Abstract
Apnea and poor respiratory drive increase the risk of extubation failure (EF) and prolonged invasive mechanical ventilation (IMV) in preterm neonates (pre-nates) with respiratory distress. Caffeine citrate (CC) is often prescribed for pre-nates in doses of 5–10 mg/kg in 24 h. This study aimed to evaluate the most effective dosage regimen (5 mg/kg/day vs >5-10 mg/kg/day) to prevent apnea and EF with minimal caffeine-associated potential side effects (CC-APSEs) in pre-nates. This one-year retrospective cohort study included all the eligible neonates admitted to NICU and received CC-therapy till 28 days of life (DOL) or discharge. Based on CC-daily dose formed LD-caffeine-group (5 mg/kg/day) and HD-caffeine-group (>5-10 mg/kg/day). Antenatal, prenatal, and postnatal characteristics, CC-regimen, comorbidities, and CC-APSEs were compared between the groups. Predictors of apnea and EF were analyzed through logistic regression. There were 181 and 72 neonates in the LD and HD-caffeine-groups respectively. In HD-caffeine-group daily CC-dose was 7 to 7.5 mg/kg/day in 93% of neonates and >7.5 to 10 mg/kg/day in only 7%. Significantly fewer neonates experienced apnea and EF in the HD-caffeine-group till 28DOL or discharge. This difference was even greater in the subgroup of ≤28 weeks GA (15.6% vs 40.0%; P < .01). In HD-caffeine-group the incidence of severe/moderate-BPD was significantly lower and the frequency of CC-APSEs was higher. Multivariate analysis showed that; the smaller the GA higher the risk of apnea (AOR = 0.510, 95% CI 0.483-0.999) and EF (AOR = 0.787, 95% CI 0.411-0.997). The HD-caffeine was inversely associated with developing apnea (AOR = 0.244, 95% CI 0.053-0.291) and EF (AOR = 0.103, 95% CI 0.098-2.976). IMV-duration before extubation (AOR = 2.229, 95% CI 1.672-2.498) and severe/moderate-BPD (AOR = 2.410, 95%CI 1.104-2.952) had a high risk of EF. Initiating early HD-caffeine may prevent apnea and extubation failure in preterm neonates. Optimization of caffeine initiation time and dosages can be a safe and feasible approach to decrease the burden of neonatal respiratory morbidities.
Keywords
Caffeine citrate is often prescribed for preterm neonates for managing apnea in the daily dose of 5-10 mg/kg in 24 h but the most effective dosage regimen with minimal adverse effects is controversial.
This study contributes to defining a more precise prophylactic daily dose of caffeine citrate to prevent apnea and extubation failure in neonates with minimal adverse effects.
This research study will help practitioners adopt the safest possible practices in neonatology and make policies accordingly.
Background
Preterm neonates have immature respiratory control mechanisms and more than 85% of neonates born at gestational age (GA) ≤34 weeks, experience a developmental disorder known as apnea of prematurity (AOP).1 -3 Preterm neonates have a higher probability of developing retinopathy of prematurity (ROP) and neurodevelopmental consequences due to apnea-associated intermittent hypoxemia.4,5 Likewise, AOP and poor respiratory drive increase the risk of extubation failure (EF) and prolonged invasive mechanical ventilation (IMV) in newborns with respiratory distress. 6
Caffeine citrate (CC) has been recognized as a standard for managing AOP due to a better therapeutic index, comparable enteral bioavailability, and longer half-life.7 -9 Its use in preterm neonates is related to shorter IMV dependence and lower chances of EF. 7 Neonatal studies reported a reduced risk of bronchopulmonary dysplasia (BPD) and an improved rate of very premature neonates’ survival with no neurological disabilities at 18 to 21 months. 10 Apnea of prematurity might persist beyond 37 weeks postmenstrual age (PMA) in preterm neonates born at GA <28 weeks. 11
Caffeine citrate stimulates the medullary respiratory center in premature neonates, enhancing respiratory functions by blocking adenosine receptors, increasing CO2 sensitivity in chemoreceptors, and boosting diaphragmatic strength. Additionally, it stimulates catecholamine release, raises cardiac output, and improves oxygenation, reducing apnea and anoxic episodes. 12 Early CC-therapy can lead to improved neonatal outcomes by shortening IMV-duration, 13 reducing weaning failure and reintubation rates,14,15 and lowering the rate of BPD, ROP, and patent ductus arteriosus (PDA).16 -19 Hence, CC is often prescribed for preterm neonates until they reach a PMA of 35 to 37 weeks in doses of 5 to 10 mg/kg in 24 h.3,5,7,20,21
The current standard CC-therapy involves a loading dose of 20mg/kg following a daily dose of 5 mg/kg/day. 22 However, with this regimen, EF was reported in a third of preterm neonates. 23 Determining the most effective dosage regimen with minimal adverse effects is a crucial scientific concern. The 2019 European RDS guidelines recommend a standard loading dose of 20 mg/kg, followed by 5 to 10 mg/kg/day. 24 Yet, there is limited evidence of the effectiveness and safety of this regimen.
Consequently, this retrospective cohort study aimed to evaluate the potential influence on the occurrence of apnea and EF in premature neonates exposed to different maintenance doses (5 mg/kg/day vs >5-10 mg/kg/day) of CC-therapy for managing AOP and obtaining other respiratory and neurological benefits in preterm neonates. 25 In addition, to evaluate the onset and persistence of caffeine-associated potential side effects (CC-APSEs) and length of hospital stay.
Methods
This study was performed in a tertiary care university hospital. The study site has the facility of a 24-bed multispecialty tertiary care NICU. About 1200 neonates are admitted annually with the influx of very preterm high-risk newborns from all over the country. All the preterm neonates (GA ≤ 37 weeks) were included in the initial cohort who were admitted to the NICU, and Step-down unit and they were administered intravenous or oral CC for managing AOP and obtaining other respiratory and neurological benefits during the study period (September 2021-2022). Participants were identified through a hospital electronic database. Neonates with (i) chromosomal and congenital anomalies, (ii) incomplete data because of death or discharge before 14DOL or discharge, (iii) neonates with ≥grade-III intraventricular hemorrhage (IVH) or other CNS defects (at the risk of having apnea) (iv) babies not on the ventilator were excluded for the outcome of EF.
Unit Protocol for the Use of Caffeine
A clinical practice guideline (CPG) regarding CC use was developed in April 2017 after the availability of caffeine. According to the CPG, preterm neonates ≤34 weeks GA started prophylactic caffeine upon birth or admission with a 20 mg/kg loading dose followed by 5 to 10 mg/kg/day at 24-h intervals. This dose was adjusted weekly based on weight until discontinuation. The CPG recommended continuing caffeine until 35 to 40 weeks PMA or until the neonate no longer experienced apnea for 5 days without non-invasive mechanical ventilation (NIMV) or high flow.
Following the unit protocol, intravenous (IV)-CC was administered initially and was switched to the oral-CC subsequently when tolerating adequate enteral daily feed of >100 mL/kg/day. We included all the neonates who received CC-therapy, either IV or oral, as the bioavailability of CC is almost similar through oral and IV routes.26,27 The attending neonatologist made judgments for extubation time, replacement of IMV, and nasal-continuous positive airway pressure (nCPAP)-duration.
Data Collection
Data were extracted from institutional electronic health records and through individual chart reviews including antenatal, prenatal, and postnatal attributes such as sex, BW, and GA. CC-regimen details of route and duration of therapy and the number of neonates who needed early higher dose caffeine were obtained from pharmacy records. Data for CC-APSEs were extracted from the individual daily chart reviews, daily progress notes maintained in patients’ files and electronically maintained laboratory records. In addition, data were retrieved about the other respiratory parameters. Duration of IMV, defined as total days being intubated and receiving IMV to 28DOL or discharge.
Incidence of major neonatal comorbidities was assessed including necrotizing enterocolitis (NEC), ROP, BPD, etc. NEC is defined as Bell’s stages II or III. 28 BPD and its severity were defined based on the National Institute of Child Health and Human Development (NICHD) consensus definition, 29 with the need for supplemental oxygen and/or positive pressure at ≥28 DOL and then 36 weeks PMA or discharge, as determined by chart review. The existence of PDA was counted by reviewing the results of echocardiogram and surgical ligations by reviewing operative records, following the NICHD consensus definition. 29 Any flow of more than 2 L/min was considered positive pressure. Extubation failure was defined as not being able to extubate when planned or reintubation within 48 hours of extubation. Documented apnea was defined as episodes of apnea recorded on the individual standard record sheet.
Intervention and Outcomes
Neonates during the study period received licensed neonatal products (Peyona, CC 20 mg/mL solution by Chiesi Ltd.). All the neonates meeting the defined criteria were assigned into 2 groups, depending on the low dose and high dose caffeine used, that is, the LD-caffeine group (5 mg/kg/day) and HD-caffeine group (>5 mg-10/kg/day). As per the CPG, all the neonates received 5 mg/kg/day as the maintenance dose, and their dose was escalated to >5 mg/kg/day if developed apnea. Those neonates who received >5 mg/kg/day within 72 h of their life and maintained on the same dose were included in the HD-caffeine group. Measurement of serum CC-concentrations was not performed in this study. 30
The impact of LD and HD-caffeine therapy to prevent apnea in the first 28DOL or discharge is the primary outcome. Apnea is counted if breathing pauses for more than 20 s, along with bradycardia [heart rate (HR) reduced by ≤20%] and/or cyanosis (SpO2 ≤ 85%). 31 Standardized apnea recording sheets were placed for nurses to record the apneic episodes and exclude secondary apnea.
Neonates received at least 48 h of IMV, were analyzed to compare the impact of HD and LD-caffeine on IMV-duration and attempts of EF, as a secondary outcome. The EF was counted if failed to extubate from IMV when planned by the neonatologist according to the patient’s condition, or if the patient required re-intubation within 24 h after extubation.22,32 Another secondary outcome was the development of caffeine-associated side effects such as tachycardia (recorded HR of >200 beats/min for consecutive 4 h), and electrolytes imbalance in the first 28DOL or discharge.
Statistical Analysis
All Categorical variables were reported as numbers (%). Continuous variables were presented as mean ± standard deviation and median (interquartile range), as applicable. The χ2 test and independent-sample t-test were employed for comparing the groups for categorical and continuous variables, respectively. Subgroup analyses were designed for the stratified ≤28 weeks GA group. The generalized linear model with log link function was applied to determine potential risk factors of apnea and EF. Conducted bivariate analysis for determining the independent effect of individual predictors on outcome. Variables were accounted for in multivariate analysis for adjustment where P < .20 in bivariate analysis and retained with P < .05 using backward elimination. Results were reported as the odd ratio (OR) and adjusted OR at a 95% confidence interval. All data were analyzed through STATA 17.
Results
During the study period, 984 neonates were admitted to NICU. Based on inclusion criteria 253 were analyzed for demographic and clinical characteristics. All the neonates received a loading dose (20 mg/kg) within 24 h after birth. In the LD-caffeine group, 181 neonates received 5 mg/kg/d, and in the HD-caffeine group 72 neonates initially received 5 mg/kg/day but within 72 h of therapy escalated to the dose to >5 to 10 mg/kg/day (Figure 1).
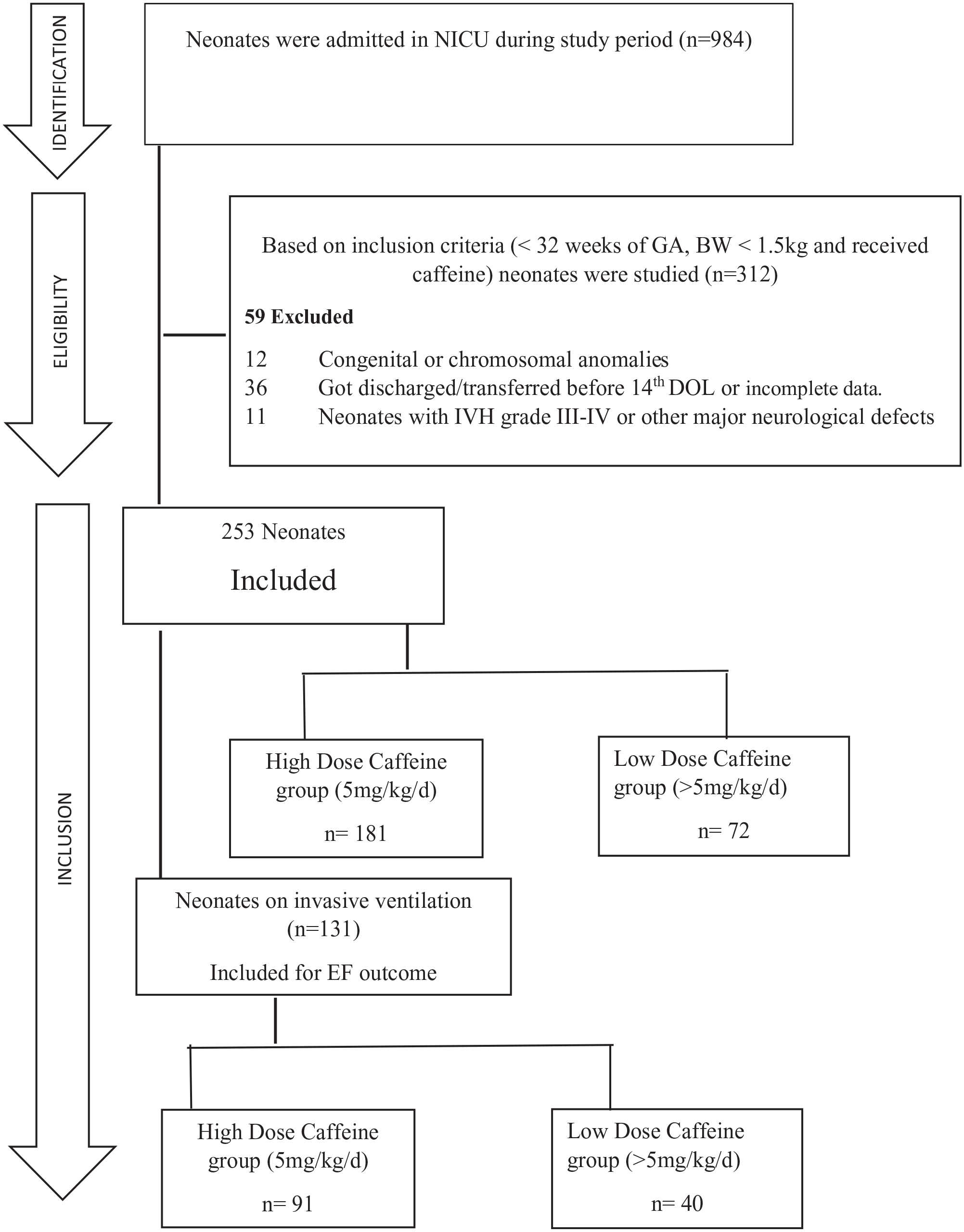
Flow chart depicting recruitment of cohort.
Patient Characteristics
The mean BW and GA were identical in both groups. Some common perinatal factors were also comparable, including mode of delivery, antenatal steroid, and 5-min Apgar scores (Table 1). Though, the HD-Caffeine group had a significantly higher occurrence of maternal chorioamnionitis (P < .034). Evaluation of some general neonatal and postnatal influencing factors, such as sex, steroid and surfactant use, PDA, and PDA surgical ligations, were similar. In the subgroup of ≤28 weeks GA, there were 30 and 32 neonates in the LD and HD-caffeine groups, respectively.
Demographic and Clinical Characteristics of Preterm Neonates Who Received Caffeine Therapy.

Note. Data is presented as n (%) unless otherwise indicated.
PPV = positive pressure ventilation.
Data presented as mean SD.
Data presented as median (IQR). A P-value less than .05 (typically ≤.05) is statistically significant.
After 72 h of starting caffeine therapy.
Included days of complete therapy.
All the bold p values present statistically significant difference.
Caffeine Regimen
The initiation time and duration of CC-therapy were similar. The daily maintenance dose in the HD-caffeine group was 7 to 7.5 mg/kg/day in 93% of neonates and >7.5 to 10 mg/kg/day only in 7% of neonates.
Respiratory Outcomes
Significantly fewer neonates experienced apnea in the HD-Caffeine group (11.11% vs 20.99%; P < 0.01) as the overall number of documented apneas till 28DOL or discharge (Table 2). The need for IMV and last pH and pCO2 before extubation were insignificantly different among the neonates.
Impact of Caffeine Citrate Daily Dose (LD vs HD) on Neonatal Respiratory Status.

Note. Data is presented as n (%) unless otherwise indicated.
LD = low dose; HD = high dose; nCPAP = Nasopharyngeal continuous positive airway pressure.
Data presented as median (IQR); A P-value less than .05 (typically ≤ .05) is statistically significant.
Data presented as mean SD.
All the bold p values present statistically significant difference.
A significant reduction in EF was observed in those neonates receiving HD-caffeine compared with LD-caffeine (20.0% vs 38.5%; P < .05). This difference was even greater in the subgroup of ≤28 weeks GA (15.6% vs 40.0%; P < .01). Though, the IMV-duration was similar between the groups, however, in the subgroup of ≤28 weeks GA significantly reduced IMV-duration was observed who received HD-caffeine (16.2 days (±11.1) vs 24.5 days (±14.2); P < .05).
Neonatal Comorbidities, Mortality, and Adverse Effects
The reported morbidities, length of NICU and hospital stay, and neonatal deaths were similar between the groups. The HD-caffeine group had a significantly (P = .038) lower incidence of moderate/severe-BPD and a higher number of culture-proven infections (P = .040). In the HD-caffeine group, a higher number of neonates experienced NEC (P = .09) (Table 3). A significantly higher number of neonates experienced tachycardia, hyponatremia, and feed intolerance in the HD-caffeine group. Similarly, a significantly longer time to regain BW was observed for neonates in the HD-caffeine group (mean [SD] days 12.6 [6.1] vs 7.3 [5.0]; P < .01; Table 4).
Morbidity and Mortality Data.

Note. Data is presented as n (%) unless otherwise indicated. A P-value less than .05 (typically ≤.05) is statistically significant.
NEC = necrotizing enterocolitis; IVH = Intraventricular hemorrhage; ROP = retinopathy of prematurity; BPD = bronchopulmonary dysplasia.
Discharged home form NICU.
All the bold p values present statistically significant difference.
Impact of Daily LD and HD Caffeine Citrate on the Onset of CC-Associated Adverse Events.
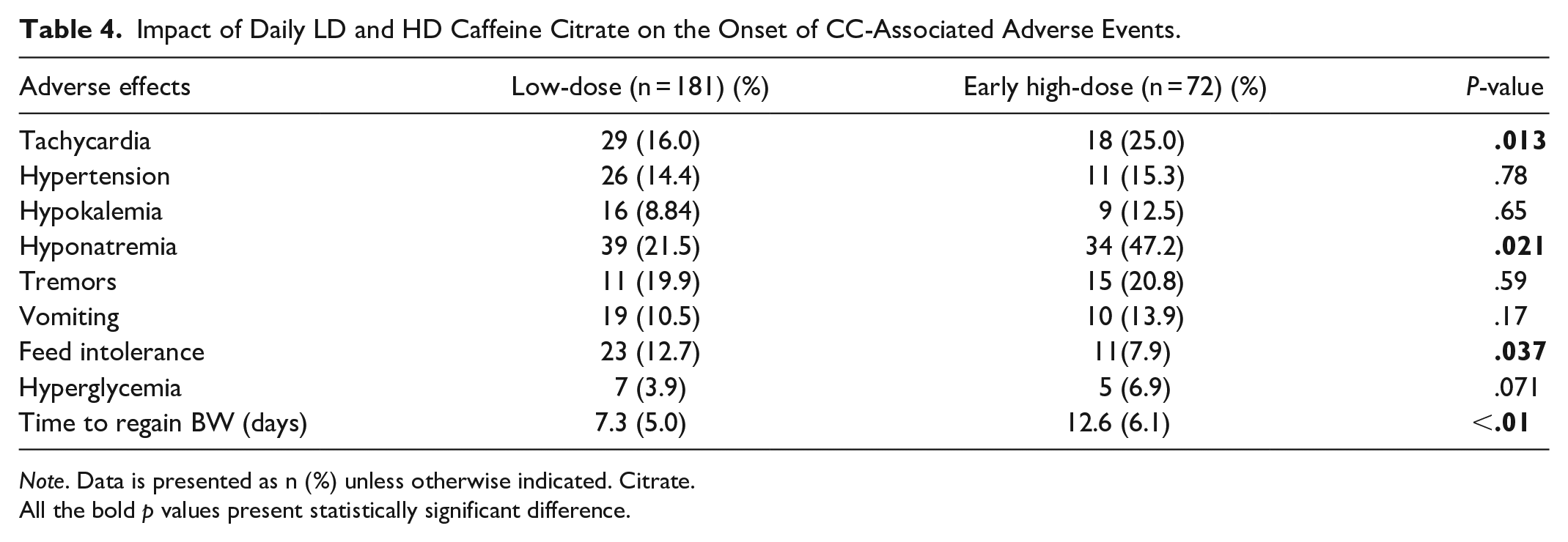
Note. Data is presented as n (%) unless otherwise indicated. Citrate.
All the bold p values present statistically significant difference.
Regression analysis
Regression analysis explains the predictors associated with experiencing apnea and EF, by multivariable logistic regression after initial univariable analysis (Tables 5 and 6). The GA and BW were inversely related to the occurrence of apnea in univariate analysis. Neonates of smaller GA were more likely to develop apnea (AOR = 0.510 95%CI 0.483-0.999) in multivariate analysis. BPD and culture-proven infection were significantly associated with experiencing apnea in univariable analysis but reported insignificant associations in multivariate analysis. The HD-caffeine therapy till 28DOL was inversely associated with the risk of developing apnea (AOR = 0.244 95%CI 0.053-0.291) in this study cohort (Table 5).
Risk Factors Associated With the Development of Apnea: Results of Univariate and Multivariable Analysis.

CL = confidence interval; OR = Odd Ratio; GA = gestational age; BW = birth weight; BPD = Bronchopulmonary Dysplasia.
Data presented as # number (%).
Median (range) values.
Risk Factors Associated With Extubation Failure: Results of Univariate and Multivariable Analysis (n = 131 Ventilated Neonates).
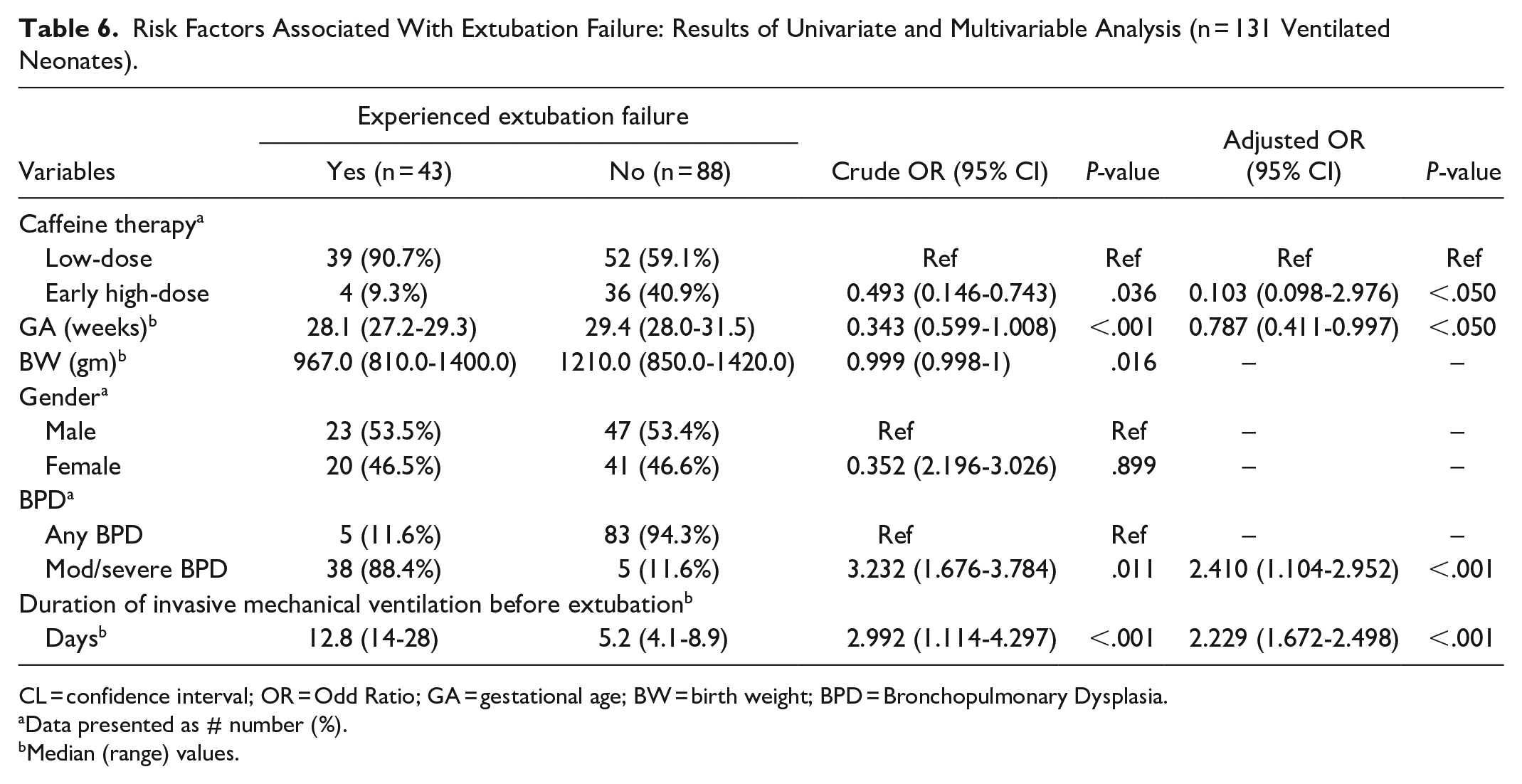
CL = confidence interval; OR = Odd Ratio; GA = gestational age; BW = birth weight; BPD = Bronchopulmonary Dysplasia.
Data presented as # number (%).
Median (range) values.
Neonates of smaller GA were more likely to experience EF (AOR = 0.787 95%CI 0.411-0.997) in multivariate analysis. Duration of IMV before extubation (AOR = 2.229 95% CI 1.672-2.498) and severe/moderate-BPD (AOR = 2.410 95% CI 1.104-2.952) were directly related to EF in multivariate analysis. Neonates with lower BW were at higher risk of experiencing EF in univariable analysis but reported insignificant association in multivariate analysis. The HD-caffeine therapy in 28DOL was inversely associated with the likelihood of developing EF (AOR = 0.103 95% CI 0.098-2.976) in this study cohort (Table 6).
Discussion
An association between caffeine dosage and the occurrence of neonatal apneic episodes and EF is reported in the present study. The incidence of these outcomes differs amongst neonatal studies conditional to study designs, sample size, population, and caffeine regimen. Early use of CC-therapy has been recognized for lowering the occurrence of PDA, EF, BPD, and apnea, shortening IMV-duration, and deaths.10,13,33 Nevertheless, CC-therapy is widely prescribed, and the optimum daily dose remains debated. To the best of our knowledge, this is the first study comparing prophylactic maintenance daily dose of 5 mg/kg with HD-group (>5 mg/kg/day) in which 93% of the neonates received 7 to 7.5 mg/kg/day.
The prophylactic use of CC-therapy initiated within 24 h of neonatal life with a loading dosage of 20 mg/kg, and a higher daily dosage of >5 to 10 mg/kg had a significant association to reduce the occurrence of apneic episodes and EF up to 28DOL or discharge compared to the standard daily dosage of 5 mg/kg, also a reduction in severe/moderate-BPD.
The use of HD-caffeine was more convincing in the subgroup of ≤28 weeks GA, where 32 neonates received HD-caffeine and only 2 neonates experienced apnea. Though IMV-duration was comparable between the study groups, a significant reduction of 7 days was observed in the subgroup of ≤28 weeks GA 23 in the HD-caffeine group. In our study the lower GA is a common risk factor for developing apnea and EF therefore, they may get more benefits of HD-caffeine therapy.
Similar results are reported in a recent trial, they compared 20 mg/kg and 5 mg/kg caffeine doses started in the peri-extubation period. 23 A very high-loading dose of 80 mg/kg was used in the HD-group and 20/kg mg in the LD-group. However, the loading dose used in our cohort was the same across the groups. Another RCT used the same loading dose of 20 mg/kg and reported similar results of significantly reduced apneic spells and EF in neonates who received maintenance caffeine 10 mg/kg/day compared to the 5 mg/k/day. 32
There is a gap in the literature, however, compared to these studies, in our study, the neonates in the HD-group received a comparatively very low maintenance dose of 7 to 7.5 mg/kg/day in 93% of neonates, and only 7% of neonates received >7.5 to 10 mg/kg/day. Even though, the number of neonates experiencing apnea and EF were significantly lower in the HD group.
On the other hand, Lamba et al. used 20 mg/kg as loading dose and compared prophylactic daily per kg dose of 10mg and 6 mg. 34 They reported no reduction in apneic spells. However, the IMV-duration was reduced significantly with no difference in IMV requirement during 28DOL. 34 Our study also reports a reduced IMV-duration but statistically not significant and the need for IMV among the neonates of HD and LD was also similar. We hypothesize that reduced IMV-duration might be the response to the pharmacological effects of HD-CC-therapy during the peri-extubation period. 15
We found no difference for IMV need till 28DOL or discharge, and this generally explains several factors linked with early-life neonatal care in NICU and the likely holistic benefits of longer use of CC-therapy. Groups were comparable for the pCO2 and pH preceding extubation, showing that the variance in IMV-duration might not because of permitting for added permissive hypercapnia or minimized vent setting during intubation.
Comparable non-invasive mechanical ventilation (NIMV)-duration between the groups might be explained to some extent due to the unit guidelines and recommendations for keeping nCPAP to 32 weeks postmenstrual age for neonatal growth optimization. The same may explain the no difference in the occurrence of any BPD between the groups. However, HD-caffeine daily exposure reduced the frequency of severe/moderate-BPD. Literature reports that severe-BPD prognosticates poor respiratory functions 35 and neurological consequences in childhood.36 -38 Therefore, we assume that prophylactic HD-caffeine can possibly benefit improved long-term neurological and respiratory outcomes.
This study reported no significant difference in the incidence of major neonatal comorbidities. However, a higher number of neonates exposed to HD-caffeine had culture-positive sepsis. Although neonatal antimicrobial guidelines across the groups during the study period were the same. When assessing the maternal history, the neonates in HD had a higher number of maternal chorioamnionitis and experienced early-onset sepsis.
Development of NEC was higher in the HD caffeine group in this study but statistically not significant. Several studies have also reported no significant difference in NEC development among neonates receiving low and high doses of caffeine.23,27,34 However, few recent studies have reported a potential association between the administration of caffeine and the development of medical or surgical NEC in premature infants.39,40 A higher risk of developing NEC with HD-caffeine can be explained by lower mesenteric tissue oxygenation in preterm neonates and caffeine-induced reduced blood flow to the superior mesenteric artery. 41
Some studies conclude that CC has a wide range of safe daily doses with no adverse effects. 42 But no consensus has been reached yet and a range of adverse effects are reported in neonates on CC-therapy.32,34,43 The most common and limiting issue is tachycardia, sometimes low at 2.3%. 44 We observed significantly higher incidents of tachycardia in the HD-caffeine group. On the other hand, few studies compared daily HD-caffeine of 10 mg/kg with LD-caffeine of 5mg/kg and reported no significant increase in the incidence of irritability, feeding intolerance, abdominal distension, and tachycardia, suggesting that 10 mg/kg/day is safe.32,34 In the present study, 93% of neonates in the HD-caffeine group received 7-7.5 mg/kg/day and only 7% received 10 mg/kg/day. Therefore, our results are suggestive of developing significantly higher CC-APSEs even at 7-7.5 mg/kg/day.
In this study, the occurrence of other CC-APSEs including hyperglycemia, hypertension, tremors, and hypokalemia was similar in both groups. Nevertheless, we observed that the HD-caffeine group took longer to regain BW. Similar results are reported in a study, by using 20 mg/k/day as HD-caffeine. 23 The delay in regaining BW can be explained by a higher number of neonates experiencing hyponatremia and feed intolerance with HD-caffeine. A study established a dose-response relationship between caffeine doses and electrolyte concentrations in serum and urine. 45 The author stated a substantially higher sodium (Na) excretion accompanying diuresis at HD-caffeine. The probable causes of caffeine-induced natriuresis are caffeine A1-adenosine receptor antagonism and proximal tubular Na-reabsorption inhibition.46 -48 Secondly through caffeine A2-adenosine receptor antagonism and reduced distal Na-reabsorption.26,49 However, the underlying mechanisms are still unexplained.
Some inherent limitations were there in the present study because of its retrospective observational nature and not an RT to make it more conclusive. The selection of CC-dose for any neonate was the clinicians’ decision ranging from 5-10 mg/kg/day and there was a likelihood of treating sicker neonates with higher doses, who were not responding to the standard dose. There was no practice of testing serum CC-concentrations in our NICU. Although the mean daily weight estimation was obtained from daily weight measurement, in a few neonates, thrice-weekly weight was measured due to some clinical limitation however, this was reflected across both neonatal groups. This study only reported short-term efficacy and CC-APSEs.
Despite a few limitations, our study has several strengths. This is the first study that compared caffeine higher daily dose group, where 93% of neonates were exposed to 7 to 7.5 mg/kg/day, with standard 5mg/kg/day, and evaluated the effect on episodes of apnea, EF, and CC-APSEs. Additionally, multiple confounders were adjusted in the multiple regression model, having a potential association with the outcomes of interest. During the study period, all the eligible neonates were included in the study and no major changes took place in the NICU practice guidelines. All the participants received similar feeding regimens. Throughout the study period, the same caffeine pharmaceutical brand was used for intravenous and oral dosages for all participants.
Conclusion
Initiating early HD-caffeine at a maintenance dosage of >7 to 7.5 mg/kg/day may prevent extubation failure and lower the incidence of apnea and moderate/severe-BPD in preterm neonates. Considering the limited availability of therapeutic choices to reduce respiratory complications in preterm neonates and their consequences, optimization of caffeine initiation time and dosages can be a safe and feasible approach to decrease the burden of neonatal respiratory morbidities.
Footnotes
Acknowledgements
Authors would like to acknowledge the support of all the neonatal faculty and team for facilitating. We are grateful to Abdul Moiz Hussain for his expert guidance and facilitation of manuscript edits and submission.
Author Contributions
SA, GA, and SR have substantial contributions to conceptualizing and designing the project. SR, ZB,RM, SAhmad and HFA were involved in data collection and data entry. GA wrote the manuscript. SA and KH were involved in supervising the project, interpreting data, and substantively revising the manuscript. HJ, SS, SAM, and AB analysed the data. GA, SA, and KH completed the final draft. All authors were equally involved in manuscript writing and critical revising.
Availability of Data and Materials
The datasets used and/or analysed during the current study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval and Consent to Participate
The research study was performed in accordance with all the relevant guidelines and regulations of the Declaration of Helsinki. Before performing this study, ethical approval was taken from the institutional ethical committee of Aga Khan University Hospital (ERC # 2023-8276-23810).
Informed Consent
Informed consent from a parent or guardian for participants was waived by Aga Khan University Hospital's ethical review committee as all the data was collected retrospectively.
Consent for Publication
Not Applicable.
