Abstract
Textile laundering is an energy-consuming and environmentally challenging process. This paper reviews the reasons for the necessity of textile laundering and its relation to odor, and alternative strategies to reduce garment washing frequency. The paper also discusses at length the various sources of odors and types of odor management techniques. It explores the current odor evaluation standards and processes, and further debates the challenges in odor management technologies in textiles.
Keywords
Laundry hygiene is essential for maintaining cleanliness, health, and comfort in everyday life. Effective laundering practices help eliminate microbes, sweat, dirt, and environmental pollutants that can accrue on clothing, preventing the spread of germs and reducing the risk of skin irritations. Odor elimination is considered an essential part of laundry hygiene that ensures garments smell fresh and pleasant, and eventually boosts personal confidence and social acceptance.
Odor can be generally defined as a distinctive smell, usually an unpleasant one. Odor is caused by one or more volatilized chemical compounds that are found in low concentrations that humans and animals can perceive by their biological system of sense of smell. It is taken as a sign of cleanliness or filthiness. Sweaty odor of clothing, attributed to sweat of the wearer, is considered as unhygienic and unclean, while fragrances and fresh odors are taken as clean.1–3 In general, odor-free garments can be worn several times before being washed4,5 and are considered fit to wear. Laundering textiles is a highly preferred approach to remove odor. However, social norms and personal habits can become more influential than actual need for laundering. 6 The perceived need of laundering takes toll on textile in terms its performance (such as a water-repellent fabric being less and less effective after every wash), shade (both in terms of hue and chroma), and durability. Washing, even the gentlest wash, can eventually deteriorate the longevity of textiles by damaging the fibers due to harsh detergents and mechanical strain. Machine washing can cause delicate fabrics to snag inside the drum and disrupt the fibers and any other embellishments on the garment. Moreover, a warm water temperature or a harsh spin cycle can wreck delicate fabrics. Henceforth, minimal washing is desirable for the longevity, look and performance of textile and clothing.
Laundering practices and human behavior
The washing of garments is associated with removing soiling and odor. The frequency of laundering is dependent on individuals’ amount of and sweat production, personal preferences, etc. Jack 6 identified that people wash their everyday clothes mostly because of their habit, rather than clothes being dirty or smelly. While 11% of the surveyed population mentioned odor as the reason for their laundering, the second main reason found by Jack was the visible dirt. Almost half of the surveyed Australians washed their cloth after every two to three wears, which created a significant amount of water and energy waste and overall impact on environment.
McQueen
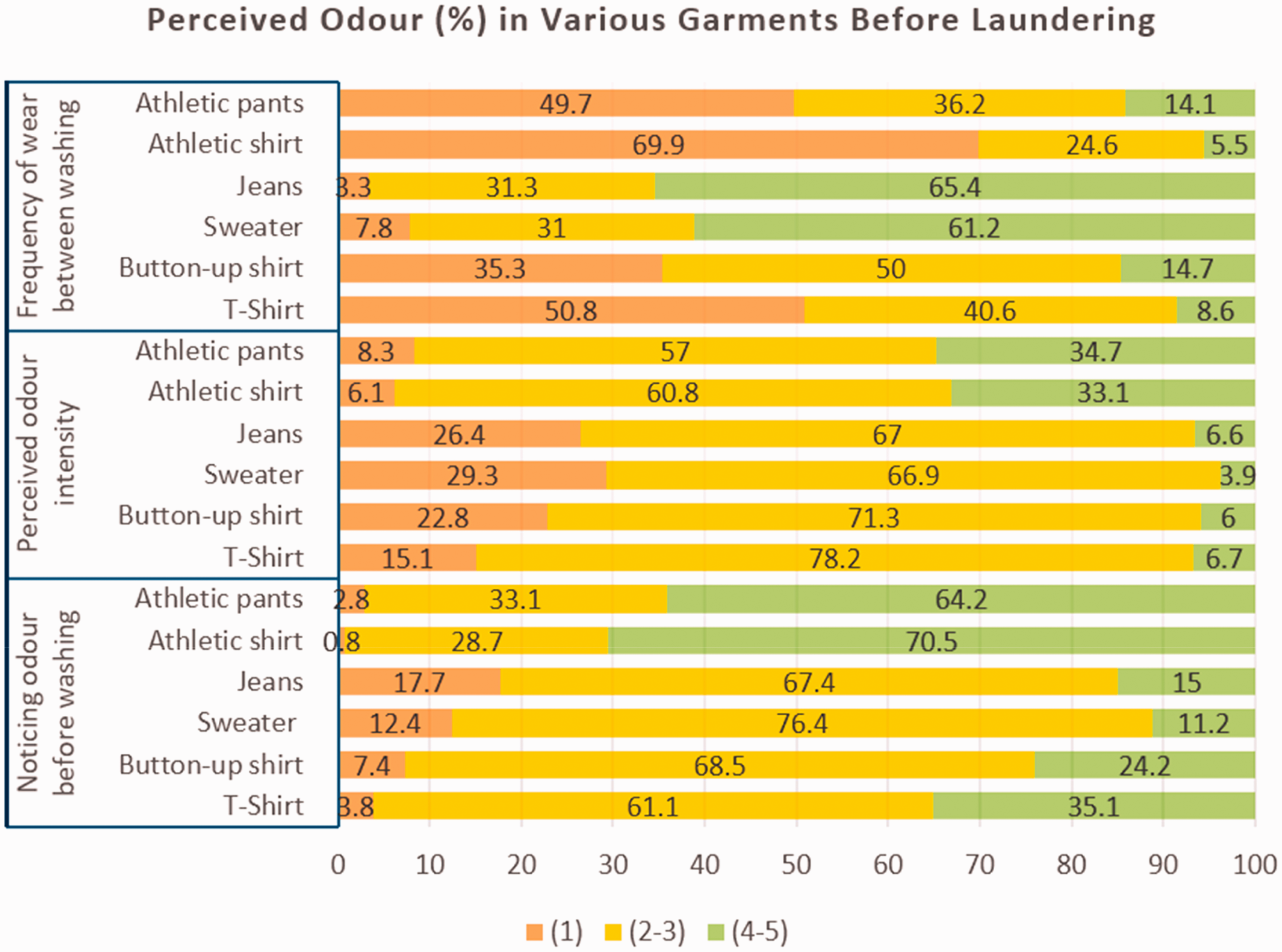
Frequency response to 1–5 Likert scale (wear frequency/perceived odor/noticing odor: 1 = once/never/no odor; 2–3 = 2–3 times/sometimes/moderate; 4–5 = 4+ times/always/high) in relation to perceived odor of various garment types prior to laundering (adapted from McQueen
Teufel
In a laundering process, the clothes are mixed with water and detergent before rinsing with fresh water and spinning out the water and moisture. The detergent contains surfactants, which lower the surface tension between water and other materials. The enzymes in the detergents cause the breakdown of sebum and other protein-based stains. The removal of pathogens from the laundry is dependent on washing and drying practices. Both surfactants and enzymes cause pathogens and malodorous bacteria to be washed away by attaching amphiphiles to the outer wall of bacteria. The reduction–release or/and inactivation of pathogens is influenced by several factors such as detergent selection, additives (such as bleach), water temperature, and drying.
Figure 2 shows the process of sebum and malodourous bacterial attachment and detachment on textiles. The sweat and other fluids present an abundant supply of sebum on the textile surface which has been worn. This results in the transfer of microbes from skin to textile surface. The bacteria thrive on sebum, which comprises cholesterol triglycerides, wax esters, fatty acids, and cholesterol esters squalene, and eventually multiply. As sweating stops, no additional moisture is transported onto the textile and bacterial growth is reduced. However, the bacteria now form biofilms, of which some can retain water. The now dried biofilm could retain malodorous compounds, and also lead to discoloration.
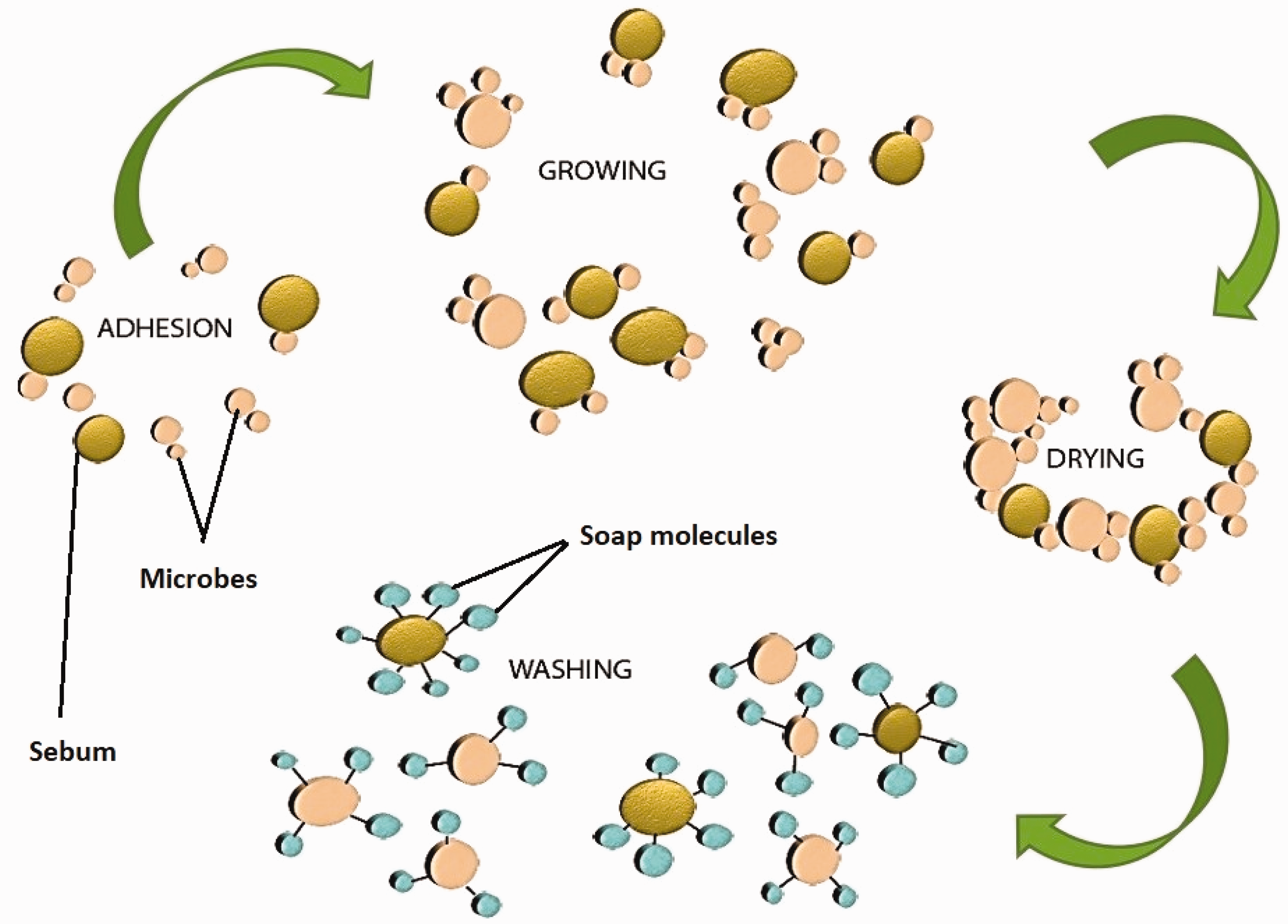
Illustration of adhesion, growth and removal of microbes and sebum during washing.
An important aspect to consider when discussing laundering practices is demographic variations and cultural practices. While the data and research quoted in articles usually relates to developed countries, the practice varies widely based on locations, climate, and belief systems. Khalid e
The laundering habits vary significantly in some cultures such as Japan, where the culture promotes cleanliness, therefore, washing clothes is more frequent than some of the other developed countries. In contrast, Nordic countries have more environmental awareness and hence practice sustainable laundering. This includes washing a combination of lower frequency, using less water, and using ecofriendly detergents. 10
Another important but less significant factor is the religious aspect of laundering practices. Some religions such as Islam prevents praying if the clothes are “filthy,” hence promoting recurrent laundering. 11 In Hindu traditions, clothes worn during religious ceremonies are to be prewashed and then rewashed separately from other clothes, thus increasing the resources usage. 12
In general, hot and humid climates often require more frequent laundering due to sweating. In countries such as Indonesia, this practice is commonly observed in addition to the social pressures of formal dress code at work. 13 Some countries such as Australia and Africa have scarcity of water. High-efficiency machines or recycled water are used as sustainable laundering practices. Furthermore, some developed countries have more awareness in terms of side effects of using harsh chemicals, allergies, and skin conditions, which often promotes less-frequent or ecofriendly laundering practices. Higher urbanization leads to greater frequency of laundering where the availability of washing machines or even laundromats is abundant. In contrast, rural settings use handwashing which is laborious and time-consuming. Some of the developing countries such as India and Pakistan have communal laundries also known as “Dhobi ghatt,” which release microfibers directly into the streams causing microfiber pollution. 14
With regards to gender, laundering is traditionally considered as a female task; however, in countries with higher gender equality, men also participate more actively. While laundering is an age-old practice, a general trend has been seen in younger generations such as millennials and Gen-Z for opting a more sustainable approach than older generations.
Skin and textiles
The interaction between textiles and human skin is a multifaceted phenomenon influenced by the properties of clothing materials, environmental conditions, and individual skin characteristics. This is further affected by the existence of skin microflora which can direct microclimate on the surface of the skin. The skin is the largest organ in humans and serves as principal defense and a sensory organ. The skin houses approximately 1012 microorganisms including bacteria, fungi, protists, and viruses.
15
The complex microclimate between skin and textile plays an important role in maintaining healthy hygiene. The microorganisms present on human skin varies with gender, ethnicity, and age.
16
The presence of moisture and temperature provides a favorable environment for skin microflora to thrive. Møllebjerg
In an event of increased activity, and/or high temperature, the body releases sweat. Sweat itself is generally odorless; however, when it interacts with bacteria on the skin’s surface, it can produce unpleasant smells. The apocrine glands, found in areas including the the armpits and groin, secrete more complex molecules. The bacteria present on skin break down these molecules into several smaller molecules. These include formation of thioalcohols, alpha-androstenone, 3-methyl-2-hexenoic acid (axillary), and isovaleric acid (feet), 18 which are responsible for forming odor and are detected by the human olfactory system.
Odor and its detection in humans
The odors detected by human nose are volatile compounds. The molecules of these compounds interact with the dendrites of olfactory receptor neurons for the olfactory system to sense odor. 19 The odor is a property of a mixture of different volatile chemical species (e.g., sulphur, nitrogen, and volatile organic compounds) that can stimulate the olfaction sense sufficiently to trigger a sensation of odor. 20 As the signal is sent to brain cells, memories and emotions can be triggered when they arrive in the amygdala and hippocampus.
Figure 3 shows the mechanism of odor detection in the human olfactory system. The odor molecules are detected by tiny hairlike structures called olfactory receptors. The receptors can bind to a restricted number of odorous compounds. The nose contains 400 kinds of olfactory receptors, and each receptor is paired with olfactory gene in individual DNA. When a compound is attached to these receptors, the olfactory nerve sends a signal to the olfactory bulb, which causes the brain to interpret the signal as odor. The classification of odor as perceived by humans can be (1) intensity, (2) degree of offensiveness, (3) character, (4) frequency, and (5) duration. It is interesting to note that odor has a high psychological perspective and varies across gender and age.
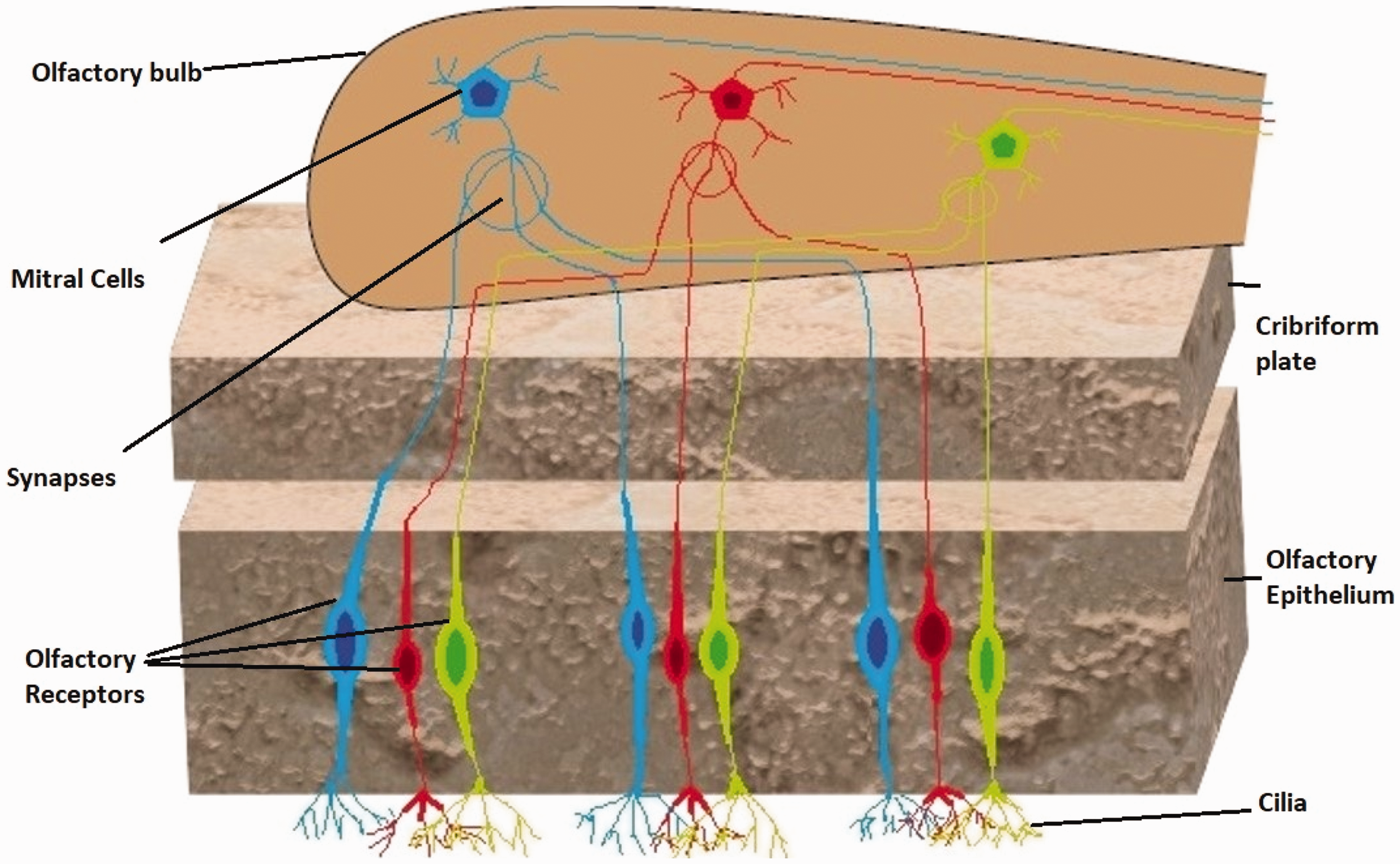
Schematic of human olfactory system.
The size and shape of an odor source are widely affected by the composition of the odor as well as patches of odorless air within the plumes. Murlis
The odor conversion mechanism includes sources such as (i) biotransformation of steroids, (ii) glutamine conjugates which discharge short-chain fatty acids, (iii) sulfanylalkanols from glycine-cysteine (or cysteine (S) conjugates), (iv) bio-transformation of long-chain into volatile short-chain fatty acids. 21 Moreover, the structure of the local environment plays a key role in shaping the perception and tracking behavior of the organisms. 22
Odorous sources
Odorous sources that may cause odor in clothing can be categorized based on their composition, processing materials, bodily substances, environmental and chemical exposure, and end usage. Understating these sources can help in addressing the odor problem and in developing specialized textiles to address this issue.
Primary odorous sources
Biofluids
Body fluids such as sweat, blood, and other secretions from the human body are the biggest cause of body odor. These fluids play critical roles in proper functioning of the body, such as removing waste from the body (blood, urine, sweat), thermoregulation (blood, urine, sweat), and other important functions. Role of each biofluid is briefly discussed in this section.
Figure 4 shows a typical sweat gland presenting the location of glands along the hair follicle in the skin layers. There are several types of sweat glands on the human body. They are found all over the human body and can be classified into three main types, eccrine glands, apocrine glands, and sebaceous glands.
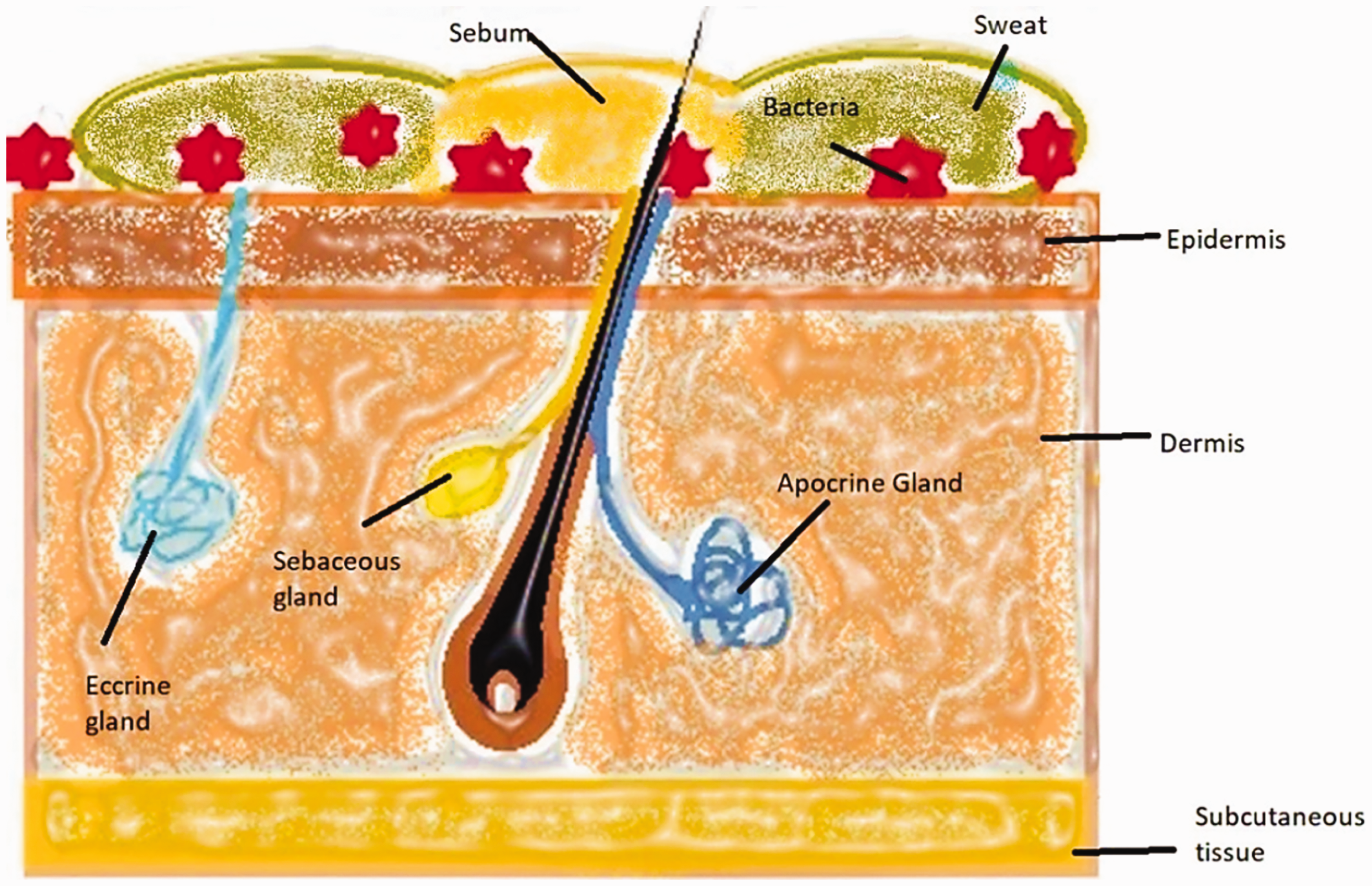
A typical sweat gland.
The eccrine glands are concentrated around the palms of hands, forehead, and soles, and the sweat is an aqueous electrolyte solution, which provides moisture for bacterial activity. The eccrine sweat glands secrete directly onto the skin's surface. The secretion from these glands is affected by hydration, diet, and metabolic rate.
23
The apocrine glands are found near in the pubic regions and axillae. The apocrine glands are connected to hair follicles, which lead to the skin surface. These glands become active at puberty and the secretions decrease with ageing. Sebaceous glands are not sweat glands, however, they secrete the viscous, oil-rich fluid sebum, which also plays an important role in skin microflora.
24
Sebum is rich in lipid materials including long-chain fatty acids.
25
Sebaceous glands are mostly found on the upper parts of the body such as face, chest, scalp, and forehead, and therefore the breakdown of these fatty acids results in a dense odor in these areas, particularly chest and scalp.
b. c.
Microorganisms
Microorganisms are abundant in our surroundings but do not become odorous until they reach a certain threshold population. The presence of different microorganisms on textile surfaces, such as bacteria thriving on sweaty garments, is the leading cause of odor in textiles. The axilla region is particularly important for malodor development due to the presence of apocrine, eccrine, and sebaceous glands, along with a diversity of skin microbes. 29 Of these microbes, the bacteria present on the axillary region (armpits) are mostly Gram-positive bacteria of the species Staphylococcus, Corynebacterium, Micrococcus, and Propionibacterium, with Corynebacterium genus being the leading cause of axillary odor. 30
Secondary odorous sources
Environmental odors
Exposure to smoke and industrial pollutants can impart odors to fabric. Storing fabrics in damp, closed environments, such as basements or poorly ventilated closets, can also lead to mustiness and mildew accumulation. Similarly, long-term storage in closets, drawers, or containers without proper ventilation can cause garments to develop musty smells.
Laundry-related odors
Detergents, softeners, and other cleaning agents can leave residues that build up over time, leading to a chemical-like smell. Improper laundering by overloading the washing machine or the odor from dirty washing machine drums can also lead to consequent odor buildup. Improper drying leads to residual moisture, which can become a breeding ground for mold and mildew, producing a characteristic stale odor. Garments that are dry-cleaned may retain the odor of the solvents used in the process, such as perchloroethylene, which has a strong chemical smell.
Activity-related odors
Garments used for physical activities (e.g., sports uniforms, gym clothes, and activewear) are highly susceptible to odor from sweat, bacteria, and the materials used (e.g., synthetic fibers such as spandex or polyester). Garments worn in industrial settings may absorb odors from chemicals, oils, or mechanical processes. Employees working in kitchens or restaurants may have clothing that absorbs food, grease, or smoke smells. Healthcare uniforms may absorb disinfectant or medicinal odors, as well as body-related smells from patients.
Chemical odors
Garments often come with a distinct “new clothes smell,” which is caused by chemicals used in manufacturing such as starches or resins applied to fabrics to give them stiffness. Often specialty chemicals are used in the textile industry to impart desired performance to fabrics that may contribute to particular odor to new garments (such as pungent smell of formaldehyde in wrinkle-resistant clothing). Some chemical treatments applied to specific garments for safety purposes, such as flame retardant finishes, could give off unpleasant chemical odor. Chemicals used to prevent mildew or mold during shipping and storage may also leave garments with a chemical smell.
Odor retention and fabric architecture
There is a strong connection between odor transfer onto textile substrate and textile architecture. Since body fluids and other microorganisms are present on human skin, they are in direct contact with the textile next to skin. Types of fiber, fabric construction, and garment design all play a significant role in this aspect to odor retention.
Effect of fiber type
Fibers are essential elements used in textile production. The fiber type plays an important role in malodor development and retention. This is due to the reaction of various textile fibers to sweat, and heat and moisture from textile laundering processes. 31 The odor intensity is higher for hydrophobic fibers regardless of the type of sweat composition; hence, polyester fabrics emit more potent odor compared to cotton and wool. 32 Some natural fibers, such as wool and cotton, have inherent odor-reduction capability. Hence, cotton and woolen textiles smell significantly less than polyester and even less than the polyester treated with odor-control treatment.33, 34 In sportswear, it is mainly polyester that requires more attention since polyester does not have any inherent odor-reduction properties. In addition, polyester is hydrophobic and does not absorb moisture; hence, sweat accumulates on skin and clothing microclimates and promotes bacterial growth.
Du
McQueen
Callewaert
The moisture sorption of wool fiber, at a given relative humidity, is comparable to human skin. 42 Wool fiber can absorb moisture up to 33% of its weight, before causing a damp sensation. This property helps keep the skin dry and reduces the amount of sweat on the skin that bacteria can break down to produce body odor. Humidity conditions can alter the rate of absorption and desorption of natural fibers. For example, wool can absorb moisture from its surroundings, including sweat from the skin. This moisture uptake helps reduce clothing odor. In contrast, synthetic fibers such as polyester that are more stable, and coupled with their hydrophobic nature, do not change much their ability of moisture absorption in humid conditions.
The glass transition (
Effect of fabric types
The effect of fabric structure on patterns of adsorption and release of odor volatiles is not properly understood. 43 This warrants further investigation using standard fabrics (and yarns) to facilitate replication of experimental work. 43 As textile properties are affected by fabric construction, odor is also perceived to be affected by fabric architecture. Fabrics with a tighter weave or higher density tend to trap odor molecules more effectively, delaying their release. Conversely, porous fabrics allow for quicker air exchange, which can facilitate the faster release of odor. Fabrics with a larger surface area, such as those with a textured or brushed finish, can adsorb more odor molecules. This increased surface area provides more sites for volatile compounds to attach, temporarily holding the odor within the fabric.
Different fabrics, such as woven, knitted, and non-woven, will all have different properties. A tight knit fabric as compared with a loose-knit fabric will certainly affect textile properties such as air permeability and water permeability, affecting odor retention. Woven fabric structures can allow movement of sweat, trapping of dirt materials and other microbes. Fabric tightness/openness, and other properties such as woven fabric design, number of warp and weft yarn per centimeter, yarn linear density, yarn twist per inch and yarn structure, will also affect odor retention in textiles. While woven fabrics are a preferred choice for many everyday activities, knitted fabrics are chosen for sports garments due to their greater stretch ability allowing ergonomic comfortability when worn against the skin.
44
To deter odor retention, the fabrics can further be treated for water-repellent finishes to enhance moisture transport, particularly for knitted fabrics. In addition to water repellent, antimicrobial agents or odor-neutralizing chemicals can also be employed to inhibit bacterial growth and reduce odor retention. These finishes can particularly help with one way moisture transport, allowing moisture to be moved from the inner side to the outer side by capillary action. Yang
Effect of garment design
The design of a garment affects how tightly it sits against the skin causing continuous textile–skin interactions and abrasion. Tighter-fitting clothes limit airflow and can trap sweat, creating an environment where bacteria thrive, leading to more odor retention. Loose-fitting garments allow for better air circulation and reduced contact with the skin, helping to minimize odor. In contrast to this, sportswear is designed to sit tightly against the skin to allow dynamic motion during exercise. This coupled with superior wicking properties of synthetic fibers allows moisture to be transported away from skin to help in thermoregulation. While moisture is being transferred away from the skin, it still allows some moisture to be trapped and, hence, causing bacterial growth, leading to odor retention. To address this, a garment can be designed to allow sportswear with a removable liner, which can be washed separately to reduce the need for laundering the actual garment. As a garment with an inner layer can be washed as needed, the outer layer can be worn for a long time. This inner liner could be a highly absorbent layer that could add comfort to the overall design.
Another design modification is to add highly absorbent lining at the sweat sites such as axilla. Socks have to be washed after every use; however, a removable insert of shoes can be washed weekly.46,47 This could be reinforced with odor trapping molecules making it more effective in odor control. Furthermore, ventilation designs such as mesh panels, underarm vents, or strategic perforations, can manage odor buildup by promoting air circulation in specific areas.
Evaluation of odor
Odor is quantifiable using several methods including objective, quantitative, and standardized scientific methods in odor testing laboratories as well as in the ambient air by trained inspectors. The odor parameters to be measured include odor concentration, intensity, persistence, and descriptors depending on samples from point, area, and volume emission sources.
34
Conti
Advantages and disadvantages of various odor sensing techniques (adapted from Conti
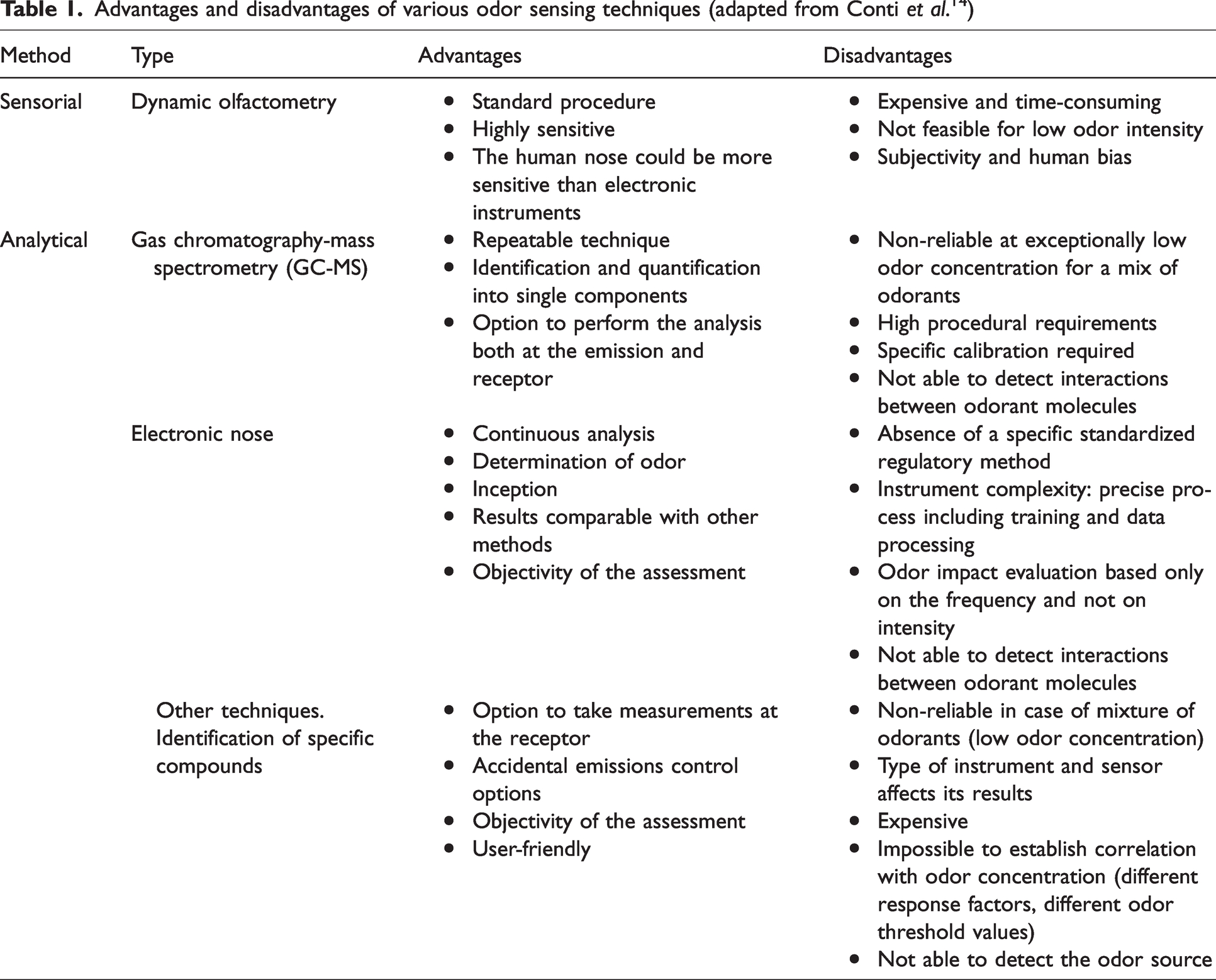
Odor can be measured and quantified directly in ambient air using one of two standard practices by trained assessors. The first method uses a standard “odor intensity referencing scale” made up of the standard odorant, n-butanol, to quantify odor intensity and the odor is then evaluated by a trained assessor, who has met predetermined repeatability and accuracy criteria.
The second method utilizes a calibrated field olfactometer, which dynamically dilutes the ambient air with carbon-filtered air in distinct dilution ratios known as “dilution-to-threshold” dilution factors. In this technique, a dilution series of the odor sample is created consisting of odor concentration at an increasing order. The calculation of dilution factors for olfactometry is based on the ratio of total volumetric flow divided by volume of odorous flow:

Standards of odor testing
One of the earliest standards of odor testing was ASTM D1391, “Standard Test Method for Measurement of Odor in Atmospheres (dilution method),” utilizing a syringe dilution technique. This was followed by ASTM E679-79, “Standard Practice for Determination of Odor and Taste Thresholds by a Forced-Choice Ascending Concentration Series Method of Limits.” When practicing ASTM 679-91 during an odor test, the odor panelist (assessor) sniffs a dilute sample of the odor as it is discharged from the olfactometer as one of three sample presentations (one presentation with the dilute odor and two with odor free air). The assessor sniffs all three of the presentations and must select one of the three that is different from the other two, even if they must guess. This statistical approach is called “triangular forced choice.” The assessor declares to the test administrator if the selection is a “guess,” a “detection” (the selection is different from the other two), or a “recognition” (the selection smells like something).
Methods of odor testing
Several odor evaluation methods have been developed worldwide. Sensory panel and instrumental methods for odor evaluation are discussed in this instance.
Sensory panel
The sensory panel method is a common method of evaluating clothing odor due to perspiration as discussed in multiple studies.34,48,49 In the study reported by Klepp
Instrumental analysis
The analytical methods of odor assessment include sensory gas chromatography (GC) analyses, closed-loop stripping analysis (CLSA), solid-phase microextraction (SPME), liquid–liquid extraction (LLE), open-loop stripping analysis (OLSA), purge and trap techniques, and steam distillation extraction (SDE) with GC/mass spectrometry (MS). 50 Of these, GC analysis method is found to be the most frequently used for textiles, while other methods are more commonly used for odors in other substrates such as water and sewage.
The principle of the GC method is the separation of the components from the odor mixture according to their affinity with the stationary phase in the column. 49 Since each type of molecule has a different rate of progression, the various components of the analyte mixture are separated as they progress along the column and reach the end of the column at various times (retention time). According to the retention time, components are qualitatively identified with MS. 48 The techniques amplifies their potential and enables lower detection limits, allowing identifying analytes in exceptionally low concentrations. 33
An alternative approach to GC–MS is GC–MS coupled with olfactory analysis (GC–MS/O). The process is equal to classical GC–MS with the only difference that at the end of GC, the sample is split between an MS detector and trained human panelists. 49
Electronic noses (Figure 5(a)) have the capability of mimicking human sensing (Figure 5(b)). The e-nose (Figure 5(a)) operates with the help of a reference odor database, like the human brain. The e-noses perform the function by interacting sensor-arrays which respond to the analytes present on an extremely sensitive material. 51 This reaction is followed by adsorption or desorption at the surface, triggering the sensor arrays to record digital messages. 52
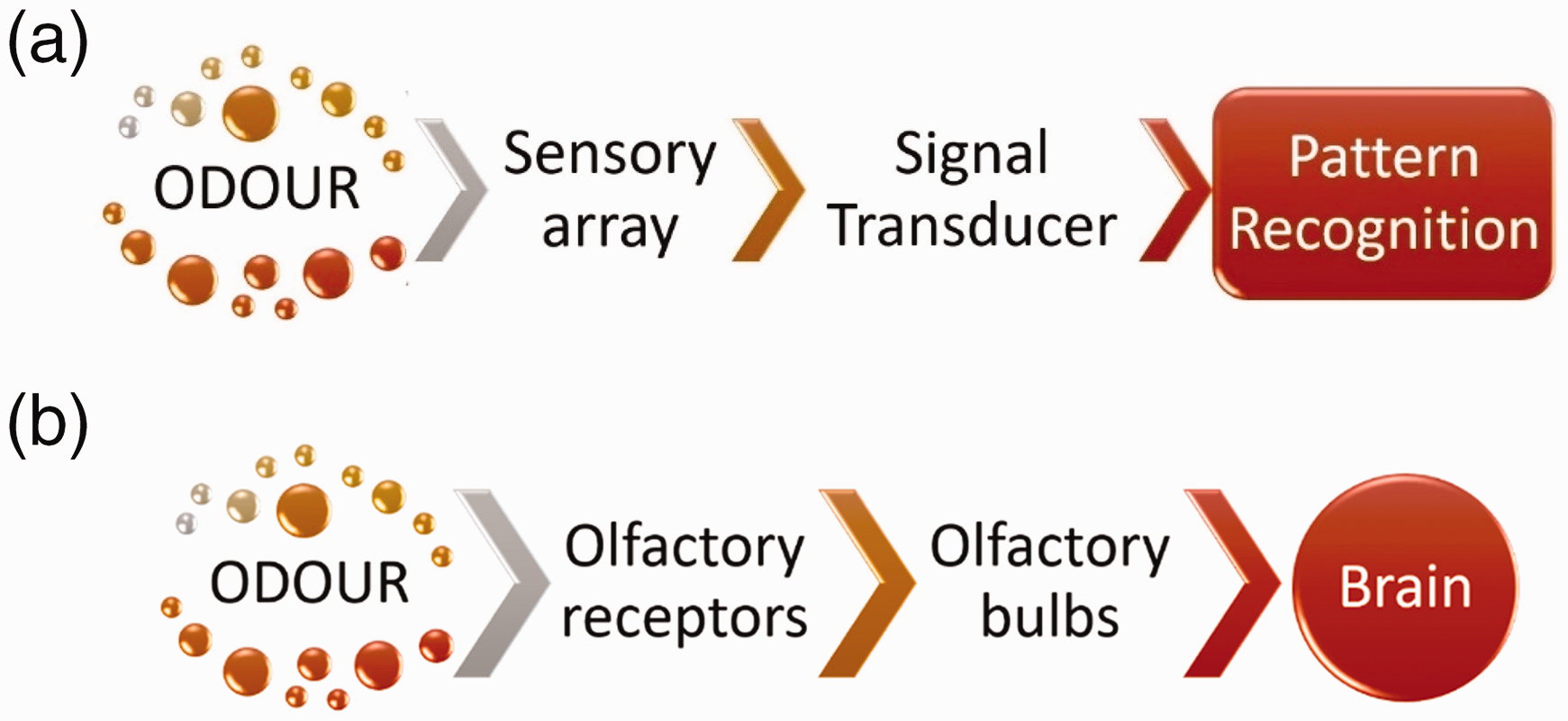
Illustration of difference between mammal olfactory and electronic noses: (a) artificial sensory system; and (b) mammalian olfactory system.
Comparison of global standards of odor evaluation
The standards used worldwide for assessing odor in textiles differ greatly due to regional and environmental priorities, industrial practices, and the existing regulatory frameworks. Although dynamic olfactory analyses using human nose (EN 13725, ASTM E679, and AS/NZS 4323.3) are widely used and accepted for their objectivity and accessibility, they are costly, rely on human assessors, and, hence, are prone to some subjectivity. Japan uses a variation of olfactometry methods (JIS Z 8100-01 and Offensive Odor Control Law) such as the triangle odor bag method, involving the collection of air samples in odor-free bags, which are then presented to a human panel in a triangular test format to identify the odor-containing bag. South Korea (Odor Pollution Prevention Act and Standard Method of Odor Evaluation) along with Japan has a separate methodology for odor intensity measurement or odor index based on measuring odor intensity and hedonic tone (pleasantness/unpleasantness), along with concentration.
China regulates odors under the GB 14554-93: Emission Standards for Odor Pollutants and GB 3095-2012: Ambient Air Quality Standards. This involves an odor intensity scale, where trained panelists rate the strength of the odor on a numerical scale sometimes supplemented by instrumental methods. The ASTM standard ASTM E679 is similar to EN 13725 and relies on trained human panels to determine odor thresholds, whereas ASTM D1391 is used for measuring odors in various atmospheres, such as environmental monitoring and industrial emissions. When choosing an odor evaluation method, odor source and thresholds should be taken into consideration. 53
Odor management in textiles
Increases in pollution, changing living standards, and resistant microbes have led to an augmented interest in odor management in textiles. The fragrant smell of fresh, clean laundry is advertised today as a sign of a healthy and pleasing lifestyle. Hence, researchers worldwide are now focused on creating smart textiles with odor management abilities. Hu
Odor neutralization
The term odor neutralization refers to reducing concentrations and mixing with new odorous compounds which are acceptable (masking). It is a passive approach and can be achieved by simply drying the fabric in the presence of fresh air. 55 The masking can be as simple as applying a fragrance or other masking agents. This method offers effective management by providing a quick and short-term solution. For a more durable solution, fabrics can be engineered to entrap or mask odors at the time of manufacture. Examples include the use of compounds such as silica, where odor molecules adhere to the silica particles, and are then broken down to carbon dioxide and water (Polygiene®). 56 Commercial brands such as Lava X2® 57 employ a zeolite carrier to deliver antibacterial finishes to target bacterium involved in odor production. Some commercial products employ more than one method such as both antimicrobial finishes as well as infused nanosilver for odor elimination, achieving promising better results.
Odor neutralization by masking is an effective approach; however, it not only is a short-term solution, but also may have an environmental impact such as indoor air pollution, and in some cases health effects.
Odor removal
It is sometimes possible to close an odorous system to prevent the release of the odor to the atmosphere. This involves stopping the source of odor, such as by perspiration plugs before it becomes a nuisance. Once on textiles, the odorous substances can be removed completely using materials such as activated charcoal or baking soda. The activated charcoal “traps” odorous plumes, leaving behind non-odorous fresh air. The presence of fresh air is vital for this process. The activated charcoal has a large surface area causing the odorous plumes to be adsorbed into the activated charcoal due to weak van der Walls forces. As the plumes pass through the tiny pores, the weak forces cause them to cling in between the pores. Another possible removal is by enzymatic cleaners which break down the odorous compounds, such as those used in pet care after spills. These methods provide a targeted approach but may only work for certain kinds of odors. For example, baking soda works well for acidic odors but is inefficient for alkaline compounds such as ammonia. These substances can become saturated and need to be replaced periodically. Furthermore, some of the odor removal compounds may contain or produce harmful byproducts and therefore proper handling and disposal is required.
Odor reduction by dilution
If an odor is not a toxic substance and has no harmful effects at concentrations below its odor threshold, dilution may be the least-expensive control technique. This technique involves a dilution of the odor sample by reducing the odor concentration gradually. An example of the method currently employed for textile odor reduction by dilution is the use of washing in water. The present odor is diluted in the presence of water and is carried away by water molecules. The effect of drying odorous clothes in the presence of fresh air will have a similar effect. Both diluting by water and air will work more efficiently in the presence of some additional catalyst to speed up the process. The presence of surfactant in water and activated charcoal in drying 55 will also have similar effects.
Odor conversion
The odor conversion method is widely used and, hence, can be regarded as a possible approach for textile odor management. In this method, odorous compounds may be converted into compounds with different odor thresholds. This may result in the formation of other compounds with an odor threshold much higher in magnitude than that of the original compound. 32 The methods currently employed for textile odor reduction by conversion include the use of washing compounds with added odorous substances such as essential oils and fragrant fabric softeners.
Odor and antimicrobials
Biochemical odor elimination works in two ways, either by deterring propagation of bacteria in the case of bacteriostats and fungistats (without altering any other bacterial properties) or by bacterial elimination through antimicrobial agents (bactericides and fungicides) such as silver. Odor control chemicals in clothing material can be activated through moisture to prevent growth of odor-producing bacteria. Silver is well known for its antibacterial properties and silver nanoparticles are extensively used in odor-control technologies. Odor control can also be achieved by non-nanoscale silver ions or triclosan. Simoncic and Tomsic 58 have discussed various organic and inorganic antimicrobial agents including natural bioactive agents which are plant based. These include chitosan, polybiguanides, N-halamines, and quaternary ammonium compounds which are used for a broad spectrum of microorganisms. Scentry® is an example of odor resistant finish which claims to be safe and sustainable odor management technology employed in clothing. 59 It is a liquid-based coating and can be employed to any product to maintain freshness up to five wears between washing. The antimicrobial agent can be bound directly to the fiber or can be affixed to a host molecule such as β-cyclodextrin or its derivatives such as 6-monodeoxy-6-mono(N-tyrosinyl)β-cyclodextrin. 60 The popular applications of odor-resistant clothing are active wear, work wear, and socks. However, the products also find their use in intimate apparel, medical textiles, footwear, and home textiles.
Antimicrobials are an emerging method of odor management, but the method does have some drawbacks. The toxicity of antimicrobial compounds remains a question and may cause health problems for users with sensitive skin. The durability of these compounds also needs to be improved as the antimicrobial effectiveness may be reduced with washing. The compounds need to be compatible with other textile processes such as dyeing and finishing, in addition to laundering. 61
Emerging trends in odor management
Alongside the order-management technologies discussed in this article, newer trends have emerged such as moisture removal, plasma technology, photocatalytic, and self-cleaning methods. One of the simplest and most effective odor control is by moisture removal. The new age of smart everyday textiles is moisture-wicking and breathable fabrics with odor control capability. The fabrics pull sweat and moisture away from the skin, thereby reducing the conditions that promote bacterial growth and subsequent odor formation. Further odor control can be reinforced with silver ions to include antimicrobial properties, hence reducing the growth of odor-causing bacteria. Another important moisture control textiles is achieved by incorporating phase change materials which aids by absorbing, storing, and releasing heat, causing a dry environment on the skin’s surface. 62
Plasma technology involves treating textile fibers with plasma (an ionized gas) to modify the fabric surface properties and generate chemically reactive sites, enhancing affinity for other substances. Thus, this can improve the antimicrobial properties of textiles, enhance dye uptake, and even impart odor control without the need for chemical coatings. 63 Plasma technology is an ecofriendly method being used to alter the surface of textile fibers at a molecular level, making them more resistant to bacteria and odors.
Photocatalytic and self-cleaning textiles incorporate materials such as titanium dioxide (TiO2) that react with light (typically UV light) to produce reactive oxygen species, breaking down organic matter, including odor-causing bacteria, mold, and pollutants which helps eliminate odors and stains when exposed to light. 64 Other compounds such as enzyme-like catalytic functions of iron (III) or cobalt (II) phthalocyanine (Fe(III)-, Co(II)-) complex derivatives may also be employed to achieve self-cleaning ability. The photocatalytic activity can also be attained using metal ions deposited on the textile surface by the process of sputtering. These metal ions include gold, silver, aluminum, tin, zinc, nickel, stainless steel, titanium, and cobalt-based alloys. 65
Laundering alternatives
The monetary value and environmental impact of washing have led several researchers to explore alternatives to laundry practices. Denim is a particularly good example of resilient clothing. It is rarely washed after every use. 47 This suggests that we can engineer clothing to impart desired characteristics such as laundry-free textiles. Soiled clothing can be fixed before it needs to be fully washed. This can reduce the need for laundering and depletion of useful resources, in addition to time and labor. In the simplest terms, one can avoid washing by rewearing a garment several times. However, this has some problems that are associated with perception as well as microbial growth. Some alternative methods to clean clothes as substitutes to washing are discussed in the following.
Steaming
Steaming a garment is an effective method for removing odors as the high temperature steam penetrates fibers in the fabric, breaking down odor-causing bacteria and other microorganisms. 47 The steam passing through the fibers allows the heat to loosen dirt, debris, and oils that can harbor smell-causing compounds. 66 This process not only sanitizes the garment but also refreshes its appearance by reducing wrinkles and enhancing its overall cleanliness. In addition, steaming can help to dissolve and remove residues from sweat and environmental pollutants that contribute to unpleasant odors. Since steamers do not use harsh chemicals, they are a gentle yet powerful alternative to traditional washing, making them highly environmentally friendly and a practical way to remove odor.
Airing
Airing a garment is the simplest and most natural method for removing odors, 47 leveraging the power of fresh air and proper ventilation. When a garment is hung outside or in a well-ventilated space, the circulation of air helps to disperse and evaporate sweat, oils, and other odor-causing substances that have accumulated in the fabric. The removal of moisture deprives microorganisms from their needed water to reproduce and multiply; hence reducing odor caused by these microbes. Exposure to sunlight can further enhance the process, as UV rays have natural antibacterial properties that help neutralize odor-causing microbes. The movement of air helps to lift and carry away the unwanted particles, leaving the garment smelling fresher. This method is particularly advantageous for delicate fabrics or items that cannot be washed frequently, as it avoids the wear and tear of mechanical washing and also avoids use of detergents while still effectively refreshing the garment.
Clothing brush
A clothing brush or lint remover can mechanically lift away particles and residues that cause unpleasant odors. When brush or lint remover is passed over the fabric, it dislodges and removes lint, dust, pet hair, and other debris that have settled into the garment over time. These particles often trap and hold odor-causing substances, such as sweat, oils, and environmental pollutants. The removal of debris also eliminates some of the sources of odors. In addition, brushing can help to refresh the fibers on fabric, allowing them to breathe better, which further aids in the dissipation of any residual aromas. This process is a quick and gentle method to revitalize the garments, keeping them clean and odor-free without the risk of damage from frequent laundering.
Stain removal
One of the key reasons why we wash clothes is because of stains attached to the fibers. Stains can be reduced by using a stain remover instead of washing the complete garment. The stain remover can be chosen based on the type of stain such as coffee, blood, red wine, or sweat. Kissa 67 has studied the mechanism of staining and stain removals. It has been observed that the permanence of stains such as coffee on textiles depends on the nature of the fibers. For example, the stain affinity with coffee stain is the highest for nylon and the lowest for polyester. This may be due to ionic interaction of carboxyl and phenolic groups with amine end-groups in nylon. Due to the inherent acidic nature of the stain, an alkaline medium is required to detach the stain.
Clothing mist
An incredibly straightforward and effortless way to reduce the odor is to mask the odor using a scented mist. A clothing freshener spray can remove odors from a garment by neutralizing and masking unpleasant smells. The spray typically contains a combination of water, alcohol, and essential oils or fragrance compounds designed to counteract odor molecules. When the freshener is sprayed onto the fabric, it disperses these agents that bind to the odor-causing particles and neutralize them. Some fresheners also contain antibacterial agents that can help eliminate bacteria responsible for producing bad smells. The misting solution also penetrates the fibers, reaching deep-seated odors that might not be removed by surface cleaning such as cleaning brush alone. In addition, the pleasant scent of the freshener leaves the garment smelling clean and refreshed. This method is particularly useful for freshening up garments between washes, removing the need for excessive laundering, while extending the life of garment.
These above-mentioned methods are currently employed as alternatives to laundry in places where water is scarce, or in extremely low temperatures including outdoor recreational activities. Hence, it can be argued that these techniques can be applied to everyday wear, reducing the water footprint and laundering bill. More investigation is, however, needed to manufacture antiodor textiles to meet the demands of the changing lifestyles globally.
Limitations in odor management
Current technologies in odor management in textiles, while improving, still face several challenges and limitations that prevent them from being entirely effective.
Several odor-control technologies, such as antimicrobial treatments, rely on chemical coatings that gradually wear off after multiple washes which result in odors returning after extended use or numerous wash cycles. Furthermore, the environmental impact of these coatings remains a critical concern. The antimicrobial coatings (AMCs) have been accused of harming the aquatic ecosystems. The silver-based AMCs have been researched extensively and are used widely. Other AMCs include titanium, zinc, chitosan copper, and quaternary ammonium compounds, of which titanium and chitosan are mainly carrier agents for release-based antimicrobial fouling compounds. 68 These compounds pose a dire threat to marine life as well as the terrestrial ecosystem by becoming part of food chains and food webs.
The use of nanosilver may have a toxic effect on the human body 69 and the nanosilver can also impair the viability of human cells.25,70 However, it has been argued that silver nanoparticles are not a health risk in general unless they come in direct contact with an injured body area. Furthermore, use of non-nanoscale silver ion-generating materials can be a better option considering this issue. 71
While odor control may be seen as a valuable addition to the textile, advanced odor-control technologies can significantly increase the cost of textile products. Antimicrobial coatings such as silver ion-based technologies or other chemical coatings impose additional cost, raising the price tag of the end product.
With washing being the primary odor-management practice, its environmental impact is highly underestimated. Moazzem
Although washing remains the most common odor management practice, it is understood that even the gentlest wash can eventually deteriorate the longevity of textiles. The other environmental concern with washing clothes is the release of microfibers into the wastewater. Henry
Other than washing, the effectiveness of the current odor-management practices is also found to be rather uncertain. After reviewing different odor control technology, Klepp
Conclusion
Laundering habits are shaped by a combination of social, cultural, environmental, and economic factors. Cultural norms and demographics around cleanliness, gender roles, environmental consciousness, and technological advancements all play a role in shaping these behaviors. Other than the clean look of textiles, odor is the driving force of frequent laundering of textile material. Odor is a manageable problem associated with textiles and is the leading cause of textile laundering. However, humans grapple with the disparity between perceived need versus the evidence based necessity for laundering. The issue of odor in textiles is intricate and is not completely understood. The odor in textiles can be associated with several factors including intensity, persistence, descriptors, type of substrate, and human perception. The odors arise from various sources in the human body and textile substrate can present both as a problem and a solution. The textiles provide incubation space for microbial build-up. They can be modified to absorb sweat, therefore taking away the moisture needed for odor development, and combating the sweat and subsequent malodor issues. However, the odor-detection and assessment methods present a challenge and need to be given attention.
Fibers and fabric structures play a key role in odor management in textiles. Natural fibers such as wool and cotton are less odorous than synthetic fabrics in clothing. Natural fibers have inherent antiodor properties due to their ability to absorb moisture, though more research is needed to better understand the mechanisms. Evidence suggests that fabric structure and design also influence odor properties in textiles. The methods of assessing odors in textiles include sensory or analytical evaluation. The type of method employed is based on textile end use such as odor development in bandages due to microbial activity from wounds or textiles in sports due to odor from sweat.
Odor management in textiles is done using several methods such as elimination, dilution, or conversion. The effectiveness of odor-management techniques depends on the context and specific application. While masking agents provide quick, short-term relief, they do not address the source of the odor and may lead to potential health concerns. New materials and techniques need to be explored to eliminate odor retention and boost odor removal performance in textiles. So far, odor management in textiles has not been explored fully and requires more research input to combat this manageable issue and to reduce environmental impact.
The limitations in odor management involve perception of soiled clothing, i.e., the need to wash. There are several alternatives to avoid washing, which can be practiced reducing environmental impact of washing as well saving time and energy. The needless washing also contributes to overconsumption of water and energy, in addition to increasing pollutants in water such as detergents that may contain suspected carcinogens and non-biodegradable ingredients. 76 Frequent or incorrect washing may also result in microplastics in complex food webs and clothes losing their shape, color, and thickness.
The future of odor management is moving toward more sustainable, multifunctional, and smart technologies that not only provide effective odor control but are also ecofriendly and cost effective. Innovations in nanotechnology, bio-based materials, plasma treatments, and photocatalytic textiles offer promising new solutions for combating odors in textiles. These emerging technologies are particularly important in industries such as medical textiles, activewear, and smart fabrics, where performance and hygiene are critical factors. However, the challenge remains in balancing effectiveness, durability, cost, and environmental impact to ensure that these solutions are accessible and sustainable in the long term and can help in optimizing the need for laundering of textiles.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is financially supported by the Australian Research Council (ARC) through grant LP190101294 and Hunting Lady Pty Ltd trading as Ventou Garment Technology.
