Abstract
The brain–gut–bone axis refers to the interactions among the brain, gut, and bones, which were developed and refined by researchers across multiple fields. This axis was complex and dynamic, and influenced overall health through any possible intervention. This narrative review primarily focused on the regulation of bone metabolism through this axis mediated by serotonin (5-hydroxytryptamine (5-HT)) and mediators. Bone metabolism, influenced by various factors, was characterized by dynamic processes occurring within bone tissue, including bone formation and resorption. To enhance bone metabolism, it was essential to consider the brain–gut–bone axis, offering new insights in clinical research. This review examined the influence of the brain–gut–bone axis on bone metabolism and its underlying mechanisms.
Introduction
Bone metabolism was a dynamic and multifaceted process, traditionally understood through the lens of local cellular activity and classical hormonal pathways. While this fundamental knowledge underpinned the clinical management of bone disorders, significant challenges remained in predicting and optimizing healing outcomes, particularly within oral and maxillofacial surgery. This limitation suggested that a strictly local or single-system perspective was insufficient, necessitating the exploration of broader physiological networks that influenced bone homeostasis.
Emerging evidence substantiated the concept of a “brain–gut–bone axis,” a bidirectional communication system linking neural, enteric, and bone functions. This axis operated via distinct pathways: the brain–bone axis, the gut–bone axis, and was mediated by systemic factors such as 5-hydroxytryptamine (5-HT) as well. Notably, 5-HT exemplifies the axis's complex regulatory role: gut-derived peripheral 5-HT inhibited bone formation, whereas central 5-HT might exert protective effects through the modulation of mood and autonomic tone. Consequently, dysregulation within this integrated system might contribute to compromised bone metabolism.
Therefore, the primary aim of this narrative review was to synthesize current evidence on the brain–gut–bone axis, with a specific focus on the function of 5-HT. By conducting a structured literature search of PubMed, this review aimed to provide oral clinicians and researchers with a novel, systemic framework for understanding bone health. We proposed that this perspective serves not merely as a theoretical model, but as a critical foundation for developing future diagnostic and therapeutic strategies for bone-related conditions.
Brain–bone axis
The “brain–bone axis” emphasized the bidirectional communication between the brain and the bones. Although research in this field was still in progress, numerous studies have already revealed that brain physiological activities, brain structure, neurological diseases, and psychological states significantly influence bone metabolism. Meanwhile, bone-derived factors like osteocalcin and FGF23 were not only vital for bone health but also affected brain functions like cognition, memory, and emotions by crossing the blood–brain barrier. 1 The axis regulated neuro-humoral balance through two pathways: the “hypothalamus–brainstem–autonomic nervous” (HBA) axis and the “hypothalamus–brainstem–endocrine” (HBE) axis. The former one used the sympathetic nervous system, neuropeptides, and neurotransmitters to facilitate the crosstalk between the central nervous system (CNS) and bone metabolism, linking the brain and bone metabolism.
The mechanism of the HBA on bone metabolism
The HBA, which included signals from the hypothalamus through the brainstem, released neurotransmitters and neuropeptides via the autonomic nervous system to exert their biological effects. This process triggered various intracellular signaling pathways that regulated activities of osteoblasts and osteoclasts, to balance bone formation and resorption.
Effects of adrenaline on bone metabolism
Adrenaline is the main neurotransmitter in the sympathetic-adrenal medullary system. β2-adrenergic receptor (β2AR) was the main receptor of it, which was highly expressed in osteoblasts and osteoclasts. After sympathetic stimulation, adrenaline bonded to β2AR to activate the expression of miR-21, targeting Pdcd4, stimulating osteoclast differentiation and resorption. 2 β2AR inhibited the proliferation and differentiation of osteoblasts. 3 Additionally, it enhanced the expression of RANKL (receptor activator of nuclear factor κ B ligand) targeting the RANK in the pre-osteoclast. RANKL-RANK promoted the differentiation and maturation of osteoclasts by activating NF-κB, MAPK, PI3K/AKT, and NFATc1. 4
Effects and mechanisms of neuropeptides on bone metabolism
Neuropeptides are large polypeptides produced by neurons, triggering intracellular signaling and affecting the behavior of cells through binding to specific receptors. 5 This section focused on two key neuropeptides: calcitonin gene-related peptide (CGRP) and neuropeptide Y (NPY).
The CGRP receptor was a complex of calcitonin receptor-like receptor (CLR) and receptor activity-modifying proteins (RAMPs), which was expressed in sensory nerve fibers of the bone and periosteum. 6 It boosted bone metabolism by directly promoting the activity of osteoblasts and indirectly inhibiting the activity of osteoclasts. It played a crucial role in bone matrix formation and mineralization. CGRP facilitated osteogenesis directly by activating the cAMP/PKA or Wnt/β-catenin. Wnt/β-catenin signaling has been shown to be particularly effective in enhancing fracture healing, especially in aged models, by promoting angiogenesis and osteogenic differentiation. Furthermore, it stimulated osteogenic differentiation by upregulating transcription factor 4 (ATF4), bone-specific alkaline phosphatase (ALP), and osteocalcin. CGRP enhanced the mineralization and maturation by enhancing the expression of osteoprotegerin (OPG) and reducing the expression of RANKL in some in vitro experiments. 7
NPY was widely distributed throughout the central and peripheral nervous systems. Peripheral NPY signaling exerted an anabolic effect on bone metabolism. In vitro, NPY bonded to NPY1R and NPY2R on osteoblasts and osteoclasts. Activation of NPY1R through Erk increased ALP and osteocalcin, stimulating osteoblast proliferation and differentiation, and downregulated the expression of RANKL and TRAP, reducing osteoclast genesis. 8 In vitro, NPY binding to the NPY1R and NPY2R enhanced the proliferation and differentiation of osteoblasts by activating the NPY1R and Erk pathway, which led to increased expression of ALP and OCN. It also reduced the differentiation of osteoclasts via downregulating RANKL and TRAP. In addition, central NPY signaling had a catabolic effect on bone metabolism, opposite to its peripheral effect. 9 NPY enhanced the production of osteoclasts by downregulating the expression of osteoblast and upregulating the osteoclast in the state of stress. The central and peripheral NPY achieved the balance in a complementary manner.
The mechanism of the HBE axis on bone metabolism
The HPG axis
HPG referred to the secretion of gonadotropin-releasing hormone (GnRH) by the hypothalamus, which stimulated the pituitary to release luteinizing hormone (LH) and follicle-stimulating hormone (FSH). These hormones acted on the gonads to promote the production of sex hormones, which inhibit the secretion of GnRH, FSH, and LH. 10 Beyond its function in the reproductive system, FSH also played a crucial role in bone metabolism. FSHRs were primarily expressed on osteoclasts; thus, FSH could upregulate several bone resorption pathways. Different concentrations of FSH had different effects on RANK. Especially, FSH significantly increased the expression of RANK during the perimenopause period to exacerbate bone resorption. The levels of FSH were inversely related to bone mineral density (BMD), particularly in women with high FSH levels. 11 In conclusion, HPG led to bone loss through multiple pathways. Playing a relative role independent of estrogen, FSH, playing a relative role independent of estrogen, made perimenopausal women more susceptible to osteoporosis (OP).
The HPA axis
The HPA was an endocrine system involving the hypothalamus, pituitary, and adrenal glands. The hypothalamus released corticotropin-releasing hormone (CRH), which stimulated the pituitary to secrete adrenocorticotropic hormone (ACTH). ACTH subsequently stimulated the adrenal glands to produce glucocorticoids (GCs), which inhibited the release of CRH and ACTH through a negative feedback loop. 10
As the final product of HPA, GCs made a big difference in bone metabolism. Long-term high levels of GCs could enhance insulin resistance and affect the function of pancreatic β cells, ultimately leading to diabetes. 12 Clinical observations revealed that diabetic patients were frequently accompanied by OP. The mechanisms included systemic and local pathways: firstly, GCs could inhibit the absorption of Ca2+, affect the metabolism of vitamin D, lead to hypercalciuria and changes in parathyroid hormone levels, all through indirect effects from systemic metabolic disorders. Secondly, it directly acted on osteoblasts and osteoclasts, accelerating osteoblast apoptosis, and enhancing osteoclast activity through upregulating RANKL, eventually leading to OP. 13
ACTH regulated the release of adrenal corticosteroids, such as GCs, by activating the melanocortin 2 receptor (MC2R) located in the adrenal cortex. MC2R was found in both the adrenal cortex and bone cells. 14 In vitro studies demonstrated that ACTH may directly affect the function and metabolism of bone cells. Further in vitro studies have shown that ACTH promoted the differentiation of osteoblasts by targeting MC2R. This process was accompanied by the production of vascular endothelial growth factor (VEGF) to stimulate bone formation. Furthermore, existing evidence from in vitro models showed that ACTH and 1, 25-dihydroxyvitamin D3 could synergistically facilitate the proliferation and activity of osteoblasts. 15 These findings revealed the potential direct role of ACTH in bone metabolism.
Gut–bone axis
The gut–bone axis refers to the communication of gut and bone through a network of biochemical signals. It highlighted the intricate connections between gut bacteria, nutrients, hormones, the immune system, and bone. Gut microbiotas managed immune responses and maintained the intestinal barrier integrity through their production of metabolites and interactions with the host. These metabolites influenced bone directly through nutrients and indirectly via immune regulation, hormone balance, and vitamin production. 15
Gut microbiota enhanced bone metabolism through the generation of SCFAs
The gut microbiota, which comprises the vast community of microorganisms that colonized in the gut and feces, is the largest and most complex ecosystem within the human body.
Accumulating animal studies indicated that gut microbiota had positive effects on bone metabolism. Schwarzer et al. 16 found animal research that, compared to age-matched healthy mice, germ-free (GF) mice exhibited slower weight gain and significantly reduced bone mass. However, their growth rate was restored to normal levels when these supplemented with Lactobacillus plantarum. This finding highlighted the important role of gut microbiota in promoting bone formation and growth. Subsequent animal studies further supported this view. Compared to GF mice, those lacking sex hormones exhibited increased intestinal permeability, primarily due to elevated levels of TNF-α, RANKL, and IL-17. Supplementing these mice with Lactobacillus rhamnosus GG (LGG) or probiotics led to a significant increase in the density of trabecular bone and effectively prevented bone mass loss. 17
In addition, gut microbiota upregulated the expression of IGF-1 to promote bone mass and growth. Honig and Gayeski found that GF mice showed a significant increase in bone formation, as well as elevated IGF-1 levels, after long-term colonization with SPF microbiota. Antibiotic treatment has an adverse effect, whereas supplementation with SCFAs can restore bone mass and IGF-1 levels. These suggested that microbiota improved bone metabolism through the secretion of SCFAs, which up-regulated IGF-1. 18
SCFAs regulate bone metabolism, targeting the immune system
SCFAs not only directly affected bone metabolism by regulating calcium absorption, but also indirectly influenced bone by modulating the immune system. Binding to free fatty acid receptors 2 and 3 (FFAR2/3), a G-protein-coupled receptor, SCFAs regulated multiple signaling pathways that affect bone metabolism, as evidenced by human, animal, and in vitro models. 19
SCFAs stimulated the differentiation and proliferation of Treg cells by targeting FFAR2. Butyrate and propionate acted on dendritic cells (DCs) via targeting FFAR2, promoting IL-10 production and inhibiting the generation of IFN-γ+ effector T cells. It secreted IFN-γ. IFN-γ activated osteoclasts and inhibited osteoblasts. Meanwhile, butyrate and propionate acted on DCs to enhance Tregs differentiation, suppressing inflammatory responses. The activation of Tregs promoted the binding of the nuclear factor of activated T cells (NFAT) and Sma and Mad-related proteins (SMAD) to the Wnt10b promoter, inducing the production of Wnt10b in CD8+ T cells, a mechanism elucidated through in vitro and animal studies. Wnt10b was a potent activator of the Wnt.20–22 Among SCFAs, in vitro experiments demonstrated that butyrate enhanced the binding of NFAT1, SMAD3, and the Wnt10b promoter, facilitating the formation of the NFAT/SMAD complex. This further activated the Wnt, thereby promoting osteoblast differentiation and bone formation.
Gut microbiota and their metabolic products regulated bone metabolism through various signaling pathways to influence bone resorption. Studies have shown that Bifidobacterium, by secreting SCFAs, acts on the TNF-α/NF-κB pathways, playing a pivotal role in regulating osteoclastogenesis. Simultaneously, the classical RANKL/RANK/OPG also played an auxiliary role in promoting osteoclast formation. The RANK/RANKL promoted bone resorption, while OPG acted as a “decoy receptor,” by competing with RANKL, thereby suppressing bone resorption. 23 SCFAs, by modulating the immune system, promoted Treg differentiation, suppressed IFN-γ secretion, enhanced the Wnt signaling pathway, and influenced multiple pathways such as TNF-α/NF-κB and RANKL/RANK/OPG, collectively exerting a positive effect on bone metabolism, particularly in enhancing osteoblastogenesis and increasing BMD.
Dysregulation of the gut mucosal barrier affected bone metabolism through inflammation and immune
The gut mucosal barrier refers to the protective barrier on the inner surface of the gut. The outermost layer included surface mucus, gut microbiota, and defense proteins; the middle layer consisted of epithelial cells; and the innermost layer 日常 in immune cells. 24 Functionally, some researchers classified the gut barrier into four main components: the mechanical barrier of epithelial cells and tight junctions; the biological barrier formed by the resident microbiota and the host microenvironment; the chemical barrier of digestive secretions (e.g. gastric acid, bile, digestive enzymes, and mucins); and finally, the immune barrier was composed of gut-associated lymphoid tissue and its immune response products. 25
The relationship between the host and the gut microbiota was a complex symbiosis. Guan et al. researched the impact of gut microbiota dysbiosis on bone loss, using an ovariectomized mouse model. Disruption of the barrier function of the gut mucosa led to an imbalance in bacterial populations and an increase in serum lipopolysaccharide (LPS) levels. Antibiotic treatment improved bone mass, bone microstructure, and bone strength in these mice, while transplantation of a microbiota adapted to ovariectomyresulted in bone loss. 26 These findings suggested that gut microbiota dysbiosis in ovariectomized mice led to bone loss.
Permeability damage induced by gut microbiota dysbiosis was often associated with abnormal immune responses, which affect Osseo-immunization and bone resorption. A comparative study showed that GF mice exhibited higher bone mass compared to CONV-R (conventionally raised) mice. It was directly associated with a significant reduction in osteoblasts, CD4+ T cells, and osteoclast precursors, as well as a decrease in the expression of TNFα and IL-6. These suggested that the normal gut microbiota may promote osteoclast activity by regulating the immune system. However, this ostensibly “negative effect” was not entirely detrimental to bone health, 27 as it reflected a physiologically adaptive interaction between the microbiome and bone remodeling processes.
SCFAs promoted bone metabolism through multiple mechanisms by regulating the immune system, especially in enhancing osteoblast formation and bone density. It influenced bone metabolism by promoting Treg differentiation, inhibiting IFN-γ secretion, enhancing the Wnt signaling pathways, and modulating TNF-α/NF-κB though the RANKL/RANK/OPG. A stable gut microbiota-maintained bone mass, while dysbiosis disrupted the gut barrier, activated immune abnormalities, and increased bone resorption. Thus, regulating the gut–bone axis and restoring gut microbiota balance could offer new treatment strategies for metabolic bone diseases like OP. In oral health, maintaining a healthy gut microbiota not only protected the alveolar bone loss but also supported its regeneration, which provided new approaches for the prevention and treatment of oral diseases.
The balanced metabolism effect of 5-HT on the gut–brain–bone axis
The gut–brain–bone axis was not a one-to-one/signal relationship but a network framework. 5-HT, the inhibitory neurotransmitter that was first discovered in serum, played a key role as a bridging substance in the axis. It regulates the CNS, gut microbiota, and bone metabolism. Both central (brain-derived) 5-HT and peripheral (gut-derived) 5-HT had dual functions in regulating bone metabolism. Specifically, central 5-HT primarily acted through hypothalamic 5-HT2c receptors, leading to the indirect suppression of bone resorption via the sympathetic nervous system. In contrast, peripheral 5-HT directly acted on local bone receptors, such as 5-HT1b, to inhibit osteoblast activity. 28
Peripherally, gut-derived 5-HT inhibited osteoblast proliferation by binding to 5-HT1b and 5-HT2b on osteoblasts. 29 Increased expression of 5-HT disrupted the balance between FOXO1 (Forkhead box O1), CREB9 (cAMP response element-binding protein), and ATF4 (activating transcription factor 4). FOXO1 tended to bind to ATF4, which in turn suppressed the expression of bone formation-related genes, impacting bone metabolism. 30
In addition, the activation of Lrp5 (LDL receptor-related protein 5) promoted bone formation through two pathways. Firstly, Lrp5 on osteoblasts could directly activate the Wnt signaling to enhance osteogenic differentiation. Secondly, the activation of Lrp5/Wnt signaling in bones inhibited the transcription factor Egr1, which in turn suppressed the activity of Tph1 (tryptophan hydroxylase 1 and the rate-limiting enzyme of enterogenic 5-hydroxylase) in the intestine, reducing the synthesis and release of enterogenic 5-HT. 31 It relieved the inhibition of gut-derived 5-HT on bone formation by binding to Htr1b receptors, thereby fostering osteogenesis.
In the central nervous system, brain-derived 5-HT, which was the ligand of 5-HT2c in the hypothalamus, 32 ultimately reduced bone resorption by inhibiting sympathetic nerve activity and suppressing the expression of RANKL.
Selective 5-HT reuptake inhibitors (SSRIs) were serving as the first-line pharmacological treatment in the field of depression, which was a major global health burden. However, their potential association with reduced BMD and increased fracture risk has been a significant concern. Numerous observational studies and meta-analyses reported a strong link between SSRIs and a higher incidence of fracture, but a rigorous target trial emulation study found no clear dose-response relationship. 33 Theoretically, SSRIs could reduce sympathetic tone in bone tissue by elevating central 5-HT, potentially benefiting skeletal health. Yet, the effects on bone seemed detrimental. There are two possible mechanisms: first, SSRIs may directly affect bone cells, such as osteoblasts and osteoclasts. These local adverse effects might outweigh any theoretical central nervous system-mediated benefits. Secondly, depression itself was a systemic condition that negatively impacts bone metabolism.
Emerging research suggested that psychoactive substances such as ketamine 34 and MDMA 35 may function as systemic modulators, which could exert beneficial effects on bone metabolism. In rodent models of depression and ovariectomy, ketamine and MDMA not only relieved behavioral abnormalities but also reversed declines in BMD. The mechanism involved a cascade: neural activation, followed by gut microbiota and metabolite remodeling, resulting in the regulation of systemic bone homeostasis.
Collectively, this evidence highlighted a novel role for psychoactive drugs in regulating systemic neuro-gut-bone crosstalk, offering an integrative perspective for understanding comorbid mechanisms and developing new intervention strategies36,37 (Table 1).
The debate, relevant evidence, and knowledge gaps regarding the role of 5-HT in bone metabolism.

Treatment
Abnormal bone metabolism causes some common diseases like OP, osteomalacia, and rickets. 38 Recovering the normal physiological activity of the hypothalamus and improving gut microbiota dysbiosis could treat these by indirectly enhancing bone metabolism. The specific methods were as follows:
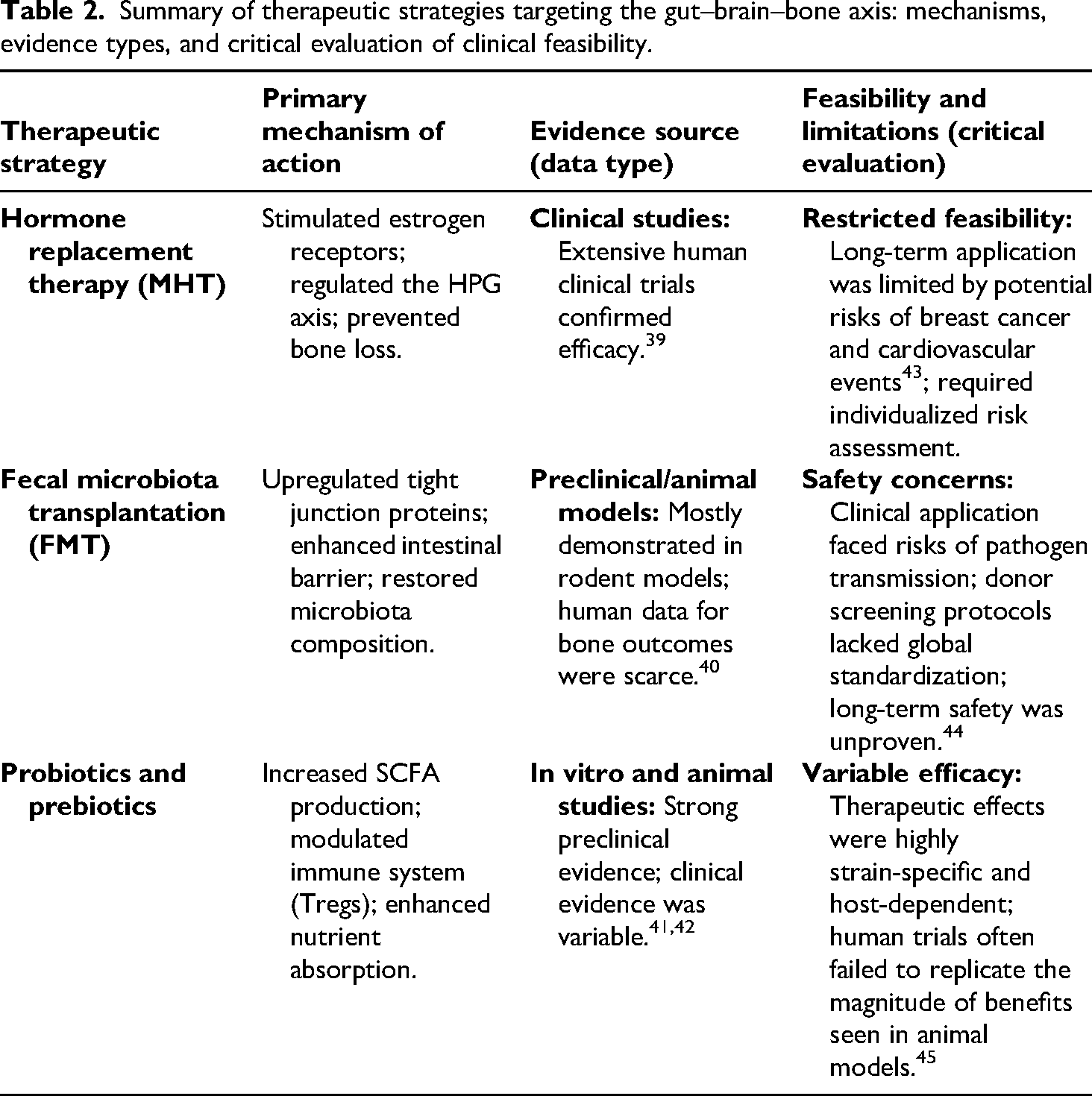
Hormone replacement therapy (MHT)
Clinical studies confirmed that MHT stimulated estrogen receptors, regulated the HPG axis, and effectively prevented bone loss. Gosset et al. 39 emphasized that although MHT effectively mitigates postmenopausal bone loss, its long-term use was limited by extra-skeletal risks, including breast malignancy and cardiovascular disease. Thus, MHT was not a universal prophylactic; current guidelines mandate strict, individualized risk assessment to ensure skeletal benefits outweigh systemic risks.
Fecal microbiota transplantation (FMT)
Preclinical research utilizing animal models demonstrated that FMT could improve gut microbiota composition, upregulate tight junction proteins (occludin, claudin, and ZO-1), and enhance intestinal barrier function to regulate bone metabolism. 40 Despite these promising results in mice, the clinical feasibility of FMT for treating OP remained controversial. It faced challenges such as the risk of pathogen transmission, lack of standardized donor screening protocols, and limited long-term clinical safety data in non-gastrointestinal diseases.
Probiotics and prebiotics
In vitro and animal studies showed that probiotics (e.g. Lactobacillus and Bifidobacterium) 41 and prebiotics 42 could increase SCFA levels, enhancing bone density. However, clinical evidence appeared more variable compared to animal data. The therapeutic efficacy was highly strain-specific and depended on individual host gut environments. Consequently, human trials indicated that not all probiotic interventions successfully translated into significant improvements in BMD.
The above treatment methods had confirmed that regulating bone metabolism through the gut–brain axis was feasible, providing new insights for future strategies (Table 2).
Summary of therapeutic strategies targeting the gut–brain–bone axis: mechanisms, evidence types, and critical evaluation of clinical feasibility.
Discussion
Unlike previous studies that looked at gut–bone or brain–bone interactions separately, or focused on a single factor, this review combined recent evidence into a unified “Brain–Gut–Bone” framework. We highlighted the key role of 5-HT in this axis, confirming the shift from a local to a systemic view of bone metabolism. This offered a vital systemic perspective for treating general bone diseases as well as oral conditions like peri-implantitis and periodontitis. However, there were limitations. As a narrative review, this study lacked the statistical rigor of a systematic meta-analysis. Also, much of the evidence on mechanisms, such as the Lrp5 feedback loop, came from mouse models. Therefore, caution was needed when applying these findings to human clinical practice.
Conclusion
This review systematically elaborated the bidirectional regulation of bone metabolism by the integrated “brain–gut–bone axis” framework. As illustrated, this axis primarily comprised two major branches: the “brain–bone axis” and the “gut–bone axis”. The neural pathway (HBA) involves the release of neuropeptides and neurotransmitters, while the endocrine pathway (HBE) primarily involves the HPA and HPG axes, regulating hormone release. Peripheral 5-HT inhibits bone formation, whereas central 5-HT may exert indirect protective effects by improving mood and reducing sympathetic tone.
Future directions
The brain–gut–bone axis represented not merely a novel theoretical concept but also provided a comprehensive systemic perspective with the potential to break through the current bottlenecks in the prevention and treatment of oral and bone-related diseases (Figure 1).
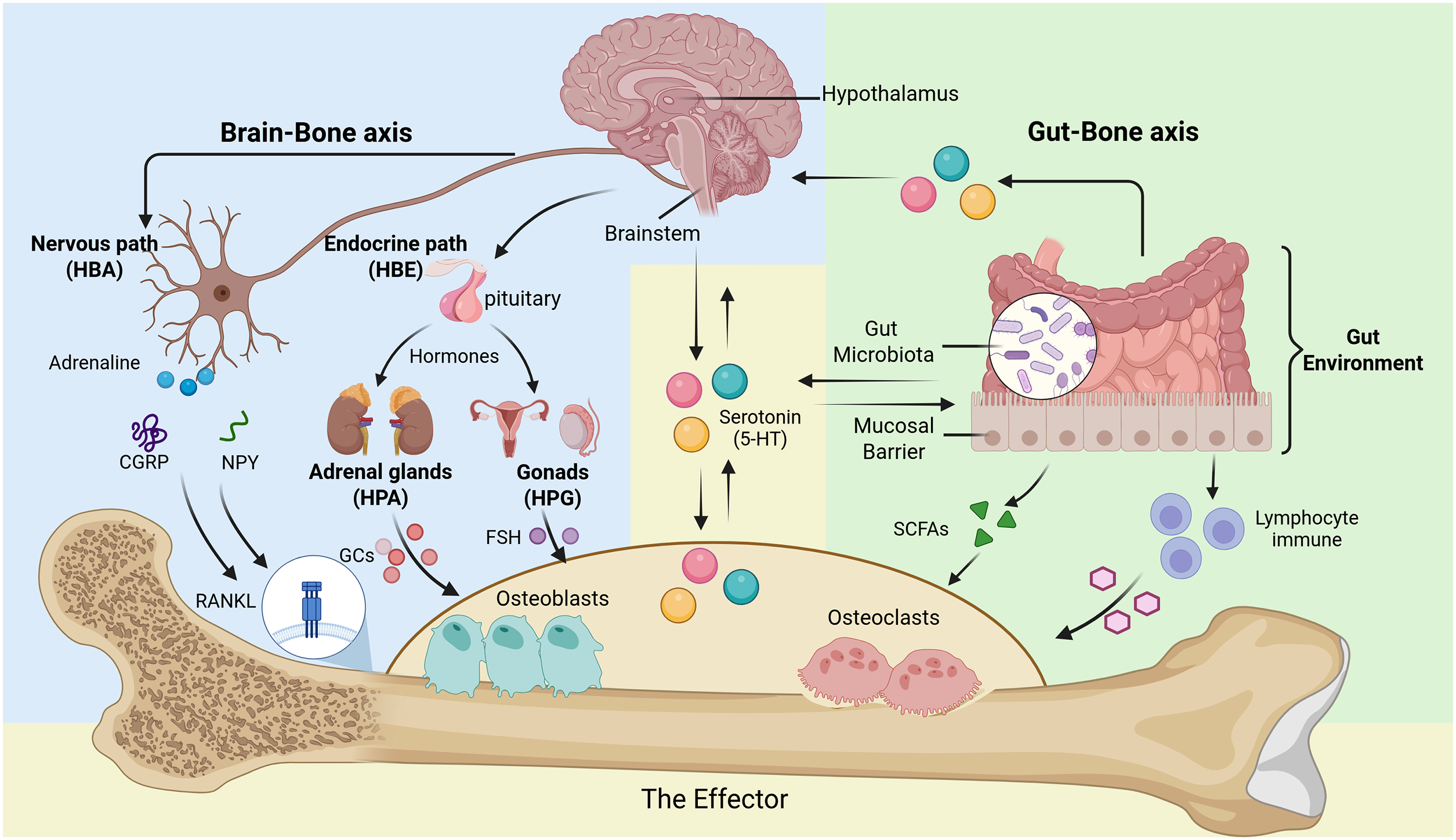
Brain–gut–bone axis and key mediators.
Footnotes
Acknowledgements
The author thanks DeepSeek AI for language improvement.
Author contributions
All authors contributed to the study conception and design. Ruimeng Shi performed the material preparation, data collection, and analysis, and wrote the first draft of the manuscript. Zunqi Yang provided critical methodological input. Zhuangzhi Zheng supervised the project. All authors commented on previous versions of the manuscript and approved the final version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
This review article is based on an analysis of publicly available research articles and data. All data and materials referenced in this review are obtained from publicly accessible sources, primarily from the PubMed database. Therefore, no new datasets were generated or analyzed during the present study. The references and citations included in this review can be accessed through the respective journals or databases.
