Abstract
Physicians’ referral networks influence the information they obtain from peers and how fast they adopt new treatment modality. This study employs a cross-sectional design. We examined how urologists’ referral network connections via primary care physicians influence the adoption of active surveillance for localized prostate cancer, using SEER-Medicare data (2001-2015). We developed a referral network index to measure urologists’ connections outside their practice. Multinomial logistic regression estimated the likelihood of active surveillance adoption, adjusting for patient, urologist, and market factors. Findings revealed that each standard deviation increase in a urologist’s referral network index was associated with a 1.90 percentage point rise in the likelihood of adopting active surveillance, with a more pronounced effect observed post-2010. The study concludes that primary care physicians may function as critical “information hubs,” accelerating the diffusion of new treatment modalities among urologists, particularly following clinical guideline updates.
Keywords
Introduction
Treatments for localized prostate cancer range from conservative disease management (eg, active surveillance) to active treatment (eg, prostatectomy or radiation therapies). 1 Evidence from randomized clinical trial for the best treatment option for patients with localized prostate cancer is limited owing to uncertainty in how best to balance quality of life and survival.2 -4 Clinical guidelines for localized prostate cancer are gradually accumulating 1 and insurance coverage policies grant urologists wide flexibility in how to treat localized prostate cancer. As early as 2002 and 2005, the Scandinavia clinical trial reported that prostatectomy and watchful waiting treatment strategies had equivalent mortality rates for patients who were 65 years and older.5,6 Active surveillance was introduced as an acceptable treatment modality for localized prostate cancer in the National Comprehensive Cancer Network (NCCN) guidelines in 2010. 1 The use of active surveillance increased gradually between 2004 and 2015,7 -9 with variations in utilization rates observed at urologist, 10 urology practice,11,12 and market levels. 13
Physicians learn from their peers and decide whether to adopt a new treatment approach based on the influence and information they obtain from peers. Physicians often rely on peer networks to acquire knowledge, exchange clinical experiences, and assess the potential benefits and risks of adopting new treatment approaches. 14 Social learning theory suggests that individuals, including healthcare professionals, are influenced by their peers when making decisions in uncertain environments. 15 In the medical field, physicians observe the clinical practices of colleagues, evaluate treatment outcomes, and integrate shared knowledge into their own decision-making processes. 16
Peer influence plays a crucial role in diffusing medical innovations, particularly in settings where clinical guidelines evolve, new technologies emerge, or treatment paradigms shift. Physicians are more likely to adopt new treatments when they observe positive patient outcomes or receive recommendations from trusted colleagues within their professional networks.16,17 Furthermore, referral networks and institutional affiliations shape physicians’ exposure to novel therapies, and influence treatment choices and variations in care patterns across institutions,18,19 as they regularly engage in case discussions, multidisciplinary teamwork, and continuing medical education programs.
Beyond direct interpersonal interactions, the structure of professional networks affects how information propagates among physicians. Those embedded in dense referral networks or affiliated with teaching hospitals may have earlier access to cutting-edge research, while those in solo practice or in resource-limited settings may rely more on peer referrals to stay updated. These dynamics highlight the importance of understanding physician learning behaviors in shaping treatment adoption patterns and healthcare delivery.
In this study, we examined how physicians’ referral network connections influence their adoption of active surveillance, an increasingly preferred conservative management approach for localized prostate cancer. Specifically, we used patients’ main primary care physicians as connections to establish a referral network connection for urologists and created a “referral network index” reflecting the number of urologist connections outside of that urologist’s practice through primary care physicians. The larger the urologist’s referral network index, the more likely the urologist is to receive information and be influenced by urologists outside their practice through patients’ primary care physicians.
We evaluated the probability for patients with localized prostate cancer to undergo active surveillance based on their urologist’s referral network index in 3 different time periods: 2001 to 2005 (period 1), 2006 to 2010 (period 2), and 2011 to 2015 (period 3). Because the clinical guideline of active surveillance was introduced in 2010, we hypothesized that urologists with a high referral network index increased their use of active surveillance more than urologists with a low referral network index between period 2 and period 3. In addition, we hypothesized that urologists in solo practice may rely on information from primary care doctors and urologists at other practices more so than do urologists who have other urologists in their practice. Therefore, we also analyzed whether the referral network index value has a larger effect on urologists working in solo practice.
Our research will offer important insights into how the adoption of a new conservative treatment is shaped by primary care physicians’ referral networks, thereby informing policy strategies to leverage the influence of primary care providers in promoting cost-effective treatment approaches.
Methods
Data
This study is a cross-sectional study using SEER-Medicare dataset. SEER-Medicare is a publicly available dataset governed by a data use agreement, and the patients included in this study were not individually identifiable. We identified patients diagnosed with localized prostate cancer between 2001 and 2015 using the Surveillance, Epidemiology, and End Results (SEER)-Medicare database, which is a population-based database containing Medicare claims for cancer patients residing within one of the 18 SEER registry regions, encompassing 34.6% of the US population. 20 We identified 704 751 men newly diagnosed with prostate cancer between January 1, 2001, and December 31, 2015. We divided patients into 3 periods (2001-2005 as period 1, 2006-2010 as period 2, and 2011-2015 as period 3) by their diagnosis date. We used the first urologist who billed the patient in SEER’s Carrier Claims dataset within 180 days before the month of diagnosis as the patient’s treatment “decision-making” urologist.
We excluded patients whose diagnosis reporting source was a hospice or nursing home, autopsy report, or death certificate, as well as patients whose month of diagnosis date was after the month of death (n = 6665). We also excluded patients who were younger than 66 years at diagnosis (n = 243 222). To ensure completeness of claims data to determine treatment modalities and to calculate baseline comorbidity, we excluded those who did not have continuous fee-for-service Medicare Part A and Part B coverage after diagnosis, enrolled in Medicare Advantage within 12 months before or after diagnosis, or died within 12 months of diagnosis (n = 178 262). We further excluded patients whose cancer was not “localized/regional” as defined by the “SEER Historic Stage A” variable 21 (n = 25 198). Prostate cancer grading follows the ISUP/WHO Grade Group system, which is derived from the Gleason Score. Particularly, Grade Group I is equivalent to Gleason score 6 (3 + 3) or lower, Grade II is equivalent to Gleason score 7 (3 + 4) and Grade Group III is equivalent to Gleason score 7 (4 + 3). Liu et al showed the use of active surveillance had been increasing among both patients with a Gleason score 6 or below and patients with a Gleason score 7 or above. 9 We categorized grades I and II as one group because patients with these grades have “well and moderately differentiated” cancer, 22 and patients whose grade was not I, II, or III were excluded (n = 40 067). We also excluded patients whose decision-making urologist’s national provider identification number or unique provider identification number was missing (n = 38 187) and those whose urologists had less than 10 patients in any of the 3 periods mentioned above to give us stable estimation (n = 68 962). Our final study population consisted of 129 384 patients and 1010 urologists.
Patient race was categorized as White, Black, or other. We calculated each patient’s Klabunde et al comorbidity index 23 using Medicare claims in the window of 12 months before the diagnosis month. We used the scoring method designed by Roux et al 24 as a proxy for patients’ neighborhood socioeconomic status at the zip-code level.
We collected the following urologist characteristics: whether they were affiliated with a teaching hospital, whether they were the only urologist at their practice (ie, “solo practice”), whether they worked at a self-referral intensity-modulated radiation therapy (IMRT) facility because urologists who worked at a self-referral IMRT facility tended to use IMRT more than other treatment method, 25 number of other urologists at their practice, and their patient volume in each period. We also determined the number of primary care physicians that each urologist had connections with, as defined by shared patients.
We also collected market-level characteristics, which included health service area level Herfindahl-Hirschman Index (HHI), calculated by the market share of each urologist for all Medicare beneficiaries with prostate cancer in the health service area. Including the Herfindahl-Hirschman Index (HHI) in the model is essential for evaluating physicians’ decisions because it quantifies market concentration, which can influence treatment patterns. A higher HHI indicates a more monopolized market, where fewer healthcare providers dominate, potentially limiting physician autonomy and reducing treatment variability. Conversely, a lower HHI reflects a competitive market, where physicians may have greater flexibility in decision-making and be more responsive to emerging practices. By incorporating HHI, we can assess how market structure impacts physician behavior, including their likelihood of adopting guideline-recommended treatments.
Treatment modalities were categorized as prostatectomy, IMRT, other treatments, active surveillance, or no intervention. Other treatments included 3D conformal radiation therapy, brachytherapy, and other kinds of treatment. We used a published algorithm to identify whether the patient underwent active surveillance9,26 if the patient had not received any treatment within 180 days after diagnosis. Specifically, patients under active surveillance were identified as those who did not receive any active treatments and underwent at least one Prostate-Specific Antigen (PSA) test or biopsy within 365 days of their localized prostate cancer diagnosis. Patients who did not receive any active treatments or undergo active surveillance were classified as having no treatments.
Construction of the Urologist Referral Network Index
We first established a connection between a urologist and a patient’s main primary care physician 27 using a published algorithm. 19 Then, we determined that 2 urologists who were not in the same practice shared 1 “referral information channel” if they were connected through a patient’s main primary care physician. Lastly, we used the number of referral information channels to calculate the weighted centrality score 28 of each urologist, and this score was defined as their referral network index. That is, a urologist’s referral network index measured the number of information channels with other urologists outside their practice through primary care physicians’ referral networks. Figure 1 illustrates the construction of this referral network index. Because urologists’ referral network index values were skewed to the left (few urologists had a large number of connections), we used the normalized log of the referral network index (ie, the Z score) as our independent variable of interest. We used any shared patients as an indication that a connection existed between a patient’s main primary care physician and a urologist, rather than at least 10 shared patients, as was done in previous studies. 19 We did this for 2 reasons. First, we were evaluating the potential information transmission among urologists by any information channel, and theories such as “structural holes” 29 and “the strength of weak ties” 30 suggested that random and weaker connections were important for information diffusion. Second, we observed only Medicare fee-for-service patients in our dataset, which limited our ability to capture the total patient volume for each urologist.

Construction of the urologist referral network index.
Econometric Model
For patient i treated by urologist j in period t, we denoted the treatment modality as Yi,j,t. We fitted multinomial logistic regression models to estimate patient i’s probability of undergoing each treatment, including active surveillance (denoted AS; other treatment as defined above is the reference group), treated by urologist j in period t:
Where k = 1, 2, 4, 5
where Z is the referral network index, our independent variable of interest. Other independent variables included patient (Xi), urologist (Pj,t), and market (HHIi,t) characteristics (Table 1). We also included period fixed effects (Ti) and health service area level fixed effects (Hi). We clustered standard error at the urologist practice level. We used the coefficient estimates to calculate the marginal effect that patients would undergo active surveillance (Table 2, column A). Selected original coefficients and 95% confidence intervals of the main regression (equation (1)) are shown in Supplemental Table 1. A sample size of 704 751 men newly diagnosed with prostate cancer provides substantial statistical power to detect meaningful associations and differences across subgroups. Given the large sample, even small effect sizes can be detected with high precision, reducing the risk of Type II errors. Additionally, we also conducted a sensitivity analysis to ensure the robustness and reliability of our main results. The manuscript followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline for cohort studies. 31 A full STROBE report is available in Supplemental File 1. All statistical analyses were conducted in STATA version 15.1 and RStudio version 1.1.456. The Emory Institutional Review Board exempted this study from human subject review.
Patient Characteristics by Their Urologist’s Referral Network Index Group.

IMRT, intensity-modulated radiation therapy.
Estimated Probability Increase of Undergoing Active Surveillance With 1 Standard Deviation Increase in the Referral Network Index.

P < .05. **P < .01.
Patient-Level Analysis
In patient-level analyses, we aggregated patient-level data into urologist-level data to obtain urologists’ rate of using active surveillance as the treatment modality for their patients with localized prostate cancer for each period. We fitted 2 ordinary least squares regressions to analyze whether urologists with a high referral network index increased their use of active surveillance more than urologists with a low referral network index between period 2 (2006-2010) and period 3 (2011-2015; equation (2)), using urologists who were consistently in the same network tertile groups in both periods. We also evaluated whether the impact of referral network index was larger for urologists working in solo practice (equation (3)), using urologists who were consistently working in solo practice or in a practice with other urologists in both periods. Equations (2) and (3) are as follows:
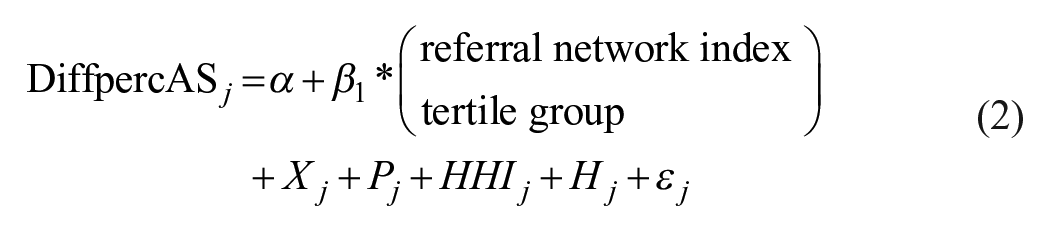
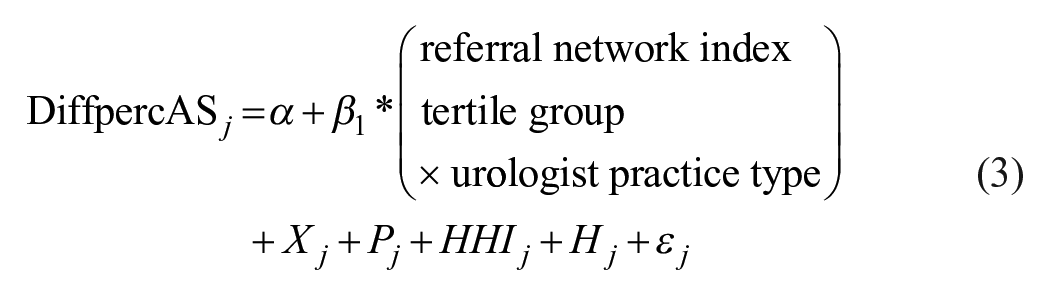
where DiffpercAS j is the difference in the rate of using active surveillance for urologist j between period 2 and 3. We categorized urologists into 3 groups by their referral network index tertile in period 3. Urologist practice type categorized urologists into 2 groups: those in solo practice and those who worked in a practice with other urologists. Xj is patient characteristics in period 3 aggregated at the urologist level, Pj is urologist characteristics in period 3, HHIj is urologist health service area characteristics in period 3, and Hj is urologist health service area fixed effects. The estimated probabilities of equations (2) and (3) are reported in Figure 2.

Active surveillance usage increase between period 2 (2006-2010) and period 3 (2011-2015) for (A) urologists with a high and low referral network index and (B) urologists with a high and low referral network index who work in solo practice or in a practice with other urologists.
Sensitivity Analysis
Our main hypothesis was that urologists were influenced by other urologists outside their practice through primary care physicians. Urologists may also have been influenced by their coworkers within the same practice through primary care physicians. In addition to controlling for coworkers’ direct impact by including the number of urologist coworkers in equation (1), we conducted our first sensitivity analysis to rule out the possibility of coworkers’ direct impact. Specifically, we created an “entire network index,” which measured urologists’ information channels with urologists both outside their practice and in their own practice through primary care physicians. Recall that the “referral network index” discussed earlier measured only urologists’ information channels with urologists outside their practice through primary care physicians. We fitted the main regression in equation (1) with urologists’ entire network index; results are shown in Table 2, column B.
Second, we conducted a sensitivity analysis by incorporating patients who received no treatment (the no intervention “none” group) into the active surveillance group. This is because urologists may suggest active surveillance to their patients but some patients may then not follow up with the suggested testing schedule because of their personal preference or other reasons such as access barrier. Results of this sensitivity analysis are reported in Table 2, column C.
Lastly, urologists’ networks in period 3 could have been expanded because of their higher use of active surveillance in the previous periods. Specifically, urologists who used active surveillance more than others in period 2 could establish more connections with primary care physicians in period 3 because of the urologists’ reputation for diversified treatment approaches, and therefore these urologists would have a higher referral network index in period 3. To explore this possibility, we fitted an ordinary least squares regression to test whether urologists’ use of active surveillance in period 2 was associated with an increase in primary care physician connections in period 3, as shown in equation (4):

where DiffPIconnection j is the difference in the number of urologists’ primary care physician connections between period 2 and period 3, AS_usage j is urologist j’s use of active surveillance in period 2, Xj is patient characteristics in period 3 aggregated at the urologist level, Pj is urologist characteristics in period 3, HHIj is urologist health service area characteristics in period 3, and Hj is urologist health service area fixed effects.
Results
Table 1 provide summary statistics of patient, urologist, and market characteristics for the full sample and stratified by tertiles of referral network index values. Figure 3 presents the unadjusted proportion of patients undertaking active surveillance by tertiles of referral network index. Urologists in the high referral network index tertile group were more likely to treat patients without Medicaid coverage, those with low cancer progression, Black patients, patients residing in urban areas, patients with comorbidities, and patients with high socioeconomic status. Urologists in the high referral network index tertile group were also more likely to be affiliated with teaching hospitals, IMRT self-referral facilities, in group practices with other urologists, and practice in health service areas with a high level of competition. In addition, urologists in the high referral network index tertile group were more likely to have large patient volume and a high number of primary care physician connections.

Share of patients receiving active surveillance from urologists with a low, medium, or high referral network index.
Table 2 reports the marginal effects of the coefficient β1 in equation (1) and the results of sensitivity analyses. As shown in column A, 1 standard deviation increase in the log referral network index value increased the probability of the patient undergoing active surveillance by 1.90 percentage points (P < .05) over the entire period studied. When classified the entire duration into 3 time periods, this effect was most obvious in period 3 (2011-2015), with an increase of 2.60 percentage points (P < .01) compared with 1.98 percentage points (P < .05) in period 2 (2006-2010) and 1.36 percentage points in period 1 (2001-2005; not significant at conventional levels).
As shown in Table 2, column B, 1 standard deviation increase in the urologist’s entire network index increased the adjusted probability of the patient undergoing active surveillance by 1.33 percentage points (P = .23). The effect of the referral network index was larger than that of the entire network index, suggesting that the information transmitted through primary care physicians from urologists in other practices has a larger impact than the information transmitted among urologists within the same practice. Column C Table 2 shows that the effects of incorporating patients with no intervention with those receiving active surveillance were not significantly different from the results shown in column A.
Figure 2A shows the estimated active surveillance usage rate increase by urologists who were in the high referral network index tertile group compared with those in the low tertile group between period 2 (2006-2010) and period 3 (2011-2015), estimated by equation (2). Specifically, urologists in the high referral network index tertile group increased their use of active surveillance by 9.35 percentage points (P < .001) and urologists in the lowest referral network index tertile group increased their use of active surveillance by 2.18 percentage points (P = .32). Urologists in the high tertile group increased their use of active surveillance 7.17 percentage points more than those in the low tertile group between period 2 and period 3, although the difference was not significant at conventional levels (P = .103).
Figure 2B reports the effects of the referral network index on active surveillance usage rate increase between period 2 (2006-2010) and period 3 (2011-2015) by urologists who were working in solo practice compared with those working in group practice, estimated by equation (3). For urologists working in solo practice, those in the high referral network index tertile group increased their active surveillance usage 4.82 percentage points more than those in the low referral network index tertile group between period 2 and period 3 (not significant at conventional levels). For urologists working at practices with other urologists, the difference was 2.01 percentage points (not significant at conventional levels).
Selected coefficients and 95% confidence intervals of equation (4) are reported in Supplemental Table 2. These results showed that urologists’ use of active surveillance in period 2 was not associated with an increase in primary care physician connections between period 2 and period 3.
Discussion
Physicians often learn new treatment modalities from their peers. Physicians’ network positions are associated with the information and influence they receive.16,18,28,32 -38 Although active surveillance for localized prostate cancer is not a new treatment method requiring urologists to invest in new equipment or for primary care physicians to acquire cutting-edge clinical knowledge, urologists have gradually adopted this less invasive treatment modality for localized prostate cancer over time as clinical evidence has accumulated.
Our paper makes several important contributions to the literature. First, our work extended current physician network analysis by using patients’ primary care physicians as connections to establish specialists’ referral networks and information sharing channels. Second, we investigated the impact of urologists’ referral network information channels on their adoption of a more conservative treatment modality. The adoption of a less invasive treatment modality implied that urologists de-adopted more invasive treatment modalities, for example, robotic prostatectomy and IMRT, for localized prostate cancer. De-adoption of technology offers a potential strategy to reduce technological overuse and lower healthcare costs in the United States and beyond. De-adoption initiatives could be integrated into healthcare policies by promoting evidence-based guidelines, incentivizing value-driven care, and enhancing physician education on the risks of low-value interventions. 39 Policymakers might consider reimbursement reforms that discourage unnecessary technology use while ensuring access to essential innovations. Additionally, fostering data-driven decision-making through real-time feedback mechanisms can support clinicians in optimizing treatment choices. Future research should explore the barriers and facilitators of de-adoption, including physician-level, institutional, and systemic factors that influence decision-making.
Third, our results highlight the critical role of primary care physicians (PCPs) as an “information hub” in disseminating new treatment approaches within specialist networks. This effect was particularly pronounced following the release of clinical guidelines and among urologists in solo practice. While the broader debate continues on whether increased investment in PCPs lowers healthcare costs in the United States, 40 our findings suggest a new direction for analyzing their impact on optimizing healthcare resource allocation. In the era of personalized medicine, PCPs play an increasingly influential role in treatment decision-making,41,42 given their longitudinal relationships with patients and deep understanding of patients’ chronic conditions and preferences. This raises important questions about how referral networks operate at different levels, particularly when comparing the referral network index (focusing on direct specialist connections outside institutions) versus the entire network index (capturing broader professional relationships within institutions). The differential findings for these indices suggest distinct mechanisms of information flow and treatment adoption. A strong direct referral network may indicate that urologists rely more heavily on a trusted group of PCPs for referrals and treatment updates, leading to faster dissemination of new clinical practices. Conversely, a broader entire network index might reflect a more diffuse, institutional influence, where policies and peer interactions within institutions primarily shape decision-making. Understanding these nuances is particularly relevant as health systems transition to value-based care models, where effective coordination between PCPs and specialists can improve treatment adherence, reduce unnecessary interventions, and optimize resource utilization.
Our findings have significant policy implications, emphasizing the need to strengthen PCP-specialist collaboration through integrated care models, improved access to shared electronic health records, and incentivized PCP engagement in care coordination via value-based payment structures. From an operational perspective, healthcare systems could leverage PCPs’ central role by implementing targeted educational initiatives, clinical decision-support tools, and multidisciplinary case reviews to enhance the dissemination of best practices.
Future research should explore the mechanisms through which PCPs influence specialists’ decisions, variations in their impact across healthcare settings, and how referral network structures affect patient outcomes. Network-based studies could further clarify information flow within healthcare networks and identify strategies to optimize referral pathways for improved care quality and efficiency.
Our study has some limitations. First, we used patients from a single-payer system (Medicare) to measure urologists’ referral network index. Although there is a high correlation between the number of Medicare patients and the number of privately insured patients shared by physicians, 43 it is possible that urologists’ referral networks could look different when determined by all patients compared with Medicare patients alone. Second, we grouped patients into three 5-year periods because of the limited number of cancer cases. Urologist characteristics, such as practice location and academic affiliation, may change within a 5-year period. We used plural claims to identify urologists’ main practice location, academic affiliation, and other characteristics for the period; hence there may be measurement errors. Lastly, as with most studies utilizing claims-based data, we are unable to directly observe or measure patient preferences. Future research could explore the impact of patient preferences on the adoption of conservative treatment approaches.
Despite these limitations, our study points to an important direction for future research – the understanding of information diffuses among specialists’ networks through their connections with primary care physicians.
Conclusion
Urologists with a larger referral network through primary care physicians were more likely to adopt active surveillance as a treatment strategy compared to those with a smaller referral network. This trend became particularly pronounced after the NCCN officially recommended active surveillance for localized prostate cancer. These findings highlight the influential role of primary care physicians in promoting conservative treatment approaches.
Supplemental Material
sj-docx-1-inq-10.1177_00469580251342466 – Supplemental material for Referral Network Increases Physicians’ Adoption of Active Surveillance for Localized Prostate Cancer: A Cross-Sectional Study Using SEER-Medicare
Supplemental material, sj-docx-1-inq-10.1177_00469580251342466 for Referral Network Increases Physicians’ Adoption of Active Surveillance for Localized Prostate Cancer: A Cross-Sectional Study Using SEER-Medicare by Yu Liu, David Howard and Ya-chen Tina Shih in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-docx-2-inq-10.1177_00469580251342466 – Supplemental material for Referral Network Increases Physicians’ Adoption of Active Surveillance for Localized Prostate Cancer: A Cross-Sectional Study Using SEER-Medicare
Supplemental material, sj-docx-2-inq-10.1177_00469580251342466 for Referral Network Increases Physicians’ Adoption of Active Surveillance for Localized Prostate Cancer: A Cross-Sectional Study Using SEER-Medicare by Yu Liu, David Howard and Ya-chen Tina Shih in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-docx-3-inq-10.1177_00469580251342466 – Supplemental material for Referral Network Increases Physicians’ Adoption of Active Surveillance for Localized Prostate Cancer: A Cross-Sectional Study Using SEER-Medicare
Supplemental material, sj-docx-3-inq-10.1177_00469580251342466 for Referral Network Increases Physicians’ Adoption of Active Surveillance for Localized Prostate Cancer: A Cross-Sectional Study Using SEER-Medicare by Yu Liu, David Howard and Ya-chen Tina Shih in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Ethical Considerations
This study qualifies for exemption from IRB review at Sam Houston State University, as it does not involve identifiable human subjects, based on the U.S. Department of Health and Human Services (HHS) regulations for the protection of human subjects, specifically 45 CFR 46.
Author Contributions
YL: Conceptualization, Methodology, Formal Analysis, Writing – Original Draft; DH: Writing – Review & Editing, Supervision, Funding Acquisition; YTS: Writing – Review & Editing, Supervision, Funding Acquisition.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially funded by the Core Facility Administrative Council of the Data Management and Analysis Core (DMAC) Pilot Grant for Comparative Effectiveness Research on Cancer in Texas, and partially funded by the Sam Houston State University College of Health Sciences Internal Research Grant Program.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Access to SEER-Medicare data requires an approved data use agreement (DUA) with the National Cancer Institute (NCI).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


