Dema Altouri, Dalya Yasin, Taysir Alsadder, Mohammad Jaber and Ramzi Shawahna in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-docx-3-inq-10.1177_00469580261436352 – Supplemental material for Maternal Practices and Knowledge on Mixing Pediatric Medications with Food: Evidence of Counseling Gaps and Educational Needs in Primary Care
Supplemental material, sj-docx-3-inq-10.1177_00469580261436352 for Maternal Practices and Knowledge on Mixing Pediatric Medications with Food: Evidence of Counseling Gaps and Educational Needs in Primary Care by Ayman Hassan, Ali Titi, Dema Altouri, Dalya Yasin, Taysir Alsadder, Mohammad Jaber and Ramzi Shawahna in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-docx-4-inq-10.1177_00469580261436352 – Supplemental material for Maternal Practices and Knowledge on Mixing Pediatric Medications with Food: Evidence of Counseling Gaps and Educational Needs in Primary Care
Supplemental material, sj-docx-4-inq-10.1177_00469580261436352 for Maternal Practices and Knowledge on Mixing Pediatric Medications with Food: Evidence of Counseling Gaps and Educational Needs in Primary Care by Ayman Hassan, Ali Titi, Dema Altouri, Dalya Yasin, Taysir Alsadder, Mohammad Jaber and Ramzi Shawahna in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-docx-5-inq-10.1177_00469580261436352 – Supplemental material for Maternal Practices and Knowledge on Mixing Pediatric Medications with Food: Evidence of Counseling Gaps and Educational Needs in Primary Care
Supplemental material, sj-docx-5-inq-10.1177_00469580261436352 for Maternal Practices and Knowledge on Mixing Pediatric Medications with Food: Evidence of Counseling Gaps and Educational Needs in Primary Care by Ayman Hassan, Ali Titi, Dema Altouri, Dalya Yasin, Taysir Alsadder, Mohammad Jaber and Ramzi Shawahna in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-docx-6-inq-10.1177_00469580261436352 – Supplemental material for Maternal Practices and Knowledge on Mixing Pediatric Medications with Food: Evidence of Counseling Gaps and Educational Needs in Primary Care
Supplemental material, sj-docx-6-inq-10.1177_00469580261436352 for Maternal Practices and Knowledge on Mixing Pediatric Medications with Food: Evidence of Counseling Gaps and Educational Needs in Primary Care by Ayman Hassan, Ali Titi, Dema Altouri, Dalya Yasin, Taysir Alsadder, Mohammad Jaber and Ramzi Shawahna in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Acknowledgements
The authors would like to thank the patients who participated in this study. An-Najah National University (www.najah.edu) and An-Najah National University Hospital (![]() ) are acknowledged for making this study possible. The authors would like to thank the participating hospitals.
) are acknowledged for making this study possible. The authors would like to thank the participating hospitals.
Ethical Considerations
The study was carried out in accordance with international and local ethical standards, following the principles of the Declaration of Helsinki and its later revisions. Approval was secured from the university’s Institutional Review Board (IRB) of An-Najah National University (Approval #: NNU-IRB: Pharm-Feb-25/6) before data collection began. Mothers were provided with clear information about the study’s objectives, procedures, possible risks, and anticipated benefits. All mothers provided written informed consent. Participation was voluntary, with the option to withdraw at any stage without penalty. Personal data were treated confidentially, stored securely, and accessed only by the research team.
Consent to Participate
Written informed consent was obtained from each mother before enrollment. The consent process emphasized transparency, ensuring that participants understood the scope of the study and the intended use of their data for academic and scientific purposes.
Author Contributions
Ayman Hassan: Data curation; Investigation; Methodology; Formal analysis; Writing – original draft; Writing – review & editing. Ali Titi: Data curation; Investigation; Methodology; Formal analysis; Writing – original draft; Writing – review & editing. Dema Altouri: Data curation; Investigation; Methodology; Formal analysis; Writing – original draft; Writing – review & editing. Dalya Yasin: Data curation; Investigation; Methodology; Formal analysis; Writing – original draft; Writing – review & editing. Taysir Alsadder: Conceptualization; Data curation; Investigation; Methodology; Supervision; Writing – original draft; Writing – review & editing. Mohammad Jaber: Conceptualization; Data curation; Investigation; Methodology; Supervision; Writing – original draft; Writing – review & editing. Ramzi Shawahna: Conceptualization; Data curation; Investigation; Methodology; Formal analysis; Supervision; Writing – original draft; Writing – review & editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data is available upon request from the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
Declaration of Generative AI and AI-Assisted Technologies in the Writing Process
In line with the journal’s policy on transparency, the authors wish to declare that during the preparation of the manuscript, the authors used Grammarly (Superhuman Platform Inc., San Francisco, California), Copilot (Microsoft Inc., Redmond, Washington), and ChatGPT (OpenAI, San Francisco, California) solely to edit the language and improve grammar, spelling, punctuation, readability, and style of the manuscript. After using these tools/services, the authors reviewed and edited the content as needed and take full responsibility for the scientific content, accuracy, and integrity of the manuscript. Artificial intelligence tools/services were not used for generative editorial work, autonomous content creation, data analysis, statistical interpretation, or the generation of scientific content.
