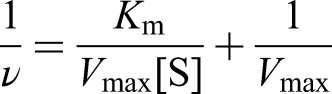Abstract
Introduction
Hydrogen peroxide (H2O2) and glucose serve as crucial biomarkers in human physiology, and their detection is essential for the diagnosis and treatment of numerous diseases. 1 Peroxidases have significant application value in H2O2 detection and glucose biosensing due to their high catalytic efficiency in decomposing H2O2.2–4 Among natural enzymes, horseradish peroxidase stands as the most representative peroxidase, widely utilized for its abundant availability, low molecular weight, and ease of purification.5,6 However, natural enzymes suffer from inherent limitations in practical applications, including poor stability, high cost, and difficult recovery, which restrict their large-scale utilization. 7 To address these challenges, nanotechnology has given rise to a novel class of artificial enzymes, namely nanozymes. 8 These nanomaterial-based enzyme mimics not only retain the catalytic activity and substrate specificity of natural enzymes but also demonstrate remarkable advantages, such as enhanced stability, low cost, and easy recyclability, thereby exhibiting tremendous potential in biomedical diagnostics and therapeutics.9–11 Since the first report of Fe3O4 nanozymes in 2007, researchers have developed various nanozymes with peroxidase-, superoxide dismutase-, catalase-, and oxidase-like activities, which have been successfully applied in biosensing, environmental remediation, antibacterial, and antitumor therapies.12–19
Among diverse nanozyme materials, two-dimensional transition metal dichalcogenides (2D TMDCs) have emerged as a current research focus due to their exceptional catalytic stability, facile synthesis methods, and low production costs.20,21 Tungsten disulfide (WS2) nanosheets, a typical 2D TMDC, owing to their unique photophysical and chemical properties, have been extensively used in biomedical applications.22,23 However, unmodified WS₂ often demonstrates unsatisfactory performance, because of its rather low intrinsic catalytic activity and limited electron transfer efficiency. Fortunately, it has been recently reported that metal ion (including Ag, Ni, Pt, Mn, Pd, etc.) doping in the crystal lattice is a powerful modification approach to precisely regulate the electronic structure and catalytic performance of nanozymes.24–28
Iron is a typical active center element of many natural peroxidases with unique Fenton activity. 29 Additionally, Fe ions can minimize lattice distortion during doping and preserve the structure of the host semiconductor, due to their appropriate ionic radius. Motivated by the catalytic and biomedical advantages of iron, we have envisaged that the Fe-doped WS2 nanosheets may exhibit efficient peroxidase-like (POD-like) nanozyme activity for biosensing. Therefore, we prepared iron-doped WS2 (Fe-WS2) nanozymes based on a hydrothermal method. The as-synthesized Fe-WS2 can catalyze the decomposition of H2O2 into hydroxyl radicals (·OH), exhibiting excellent POD-like activity and high sensitivity. Furthermore, in vitro colorimetric biosensing of glucose and H2O2 by Fe-WS2 as a catalytic material exhibited excellent specificity and ultra-sensitivity. Thus, a novel Fe-WS2 nano-biosensor based on Fe-WS2 nano-sensor was not only developed by such work, but also promoted further applications of inorganic nanozymes in biomedical diagnosis and therapy.
Experimental section
This research complies with all relevant safety protocols approved by Jinan University.
Materials
Commercial WS2 (99.8%) was obtained from Alfa Aesar and used without further purification. FeCl3·6H2O, H2SO4 (95.0% to 98.0%, analytical reagent), potassium bromide, and ascorbic acid were obtained from Chron Chemical Co. Ltd (Chengdu, China). 3,3′-diaminobenzidine (DAB), 3,3′,5,5′-tetramethylbenzidine (TMB), 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS), and o-phenylenediamine (OPD) were all from Aladdin Company (Shanghai, China). H2O2 (30%) was procured from Jinshan Chemical Reagent Co. Ltd (Chengdu, China). Glucose oxidase (GOx) was acquired from Shanghai Yuanye Bio-Technology Co. Ltd (China). Deionized (DI) water was used in the whole process.
Preparation of WS2 nanosheets
The synthesis of WS₂ nanosheets was performed via ultrasound-assisted liquid exfoliation by using H2SO4 as an intercalation agent according to procedures previously reported with necessary optimizations. 30 First of all, the commercially available WS2 bulks were ground with a ball mill for 6 h. Afterwards, the ground WS2 (100 mg) powder was added to H2SO4 (100 mL) solvent and treated with an oil bath at 90 °C for 24 h. Subsequently, the H2SO4 remaining in the product was removed with DI water, and the product was sonicated for 8 h and then centrifuged at 6000 r/min for 10 min to acquire WS2 nanosheets.
Preparation of Fe-WS2 nanozymes
Fe-WS2 was synthesized by the solvothermal method according to the previously reported method. 31 In this protocol, ascorbic acid was used as an efficient reducing agent and stabilizer.32,33 Briefly, WS2 (15 mg), FeCl3·6H2O (15 mg), and ascorbic acid (15 mg) were solubilized in 30 mL of water under severe stirring with magnetic force, and finally, a mixed solution was formed. NaHCO3 (10 mg) was inserted after 1 h and continued to stir magnetically for 20 min. Then the blended solution was placed into a 100 mL polystyrene-lined and highly pressure pot of stainless steel and heated in an oven at 150 °C for 6 h. It was washed three times with water for further use.
Basic characterization
The morphology and energy-dispersive X-ray spectroscopy (EDS) elemental mapping of samples were visualized by transmission electron microscopy (TEM) (JEM-2100F, JEOL). The diameter of the samples was measured with ImageJ software. The hydrodynamic diameter, zeta potential, and stability analysis on samples were performed at room temperature (25 °C) by a dynamic light scattering (DLS) analyzer (Zetasizer ZS90, Malvern). Before measurement, the samples were dispersible in ultrapure water (100 μg/mL) by an ultrasonic dispersing technology using an ultrasonic cell crusher (300 W, 40 min). The crystalline structure was obtained using an X-ray powder diffraction (XRD) system with irradiation of Cu-Kα. Fourier transform-infrared (FT-IR) spectrometer (IR 200, Thermo Fisher Scientific, USA), ultraviolet (UV)-visible spectrophotometer (UV-6100, Mapada, China), and Raman spectrophotometer (Invia Reflex, Renishaw, UK) were used to determine the structure of the samples.
POD-like enzyme activity analysis
It was chosen to use TMB to evaluate the ability of Fe-WS2 nanozyme to transform H2O2 into ·OH under diverse conditions in citric acid and Na2HPO4·12H2O buffer solutions. The change in absorbance in the Fe-WS2 nanozyme/H2O2/TMB system was examined by varying the pH, reaction temperature, TMB, H2O2, and material concentration. Measurements of UV absorbance of oxidized TMB (oxTMB) were carried out by adding H2O2 (30 mM), TMB (0.5 mM), and Fe-WS2 nanozyme (50 µg/mL) to the reaction buffer and leaving them for 15 min at different reaction temperatures.
Kinetic assay
The steady-state dynamics behavior of Fe-WS2 nanozyme was evaluated by changing the concentrations of TMB and fixing the H2O2 concentration, and conversely. The final concentration of Fe-WS2 nanozyme was 50 µg·mL−1, and the pH of the reaction system was adjusted to 4.0 with 0.1 M HAc-NaAc buffer solution. The reaction was monitored with the spectrophotometer at 652 nm. Which kinetic variables were obtained from the double inverse equation based on the Michaelis–Menten equation:
Glucose and H2O2 detection with Fe-WS2 nanozyme
An appropriate amount of glucose was mixed with concentrations of GOx (1 mg/mL), TMB (0.5 mM), and Fe-WS2 nanozyme (50 µg/mL) in the order from 1 to 10 mM. The reaction was carried out at 37 °C for 1 h under dark conditions. The absorbance of the mixed solutions at 652 nm was recorded by a spectrophotometer.
The detection was next performed using different concentrations of H2O2. And appropriate amounts of H2O2 were mixed with TMB (0.5 mM) and Fe-WS2 nanozyme (10 µg/mL). The reaction was carried out at 37 °C for 5 min in the dark. As the reaction is relatively rapid, we used the nanomaterial concentration of 10 µg/mL for the H2O2 assay.
Meanwhile, in order to study Fe-WS2 nanozyme as selective to H2O2, this experiment was set up to detect the material's resistance to H2O2 interference. Fe-WS2 solution (50 μg/mL), TMB solution (0.5 mM) buffer (disodium hydrogen phosphate-citrate, pH = 4) was added to the solutions (1 mM) containing NaCl, MgCl2, MnSO4, CaCl2, ZnCl2, KCl, L-cysteine, amino acid, bovine serum albumin, glucose, and glutathione-S-transferase, respectively, and the reaction was carried out away from light to determine the absorption at 652 nm.
Results
The as-synthesized Fe-WS2 nanozymes were prepared by a hydrothermal method (Figure 1). In this protocol, ascorbic acid was used as an efficient reducing and stabilizing agent, and FeCl3·6H2O was selected as the iron supply.30–33

Schematic illustration of the Fe-WS2-based biosensing system for H2O2 and glucose detection.
Concretely speaking, benefitting from the POD-like catalytic performance of Fe-WS2, versatile enzyme cascade-based colorimetric bioassays for the detection of H2O2 and glucose have been devised using Fe-WS2-triggered reaction of TMB as an amplifier. In this study, GOx catalyzes the oxidation of glucose in the presence of O2, yielding gluconic acid and H2O2 as byproducts. Subsequently, the as-synthesized Fe-WS2 nanozyme decomposes the generated H2O2 via POD-like activity, producing •OH that oxidizes colorless TMB into a blue-colored product (oxTMB). Notably, the POD-like activity of Fe-WS2 is H2O2-concentration-dependent, resulting in a progressive intensification of the colorimetric signal with elevated H2O2 and glucose concentrations.
The morphological and structural characterizations of both WS2 nanosheets and Fe-WS2 were systematically investigated. TEM images revealed that the lamellar morphology of both WS2 nanosheets and Fe-WS2, with differences in their physical dimensions (Figure 2(a) and (b)). Quantitative analysis from TEM images demonstrated that Fe doping induced a size increase, from an average lateral dimension of 122.47 ± 3.95 nm for WS₂ to 211.72 ± 45.35 nm for Fe-WS₂ (Supplemental Figure S1). This dimensional enhancement was further corroborated by DLS measurements, which showed a consistent shift in hydrodynamic diameter distribution (Figure 2(e) and (f)), suggesting successful Fe integration and potential surface modification. Furthermore, EDS elemental mapping analysis confirmed the homogeneous distribution of Fe throughout the WS2 nanosheets, with quantitative analysis revealing an Fe loading of 1.35% (Figures 2(c) and (d)). The zeta potential measurements revealed significant alterations upon Fe incorporation (Figure 2(g)). The shift from −37.45 mV for WS₂ nanosheets to −14.2 mV for Fe-WS₂ indicates modified surface properties. Besides, comprehensive structural characterization was performed using FT-IR, XRD, and Raman spectroscopy (Figure 2(h) and (i) and Supplemental Figure S2). FT-IR spectra confirmed the preservation of characteristic WS₂ vibrational modes while revealing new Fe–S bonding signatures (674 cm−1) (Supplemental Figure S2). XRD patterns maintained all major WS2 diffraction peaks with inconspicuous peak shifting, indicating that Fe doping occurred without disrupting the host crystal structure (Figure 2(h)). Raman spectroscopy showed the distinctive E2g and A1g modes of WS₂ at ∼350 cm−1 and ∼416 cm−1, respectively, with the weakened peaks of Fe-WS2 attributable to Fe doping (Figure 2(i)). As evidenced by spectroscopic characterizations above, this controlled doping level was achieved while maintaining the structural integrity of the WS2 nanosheets.

Structural characterization analysis of WS2 nanosheets and Fe-WS2. TEM images of WS2 nanosheets (a) and Fe-WS2 nanozymes (b). (c) The elemental mapping of Fe-WS2 nanozyme. (d) EDS analysis of synthesized Fe-WS2. Hydrodynamic diameter distributions of WS2 nanosheets (e) and Fe-WS2 (f) (n = 3). (g) The zeta potentials of WS2 nanosheets and Fe-WS2. XRD patterns (h) and Raman spectra (i) of WS2 nanosheets and Fe-WS2.
After the successful fabrication of the Fe-doped tungsten disulfide nanozyme, the POD-like activity of Fe-WS2 was systematically evaluated using TMB as the chromogenic substrate. Upon oxidation, TMB undergoes a characteristic colorimetric transition, producing a blue-colored oxidized product (oxTMB) with a distinct absorption maximum at 652 nm (Figure 3(a)). Negligible oxTMB absorption enhancement in the H2O2- or Fe-WS2-treated group could be observed, while the Fe-WS2 + H2O2 + TMB reaction system exhibited the strongest absorbance at 652 nm after just 5 min of incubation at room temperature. In addition, control experiments with Fe ions alone and WS2 without doping have now been included in Supplemental Figure S3. A negligible absorption change with the WS₂ alone-treated group could be observed, while an enhanced absorption can be found in the group of Fe3+ treatment. Besides, after Fe was doped with WS2, the oxTMB of the Fe-WS2-treated group showed the highest absorption peak, confirming the synergistic role of Fe-doped WS2. The underlying mechanism of Fe-doping-facilitated POD-like activity illustrates that the electron cloud is affected by Fe embedded in the crystal lattice, resulting in more active electron-deficient or electron-rich centers for the catalytic process. To further confirm the POD-like activity of Fe-WS2 nanozyme, comparative chromatography experiments with other typically peroxidase substrates (e.g. ABTS, OPD, and DAB) were also carried out (Figure 4(c)), where similar experimental phenomena were observed, demonstrating efficient catalytic activity of Fe-WS2.
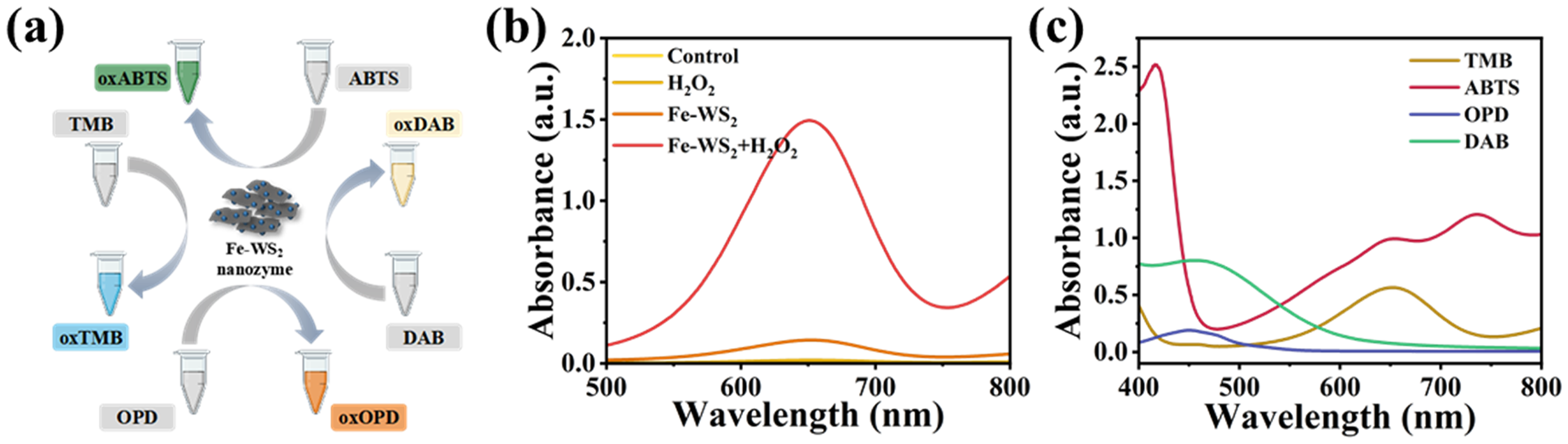
(a) Schematic illustration of the catalytic oxidation of different peroxide substrates between Fe-WS2 nanozyme and H2O2. (b) Absorption spectra of oxTMB in various reaction systems. (c) UV–Vis absorption spectra of the oxides of TMB, OPD, ABTS, and DAB in the presence of the Fe-WS2 nanozyme and H2O2.

The relative POD-like catalytic activity of WS2 and Fe-WS2 nanozyme depends on (a) concentration, (b) H2O2, (c) TMB, (d) pH, and (e) temperature. The maximum value in each curve is set to 100%.
Similar to natural enzymes, the catalytic activity of nanozymes is highly dependent on both substrate concentration and reaction conditions. 8 To systematically evaluate the POD-like activity of WS2 nanozymes before and after Fe loading, we conducted parallel experiments under identical reaction conditions. As demonstrated in Supplemental Figure S3, Fe-WS2 exhibited significantly enhanced enzymatic activity compared to its unmodified counterpart (WS2 and free Fe3+), confirming the crucial role of iron incorporation in boosting catalytic performance. To gain deeper insights into the catalytic behavior of the Fe-WS2 nanozyme, we conducted comprehensive investigations on several key parameters affecting its POD-like activity, including Fe-WS2 concentration, H2O2 concentration, TMB concentration, reaction temperature, and pH value. As illustrated in Figure 4 and Supplemental Figures S4 to S8, the enzymatic activity showed a positive correlation with increasing concentrations of the nanozyme material, H2O2 substrate, and TMB chromogenic substrate, as evidenced by the progressively intensified UV–visible (UV–vis) absorption corresponding to the oxidation product oxTMB. Besides, this concentration-dependent activity enhancement eventually reached a plateau at higher concentrations. Through careful optimization of environmental factors, we identified that the Fe-WS2 nanozyme achieved maximum relative activity at pH 4.0 and 55 °C, with activity declining outside these optimal conditions. The experimental results show that the nanozyme not only possesses excellent enzyme-like activity, but also provides some experimental basis for its use in biological assays.
To elucidate the catalytic mechanism of Fe-WS2 nanozymes and quantitatively assess the POD-like activity, we conducted a comprehensive steady-state kinetic analysis. The kinetic studies were performed using a well-established methodology where we systematically varied the concentration of one substrate (either H2O2 or TMB) while maintaining the other substrate at a constant concentration. This experimental approach allowed us to obtain classical Michaelis–Menten saturation curves across an optimized range of substrate concentrations, as shown in Figure 5(a) and (c). From these kinetic analyses, we derived two fundamental enzymatic parameters: the maximum reaction velocity (Vmax) and the Michaelis constant (Km). The Km value serves as a crucial indicator of enzyme-substrate affinity, with lower values signifying stronger binding interactions between the nanozyme and its substrate. Our detailed kinetic measurements revealed that the Fe-WS2 nanozymes exhibited significantly higher affinity (lower Km) for H2O2 compared to TMB, suggesting that the catalytic oxidation process is likely initiated through preferential activation of H2O2 molecules at the active sites (Figure 5(b) and (d) and Supplemental Table S1). The calculated Vmax values further confirmed the robust catalytic efficiency of the Fe-WS2 nanozymes, demonstrating their capacity to achieve high catalytic efficacy under optimal conditions. The superior catalytic performance, combined with the inherent stability of the nanomaterial, establishes Fe-WS2 as a promising candidate for developing reliable and efficient molecular detection platforms. This advancement provides a solid foundation for designing novel nanozyme-based biosensing systems with potential applications in clinical diagnostics, environmental monitoring, and biomedical research.

The steady-state kinetic assay of Fe-WS2 nanozyme. Reaction rates were correlated with different concentrations of H2O2 ((a) 30 mM H2O2) or TMB ((c) 0.5 mM TMB). Double inverse plots of velocity versus different concentrations of (b) H2O2 and (d) TMB.
The above experiments demonstrated the excellent POD-like activity of Fe-WS2 nanozymes, and we constructed a colorimetric biosensing system for H2O2 based on Fe-WS2 nanozymes, as shown in Figure 6(a). The absorbance intensity increased dramatically with increasing H2O2 concentration (Figure 6(b) and (c)), and simultaneously, the Fe-WS2 nanozymes-H2O2–TMB system was able to detect 0.2 to 1 mM H2O2 with excellent linearity (Figure 6(d)), and the limit of detection (LoD) (signal-to-noise ratio = 3) was calculated to be 7.27 × 10−7 M, which superior to other nanomaterials (Supplemental Table S2). Furthermore, the selectivity of the Fe-WS2 nanozyme for H2O2 was assessed by performing control experiments using other proteins, biomolecules, and metal ions (Figure 6(e)). The high selectivity of the biosensor for H2O2 was demonstrated by the fact that only H2O2 generated significant UV–Vis absorption at the characteristic peak of 652 nm compared to other proteins, biomolecules, and metal ions. This result indicates that the developed colorimetric method has favorable selectivity for the detection of glucose due to the high specificity of GOx in catalyzing the oxidation of glucose.

Schematic diagram of the response mechanism of the H2O2 bioassay. (a) Reaction mechanism for the detection of H2O2. UV–Vis absorbance in Fe-WS2/TMB systems containing different H2O2 concentrations (b, c) (n = 3). (d) The corresponding calibration curves (n = 3). (e) Selectivity of Fe-WS2 nanozyme for the detection of H2O2 (n = 3).
Glucose is the main source of energy supply in living organisms, and changes in blood levels, either low or high, can lead to disease. Therefore, it is important to know how to effectively monitor the level of glucose in the blood, and the results obtained from the test allow for effective management and control of the patient. We assayed glucose by glucose oxidation catalyzed with GOx and TMB oxygenation catalyzed by Fe-WS2 nanozymes (Figure 7(c)). It also should be noted that this sensing system exhibits an excellent selectivity toward glucose among different saccharides, profiting from the high specificity of GOx (Supplemental Figures S9 and S10). As shown in Supplemental Figure S9, single GOx or glucose cannot evoke the chromogenic reaction of TMB due to the lack of H2O2. Only when GOx and glucose are present conjointly will a significant colorimetric signal be generated. Firstly, GOx catalyzed the glucose oxidation to generate H2O2 in the presence of O2; subsequently, Fe-WS2 nanozymes catalyze the oxidation of H2O2 generated in situ. Therefore, glucose can be assayed by measuring the absorbance of TMB at 652 nm. As shown in Figure 7(a) and (b), it demonstrated promising results by detecting glucose at different concentrations. This result indicates that the developed colorimetric method has promising selectivity for the detection of glucose due to the specificity with which GOx catalyzes the oxidation of glucose. Therefore, compared with other nanomaterial-based bioassays that have been reported, the Fe-WS2 nanozyme-based catalytic system possesses great potential for detection applications with higher selectivity and sensitivity in glucose biosensing. Therefore, compared with other reported nanomaterial-based bioassays, the Fe-WS2 nanozymes-based catalytic system possesses significant potential for detection applications with higher selectivity and sensitivity in glucose biosensing.
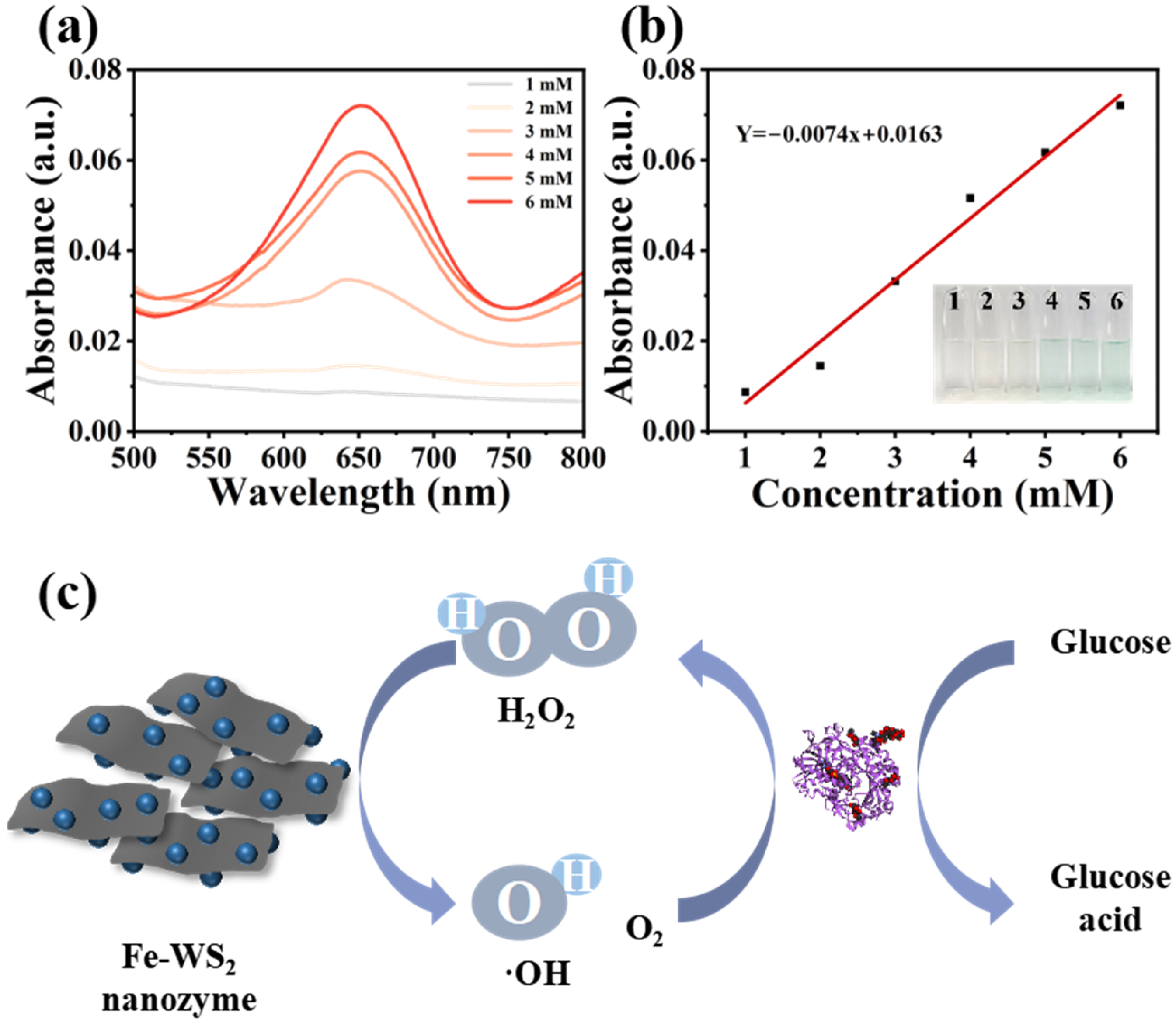
The response mechanism and detection capability of the glucose bioassay. (a) UV absorbance in Fe-WS2 nanozyme/TMB systems containing different glucose concentrations. (b) Glucose concentration-dependent UV absorbance calibration curves and images. (c) Response mechanism of the glucose assay bioassay.
Further, we conducted a CCK-8 assay to explicitly highlight the biocompatibility of Fe-WS2 nanozymes. As shown in Supplemental Figure S11, Fe-WS2 nanozymes exhibit a high tolerance dose in the mouse fibroblast cell line L929, and the relative cell viability remains above 80% even at concentrations of 100 μg/mL, respectively. These findings demonstrate its promising potential for in vivo biosensing applications.
Discussion
Although the Fe-WS2 nanozymes demonstrate high selectivity in the detection of H2O2 and glucose, their performance in complex biological samples (e.g. serum and urine) may be affected by interfering substances. Additionally, long-term stability remains a challenge, as Fe-WS2 nanozymes may undergo aggregation or catalytic deactivation during prolonged storage and usage, potentially compromising their enzymatic activity. Besides, the pH and temperature optima (pH 4, 55 °C) are far from physiological conditions (pH 7.4, 37 °C).
To address these limitations and further advance the practical application of Fe-WS2 nanozymes, the following research avenues are proposed:
These future studies will be crucial in bridging the gap between laboratory research and clinical implementation, ultimately facilitating the development of reliable nanozyme-based diagnostic tools.
Conclusions
In conclusion, iron-loaded WS2 nanozymes with outstanding POD-like activity were fabricated successfully by the hydrothermal method, which can be used for ultrasensitive biosensing. Due to the successful loading of Fe ions on WS2 nanosheets, Fe-WS2 nanozyme enabled enhanced catalytic activity of its inherent POD-like enzymes and excellent stability. We have the advantages of easy preparation, high stability, low cost, and favorable biocompatibility of Fe-WS2 nanozymes. Most significantly, the fabricated Fe-WS2 nanozyme catalytic system exhibits satisfactory sensitivity and specificity in the ultrasensitive bioassay of H2O2 and glucose. Thus, our work not only provides a novel inorganic nanozymes biosensor that can be used in various fields, but also provides some experimental basis for the application of other inorganic nanozymes biosensors in biomedical diagnostic fields.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251362318 - Supplemental material for Iron-doped tungsten disulfide nanozyme with peroxidase-like activity for hydrogen peroxide (H2O2) and glucose detection
Supplemental material, sj-docx-1-sci-10.1177_00368504251362318 for Iron-doped tungsten disulfide nanozyme with peroxidase-like activity for hydrogen peroxide (H2O2) and glucose detection by Jing Zhou, Guobo Du, Yi Li, Shiyao Liu, Xiaoyu Chen, Yue Peng and Xiaoming Wang in Science Progress
Footnotes
Acknowledgments
We gratefully acknowledge the Institute of Nanomedicine Innovation Research and Transformation at Affiliated Hospital of North Sichuan Medical College for providing experimental support and technical assistance.
Ethical statement
Ethical approval was not required for the use of the cell line in this study, as it is a commercially available, well-established cell line (L929, CL-0137 from Wuhan Pricella Biotechnology Co. Ltd).
Author contributions
YL, SL, and XC performed the experiments. JZ contributed to the scheme and figures. GD and YL discussed the data. JZ prepared the original draft. GD and XW contributed to the revision of the original draft. SL and YP conceived the idea and designed the experimental plan. GD acquired the funding.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Research project of Affiliated Hospital of North Sichuan Medical College (2022JC012), Young Elite Scientists Sponsorship Program by CAST (YESS) (2022-2024QNRC002), Central Nervous System Drug Key Laboratory of Sichuan Province (230011-01SZ), Key Research and Development Program of Chengdu (2022-YF05-02071-SN), Research and development project of Affiliated Hospital of North Sichuan Medical College (2023-2ZD004), the Fundamental Research Funds for the Central Universities, Southwest Minzu University (ZYN2024006), and Scientific Research Development Plan of The Affiliated Hospital of North Sichuan Medical University in 2024 (2024PTZK003).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available within the paper and its supplementary information files. Data are from three independent experiments and presented as mean ± SD (n = 3).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.