Abstract
Background
Cardiovascular disease affects over 7 million people in the UK and statins are often prescribed to mitigate cardiovascular risks. The effect of statins on a number of cancers is debated and their effect on Bacillus Calmette-Guérin (BCG) responsiveness in non-muscle invasive urinary bladder cancer (NMIBC) is not fully understood.
Aims
This study aims to explore the difference in HMG Co-A reductase (HMGCR) expression in NMIBC on immunochemistry in BCG responders and non-responders while on statins.
Method
Three hundred and thirty-two cases of intravesical BCG treatment for high-risk NMIBC between November 2003 and December 2017 were identified. Patients taking statins for at least 12 months before the diagnosis of NIMBC and with a follow-up of at least 5 years were included. They were divided into BCG responders and non-responders. Tumour tissue from these patients was immunohistochemically stained and quantitative image analysis carried out to assess and compare HMGCR expression in the groups.
Results & Conclusion
This study showed a differential expression of HMGCR in responders vs. non-responders to BCG for high-risk NMIBC on statins. This data should form the basis of a further research and multi-centre study in a larger cohort, using HMGCR as a biomarker of response in patients on statins.
Introduction
Bladder cancer accounts for around 3% of all new cancer cases annually in the UK. Over 90% of cases are urothelial in nature. It is the 11th commonest cancer in the country and 8th commonest among men. As of 2018, it was the 10th most common cause of cancer-related mortality with around 25% of cases being diagnosed at a late stage in England and Northern Ireland. 1 The majority, however, present with non-muscle invasive bladder cancer (NMIBC) which has a natural history of high recurrences without adjuvant intravesical treatment. Following transurethral resection of bladder tumour (TURBT), histological assessment and risk stratification, further management and follow-up plans are formulated according to guidelines. TURBT alone for high-risk NMIBC is associated with recurrence rates of 50–70% and progression rates of 10–20%. Hence, high-risk group patients are offered intravesical immunotherapy with Bacillus Calmette-Guérin (BCG) in a bid to reduce the risk of disease recurrence and progression.
Statins are one of the most commonly prescribed drugs in the UK, according to British Heart Foundation, to reduce the risk of cardiovascular disease and hypercholesterolaemia. 2 Controversy exists as to the overall effect of statins in the treatment of bladder cancer. In-vitro models have suggested both anti-cancer and pro-cancer effects.3,4
Statins specifically inhibit the activity of HMG Co-A reductase (HMGCR), which is the rate-limiting enzyme of the mevalonate pathway (Figure 1) for cholesterol synthesis. Studies using cell lines have shown statins suppress cell proliferation5,6 and reduce cell migration, 7 invasiveness and metastasis formation 8 in murine models. 4 Many of these effects are a consequence of HMGCR inhibition and resulting interruption to mevalonate pathway-produced metabolites, including isoprenoid intermediates farnesyl (FPP) and geranylgeranyl-pyrophosphate (GGPP) as well as cholesterol. 5
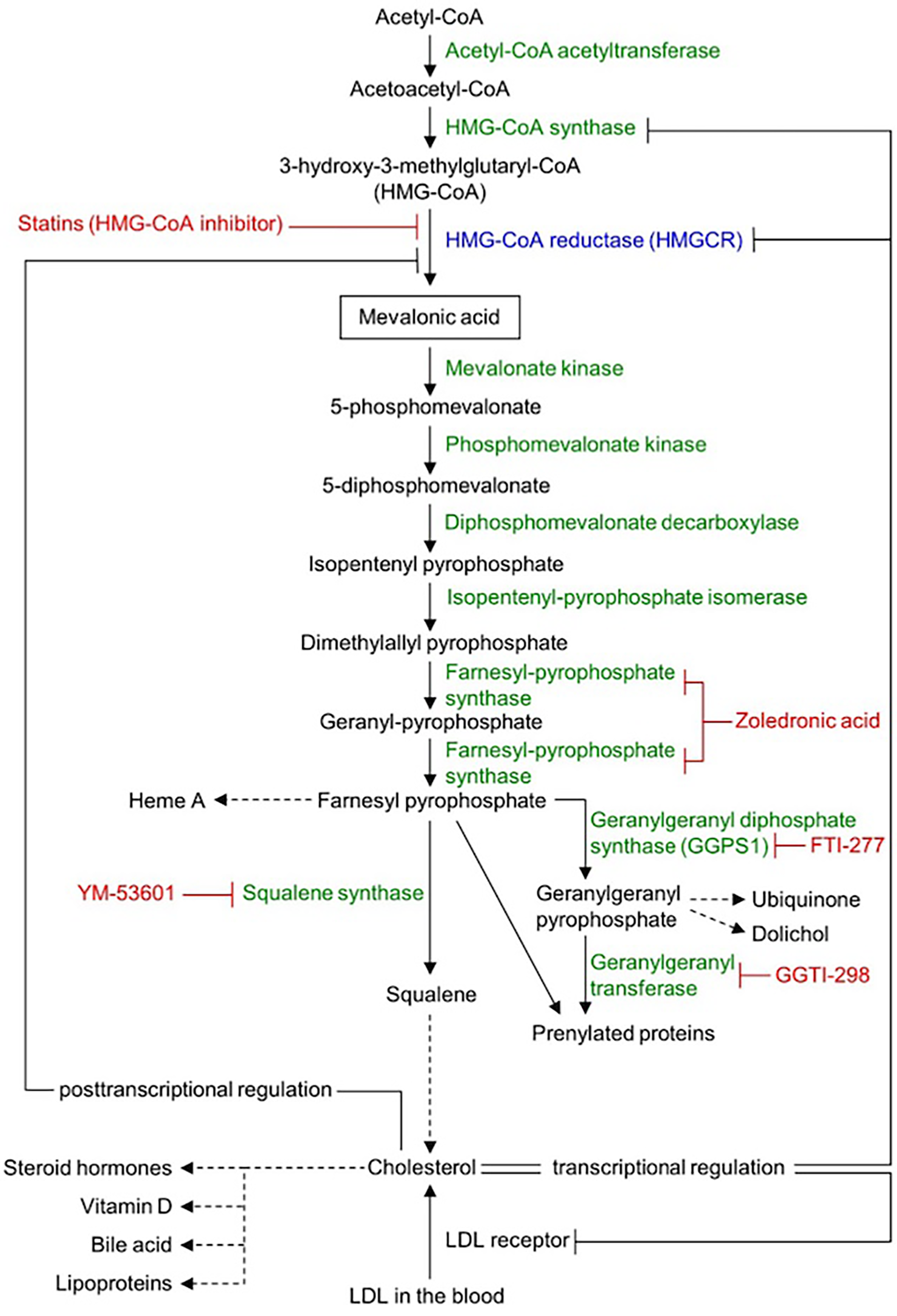
Mevalonate pathway.
The reduction in GGPP leads to reduced activity of RhoGTPase and inactivation of the Hippo pathway transcription factors YAP and TAZ. 7 The end result of this is reduced tumorigenesis and metastasis formation. 9 In addition, there is reduced prenylated RAS activity as a result of downregulation of GGPP. This leads to reduced activity of canonical MEK/erk/P13 kinase/Akt/mTor signalling pathways. 10
Statins have been shown to reduce inflammation by skewing the response from a T-helper-1 to a T-helper-2 response, which may reduce the effectiveness of immunotherapies, such as BCG.11,12 Intravesical BCG favours a T-helper-1 cell response involving CD8+ lymphocytes and natural-killer cells, so a shift to a T-helper-2 response could in theory be counterproductive. 13 As a result, some debate exists regarding the response to BCG instillations in patients with high-risk NMBIC on statins.
A number of retrospective studies have specifically assessed the impact of statins on BCG responsiveness. While Hoffman et al. concluded the medication was associated with poorer BCG responsiveness, 14 studies by Kamat and Wu, Berglund et al. and Skolarus et al. found no significant difference among statin and non-statin users.15,16,17
A similar single-centre study by Hillen et al. also carried out at Ninewells Hospital concluded patients should continue taking statins while receiving their BCG therapy as no effect was seen on recurrence-free or overall survival between cohorts. 18 These findings were similarly supported by Crivelli et al. 3
In a proof-of-concept study, we analysed the difference in HMGCR expression in BCG responders and non-responders with high-risk NMIBC while on statins.
Patients and methods
Cohort identification
We used Tayside Urological Cancers (TUCAN) database to retrieve electronic patient records. A cohort of 322 patients who received BCG therapy for high-risk NMIBC between November 2003 and December 2017 was identified. Patients taking statins for at least 12 months prior to diagnosis of NMIBC and who had complete follow-up of at least 5 years were included. Patients with upper tract disease and those who received radiotherapy were excluded. The patients were then divided into responders (n = 10, no recurrences or progression) and non-responders (n = 9, with disease recurrences or progression). Data collected included patient identifiers, tumour stage and grade of original tumours for both groups.
Figure 2 details the follow-up schedule for high-risk NMIBC used in NHS Tayside. Following initial TURBT, early re-resection is offered at 4–6 weeks, after which cases are discussed at a Urology Multidisciplinary Team Meeting. Typically, patients are then preferentially offered intravesical BCG immunotherapy. Alternative options that may be offered include cystectomy or endoscopic surveillance depending on the patients’ lower urinary tract symptoms, recurrent urinary tract infections, anaesthetic fitness and patient choice. Subsequent follow-up includes CT-Urogram at 18, 36 and 60 months and regular check cystoscopies for 10 years.
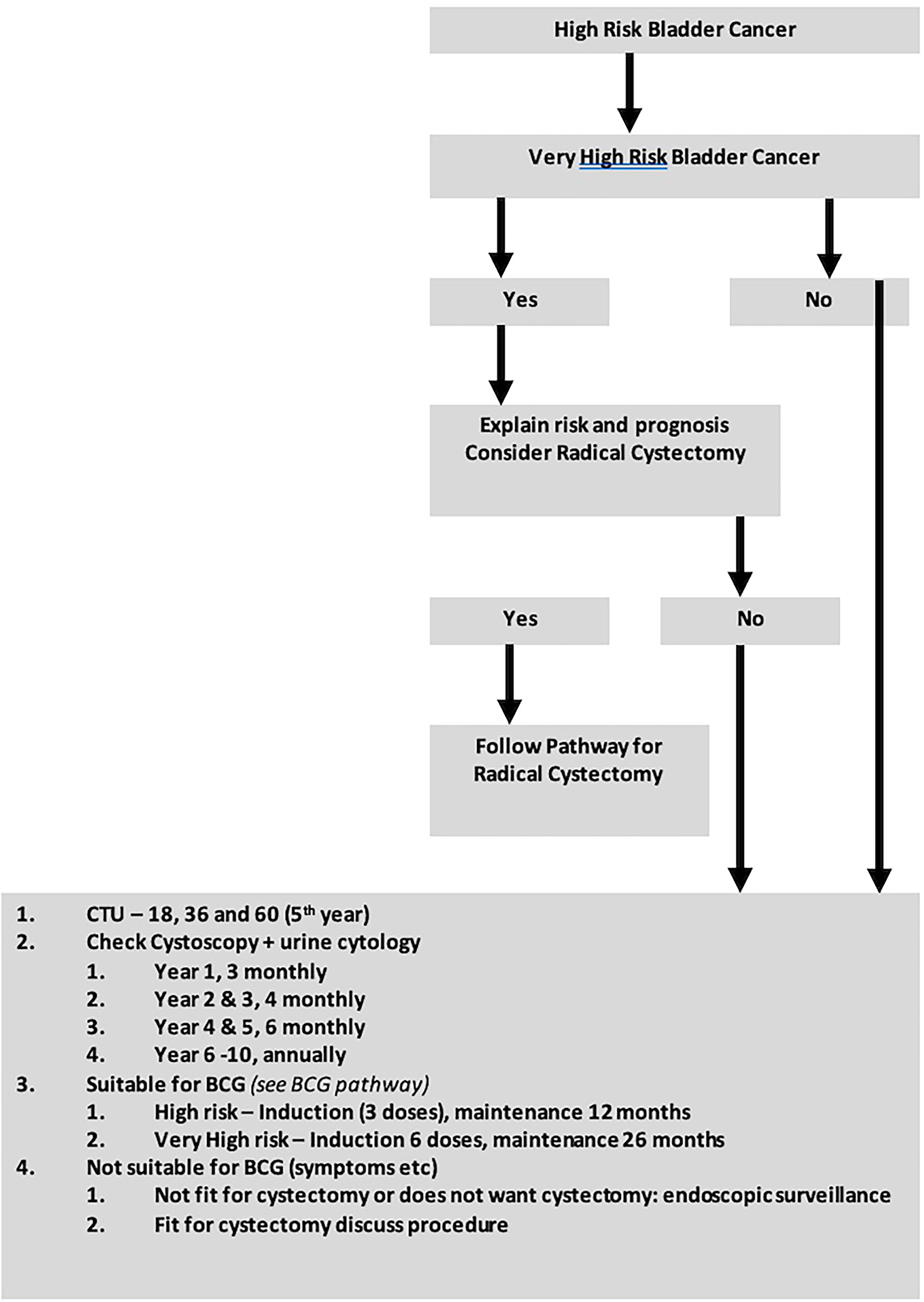
NHS tayside follow-up schedule for high-risk NMIBC. NMIBC: non-muscle invasive bladder cancer.
Immunohistochemistry
We retrieved histopathology blocks for both responders and non-responders using Tayside Tissue bank protocol. Antigen retrieval and de-paraffinisation was performed using DAKO EnVision™ FLEX Target Retrieval solution (high pH) buffer (50× conc) (K8004) in a DAKO PT Link (serial number PT2794Y1205) for 20 min at 97°C. Immunostaining using DAKO EnVision™ FLEX system was performed using a DAKO Link Autostainer. Sections were initially washed in Flex Wash Buffer (K8006). The following steps were then performed manually:
1. Flex Peroxidase-Blocking Reagent (SM801) applied for 5 min 2. Incubation with HMGCR C-1 (Santa-Cruz Biotechnology sc-271595) diluted at 1–25 in Flex Antibody Diluent (K8006) overnight at 4 degrees centigrade. 3. Flex/HRP labelled polymer (SM802) for 20 min 4. Flex DAB + working solution (SM803) for 2 × 5 min 5. Copper Sulphate solution for 5 min 6. Flex Haematoxylin for 5 min
The following steps were then performed using the automated platform:
In between steps, sections were rinsed with Flex Wash Buffer with a final wash dH2O.
Sections known to stain positively were included in each batch and negative controls were prepared by replacing the primary antibody with DAKO antibody diluent.
Slides were manually washed in tap water before being rinsed in graded concentrations of alcohol, with a final rinse in Xylene. Glass coverslips were applied.
Quantitative image analysis carried out using software (Leica Aperioà slide scanner at 40× magnification). The positive pixel count algorithm was used to compare HMGCR expression between the groups. Number and intensity of pixels were quantified and correlated with clinical outcomes. Positive pixel count algorithm is designed to quantify the amount of a HMGCR stain in a scanned slide image by segmenting each pixel within the outlined region into a range of intensities. These intensity ranges were classified into background (blue), weak positive (yellow), medium positive (orange) and strong positive (red). Colour quantification was correlated between two the groups.
Results
Table 1 summarises the details of urinary bladder cancer in the two groups depending upon the response to intravesical BCG: responders and non-responders. There were no significant differences in the clinicopathological characteristics between the two groups. Briefly, in the responders group (n = 10), one patient had a high grade (HG) G2pT1 tumour on their original diagnosis, while four had G3pTa and the remaining five had G3pT1 tumours. In total, eight patients had concomitant carcinoma in situ (CIS) at the time of their original diagnosis. Eight of the responders were taking Simvastatin and the remaining two Atorvastatin. Among the non-responders (n = 9), eight were taking Simvastatin and one Rosuvastatin; five had original diagnoses of G3pT1 tumours, one with HG G2pTa, two with HG G2pT1 disease and one with CIS only. Additionally, six of the non-responders had concomitant CIS on their original diagnosis.
Comparison of responder and non-responder groups.
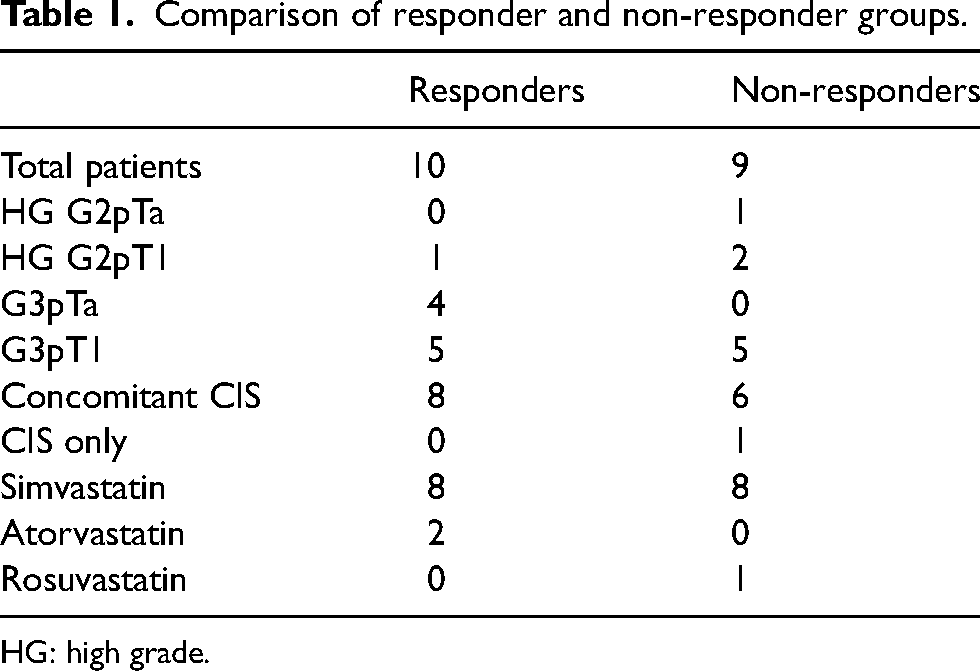
HG: high grade.
The dose of statins being taken by the responder group ranged from 10 mg to 40 mg while in the non-responder group the range was from 5 mg to 40 mg.
A differential expression of HMGCR was observed between the responder and non-responder groups, with higher HMGCR immunopositivity in non-responders (Figure 3). The differential expression of HMGR between the two groups demonstrated a correlation trending towards but not reaching statistical significance (Figure 4).
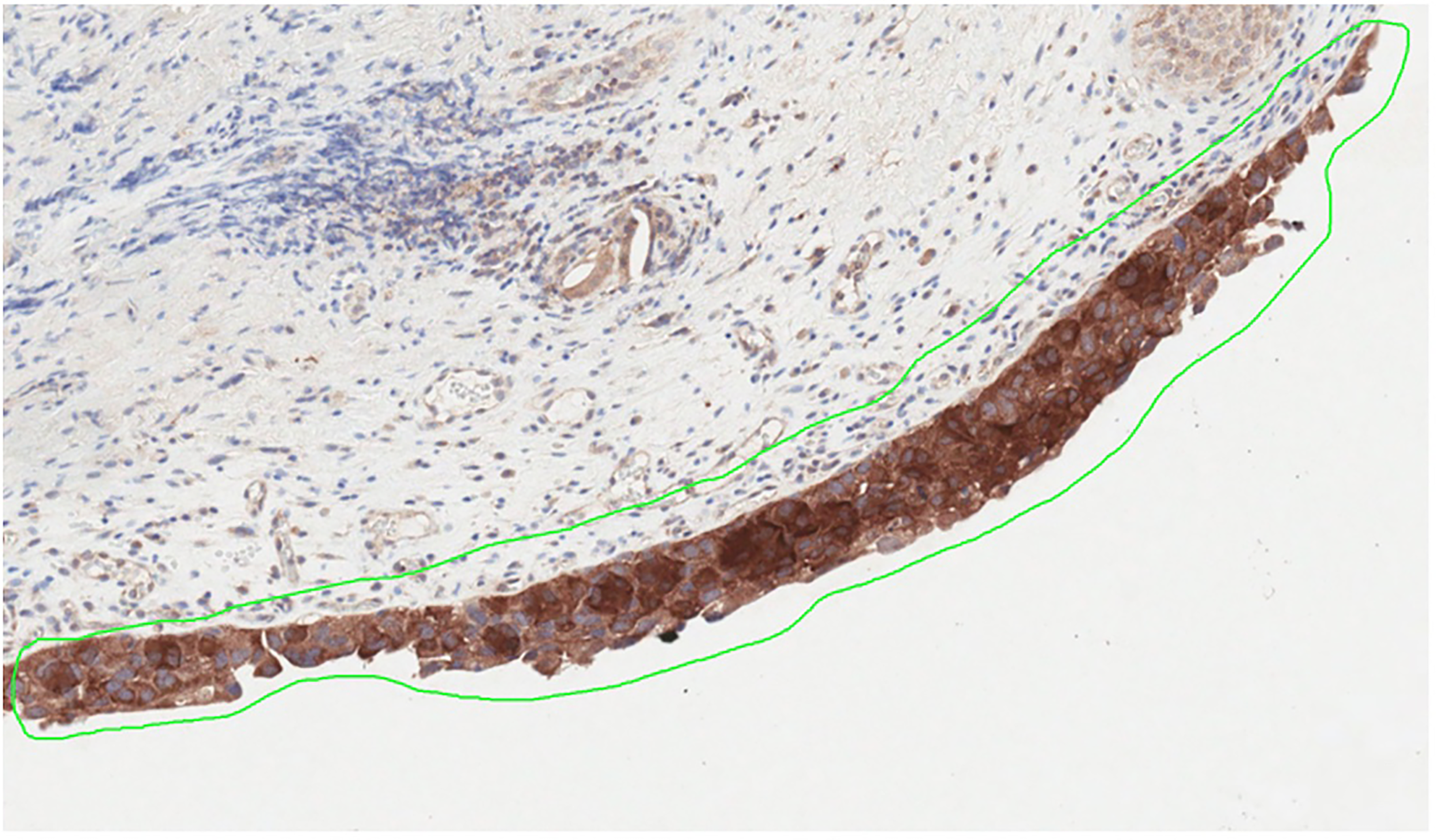
Histopathological slide demonstrating HMGCR immunopositivity. HMGCR: HMG Co-A Reductase.
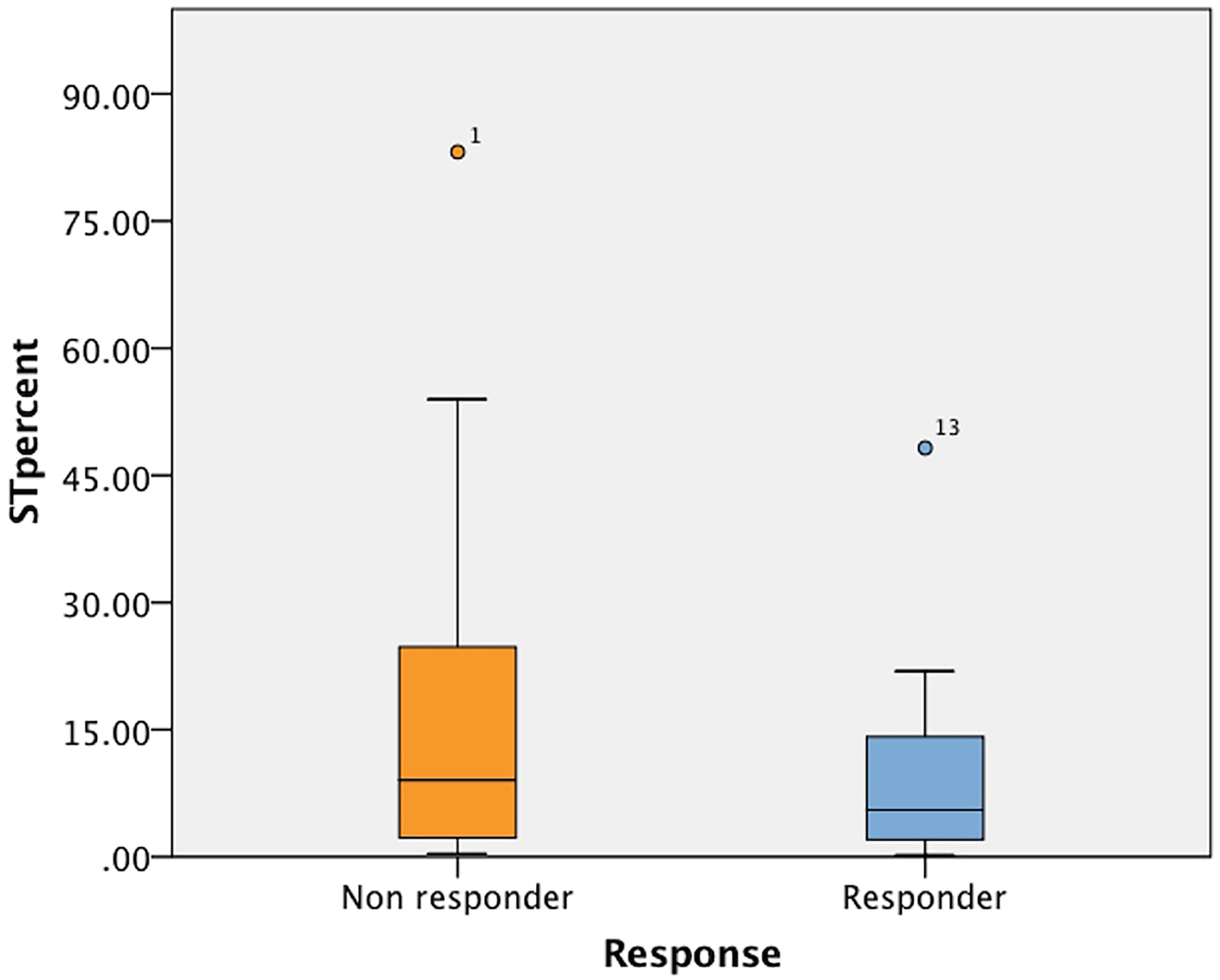
Box plot demonstrating differential expression of HMGR between responders and non-responders.
Discussion
This is a first proof of concept study measuring expression of HMGCR in NIMBC using patient paraffin fixed tissue blocks as histopathological resources. The study showed some differential expression of HMGCR between those who responded to intravesical BCG in comparison to those who did not. Non-responders had a slightly higher level of expression; however, this was not statistically significant. Observations of our study – first using clinical patients’ sample, are supported by basic ex-vivo experiments. Cell line experimental work by Ishikawa et al. demonstrated HMGCR expression levels remained insignificant with and without statin therapy. 4 Subcellular distribution of HMGCR also remained unaltered following commencement of atorvastatin therapy, in both statin-sensitive and resistant cell lines. Furthermore, it was suggested that statins exert their effect by inhibiting HMGCR enzyme activity. Exogenous downregulation of HMGCR expression in statin-resistant cell lines did however improve the anti-cancer effect of atorvastatin. This was achieved by inhibiting synthesis of GGPP, thereby inhibiting cell growth. This would propound that a joint targeting of FPP and GGPP alongside HMGCR enzyme blockade could have a synergistic anti-cancer effect. Such downregulation of HMGCR expression may be of benefit in BCG immunotherapy for NMIBC, particularly for non-responders. Our findings focus future research in this direction.
Contemporary literature from other site cancers suggests a role of HMGCR inhibition in delaying cancer progression. Kimburg et al. showed that higher HMGCR levels were linked with atorvastatin resistance in breast cancer. 19 The use of statins is linked with significantly reducing the risk of developing lung and colorectal cancers compared to non-users in a retrospective cohort study of USA Veteran population. This study also suggests a ‘dose-response relationship’, as the cancer incidence risk decreased with higher doses of Simvastatin. 20
This appears to be in keeping with results reported by Poynter et al. and Khurana et al.21,22 The former, in a retrospective case–control study, found that long-term statin use was associated with a reduction in the relative risk of colorectal cancer, while the latter noted similar results in relation to lung cancer in a Veteran population.
While inhibitory effect of statins on cancer progression and cancer-related death has been reported, their role in potentiating or otherwise response to intravesical BCG treatment in NMIBC remains unresolved and needs further research. Our study showed expression of HMGCR protein on the cells of both responders and non-responders, albeit marginally higher in the latter. It may be possible that non-responder's bladder cancers to BCG treatment are statin resistant. Göbel et al. in a recent study of HMGCR expression in breast cell lines have shown an increased level of HMGCR expression in statin-resistant cells. 23 Similarly, Longo et al. have shown a higher level of HMGCR expression was associated with poor prognosis in prostate cancer. 24 The role of statin resistance and its impact on response to intravesical BCG in NMIBC needs further research including identification of targets to reverse acquired resistance. The approach may improve outcome of intravesical treatment using BCG.
Future role of statins in high-risk NIMBC along with intravesical BCG needs focused hypotheses and exploration of its role in potentiating reduced recurrences and cancer progression. Inasu et al. in a large breast cancer cohort have reported a reduced rate distant metastases in women on statin vs. those who were not. However, there was no impact on the locoregional recurrence rate. They hypothesised that statins reduce transformation of epithelial to mesenchymal phenotyping of cancer cells and thereby reduce distant spread. 25 This observation has been supported by an experimental study by Beckwitt et al. 26
A similar observation has been reported in muscle invasive urinary bladder cancer by Wissing et al. 27 This suggests that the role of statin as a long-term therapy to reduce distant metastasis and bladder cancer mortality needs further research. An increased HMGCR expression seen in our study in non-responders indicates that statins induced inhibition of HMGCR and inhibited the mevalonate pathway within the tumour or by lowering circulating levels of cholesterol negatively increased expression of HMGCR proteins. An observation similar to this is reported in breast cancer by Clendening et al. 5
Our study has the limitation of having small numbers and a retrospective design. Future studies using a larger cohort may require multi-centre recruitment with careful documentation of follow-up. This proof-of-concept study provides a further insight into an area of further research including re-purposing of drugs using new targets.
Conclusions
A difference in the expression of HMGCR between BCG responders and non-responders was noted and, although not statistically significant in the small numbers presented here, findings form a basis for further investigation and research.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
