Abstract
The use of chiral medicines (possessing center(s) of asymmetric carbon) may cause adverse drug reactions (ADRs). The safety assurance of these medicines is critical. We aimed to evaluate registered and commonly used anti-infective chiral medicines circulating in the Tanzanian market to establish their safety profile to protect public health. A mixed prospective-retrospective cohort study was conducted to assess the safety profile of amoxicillin, amoxicillin-clavulanic acid and ceftriaxone injection. ADRs causality assessment was conducted by using World Health Organization (WHO)-Algorithm criteria. Data were collected from 7 tertiary hospitals: Muhimbili National Hospital (MNH), Kilimanjaro Christian Medical Centre (KCMC), Bugando Medical Centre (BMC), Ligula Referral-Regional Hospital (LRRH), Kitete Referral-Regional Hospital (KRRH), Dodoma Referral-Regional Hospital (DRRH), and Mbeya Zonal-Referral Hospital (MZRH). Data were supplemented by those recorded in the WHO-Vigiflow/VigiLyze database within the same monitoring period. Data were analyzed using STATA version-15. The results were considered statistically significant when P < .05. A total of 2522 patients were enrolled in hospitals: MNH (499), KCMC (407), BMC (396), LRRH (387), KRRH (345), DRRH (249), and MZRH (239). Among those, 1197 (47.5%) were treated with ceftriaxone, 585 (23.2%) amoxicillin and 740(29.3%) amoxicillin-clavulanic acid. Out of those, 102 (4.5%) experienced adverse events (AEs), 49 (48%) were due to ceftriaxone, 37 (36.3%) amoxicillin-clavulanic acid and 16 (15.7%) amoxicillin (P-value .012). A total of 443 participants from the enrolled and WHO-Vigiflow/VigiLyze database were experienced with ADRs. The ADRs affected mainly gastro-intestinal system 234 (53%), skin and subcutaneous tissue 85 (19%), nervous system 49 (11%), respiratory thoracic 22 (5%), and general disorders 18(4%). In this study, approximately 90% of reported AEs were ADRs possible-related to the monitored medicines, with few plausible and certain. Ceftriaxone injection caused more ADRs. Amoxicillin-clavulanic acid was associated with more ADRs than amoxicillin alone. The safety profile of these medicines is still maintained; however, comprehensive monitoring of ADRs is recommended to improve patient safety and enhance overall treatment outcomes.
Keywords
Anti-infective chiral medicines namely ceftriaxone injections, amoxicillin and amoxicillin-clavulanic acid or sometimes known as co-amoxiclav are safe with no serious adverse drug reactions (ADRs).
The research contributes to the science of pharmacovigilance or safety monitoring of medicines that:
Chiral medicines, specifically amoxicillin, amoxicillin-clavulanic acid, and ceftriaxone have (ADRs) with ceftriaxone associated with more serious ADRs.
This underscores the importance of comprehensive pharmacovigilance in clinical practice to improve patient safety and enhance overall treatment outcomes.
Thus, healthcare professionals should remain vigilant in monitoring ADRs of chiral medicines and also use evidence-based prescribing practices to mitigate risks and optimize therapeutic outcomes for their patients.
Due to the existence of some identified potential adverse drug reactions that were not reported previously, these medicines especially ceftriaxone injection should be monitored closely under active method of reporting. Healthcare workers and patients should continuously report any adverse events (AEs) that occurred during the use of medicines to their respective national regulatory agencies to monitor the safety profiles of medicines.
Introduction
Pharmacovigilance (PV) activities, with the ultimate goal of minimizing risks and maximizing the benefits of medicinal products, are essential in providing key information for assessing and monitoring safety. 1 Safety monitoring of the use of chiral medicines (medicines that possess one or more centers of an asymmetric carbon) is crucial, as it may pose adverse events (AEs).2 -4 These medicines may give rise to enantiomers (either active, inactive, or toxic enantiomers) that exhibit marked differences in biological activities, such as pharmacology, toxicology, and pharmacokinetics.5 -7 Thus, the safety assurance of these medicines is a key issue in protecting public health.
Findings from various studies evaluating the safety profile of different chiral medicines have been reported8,9 and are mainly associated with the side effects of the active pharmaceutical ingredients (API) and inherent properties of the API.8,9 In a study conducted in Tanzania, most of the anti-infective medicines registered by the Tanzania Medicines and Medical Devices Authority (TMDA) and those listed in the National Essential Medicines List (NEMLIT) were racemic mixtures. 4 The presence of these medicines in the NEMLIT means that these types of medicines are used more in each level of healthcare facilities within the country, and therefore, monitoring of their safety is necessary. Of the registered anti-infectives, amoxicillin capsules and suspension, amoxicillin-clavulanic acid injection, tablets and suspension and ceftriaxone powder for injection are among the most commonly used chiral medicines. 4 Again, the reported individual case safety reports (ICSRs) from Tanzania in the VigiLyze database owned by Uppsala Monitoring Centre has indicated that 56.4% (974) of the reported adverse drug reactions (ADRs) in 2018 were caused by the use of anti-infective medicines, of which majority were chiral medicines. 4
Despite reports on the presence of racemic mixture medicines and the existence of ADRs, no study has been conducted in Africa including Tanzania to investigate ADRs caused by chiral medicines circulating in the market. This study, aimed to investigate ADRs associated with the use of 3 antibiotic chiral medicines in Tanzania, namely amoxicillin capsules and suspension, amoxicillin-clavulanic acid injection, tablets and suspension, and ceftriaxone powder for injections. The drug safety information obtained from this study will help in updating the safety profiles of selected chiral medicines. In addition, this information will be useful in formulating policies and protecting individuals at risk from serious ADRs.
Materials and Methods
Study Setting
This study was conducted in 7 tertiary hospitals hosting pharmacovigilance centers. The centers were located at the Muhimbili National Hospital (MNH), Kilimanjaro Christian Medical Center (KCMC), Bugando Medical Centre (BMC), Ligula Regional Referral Hospital (LRRH), Dodoma Regional Referral Hospital (DRRH), Kitete Region Referral Hospital (KRRH) and Mbeya Zone Referral Hospital (MZRH). The selection of the study sites was based on the existence of established PV centers, as recognized by the TMDA.
Study Design
This was a mixed prospective-retrospective cohort study that assessed the ADRs associated with selected commonly used chiral medicines in Tanzania. We analyzed data collected from January 2019 to June 2020 using the structured stimulated spontaneous safety monitoring method (SSSSM). 10 During the same period, apart from the participants enrolled in the study, patients from different healthcare facilities experiencing AEs caused by the same medicines whose data (ICSR) were entered in the Vigiflow were reviewed. Therefore, the analyzed data were supplemented by those data extracted from the WHO- Vigiflow/VigiLyze database for Tanzania.
Medicines Selection
Medicines were selected based on two criteria: firstly, they were among the most commonly used chiral medicines in Tanzania, and; secondly, they exhibited the highest rate of reported ADRs, as per the WHO VigiLyze Global database for Tanzania. 4 All selected chiral medicines are commonly prescribed antibiotics to treat bacterial infections. They are usually prescribed depending on the type of illness or infection being treated, and the bacteria responsible for it.4,11 Detailed information on how the medicines were selected has been published. 3
Safety Data Collection
Prospective data collection of suspected ADRs was conducted using the published method SSSSM. 10 The method was developed to strengthen safety data collection in a pharmacovigilance system. Data collection was conducted at 7 hospitals located in the 7 regions mentioned above. Patients using the selected chiral medicines were identified by healthcare providers in their respective hospitals. Data from the WHO-Vigiflow/VigiLyze database were collected retrospectively.
All AEs were recorded and closely monitored by respective trained healthcare providers and pharmacovigilance officers. Patients with reported AEs were informed to receive medical care services following existing healthcare procedures.
Data Collection Tools
Yellow forms for collecting AEs were used. This form was used for spontaneous reporting. All information, as indicated in the yellow form, were recorded to include particulars of the patient, details of adverse reactions, details of suspected medicines used, management of adverse reactions, therapeutic failure (if any), medication error or overdosage (if any), and particulars of the reporter.
All information on social demographic data, such as contact address of the patients, names, and dosage regimen of the selected chiral medicines, were collected using a newly designed, validated, and coded case report form (CRF).
Data Collectors
Trained PV officers from the TMDA headquarters, zone offices, PV centers, and healthcare providers assisted by a researcher who participated in data collection as a research assistant. The training was conducted before data collection.
Study Participants
The study participants were patients using either amoxicillin capsules, suspension, amoxicillin-clavulanic acid tablets, suspcension, or ceftriaxone injection as identified by the trained healthcare providers (physicians, nurses, pharmacists).
The identified patients were explained about the study. The written consent forms were given to those who were willing to join. The adverse events were detected by the healthcare providers. For those outpatient or discharged patients, communication via mobile phones was done, and if there were any adverse events, instructions were given to go back to the respective healthcare facilities. All participants were followed up for 14 days after taking the medicines. In all healthcare facilities, there were general pharmacovigilance focal personnel and in each hospital ward, there was a focal person to oversee the follow-up and monitoring.
The inclusion criteria: All patients regardless of their age attended and/or admitted at any of the 7 healthcare facilities and who were prescribed any formulation of amoxicillin, co-amoxiclav or ceftriaxone medicine as a routine treatment were eligible to be included in the study.
The exclusion criteria: Patients who were not prescribed the selected medicines, terminally ill patients, patients with renal and liver failure, patients with more than 1 co-morbidity.
Sample Size
Sample size calculation was done by using a design effect formula 12 as indicated below:
where n is the desired sample size, deff is the design effect, N is the population size, and p is the proportion of the target population estimated to have a particular characteristic (in this study, we referred to patients using the selected chiral medicines). Because there is no proportion in the target population estimated to have a particular characteristic, the assumption was estimated to be 0.50, q = 1 − p, d = degree of accuracy desired, .05, Therefore; population size (for finite population correction factor or fpc) (N) was considered to be 1 000 000, hypothesized % frequency of outcome factor in the population (p) was 50% ± 5, confidence limits as % of 100 (absolute ± %) (d) was 5%, and design effect (for cluster surveys-DEFF) was 2. The sample size (n) was 768 participants. The study estimated 30% for non-response; therefore, the total sample size was rounded to 1000 participants for each selected chiral medicine.
The design effect formula was used as a standard method in epidemiological studies to adjust for the design complexities of the study, such as clustering effects. This method was chosen to ensure adequate power for detecting significant differences or associations given the study’s complex design, as it involves multiple healthcare facilities and a heterogeneous patient population.
Data Management and Statistical Analysis
The data was cleaned and checked for any inconsistencies, entered into Microsoft Excel spreadsheets (version 2010), verified and validated using the source data/information recorded in the patient files at the respective hospitals. The ICSR data were generated in Excel from the WHO-VigiLyze/ Vigibase database. All data were exported to STATA version 15 for statistical analysis. Data duplicates and missing values within the dataset were identified and resolved. Descriptive statistics were used to summarize the characteristics of patients and to determine the occurrence of adverse drug reactions (ADRs). The adverse events (AEs) were categorized according to age, sex, and organ system involvement. The number of patients with ADRs was calculated to determine the age and sex distribution of ADRs and were presented as numbers and/or percentages. Events were classified according to System Organ Class (SOC). A list of reactions showing the percentage of each reaction within the SOC, the percentage of each reaction among all reports, and the rate of occurrence of each reaction was calculated. The significance level was set at P < .05 in all statistical tests done in this study. Pearson’s Chi-square or Fishers exact’s test was used where appropriate to identify significant differences between categorical variables.
Univariate and multivariate logistic regression was utilized to evaluate the influence of patients’ characteristics on the likelihood of experiencing ADRs. The odds ratio (OR) and its corresponding 95% confidence interval (CI) were estimated.
The Hosmer and Lemeshow test were performed to assess the goodness-of-fit of the logistic regression model fitness, and the receiver operating characteristic (ROC) analysis was used to evaluate the model’s prediction ability.
The events were named using the Preferred Term (PT) according to the Medical Dictionary for Regulatory Activities (MedDRA) Terminology to name events. 13 MedDRA is an international medical terminology developed under the auspices of the International Conference on Harmonization (ICH) of Technical Requirements for Registration of Pharmaceuticals for Human Use.
Causality Assessment
Individual reports from each patient were subjected to causality assessment to establish the relationship between the event and the use of chiral medicine. Causality assessment was done by using the WHO alogarithm criteria 14 which were classified as “certain,” “probable,” “possible,” “unlikely,” “unclassified,” or “unassessable.” According to WHO definitions, 14 causality was defined certain when the clinical event had a plausible time relationship to medicine, which could not be explained by other factors such as concurrent disease, other medicines, or chemicals and re-challenge satisfaction (if necessary). Causality was defined probable when the clinical event had a reasonable time relationship to medicine administration, was unlikely to be attributed to concurrent disease or other medicines and de-challenge was positive. Causality was possible when the event had a reasonable time relationship with medicines, but this could also be explained by concurrent diseases or other medicines. Causality was unlikely when clinical events whose time relationship to medicine administration made a causal connection improbable but could be plausibly explained by underlying disease, other medicines, or chemicals. Causality was defined un-classifiable when the event had insufficient information to permit assessment and identification of the cause. Several factors were considered during the causality assessment, such as time to onset of events, pharmacological class, other diseases and medications, and re-challenge and de-challenge information. The presence of events similar to the clinical features of the disease at the baseline was also considered.
In addition, in order to avoid misinterpretation/misclassification of ADRs, causality assessment was done in duplicate and each was concluded by a group of clinicians and pharmacists involved in the study.
Seriousness of the ADRs
The seriousness of the ADRs was filled in at the healthcare facility level by healthcare providers and were evaluated by clinicians. A serious adverse event or reaction is any untoward medical occurrence associated with the use of a medical product in a patient that at any dose, the outcome is related to its life threatening condition. 15 It was considered serious when the patient died, or had discomfort condition and could not work, or the patient required prolonged hospitalization, or had a life-threatening condition, or the reported medicine caused persistent disability or incapacity, a congenital anomaly. 15
Classification of Frequency of Occurrence of ADRs
The classification categories of frequency were based on categories used by manufacturers as indicated in summary of product characteristics.16 -18 The classification categories of frequency used by manufacturers were: very common (≥1/10): if the medicine is affecting more than 1 in 10 people (>10%), common (≥1/100-<1/10): if it affects between 1 in 10 people to 1 in 100 people (1% to 10%), uncommon (≥1/1000-<1/100): if it affects between 1 in 100 to 1 in 1000 people (1% and 0.1%), rare (≥1/10 000-<1/1000):if it affects between 1 in 1000 to 1 in 10 000 people (0.1% to 0.01%).
Ethical Consideration
Ethical clearance was granted by the National Institute for Medical Research, Tanzania (certificate number NIMR/HQ/R.8 a/vol. IX/3086) and the Institutional Review Board of Muhimbili University of Health and Allied Sciences (MUHAS; certificate number DA.282/298/01.C). Data collection request letters were sent by the authors to all participating hospitals. Approval letters were obtained from each participating pharmacovigilance center before the start of the study at the respective hospital.
A regulatory requirement for all patients and healthcare workers is to report any ADR(s) caused by the use of any medicine(s). However, in this study, patients who used the selected chiral medicines were identified and filled-in consent form to participate in the study, and requested to report AEs/ADR(s) (if any). The patients were followed-up for 14 days after starting the use of chiral medicines. Physical follow-up was performed in wards (for admitted patients) or via phone calls for discharged/outpatients.
The research methods were conducted in accordance with relevant guidelines and regulations pertaining to the safety monitoring of medicines in Tanzania.
Results
In this study, a total of 2522 patients were enrolled, 1476 (58.5%) were female. The median patient age was 30 years, with an interquartile range of 19 to 48. The median weight was 61 kg with an interquartile range of 40 to 70 kg. The patients were recruited from 7 healthcare facilities in Tanzania (Table 1). Our findings showed 1461 (57.9%) of patients aged between 20 and 59 years while 381(15.1%) aged between 1 and 9 years followed by 377 (14.9%) for the age group above or equal to 60 years.
Social-Demographics Characteristics of the Participants.

Note. MNH = Muhimbili National Hospital; BMC = Bugando Medical Centre; MZRH = Mbeya Zonal Referral Hospital; KCMC = Kilimanjaro Christian Medical Centre; LRRH = Ligula Regional-Referral Hospital; DRRH = Dodoma Regional-Referral Hospital; KRRH = Kitete Regional-Referral Hospital; IQR = Interquartile range.
Furthermore, age and sex distributions were analyzed at the facility level, as summarized in Table 1. The results revealed that, in all 7 hospitals, more than half of the patients were female, as was the overall cohort. Similarly, the age group distribution across all facilities was in-line with the overall trend of having a majority in the 20 to 59 years age group.
The number of patients treated with selected chiral medicines was 1197 for ceftriaxone injections and 585 (capsules 557 and suspension 29) for amoxicillin and 740 (tablets 697, suspension 43) for amoxicillin-clavulanic acid, as summarized in Table 2.
Characterization of Pre-Treatment Symptoms and Medicines Used by Participants at the Facility.
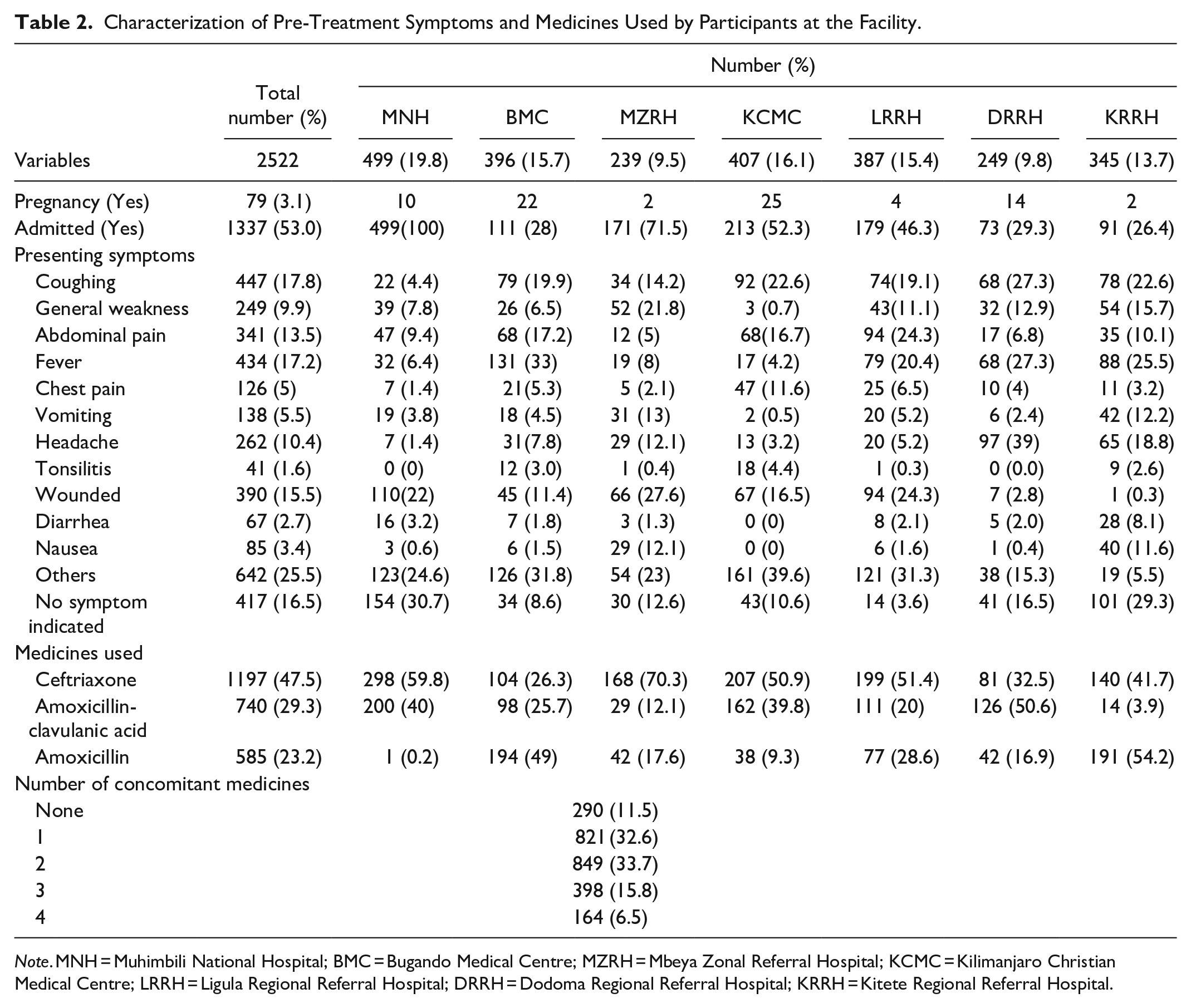
Note. MNH = Muhimbili National Hospital; BMC = Bugando Medical Centre; MZRH = Mbeya Zonal Referral Hospital; KCMC = Kilimanjaro Christian Medical Centre; LRRH = Ligula Regional Referral Hospital; DRRH = Dodoma Regional Referral Hospital; KRRH = Kitete Regional Referral Hospital.
Results showed a slight majority of patients admitted to the hospital 53.0% over the 47.0% who were not. The patients in the cohort had reported several symptoms, with majority 17.8% (n = 447) reporting to have coughing, followed by fever 17.2% (n = 438) and wounds 15.5% (n = 390). At MNH, the majority of the patients 22% (n = 110) presented with wounds. At BMC, the majority of the patients 33% (n = 131) presented with fever. At MZRH, the majority of the patients 27.6% (n = 66) presented with wounds. At KCMC, majority of the patients 22.6% (n = 92) presented with coughing. At LRRH, the majority 24.3% (n = 94) of the patients presented with either abdominal pain or wounds. At DRRH, the majority 39% (n = 97) of the patients presented with headaches. At KRRH, the majority of the patients 25.5% (n = 88) presented with fever (Table 2).
Moreover, 3.1% (n = 79) of the respondents in the cohort were pregnant. KCMC had the highest number of pregnant women 31.6% (n = 25) of all pregnant women in the cohort, followed by BMC 27.8%.
Most of the patients in the cohort 47.5% (n = 1197) were on ceftriaxone. In MNH majority, 59.8% (n = 298) were on ceftriaxone, while at BMC, 49% (n = 194) were on amoxicillin. At MZRH 70.3% (n = 168) were on ceftriaxone, while at KCMC and LRRH over half of the patients 50.9% (n = 207) and 51.4% (n = 199) respectively were on ceftriaxone. Almost half of the patients 50.6% (n = 126) at DH were on amoxicillin-clavulanic acid, and at KRRH majority 54.2% (n = 191) of the patients were on amoxicillin (Table 2 above).
Incidence of Adverse Drug Reactions (ADRs)
In all 7 healthcare facilities, after the use of monitored medicines, out of 2522, only 4.04% (n = 102) from 5 healthcare facilities experienced some ADRs, as indicated in Table 3. This distribution highlights the relatively low incidence of ADRs among the population studied, suggesting that while ADRs do occur, the vast majority of treatments with these medications do not result in adverse events.
Number of Participants Experienced With ADRs After the Use of Ceftriaxone Injection, Amoxicillin-Clavulanic Acid and Amoxicillin per Facility.

Note. LRRH and DRRH had 0 adverse event.
MNH = Muhimbili National Hospital; BMC = Bugando Medical Centre; MZRH = Mbeya Zonal Referral Hospital; KCMC = Kilimanjaro Christian Medical Centre; LRRH = Ligula Regional Referral Hospital; DRRH = Dodoma Regional Referral Hospital; KRRH = Kitete Regional Referral Hospital; AEs = Adverse Events.
Association Between ADRs Occurrence and Medicines Used
The association between the type of medication and the occurrence of ADRs was evaluated using a chi-square test. The medications tested were Ceftriaxone, Amoxicillin–Clavulanic Acid, and Amoxicillin. The results of the chi-square test presented in Table 4 indicate that there is no statistically significant relationship between these medications and the incidence of ADRs at the .05 level of significance (χ2 = 3.922; P = .146).
Occurrence of Adverse Drug Reactions (ADRs) After the Use of Different Medications.

The results in Table 5 compare the types of ADRs associated with the use of ceftriaxone injection, amoxicillin, and amoxicillin-clavulanic acid. Fisher’s exact test was employed to evaluate the statistical significance of differences in ADR occurrences among these medications. The analysis revealed that nausea and vomiting were significantly more common with ceftriaxone compared to amoxicillin and amoxicillin-clavulanic acid, with P-values of .001 and .030, respectively. Similarly, diarrhea and cough were more frequently associated with amoxicillin-clavulanic acid than the other medications, with P-values of .006 and .004 respectively. Other ADRs, such as headache, rashes, chest pain, itching, tachycardia, micturition, and dizziness, did not show statistically significant differences among the medications, with P-values greater than .05.
Comparison Between the Type of ADRs With the Type of Monitored Medicines: Ceftriaxone Injection, Amoxicillin, Amoxicillin-Clavulanic Acid.

*P < .05 suggesting a statistically significant difference.
The comparison of onset latency, event follow-up, and outcomes between ceftriaxone injection, amoxicillin-clavulanic acid tablets/suspension, and amoxicillin capsules is detailed in Table 6. The median onset latency for adverse reactions was 24 h across all 3 medications, with interquartile ranges varying slightly. Among the 3 medicines, serious adverse reactions were reported in 3 cases: 2 following the use of amoxicillin-clavulanic acid and 1 after the use of amoxicillin. Fisher’s exact test was employed to assess the significance of differences in the seriousness of adverse reactions, prolonged hospitalizations, and hospitalizations between the medications. The results indicate that there was a statistically significant difference in the rate of hospitalizations (P = .024). In this study, prolonged hospitalization is a serious adverse reaction corresponding to any untoward medical occurrence that at any dose results in the prolongation of existing hospitalization. 19 Additionally, event outcomes such as recovery status showed significant differences, with a P-value of <.001. Most patients (63.3%) recovered from the adverse reactions, while 24.5% received treatment for their ADRs.
Comparison of On Set Latency, Event Follow-up and Event Outcomes Between Ceftriaxone Injection, Amoxicillin-Clavulanic Acid Tablets and Amoxicillin Capsules.
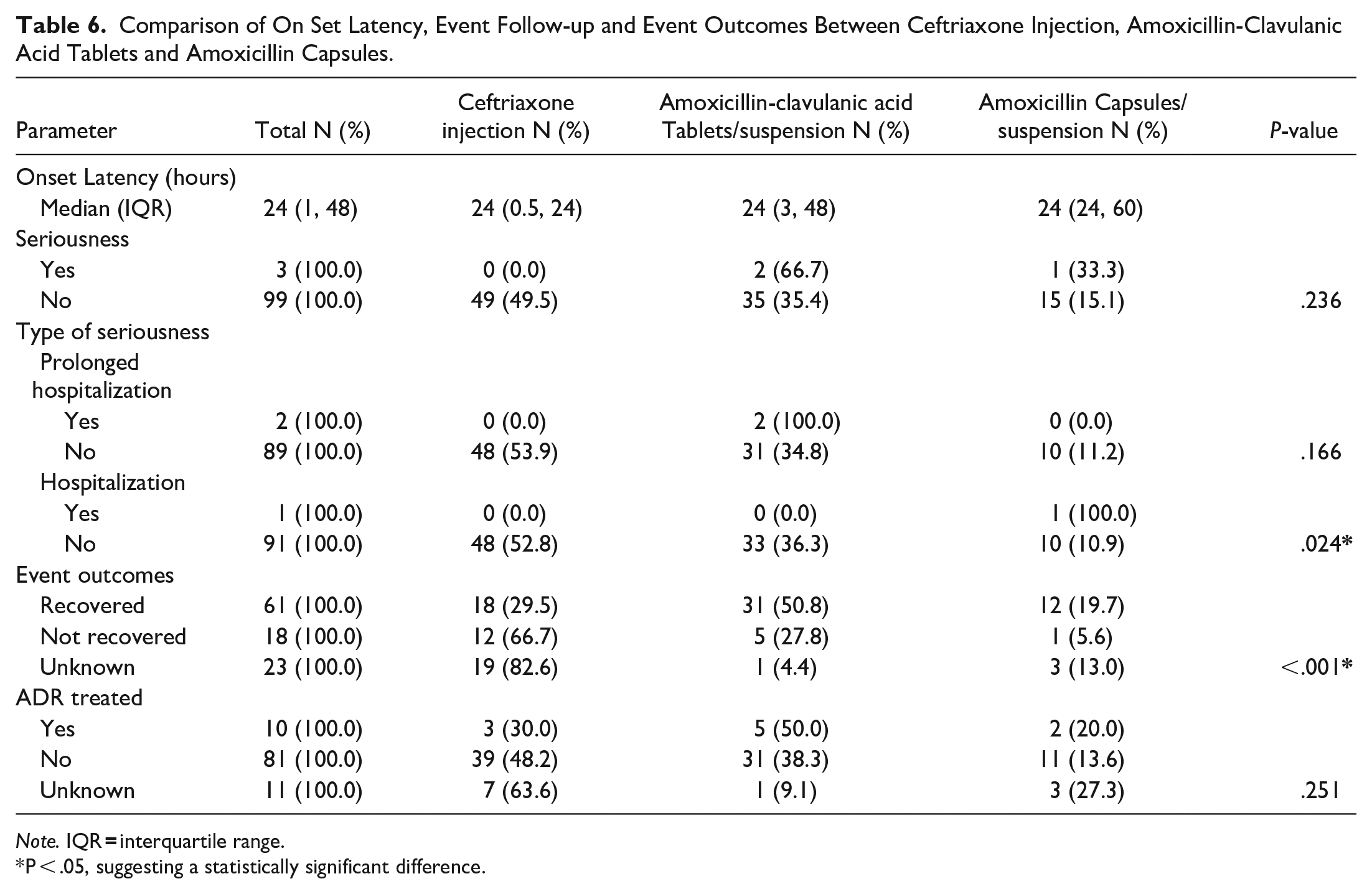
Note. IQR
P < .05, suggesting a statistically significant difference.
Only as few as 5 pregnant women experienced ADRs with a notable occurrence of tachycardia in 2 cases following ceftriaxone injection. Other ADRs were abdominal pain and diarrhea, weakness, blurred vision, ulceration of the mouth, sores and white spots on the lips and in the mouth, and facial swelling. One pregnant woman was reported from BMC (amoxicillin-clavulanic acid) and 4 from KCMC (2 ceftriaxone, 1 amoxicillin-clavulanic acid, and 1 amoxicillin).
Factors Influencing the Incidence of Adverse Drug Reactions
Univariate and multivariate binomial logistic regression analyses were performed to ascertain the effects of gender, age group, number of concomitant medications, and admission status on the likelihood of participants experiencing ADRs (as shown in Table 7). The logistic regression model included the following variables: gender (male as reference), age group (under 1 year as reference, with other groups being 1-9, 10-19, 20-59, and 60 years and above), number of concomitant medications (none as reference, with categories for 1, 2, 3, and 4 or more medications), and admission status (not admitted as reference). The Hosmer and Lemeshow test indicated a good fit (P > .446), and the ROC analysis yielded an area under the curve (AUC) of 0.78, demonstrating adequate predictive performance.
Results of Univariate and Multivariate Logistic Regression Analysis of Significant Influencing Factors for the Occurrence of an Adverse Drug Reaction.

Note. OR = odds ratio; CI = confidence interval; Ref = reference group.
P < .05, suggesting statistical significance.
Of the variables examined in the logistic regression analysis, 3 showed statistically significant associations with the incidence of adverse drug reactions (ADRs): age group 10 to 19, and the number of concomitant medications taken—specifically 1 and 3 medications. Individuals in the age group 10 to 19 years had significantly lower odds of experiencing ADRs than those under 1 year-old, with an adjusted odds ratio (OR) of 0.385, 95% CI [0.163, 0.911], P = .030. For patients taking one concomitant medication, the odds of experiencing ADRs were reduced, with an adjusted OR of 0.386, 95% CI [0.158, 0.941], P = .036. Similarly, taking 3 concomitant medications was associated with reduced odds of ADRs, with an adjusted OR of 0.493, 95% CI [0.248, 0.980], P = .044. In contrast, gender, other age groups, admission status, and taking 2 or 4 concomitant medications were not significantly associated with the incidence of ADRs.
Individual Case Safety Reports (ICSRs) From the Vigiflow/VigiLyze Database
Analysis of AEs extracted from WHO-Vigiflow/VigiLyze database for Ceftriaxone Injection, Amoxicillin capsules/suspension, and amoxicillin-clavulanic acid tablets was conducted. The demographic characteristics of those reported with AEs are shown in Table 8. The comparison of adverse events in the ICSRs between the 3 medicines is shown in Table 9.
Demographics Characteristics of the Participants Reported in the ICSRs With ADRs of Ceftriaxone Injection, Amoxicillin Capsules/Suspension and Amoxicillin-Clavulanic Acid Tablets (N = 341).

Note. IQR = interquartile range between the type of ADRs with the type of monitored medicines.
*Health facilities other than the studied facilities.
Comparison Between the Type of ADRs in the ICSRs With Ceftriaxone Injection, Amoxicillin Capsules and Amoxicillin-Clavulanic Acid Tablets.
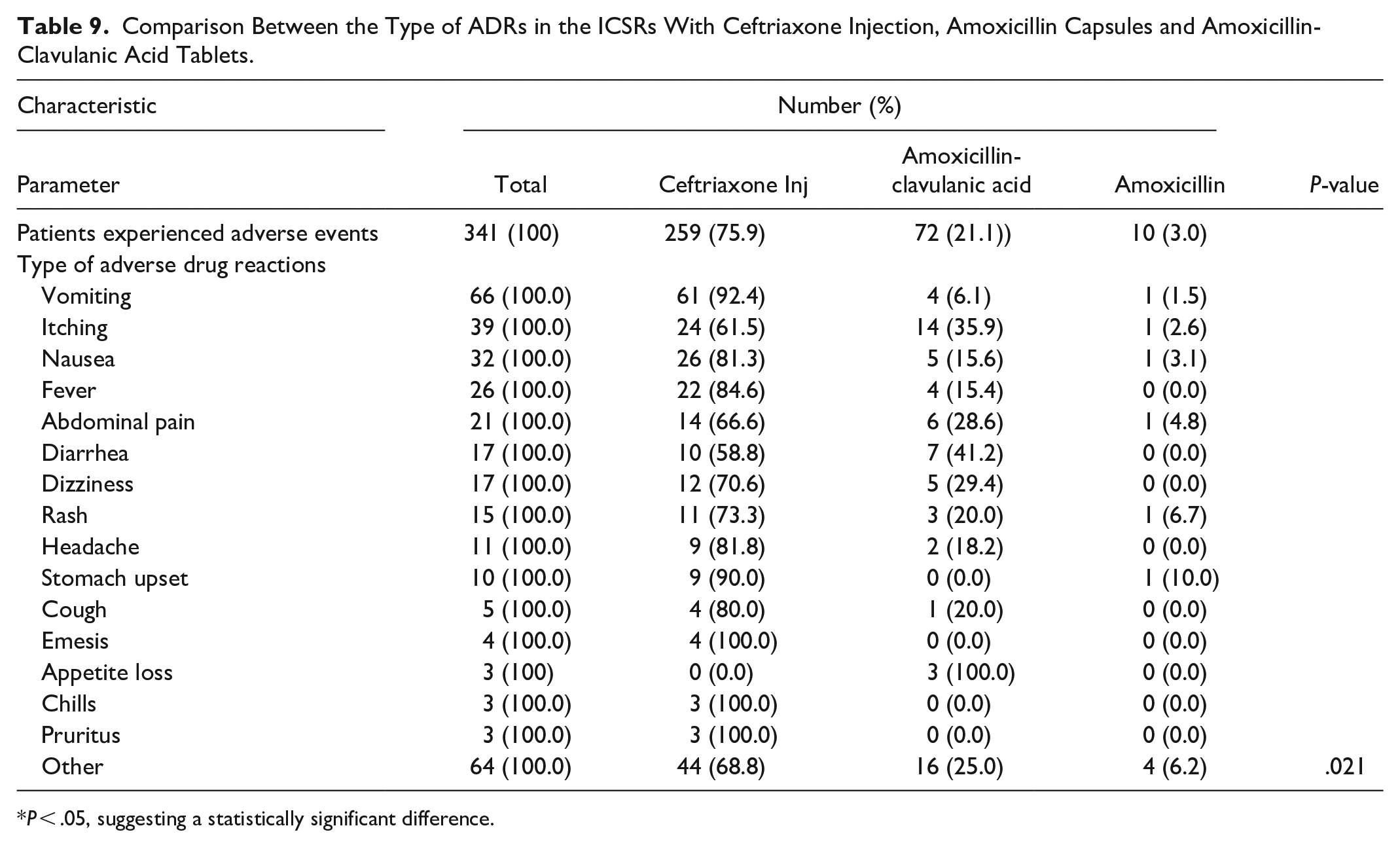
P < .05, suggesting a statistically significant difference.
The comparison between the type of ADRs reported in individual case safety reports (ICSRs) for ceftriaxone injection, amoxicillin capsules and amoxicillin-clavulanic acid tablets is detailed in Table 9. Fisher’s exact test was used to determine the statistical significance of differences in ADR occurrences among monitored medicines. The results indicated that, there is a statistically significant difference (P-value < .05) in the distribution of ADRs among the 3 antibiotics. This suggests that the type of ADRs experienced by patients may differ depending on the antibiotic they are taking. Ceftriaxone was associated with a higher incidence of vomiting (92.4%), nausea (81.3%) and fever (84.6%) compared to amoxicillin-clavulanic acid and amoxicillin with statistically significant P-values of .021. Additionally, ceftriaxone showed a significantly higher incidence of diarrhea (58.8%) and dizziness (70.6). Overall, ceftriaxone injection had a broader range of ADRs compared to the other medicines, suggesting a need for careful monitoring.
Other events due to ceftriaxone injection were 44, including anuria (n = 2), confusion (n = 2), seizure (n = 2), skin irritation (n = 2), swelling (n = 4), heavy sweating (n = 2), tachycardia, increased blood urea, and difficulty in breathing. Other events included constipation, dysarthria, armpit pain, dyspnea, eosinophilia, fatigue, gum bleeding, Stevens-Johnson syndrome, hyperhidrosis, increased serum creatine, metallic taste, inflammation, thirst, thrombocytopenia, thrombocytosis, loss of taste, tinnitus, vertigo, edema, pale skin, excessive perspiration, pulmonary congestion, reflux gastritis, salivary hypersecretion, and tachypnea.
For amoxicillin-clavulanic acid, 16 other reported events included acute allergic urticaria, bloating, constipation, difficulty in breathing (n = 2), demyelinating polyneuropathy, hives, increased heart rate, palpitations (n = 2), tremor, toothache, tiredness (n = 2), and weakness. Amoxicillin included 4 events namely loose stools, pancytopenia, swelling, and vaginal infection.
The seriousness and event outcomes between ceftriaxone injection, amoxicillin-clavulanic acid and amoxicillin capsules are detailed in Table 10. Among the 341 individual case safety reports (ICSRs), 12 cases were identified: 66.7% were associated with ceftriaxone, and 16.7% each were linked to amoxicillin and amoxicillin-clavulanic acid. Fisher’s exact test was used to determine the statistical significance of differences in the seriousness of adverse events and event outcomes between medications. Results suggest that there is a statistically significant difference in the seriousness (P = .016) and type of seriousness (P = .044) of adverse events experienced by patients taking the different medications. However, the results for event outcomes were not statistically significant (P = .211).
Comparison of Seriousness and Event Outcomes Between Ceftriaxone Injection, Amoxicillin Capsules and Amoxicillin-Clavulanic Acid Tablets.

P < .05, suggesting a statistically significant difference.
MedDRA System Organ Classification (SOC) Affected by Adverse Drug Reactions (ADRs)
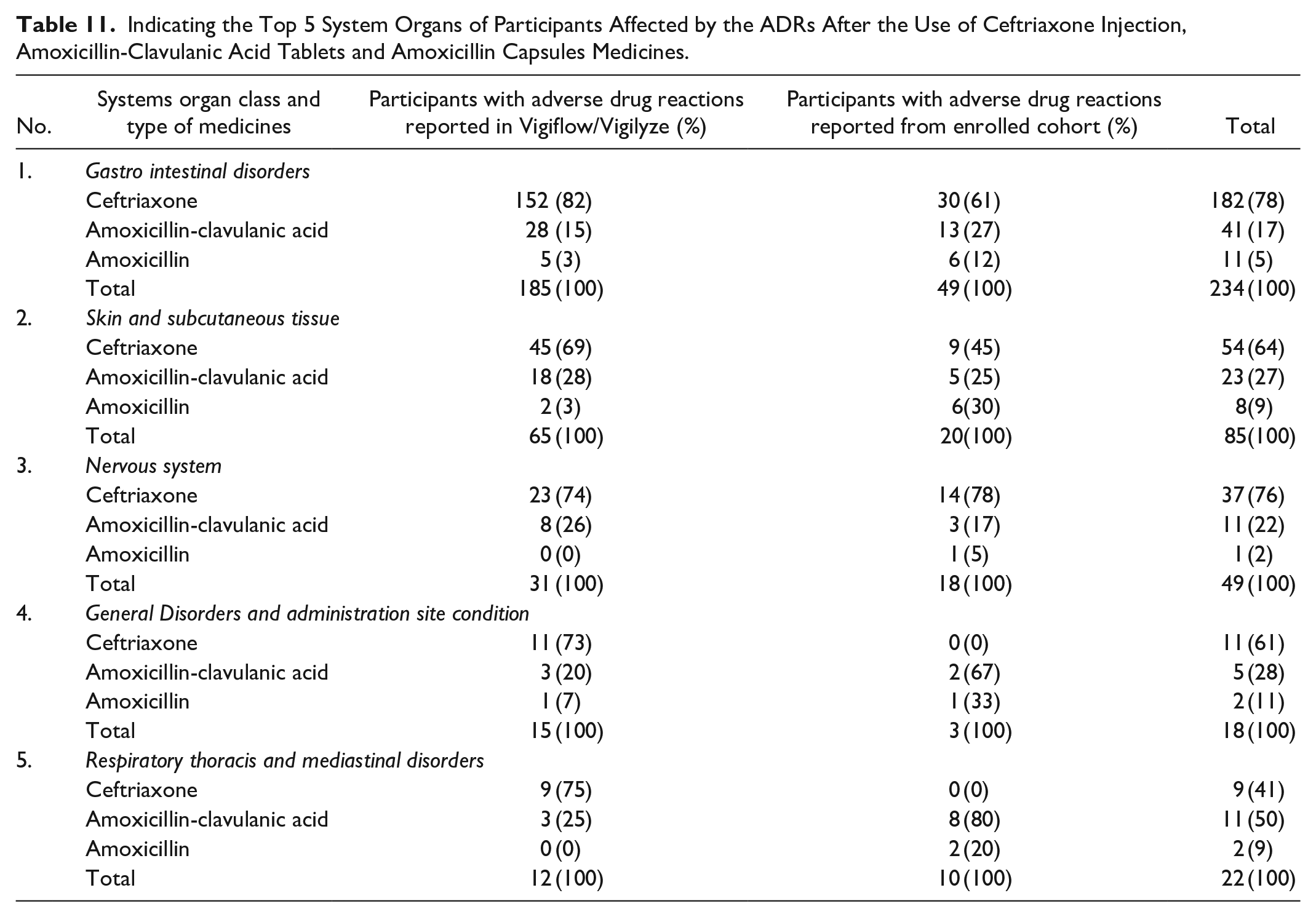
All enrolled and Vigiflow/Vigilyze participants were grouped according to the affected systemic organ as classified in MedDRA system organ classes (SOC). A total of 14 out of 27 MedDRA system classified organs were identified affected by ADRs after either the use of ceftriaxone, amoxicillin-clavulanic acid or amoxicillin or both 3 or 2 of them. The participants were affected in gastrointestinal, skin and subcutaneous tissue, nervous system, general disorders and administration site conditions, respiratory, thoracic and mediastinal systems, cardiac, renal and urinary, and blood and lymphatic systems. Other affected system organs were ear and labyrinth, immune system, investigations, metabolism and nutrition disorders, musculoskeletal and connective tissue, and psychiatric. The number of participants affected by ADRs in the top 5 system organ classes were as indicated in Table 11.
Indicating the Top 5 System Organs of Participants Affected by the ADRs After the Use of Ceftriaxone Injection, Amoxicillin-Clavulanic Acid Tablets and Amoxicillin Capsules Medicines.
Causal Relationship Between Medicines Used and the ADRs
The causality assessment results revealed that, all reported AEs were ADRs. Out of all reported ADRs from both enrolled participants and Vigiflow/VigiLyze, about 90.3% of reported ADRs were possible, 9%, plausible and 0.7% certain related to the use of medicines. For a certain category, 2 patients who used ceftriaxone, experienced vomiting, nausea, and abdominal pain, while one who used amoxicillin-clavulanic acid experienced vomiting only. The re-challenge and de-challenge were conducted for all 3 patients. Furthermore, the positive de-challenge was observed in 40 patients, who used ceftriaxone (27), Amoxicillin-clavulanic acid (14), and amoxicillin (2). The possible categorization was due to the fact that participants under treatment were also using other concomitant medicines, thus, events could also be explained by other factors.
Classification of Frequency of Occurrence of ADRs
The results indicated that, for the enrolled cohort 2522, a total of 49 ADRs were reported among the 1197 participants who were exposed to Ceftriaxone, representing a prevalence of 4.1% (49/1197). The most frequent category for reported ADRs was gastrointestinal disorders (30 cases), accounting for 61.2% (30/49) of all ADRs caused by the use of ceftriaxone. This translates to common occurrence with a rate of 2.50% (30/1197) within the ceftriaxone-exposed group. Skin and subcutaneous disorders followed with 9 cases with a rate of 0.75% (9/1197). This rate indicates that, skin and subcutaneous disorders is uncommonly disorders to occur. Nervous system disorders were reported in 14 cases with a rate of 1.17% within the ceftriaxone-exposed group. This means is a common category to occur. Three cases with a rate of 0.25% involved cardiac disorders, which is uncommon to occur. No ADRs were reported within the general disorders or respiratory thoracic categories.
For Co-Amoxiclav, a total of 37 ADRs were reported (37/740 = 5.00% prevalence). Gastrointestinal disorders were the most common (13 cases, 1.76%), followed by skin and subcutaneous (5 cases, 0.68%), nervous system disorders (3 cases, 0.41%), general disorders (2 cases, 0.27%), respiratory thoracic disorders (8 cases, 1.08%), renal & urinary disorders (2 cases, 0.27%) and cardiac disorder (1 case, 0.14%). These rates indicate that gastrointestinal and respiratory thoracic disorders are common, while skin and subcutaneous, nervous system, renal & urinary, general disorders and cardiac disorders are uncommon to occur.
For Amoxicillin, a total of 16 ADRs were reported (16/585 = 2.74% prevalence). Similar to Amoxiclav, the most frequent category was gastrointestinal disorders (6 cases, 1.03%) and skin and subcutaneous disorders (6 cases, 1.02%), respiratory thoracic disorders (2 cases, 0.34%), nervous system disorders (1 case, 0.17%), and general disorders (1 case, 0.17%). This means gastrointestinal and skin and subcutaneous systems are commonly affected by the use of amoxicillin while respiratory thoracic disorders, nervous system and general disorders are uncommon to occur. Cardiac disorders and renal and urinary are rare. The comparison ratios of ADRs frequencies as specified by manufacturer categories have been attached as Appendix 1.
However, the frequency as per manufacturer categories for the participants from the Vigiflow/vigibase was not calculated because, in the database there was no reported number of populations utilized the medicines per specific area. Currently, Tanzania has no utilization system to capture a total number of patients who used the specific medicine at a specific time in a specific facility or region.
Discussion
In this study, the mixed prospective-retrospective cohort method was used to assess the safety profile of selected commonly used registered anti-infective chiral medicines circulating in the market. The monitored chiral medicines were amoxicillin capsules and suspensions, a combination of amoxicillin and clavulanic acid (co-amoxiclav) tablets/suspension, injections, and ceftriaxone injections. Our results indicated that the majority of enrolled patients were between 20 and 59 years of age. This indicates that the majority of patients fall within the working-age range.
Our study revealed that most of the enrolled participants were treated with ceftriaxone injections. This indicates that ceftriaxone injection is more prescribed in healthcare facilities than amoxicillin and co-amoxiclav medicines. These results are in-line with those studies conducted in tertiary hospitals in Tanzania and other countries.20 -22 In addition, amoxiclav was the most commonly used compared to amoxicillin alone. This is in-line with some studies that showed that the use of a combination of amoxicillin and clavulanic acid is preferred and prescribed for the treatment of bacterial infections compared with amoxicillin alone.23,24
Our study also showed that, the overall incidence of ADRs was low at 4.0%, suggesting a generally favorable safety profile for these medications. Other studies on similar medications have reported varying ADR rates, but generally manageable risks. For example, one study reported a slightly higher ADR incidence of 7.3% among a similar demographic, which could be due to differences in drug formulations or patient health statuses. 25 These findings are in-line with some other studies which indicated the medicines are safe and few patients may experience ADRs.25 -27
Moreover, our study indicated that in all cohorts (enrolled participants and ICSRs in the Vigiflow/VigiLyze) more than 50% of ADRs were reported in females. However, there was no significant difference between them. These findings were similar to those studies conducted in different countries such as India, Japan, Iran, and the UAE, which reported that females had more AEs than males.28,29 These results have also been evidenced in the study conducted on the global post-marketing surveillance data on spontaneous reports which indicated that women report more ADRs than men. 30 However, in this study, the case is not the same in the use of amoxicillin, where more males reported AEs than females. The reason why females reported more ADRs is still under today’s pharmacovigilance discussion. Other studies indicated that, it might be due to gender-related differences in physiological, pharmacokinetic, immunological, and hormonal factors.28,29 Other studies indicated that, it might be due to the fact that, during the conduct of clinical trials, more men are often recruited compared to women. 31 Another study, reported that, it might be due to the sex differences in how the medicines work in the body. 32 While another study reported that, among other factors it might be due to the epidemiological population distribution. 33 Under this study, we recommend more studies to be conducted globally to investigate why more women are observed with more ADRs compared to women. This will help to reduce potential risks of having ADRs to women.
Logistic regression analysis revealed that age and the number of concomitant medications significantly affect the likelihood of experiencing ADRs. Patients aged 10 to 19 years had significantly lower odds of ADRs compared to those under 1 year, and patients on 1 to 3 concomitant medications had reduced odds of ADRs. This contrasts with the study which found there were no significant age-related differences in ADR occurrences. 34 Furthermore, the study suggests a potential protective effect associated with certain numbers of medications, a finding that was similarly discussed in a polymedication study34,35 which highlighted that polypharmacy could either increase or decrease ADR risks based on medication interactions. The study’s findings have significant implications for clinical practice and suggest the potential for targeted patient education and monitoring strategies. This could particularly benefit younger and polypharmacy patients. Additionally, healthcare providers might consider these factors when prescribing these medications to optimize safety and effectiveness.
In this study, approximately half of the reported ICSRs of enrolled participants had experienced ADRs due to the use of ceftriaxone injection, followed by amoxicillin-clavulanic acid and amoxicillin. The same tendency was observed in the ICSR reported from Vigiflow/VigiLyze after the use of these medicines, where approximately 3-quarters of ADRs were caused by ceftriaxone injection. The high number of ADRs caused by ceftriaxone injections is in-line with other studies,29,36 conducted in Iran that reviewed ADRs for 10 years and in China respectively. Due to the high utilization and existence of ADRs, there is still a need for this medicine to be actively monitored.
Our study revealed that, the use of these 3 medicines can cause ADRs in 14 of 27 MedDRA SOC. Ceftriaxone can cause events in 13 systemic organs, amoxicillin-clavulanic acid in 10 SOC, and amoxicillin in 7 SOC. The results revealed that ADRs caused by ceftriaxone injection mostly affected the gastrointestinal system with significantly common nausea and vomiting. Other ADRs included abdominal pain, diarrhea, stomach upset, constipation, and reflux gastritis. This is in contrast with the summary of product characteristics (SmPC) provided by the manufacturer which showed that ceftriaxone may commonly cause diarrhea and loose stools, whereas nausea and vomiting are uncommon. 16 In the SmPC, the manufacturer declared that it is unknown if the medicine can cause pancreatitis, stomatitis, or glossitis. 16 In a randomized, double-blinded multicentre trial, ceftriaxone was reported to be safe and well-tolerated.37,38 The most commonly reported ADRs were injection site reactions, diarrhea, and skin rashes.37,39 ADRs to ceftriaxone have been reported to occur in 1% to 10% of patients, with hematologic signs of thrombocytosis and eosinophilia being the most common reported.40,41
Other affected systemic organs were, skin and subcutaneous tissue disorders, nervous system disorders, general disorders and administration site conditions, respiratory, thoracic, and mediastinal disorders, and cardiac disorders. These results are also in contrast with the manufacturer SmPC which indicated that, the medicine does not affect the cardiac system,
16
however, our findings revealed that, the medicine can uncommonly affect the cardiac system by causing tachycardia. Therefore, tachycardia is a new ADR reported in 4
In addition, there are some potential ADRs in which their frequencies were not declared by manufacturers, such as the Stevens-Johnson Syndrome, which has also been reported in some studies.29,36,45,46 In SmPC, 16 the Stevens-Syndrome event was reported with an unknown frequency. It was also documented in a study that estimates varying degrees (1-10 patients per million) of incidence in the U.S.A, France, Japan, and Germany, but little is known about its incidence in Africa. 47
The administration of amoxicillin also resulted in some ADRs. Our findings indicated that, the most common occurrence of ADRs affected gastrointestinal, and skin and subcutaneous tissue systems. This is in-line with manufacturer SmPC, 18 where gastrointestinal, skin and subcutaneous tissue disorders were the only ADRs with common occurrence. However, in our study, there were 3 identified ADRs in the use of amoxicillin that affected 2 systemic organs; respiratory, thoracic and mediastinal disorders and general disorders. These organs were not reported in the SmPC 18 that can be affected by the ADRs.
Moreover, the study indicated that the fixed-dose combination of amoxicillin and clavulanic acid had significantly more ADRs compared with amoxicillin alone. These results are in line with those of the comparison study, which indicated that the ADRs for amoxicillin were less than those for amoxicillin in combination with clavulanic acid.48,49 The most commonly reported ADRs after the use of Amoxicillin-clavulanic acid affected the gastrointestinal system, followed by skin and subcutaneous system organs. Gastrointestinal disorders were reported as nausea, vomiting, and diarrhea. In comparison with SmPC, 17 diarrhea was a very common ADR, while skin disorders were reported as an uncommon occurrence. No new reported ADR was identified with the use of Amoxicillin-clavulanic acid. These results are in-line with those of previous studies, which showed that amoxicillin-clavulanic acid is associated with a higher risk of gastrointestinal complications and hepatotoxicity than amoxicillin.50 -52 The most reported ADRs for amoxicillin and amoxicillin-clavulanic acid include diarrhea, rash, vomiting, nausea, rash, urticaria, pruritus, pharyngitis, bronchitis, rhinitis, fever, angioedema, dyspepsia, dyspnea, dizziness, abdominal pain, otitis media, allergy.51,53
The results also revealed that, approximately 3% of the enrolled participants in the cohort were pregnant women. Out of these, 6.3% were reported with ADRs. Of note, 2 of the ADRs reported among those who used ceftriaxone injections were tachycardia. Ceftriaxone, a beta-lactam antibiotic medicine, has not been studied in pregnant women; however, it has been administered to pregnant women if the benefits to pregnant women outweigh any possible risks to the unborn child.16,54
The study indicated that, though more ADRs were observed in patients who used ceftriaxone injection; there was no serious reported ADR. This is different from a study which indicated that amoxicillin had more ADRs compared to others. 55 Moreover, only a few serious ADRs were reported among patients who used penicillin medicines. These serious ADRs included 2 cases of prolonged hospitalization after the use of Amoxicillin-clavulanic acid tablets and one case of hospitalization after the use of amoxicillin capsules. The majority of the patients who experienced ADRs (63.3%) recovered from the ADRs at the time of reporting. Approximately 1-quarter of patients received treatment for ADRs. This indicates that amoxicillin and amoxicillin in combination with clavulanic acid are still safe. However, amoxicillin alone is safer than amoxicillin combined with clavulanic acid. These findings have also been reported in other studies.52,56
In addition, our results indicated that females had been more enrolled (58.3%) compared to males. This was also observed in each healthcare facility. The results indicate that women use more healthcare services than men do, as previously reported in other studies. 57 This might be due to differences in treatment-seeking behavior between women and men who are willing to engage in healthcare. 58
Limitations and Strengths of the Study
Our study has strengths and limitations, one of the strengths is that, there were no missing data. All data for the enrolled participants and the vigiflow/vigiLyze were verified, validated, and considered during the analysis. In addition to the fact that, data for the enrolled participants were collected from 7 pharmacovigilance centers, the study also captured other data on the same medicines reported directly in the vigiflow/vigiLyze during the same period to supplement the collected data. Thus, the events reported for the same medicines from other healthcare facilities were also considered. The limitations were that the reported events from this study were those reported using the spontaneous method under the SSSSM program; thus, laboratory testing before and after medications was not performed. Causality assessment also considered the presence of other factors contributing to the occurrence of ADRs; hence, most of the reactions were possibly related to medication use. Another limitation is that, Tanzania currently has no medicines utilization system to capture the total number of patients who used a specific medicine at a specific time in a specific facility among the specific population in the region or country. Therefore, the occurrence frequency of ADRs for the data reported in Vigiflow/vigilyze was not calculated.
Conclusion
In this study, approximately 90% of all reported ADRs were possible ADRs related to the monitored medicines, with very few proportions of plausible and certain. Ceftriaxone injection caused more ADRs than the other medicines. Amoxicillin-clavulanic acid was associated with more ADRs than amoxicillin alone. Although, these monitored medicines are generally well-tolerated, they may lead to ADRs. Nausea and vomiting were significantly more common with ceftriaxone compared to other medicines. Whilst, diarrhea and cough were more frequently associated with co-amoxiclav than other medicines. The tachycardia, a new potential ADRs can be caused after the use of ceftriaxone injection in patients including pregnant women. This underscores the importance of comprehensive monitoring of ADRs in clinical practice to improve patient safety and enhance overall treatment outcomes.
Besides the fact that, the safety profile of the medicines is still maintained; active studies are recommended for the ceftriaxone injection for further investigation of the rare and uncommon events reported during the study.
Footnotes
Appendix
Comparison ratios of ADRs frequency as specified by manufactures categories.
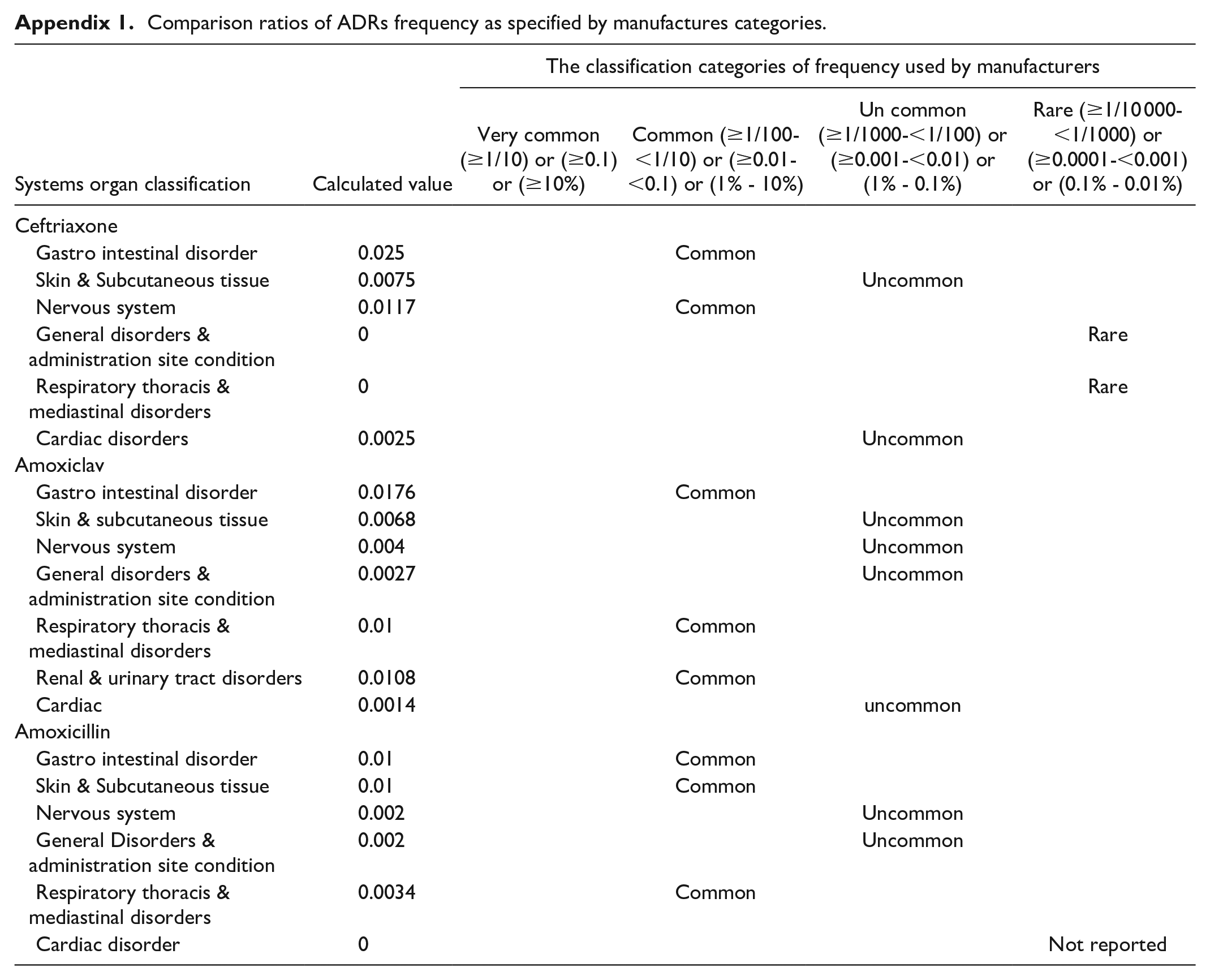
| Systems organ classification | Calculated value | The classification categories of frequency used by manufacturers | |||
|---|---|---|---|---|---|
| Very common (≥1/10) or (≥0.1) or (≥10%) | Common (≥1/100-<1/10) or (≥0.01-<0.1) or (1% - 10%) | Un common (≥1/1000-<1/100) or (≥0.001-<0.01) or (1% - 0.1%) | Rare (≥1/10 000-<1/1000) or (≥0.0001-<0.001) or (0.1% - 0.01%) | ||
| Ceftriaxone | |||||
| Gastro intestinal disorder | 0.025 | Common | |||
| Skin & Subcutaneous tissue | 0.0075 | Uncommon | |||
| Nervous system | 0.0117 | Common | |||
| General disorders & administration site condition | 0 | Rare | |||
| Respiratory thoracis & mediastinal disorders | 0 | Rare | |||
| Cardiac disorders | 0.0025 | Uncommon | |||
| Amoxiclav | |||||
| Gastro intestinal disorder | 0.0176 | Common | |||
| Skin & subcutaneous tissue | 0.0068 | Uncommon | |||
| Nervous system | 0.004 | Uncommon | |||
| General disorders & administration site condition | 0.0027 | Uncommon | |||
| Respiratory thoracis & mediastinal disorders | 0.01 | Common | |||
| Renal & urinary tract disorders | 0.0108 | Common | |||
| Cardiac | 0.0014 | uncommon | |||
| Amoxicillin | |||||
| Gastro intestinal disorder | 0.01 | Common | |||
| Skin & Subcutaneous tissue | 0.01 | Common | |||
| Nervous system | 0.002 | Uncommon | |||
| General Disorders & administration site condition | 0.002 | Uncommon | |||
| Respiratory thoracis & mediastinal disorders | 0.0034 | Common | |||
| Cardiac disorder | 0 | Not reported | |||
Acknowledgements
The author would like to thank the Tanzania Medicines and Medical Devices Authority (TMDA) management for allowing the main author to conduct this safety study. Permission to use pharmacovigilance centers established by the authority to collect data and some data from the WHO vigiflow/VigiLyze database (data owned by TMDA). We would also like to acknowledge the management of different healthcare facilities and HCP from pharmacovigilance centers and regional hospitals for accepting and participating in the study. The authors would like to acknowledge the European and Developing Countries Clinical Trials Partnership (EDCTP2), which funded the PhD studies.
Author Contributions
KWM contributed to conception, design, funding acquisition, project administration, design, data collection and validation, analysis, writing, and approval of the manuscript. EMB contributed to the data analysis, writing, and approval of the manuscript. BAM contributed to data validation and analysis and writing and approval of the manuscript for publication. CM contributed to the data validation and analysis, writing, and approval of the manuscript for publication. AMF and WS contributed to review and approval of the manuscript for publication. DB and EPM contributed to data collection, review, and approval of the manuscript and DHS. MCS, ADS, and EAK supervised, wrote, and approved the manuscript for publication. All authors commented on the previous versions of the manuscript. All the authors have read and approved the final manuscript.
Data Availability
The datasets generated and analyzed during the study are available at the TMDA office. All data are available with permission from the Tanzania Medicines and Medical Devices Authority (TMDA).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was part of the EDCTP2 Program supported by the European Union partly under the SMERT project (CSA2016ERC-1432) and ASCEND project (CSA2019ERC-2683); and partly under TMDA. The funder had no role in study design, collection, analysis, or manuscript writing. However, the report of the study and progress of what has been done was of interest to the funder to ensure that the study results are useful in protecting public health.
Ethical Consideration
Ethical clearance was granted by the National Institute for Medical Research, Tanzania (certificate number NIMR/HQ/R.8 a/vol. IX/3086) and the Institutional Review Board of Muhimbili University of Health and Allied Sciences (MUHAS) (certificate number DA.282/298/01.C). Data collection request letters were sent by the authors to all participating hospitals. Approval letters were obtained from each participating pharmacovigilance center before the start of the study at the respective hospital.
A regulatory requirement for all patients and healthcare workers is to report any ADR(s) caused by the use of any medicine(s). However, in this study, patients who used the selected chiral medicines were identified and filled-in consent form to participate in the study, and requested to report AEs/ADR(s) (if any). The patients were followed-up for 14 days after starting the use of chiral medicines. Physical follow-up was performed in wards (for admitted patients) or via phone calls for discharged/outpatients.
The research methods were conducted in accordance with relevant guidelines and regulations pertaining to the safety monitoring of medicines in Tanzania.

