Abstract
Foot self-care has been commonly studied among people with diabetes. Previous research on foot self-care among those with diabetes-related high-risk feet is almost unavailable or very limited. The current study aims to fill this gap and provide a contemporary unprecedented analysis of this area of specialization. To assess the levels of, and factors associated with foot self-care among people with diabetes-related high-risk feet. A multi-center cross-sectional study from Jordan assessed the foot self-care of 107 participants with diabetes-related high-risk feet. Multiple socio-demographic, physiological, and psychosocial factors were collected, and the Arabic version of the diabetes foot self-care behavior scale was used to estimate the foot self-care of the study population. A multiple linear regression model was employed to identify factors associated with foot self-care. The mean score of foot self-care was 25.4 ± 7.1 (35 is the highest) indicating ~73% of adherence to foot self-care. Factors associated with higher foot self-care were being treated at the King Abdullah University Hospital (β = .30, P < .01) and reporting higher scores of patients’ interpretations about neuropathy physical causes of foot ulcers (β = “.22,” P = .02). People with diabetes-related high-risk feet reported a relatively adequate foot self-care practice. However, clinicians are still required to enhance foot self-care among this population. Health promotion programs may benefit from engaging people in understanding the pathophysiology of diabetes-related foot ulcers to improve foot self-care practices.
Foot self-care is recommended for people with diabetes to decrease the risk of complications.
This research promises to guide future health promotion interventions to enhance foot self-care in people with diabetes particularly those with high-risk feet.
This research tested several psychosocial hypotheses that predict health behavior such as foot self-care. This research has implications for practice as it can guide clinicians in which factors they need to consider to improve foot self-care when providing education for their patients.
Introduction
Diabetes-related foot disease including foot ulcer has been a global burden in recent decades. 1 Diabetes-related foot ulcers (DFUs) can result in devastating complications making amputations the chosen treatment option in many cases, and adding a high-cost burden in high-income countries such as China, the United Kingdom, and the United States,2 -4 and low\ middle-income countries such as Bangladish and Jordan.5,6 In addition, people with DFUs are at high risk of premature death which can lead to higher mortality than several cancers, 4 and poor health-related quality of life. 7
Diabetes may cause micro and macrovascular complications, leading to devastating changes in the foot, and then increasing the risk for ulceration. These changes include peripheral neuropathy (loss of sensation and foot deformities related to motor neuropathy) or peripheral arterial disease (PAD). 8 Detecting such complications in routine practice has been well-known to identify the foot at risk for ulceration. 9 However, when people have the aforementioned complications in line with a history of previous DFU, the feet of those people are considered at higher risk of recurrent ulceration. This has been demonstrated in risk category classification by the International Working Group of Diabetic Foot (IWGDF) in which people with high-risk feet are classified under risk category 3 and those should visit health care providers every 1 to 3 months. 9
In addition to the need for regular foot screening, self-care practices for the foot at risk have been strongly recommended by the IWGDF despite the lack of high-quality evidence to support its effectiveness in preventing DFUs. Daily routine practices include wearing preventive shoes, foot inspection, foot hygiene, skin moisturizing, and callus removal. 9 Therefore, it is valuable to understand the magnitude of the daily routine practice of self-care activities and the related factors, particularly among those with high-risk feet and this can help to guide future educational interventions on foot self-care.
Foot self-care behavior can be a result of multiple cognitive factors such as knowledge, beliefs, or controllability according to a recent qualitative systematic review. 10 Such cognitive factors have been discussed in popular theories and models explaining health behavior such as social cognitive theory (ie, self-efficacy and outcomes expectations) and health belief model.11,12 Previous research showed that cognitive factors including patients’ understanding of the causes of DFUs,10,13 self-efficacy to perform foot care,11,14 and the expected benefits (outcomes expectations) from performing foot self-care activities,11,15 can predict foot self-care among people with diabetes. Therefore, all the above-mentioned variables seem promising hypotheses.
Globally, it appears that the previous research on foot self-care only targeted the general population of diabetes not the specific population of high-risk foot. 16 Similarly, in Jordan the present data on foot self-care among people with diabetes-related high-risk foot is insufficient. Although 2 previous large cross-sectional studies showed poor foot self-care in people with diabetes in Jordan,17,18 the data represent the overall population of diabetes including people with low-risk feet while, as mentioned, people with diabetes-related high-risk feet need much more attention to reduce the high risk and hopefully preventing DFUs and the related amputations. Thus, our study aimed to provide an initial understanding of foot self-care among people with diabetes who have high-risk feet.
Research Design and Methods
Study Design and Settings
A cross-sectional multi-center study was conducted in 2 of the largest referral diabetic foot clinics in the north and middle of Jordan: i) the National Centre for Diabetes, Endocrinology, and Genetics (NCDEG) (Amman – Jordan), and ii) King Abdulla University Hospital (KAUH) affiliated with Jordan University of Science and Technology (Irbid-Jordan). Data were collected in the period between October 2022 and March 2023.
Participants
The study included adult Jordanian participants (age > 18 years) with diabetes mellitus (type 1 or 2). Participants had at least one high-risk foot (risk category 3) which was defined as the presence of either loss of protective sensation or peripheral arterial disease (PAD) in addition to the presence of either previous ulceration, amputation (minor or major) or end-stage renal disease 9 (Table 1). Illiterate people or those who had a mental illness were excluded.
Definitions, Supported Citations, and Justifications of the Sociodemographic, Health, and Foot Variables.
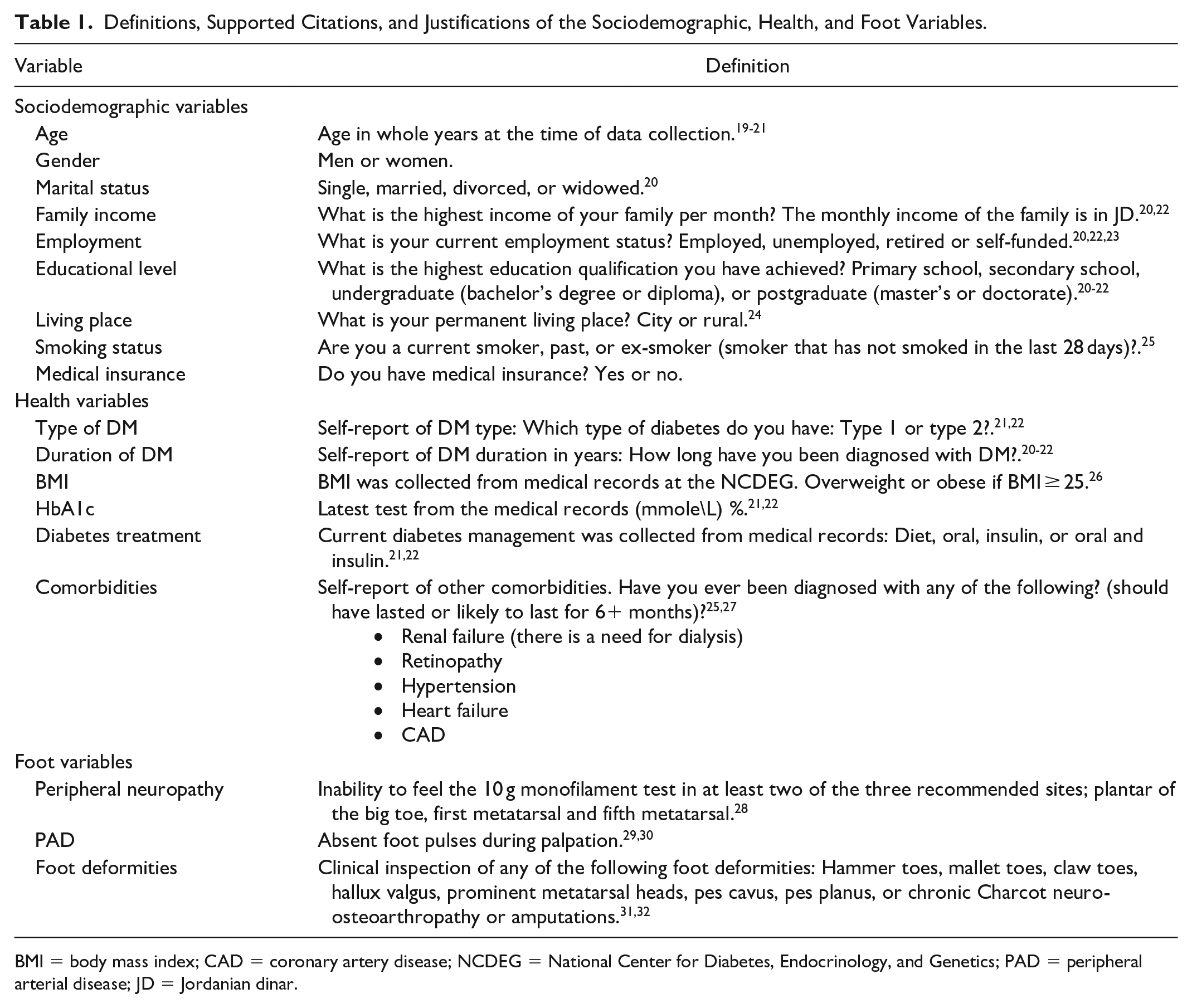
BMI = body mass index; CAD = coronary artery disease; NCDEG = National Center for Diabetes, Endocrinology, and Genetics; PAD = peripheral arterial disease; JD = Jordanian dinar.
Sample Size
A minimum sample size of 60 participants was needed. The main outcome (foot self-care) was a continuous variable, and thus multiple linear regression was utilized to estimate an expected 5 to 6 independent variables. A minimum of 10 participants was necessary for each factor so as not to overfit the anticipated regression model. 33 According to VanVoorhis and Morgan, 34 a rule of thumb to detect relationships through correlations or regression can be a sample size of around 50 participants. However, 107 participants were recruited which is much higher than the needed minimal sample size as we aimed to reduce type 1 or 2 errors that can be related to insufficient sample power.
Explanatory Variables
Sociodemographic, health, foot, and psychosocial variables were collected. Sociodemographic variables included age, gender, marital status, family income, employment, education level, living place, presence of health insurance, and smoking status. Health variables included type and duration of diabetes, body mass index (BMI), hemoglobin A1c (HbA1c), diabetes treatment, and presence of comorbidities. Foot variables included peripheral neuropathy, PAD, and the presence of foot deformities or amputations (see Table 1).
Psychosocial variables included: (i) the perceived self-efficacy of foot self-care which was measured using the foot care confidence scale (FCCS). 35 This is a 12-item scale (5-point Likert items, maximum score = 60) that measures patients’ confidence to undertake foot self-care activities (eg, washing feet or cutting nails); (ii) the perceived benefits (outcomes expectations) of foot self-care: this was measured using footcare outcomes expectations scale (FCOES). 36 This a 15-item (5-point Likert items, maximum score = 75) that measure patients’ expectations of the outcomes of performing foot self-care in preventing DFUs (eg, moisturizing feet or wearing proper footwear can prevent DFU); and (iii) understanding the causes of neuropathy and DFUs: this was measured using the patient interpretation of neuropathy scales (PINS). 13 These are 11-item (5-point Likert items) sub-scales that measure patients’ knowledge of the potential causes of neuropathy and DFUs including self/practitioner blame (maximum score = 20), physical causes of DFUs (maximum score = 20), and duration and time of the onset of DFUs (maximum score = 15). The Arabic version of the above scales has shown reasonable reliability in use. 37
Outcomes of Interest
The outcome of interest was the behavior of foot self-care which was measured using the diabetes foot self-care behavior scale (7 items, maximum score = 35 indicating perfect foot self-care). 38 This scale has 2 subscales (A&B). Subscale A has 4 Likert items that measure the frequencies of several foot self-care activities (eg, examining the bottoms of feet and between toes, and washing between toes) in the last 7 days (1 = never [0 days] to 5 = always [7 days]). Subscale B asks participants how often (1 = never to 5 = always) they practice foot self-care activities (eg, applying a moisturizing lotion, checking the inside of the shoes, and breaking in new shoes slowly. Arabic version of the DFSBS was used in this study which is valid and reliable (Cronbach’s Alpha = .75). 39
Procedure
First, ethical approvals were obtained from the clinical settings and the institutional review board committee at the Applied Science Private University, Amman, Jordan (Approval No. 2021-2022-3-1), NCDEG (No. 1-2022), and KAUH (No. 474-2022). This research was considered a low-risk project with no physical or financial harm. The first author (expert in diabetes-related foot disease, PhD) provided training for the 2 co-authors (SM & YA) (experienced practitioners in diabetic foot) who collected the data independently from the 2 different research settings (KAUH & NCDEG). This training included clarification of the inclusion criteria, instructions on how to use the study questionnaire (eg, scoring the psychosocial scales), and assessment of foot variables. The study was then advertised by informing clinicians in the research settings to facilitate recruiting potential participants. The interested participants, who met the inclusion criteria, were asked to sign a consent form after reading the information sheet. Participants were then interviewed in a private room or in the clinic to collect their sociodemographic information and medical history. Further, participants’ feet were examined to collect the foot variables (peripheral neuropathy, PAD, and foot deformities). Once, the interviews finished, the anticipated participants were provided with the study questionnaire (around 30 min). Participants were encouraged to ask if there was any non-clear phrase or item and to answer all the questionnaire items unless they felt uncomfortable answering any specific item.
Statistical Analysis
Statistical analysis was carried out using Statistical Package for Social Sciences IBM SPSS version 22. Descriptive statistics used frequencies (proportions) for categorical data and mean (standard deviation (SD) or median (range) for continuous variables. Independent sample t-test, one-way ANOVA, Person’s chi-square tests, and Pearson or Spearman’s correlations were used to test the bivariate associations. Multiple linear regression was conducted to eliminate confounders and to estimate a valid prediction model of the study variables. Multicollinearity testing between independent variables was tested. A P-value < .05 was considered statistically significant. Cases with missing data (eg, not reporting the main outcome) were excluded and this was minimal.
Results
A total of 107 participants participated in this study; 57 from the KAUH and 50 from the NCDEG. Table 2 presents participants’ socio-demographic, limb, and psychosocial characteristics including each research setting. Overall, the mean age was 58 ± 12.8 (SD) years, and of those, 13.8% were women, 73.8% were married, 30.8% were highly educated, 81.3% were unemployed, and 85% had type 2 diabetes.
Participant Characteristics (Number (%) or Mean ± Standard Deviation (SD)).

BMI = body mass index; CAD = coronary artery disease; DFU = diabetes-related foot ulcers; FCCS = foot care confidence scale; FCOES = footcare outcomes expectations scale; KAUH = King Abdulla University Hospital; NCDEG = National Center for Diabetes, Endocrinology, and Genetics; PAD = peripheral arterial disease; PIN = patient interpretation of neuropathy; SD = standard deviation; JD = Jordanian dinar.
Represents a significant difference between the 2 research settings (P < .05).
Table 2 also presents significant differences in participants’ characteristics between the KAUH and the NCDEG and these included living place, family income, having medical insurance, having chronic conditions (eg, end-stage renal failure, heart failure, CAD, retinopathy, neuropathy), and the total score of PIN: self/practitioner blames scale (all P < .05).
The mean overall total score of the DFSBS (foot self-care) was 25.4 ± 7.1(SD). There was a significant difference between the mean total score of foot self-care between the KAUH (27.6 ± 7.6 (SD)) and the NCDEG (22.9 ± 7.6 (SD)) (P = .001) (Table 2).
The bivariate analysis demonstrated that the total score of foot self-care (n = 107) was significantly associated with the research setting (KAUH), the total score of each FCOES, and the PIN: physical causes of DFU (P < .05) (Table 3). Variables including the total score of FCCS and PIN: self/practitioner blames were eligible for the final regression model (P < .1). However, the multicollinearity testing found that research setting (KAUH) correlated with PIN: self/practitioner blames, FCCS correlated with FCOES and PIN: physical causes of DFU, and PIN self/practitioner blames correlated with PIN: physical causes of DFU (all P < .05). Thus, variables with the least significant association with the DFSBS were excluded leaving the research setting (KAUH) and the PIN physical causes the only eligible independent factors for the final regression model.
Unadjusted and Adjusted Associations Between Variables and the Mean Score of Foot Self-Care.

BMI = body mass index; CAD = coronary artery disease; DFU = diabetes-related foot ulcers; FCCS = foot care confidence scale; FCOES = footcare outcomes expectations scale; KAUH = King Abdulla University Hospital; PAD = peripheral arterial disease; PIN = patient interpretation of neuropathy; SD = standard deviation; JD = Jordanian dinar.
ϯ Foot self-care score is not applicable; *Variables with P-value ≤ .1 in the bivariate testing were nominated for the multiple regression model; ** P ≤ .05 in the multiple regression model;
The final multiple linear regression model was significant (adjusted R 2 = .14, P < .001) (Table 3). Two factors were independently associated with higher scores of foot self-care which included being treated at the KAUH (β = .30, P < .01) and reporting higher scores of PIN physical causes (β = .22, P = .02) (Table 3).
Discussion
We investigated foot self-care practices among people with diabetes and who have high-risk feet of developing DFUs. Our research is likely the first of its kind that targeted this specific population as the previous research examined foot self-care practices among the overall population of people with diabetes, not high-risk foot according to a recent meta-analysis. 16 Our data showed that self-reported foot self-care was almost good (~73%). We also found participants at the KAUH reported a higher score of foot self-care than participants at the NCDEG, and participants who had higher knowledge about the physical causes of DFUs reported a higher score of foot self-care.
As we could not find previous studies that examined self-care among people with diabetes who have high-risk feet, we compared our results with foot self-care of the overall diabetes population. One meta-analysis of 8 studies reported a pooled score of foot self-care of ~63% among the overall diabetes population. 16 Despite that the level of foot self-care in our study was relatively higher (~73%), there is still an urgent need to enhance self-care in this population with high-risk feet to reach almost perfect foot self-care. The foot at risk in people with diabetes has several risk factors that can contribute to the development of DFUs including loss of sensation and foot deformities related to motor neuropathy or peripheral arterial disease (PAD), 8 and therefore, a higher level of self-care is always recommended.
Interestingly, we found a variation in foot self-care levels between the 2 research settings of this study in which participants at the KAUH reported higher foot self-care compared to those at the NCDEG. The socio-demographic data of the 2 populations are not homogeneous including the living place (eg, urban or rural), family income, and having medical insurance and this could impact foot self-care levels in the 2 hospitals. For instance, one study found that people with diabetes in rural had higher foot self-care. 40 Another study found family income as a predictive factor of foot self-care. 41 Another plausible hypothesis is that the population at the KAUH, who reported higher levels of foot self-care, had significantly higher burdens of several chronic conditions including end-stage renal failure, heart failure, CAD, and neuropathy. Previous investigations found conditions such as retinopathy, heart failure, and PAD as predictors of higher adherence and foot self-care among people with diabetes-related foot disease.42 -44 However, we found that all the above variables were not associated significantly with the overall foot self-care in our study. Last, participants from the KAUH were potentially exposed to further empowerment or engagement to perform the needed foot self-care, which could have been another plausible explanation. Future research is needed to explain the differences in foot self-care outcomes between healthcare settings in Jordan.
Our research also showed that the score of the PIN: physical causes of DFU was independently associated with foot self-care practices. This is an important outcome that confirms the suggested hypothesis by Vileikyte et al 13 in which patients’ interpretation of neuropathy can impact their intention and engagement in the needed foot self-care. This also highlights the potential of the recently suggested “The Fragile Feet & Trivial Trauma model” by Jarl et al 45 as a promising strategy to enhance people’s foot self-care by increasing their awareness about the etiology of DFUs. Overall, this result also matches the commonly found relationship between individual knowledge and foot self-care,17,46 and this suggests that clinicians need to educate patients about the pathophysiology of DFUs and probably use models that simplify the education process, 45 then this can hopefully result in improving engagement in foot self-care.
Finally, our investigation did not identify an association between self-efficacy and foot self-care. This agrees with the previous research about adherence to offloading treatment among patients with DFUs in Jordan.42,43 Self-efficacy was not also associated with foot self-care among people with diabetes in previous studies. 47 However, self-efficacy is a well-known valid predictor of health behavior including diabetes and foot self-care and has been commonly known as a core of health education interventions to enhance foot self-care.22,48,49 Thus, further research is needed to test the contradiction about the role of self-efficacy in predicting foot self-care and adherence among patients with diabetes-related foot disease.
Limitations
This study has some limitations. First, the implemented cross-sectional research design is not optimal for assessing the causality of foot self-care behavior. Second, the results of promising outcomes of foot self-care in this research have to be treated with caution as it could result from the overestimation bias by the study participants which is a common limitation of self-report measures in diabetic foot disease. 50 Third, we did not implement exclusion criteria for several chronic conditions to help in matching the populations of the 2 research settings. Last, we did not estimate the time required to fill out the study questionnaire by participants.
Conclusion
Patients with high-risk feet have relatively adequate foot self-care practice but it is not perfect. Improving foot self-care among patients with high-risk feet is needed due to the burden of diabetes-related foot disease and the high risk of DFUs. Foot self-care varied between the study research settings and future research is suggested to examine these differences. Last, as patients’ interpretation of neuropathy included understanding the causal pathway of DFU significantly associated with foot self-care, future health education interventions may benefit from engaging patients in learning the pathophysiology of DFU to enhance their knowledge which may result in improvement of their foot self-care.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics and Consent Statements
Ethical approvals were obtained from the clinical settings and the institutional review board committee at the Applied Science Private University, Amman, Jordan (Approval No. 2021-2022-3-1), the National Centre for Diabetes, Endocrinology, and Genetics (No. 1-2022), and King Abdulla University Hospital (No. 474-2022). All participants signed a consent form before data collection.
