Abstract
Objective
Recent advancements in high-resolution ultrasonography (US) have established it as a critical tool for evaluating parotid lesions. However, the lack of standardized diagnostic criteria limits the utility of US in determining malignancy. This study investigates the potential role of US as a prognostic factor in parotid cancer.
Methods
Patients diagnosed with and surgically treated for parotid cancer at our tertiary referral center from January 2016 to December 2022 were included in this retrospective cohort study. We retrospectively obtained patient data including US images and clinical factors and analyzed their correlation with various adverse features and oncological outcomes, including five-year disease-free survival (5Y DFS) and overall survival (5Y OS).
Results
A total of 126 patients were included. The 5Y DFS and 5Y OS were 81.7% ± 3.7% and 81.2% ± 4.1% respectively. Multivariate analysis revealed that age (DFS; HR 2.75 [1.76–4.29], p = .023, OS; HR 3.38 [2.06–5.54], p = .014), clinical nodal stage (DFS; HR 5.87 [3.74–9.21], p < .001, OS; HR 9.34 [5.48–15.91], p < .001) and the presence of posterior enhancement artifact on US (DFS; HR 0.33 [0.21–0.53], p = .019, OS; HR 0.34 [0.20–0.57], p = .037) were significant variables. In patients with early-stage parotid cancer who showed posterior enhancement, the extent of surgery did not affect treatment outcomes.
Conclusion
Posterior acoustic enhancement on ultrasonography is a favorable prognostic factor in parotid cancer. For patients with early-stage cancer who demonstrate posterior enhancement, minimizing the extent of surgery does not compromise oncologic outcomes.
Keywords
Introduction
Salivary gland cancer is a rare entity, accounting for about 0.5% of all malignancies and 5% of all head and neck cancers.1,2 Its incidence is estimated at 1.3 per 100,000 individuals, and includes a wide variety of pathological types. 3 Among the major salivary glands, the parotid gland is the most commonly affected, with approximately one-fifth of parotid tumors being malignant. 4 The prognosis of parotid gland cancer is well known to depend on various factors such as cancer stage, grade, histological type, involvement of resection margin, etc.5–7
Parotid cancer is mainly treated with surgery. 2 Various approaches to parotidectomy exist, ranging from partial and superficial to total and radical parotidectomy. However, the optimal extent of resection remains debatable.8,9 When a parotid tumor is suspected to be malignant before surgery, thorough evaluation and staging are necessary to determine the surgical extent including the management of cervical lymph nodes. 8 For instance, as securing clear surgical margins is one of the most important prognostic factors, minimal approaches such as parotid extracapsular dissection must be avoided in treatment planning. 9 Thus, preoperative evaluation using various radiologic modalities has been widely adopted to predict the malignant potential of parotid tumors. 8
High-resolution ultrasound (US) is a key tool in the preoperative assessment of parotid tumors and plays a pivotal role in diagnosing parotid cancer. US demonstrates high diagnostic accuracy in differentiating malignant from benign parotid lesions, with 39%–88% sensitivity and 67%–90% specificity.10–13 However, its operator-dependent nature and the lack of internationally accepted standardized criteria make it impractical for clinicians to rely solely on US to assess the malignant potential of parotid lesions. Moreover, although US provides a high level of diagnostic accuracy, fine-needle aspiration biopsy (FNAB; 75% sensitivity, 95% specificity) and core-needle biopsy (CNB; 92% sensitivity, 100% specificity) offer even greater reliability in diagnosing parotid malignancies.10,11,14–17 A systematic review highlighted that the diagnostic value of US-guided CNB for parotid tumors is superior to that of FNAB in providing definitive histological subtyping, primarily because core samples preserve tissue architecture, making interpretation by experienced pathologists more reliable. Inconsistent diagnostic performance has been linked to interobserver variability among pathologists, especially in cases of rare or complex tumor types.14,18 US findings suggestive of malignancy will help surgeons plan surgical strategies and inform patients about the potential need for adjuvant treatments before surgery.
Therefore, this study aimed to assess the use of US as a prognostic factor and to determine its role in clinical decision-making in the therapeutic management of patients with parotid cancer.
Materials and methods
Study design and data acquisition
This retrospective cohort study was conducted at Asan Medical Center, a tertiary referral center in Seoul, Republic of Korea. Data including clinicopathological information, therapeutic processes, oncological outcomes, and US imaging were acquired through our electronic medical records database.
Patient selection
Patients pathologically diagnosed with parotid cancer at our tertiary hospital from January 2016 to December 2022 were included in this study. Patients who received surgical treatment with curative intention, and those who had complete medical records and US imaging data available were included. The exclusion criteria were as follows: pediatric patients under the age of 19 years at the time of diagnosis, patients initially presenting with distant metastatic lesion(s) (clinical M1 stage), recurrent cases who received initial therapy in another institution, patients diagnosed with another type of malignancy (current or past), and those without US imaging data.
Ultrasonography
All US images were obtained by board-certified radiologists within one year before surgery. We retrospectively evaluated parotid lesion US images based on the following aspects: size (cm), shape, margin, echotexture, echogenicity, acoustic artifact (posterior enhancement), microcalcification, and vascularity. Examples for each terminology are presented in Figure 1, and are defined below, with reference to previous studies.12,13,19,20
Size: Tumor measurements were obtained in centimeters in the direction parallel and perpendicular to the skin surface. The longest measurement, whether parallel or nonparallel to the skin, was designated as the longest diameter, while the shortest measurement perpendicular to this was recorded as the shortest diameter. The tumor was categorized as “parallel” if the horizontal length exceeded the vertical length, and “nonparallel” if the vertical length was greater. Shape: Tumors were classified as “round” (spherical or elliptical) or “irregular” (not round; spiculated; or microlobulated). Margin: Tumor margins were described as “well-defined” if more than 50% of the margin was clear, while all others were classified as “ill-defined.” Echotexture: Tumors with evenly distributed echogenic signals within the boundaries were described as “homogeneous.” Tumors with irregular internal signals were classified as “heterogeneous.” Echogenicity: Echogenicity was defined based on the predominant portion (occupying over 50% of the tumor). Tumors emitting a higher signal than the surrounding parenchyma were described as “hyperechoic,” while those emitting a lower signal were classified as “hypoechoic.” Acoustic artifact: The posterior echogenic signals of the parotid tumors were evaluated and categorized into either “posterior enhancement,” or “no posterior enhancement.” Microcalcification: The presence of microcalcifications was defined as small, highly echogenic spots (<1 mm in size) observed within the tumor contents. Vascularity: Tumors were classified as “vascular” if at least one blood flow signal was detected on a Doppler US imaging; otherwise, they were categorized as “nonvascular.”

Examples of ultrasound (US) terminology used in evaluation of parotid lesions. (a) An example of a round, well-defined, homogeneous, hypoechoic tumor with posterior acoustic enhancement. (b) Tumor with an irregular shape. (c) Tumor with an ill-defined margin. (d) Tumor with a heterogeneous echotexture. (e) An example of a hyperechoic tumor. (f) Tumor with posterior acoustic enhancement artifact. (g) Small highly echogenic spots indicating the presence of microcalcification. (h) Blood-flow signals on a Doppler US image, indicating its vascular nature.
Study outcomes and statistical analysis
All postoperatively diagnosed parotid cancer cases, including all pathology types, went through statistical analysis. The primary endpoints for this study were five-year disease-free survival (DFS) and 5-year overall survival (OS), defined as the time from the date of surgery to the date of recurrence or death, respectively.
Statistical analyses were performed using SPSS 22.0 (SPSS Inc, Chicago, IL, USA). Survival analysis was performed using the log-rank test for univariate analysis and the Cox proportional hazards model for multivariate analysis. Correlations among various factors were assessed using Fisher's exact test and the chi-square test. A p-value of less than .05 was considered statistically significant.
Ethical declaration
This study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki. The study protocol was approved by the institutional review board of Asan Medical Center (Approval No. 2024-1417). Owing to the retrospective nature of this study and the use of deidentified data, the requirement for obtaining patients’ informed consent was waived.
Results
A total of 126 patients who received surgical treatment for parotid cancer were included in this study. In accordance with our exclusion criteria, 47 out of the original 173 patients were excluded (Supplemental Figure S1). The demographics and clinical information of our patients are summarized in Table 1.
Basic demographics and clinical characteristics of the study population.
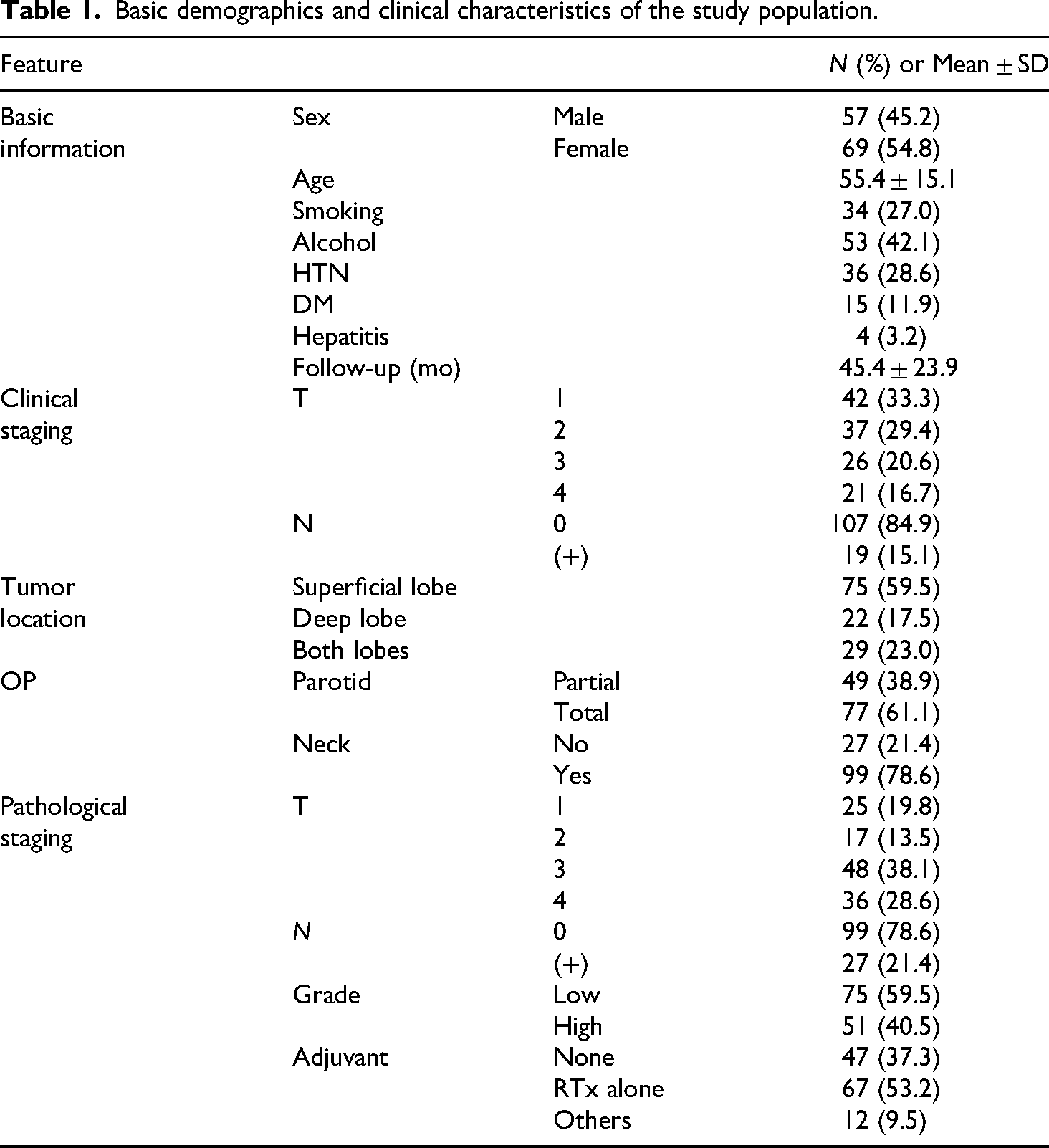
All 126 patients underwent preoperative work-up at our hospital, including US and US-guided CNB. Nine patients were initially diagnosed with benign conditions (e.g. pleomorphic adenoma, oncocytoma, basal cell adenoma), and four yielded nondiagnostic results on CNB. The former nine patients were all treated with partial parotidectomy as they did not exhibit any signs of malignant potential preoperatively, and two out of the four “nondiagnostic” patients had ill-defined margins on US and signs of capsular invasion on computed tomography and magnetic resonance imaging scans and received total parotidectomy. The remaining 113 patients were diagnosed with malignancy both before and after surgery. Our study group exhibited similar proportions of male and female patients (57 vs 69; 45.2 vs 54.8%), and more than half of the patients had tumors confined within the superficial lobe (75; 59.5%). A majority of the patients presented with clinically negative nodal status (107; 84.9%) with only eight (7.5%) found to have occult cervical metastasis after surgery. Regarding the extent of surgery, 49 (50.5%) out of the 97 patients with tumors limited to either superficial or deep lobe underwent partial parotidectomy, and only 27 (25.2%) out of 107 clinically node negative patients underwent parotid gland surgery without concurrent neck dissection. Of the 79 patients who required adjuvant therapy, 67 (84.8%) received radiotherapy alone.
All US images were obtained within one year before surgery, with a median time interval of 27 days between US and surgery (IQR: 21–38 days). The US findings and their frequencies within our dataset are illustrated in Table 2. While most features and subtypes are fairly distributed, echogenicity and microcalcification show heavily skewed distributions.
Ultrasound findings in patients with parotid cancer.

The results of survival analysis are presented as Kaplan–Meier curves of 5Y DFS and 5Y OS in Figure 2. The 5Y DFS and 5Y OS rates were 81.7% ± 3.7% and 81.2% ± 4.1%, respectively. We also analyzed the impact of various factors—including demographics, tumor stage, and US features—on patient survival outcomes, including 5Y DFS and 5Y OS. The possible prognostic factors and the results of the analyses are listed in Table 3. For practicality, age was converted into a binary variable, with a cutoff value of 65 years. It is noticeable that multivariate analysis of both 5Y DFS and 5Y OS consistently revealed age (DFS; HR 2.75 [1.76–4.29], p = .023, OS; HR 3.38 [2.06–5.54], p = .014), clinical nodal stage (DFS; HR 5.87 [3.74–9.21], p < .001, OS; HR 9.34 [5.48–15.91], p < .001), and the presence of posterior enhancement artifact on US (DFS; HR 0.33 [0.21–0.53], p = .019, OS; HR 0.34 [0.20–0.57], p = .037) as significant variables.

Kaplan–Meier survival curves depicting 5Y DFS and 5Y OS of patients with parotid cancer. (a) 5Y DFS 81.7% ± 3.7%, (b) 5Y OS 81.2% ± 4.1%.
Results of univariate and multivariate survival analyses.

Log-rank test; results of univariate analysis are described as their p-value.
Cox regression analysis.
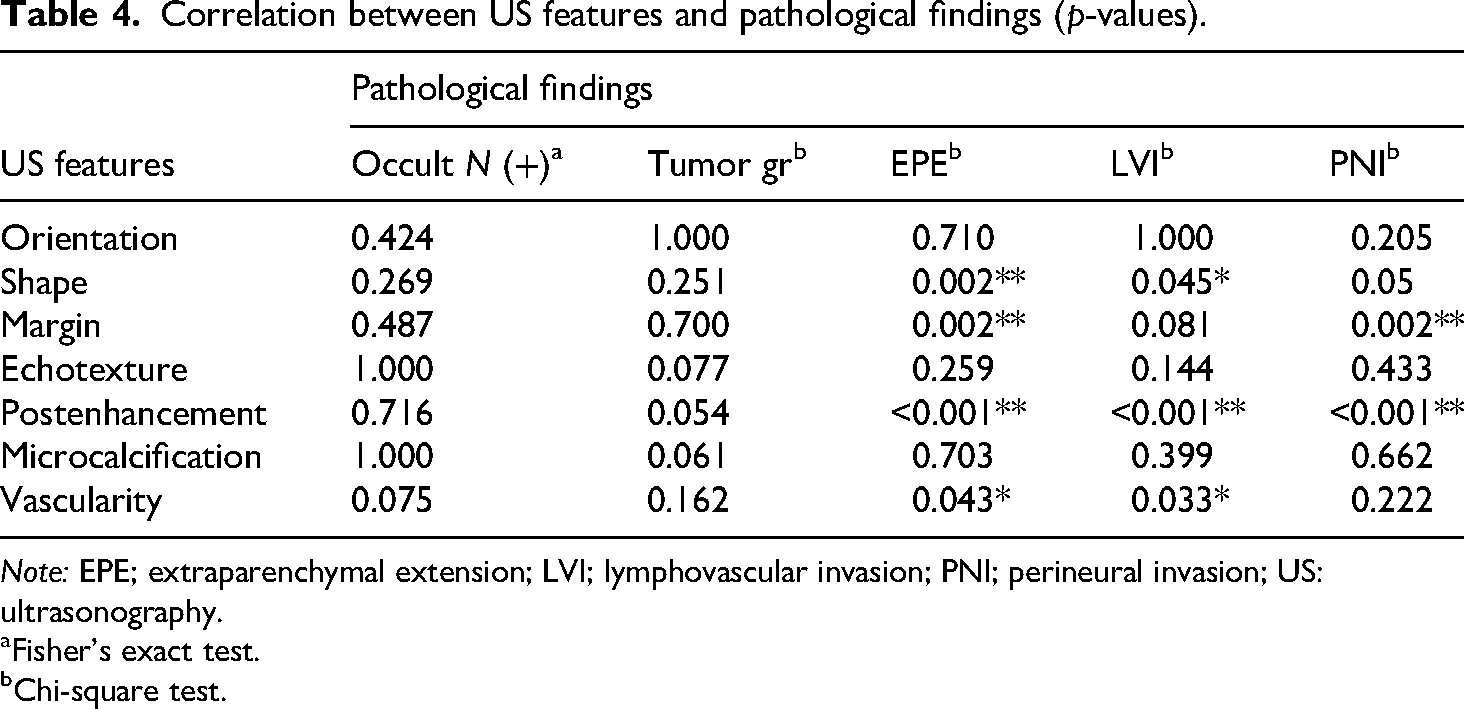
The results of the correlation analysis between US features and various adverse pathologic features are summarized in Table 4. None of the US features had any correlation with occult nodal metastasis but shape, margin, and posterior enhancement had significant correlation with adverse pathologic findings such as extraparenchymal extension (EPE), lymphovascular invasion (LVI), and perineural invasion (PNI). The p-values from Fisher's exact tests and chi square tests are listed in Table 4.
Correlation between US features and pathological findings (p-values).
Note: EPE; extraparenchymal extension; LVI; lymphovascular invasion; PNI; perineural invasion; US: ultrasonography.
Fisher's exact test.
Chi-square test.
We further investigated the potential role of posterior enhancement in determining treatment courses and their impact on survival outcomes (Figure 3). Patients with posterior enhancement had significantly better five-year outcomes for both DFS and OS (DFS; 90.0% ± 3.6% vs 68.5% ± 7.2%, p = .004, OS; 87.4% ± 4.6% vs 72.8% ± 6.9%, p = .004). Oncologic outcomes were compared among patients exhibiting posterior enhancement based on two adjustable factors related to surgical extent: the type of parotidectomy and the addition of concurrent elective neck dissection. The log-rank test revealed that in patients with early clinical T-stage parotid cancer (T1 and 2) confined to the superficial lobe, the selection between partial and total parotidectomy did not have significant impact on the outcome (DFS; 80.8% ± 7.7% vs 85.7% ± 9.4%, p = .506, OS; 86.3% ± 7.4% vs 84.4% ± 10.2%, p = .814). Similarly, performing concurrent elective neck dissection in clinically N0 stage patients had no significant benefit in oncologic outcome (DFS; 88.2% ± 7.8% vs 94.1% ± 3.3%, p = .441, OS; 100.0% vs 88.3% ± 5.0%, p = .278).

Survival analysis of patients with parotid cancer with posterior acoustic enhancement. (a, b) Kaplan–Meier curves showing five-year disease-free survival (5Y DFS) and 5-year overall survival (5Y OS), stratified by the presence of posterior enhancement; (c, d) 5Y DFS and 5Y OS of patients with parotid cancer with posterior enhancement, stratified by the extent of parotidectomy; (e, f) 5Y DFS and 5Y OS of patients with parotid cancer with posterior enhancement, stratified by the addition of concurrent elective neck dissection.
Our dataset included a variety of pathological types of parotid cancer. When divided into two groups depending on the existence of posterior enhancement, mucoepidermoid carcinoma (MEC) was the most common in both groups (41.6% and 38.8%), followed by salivary ductal carcinoma (SDC: 11.7% and 22.4%) (Supplemental Table S1). As other pathological types accounted for less than 10% in each group, we restricted the analysis to MEC and SDC to examine the differences in composition. The chi-square test revealed no statistically significant difference between the two groups (p = .173). We further investigated various factors correlated to outcome with our dataset based on the most common pathological types; MEC and SDC. The results are summarized in Supplemental Table S2.
Discussion
Numerous studies have evaluated the utility of US as a diagnostic tool to distinguish between benign and malignant parotid lesions, with conflicting results. Various US features, such as shape, margin, echogenicity, echotexture, and vascularity, have been investigated for their potential to differentiate malignant lesions. However, only a handful of them have been recognized as significant, and the results often vary among studies, with some even concluding that relying on US alone to predict malignancy is not practical, emphasizing the need for US-guided CNB for a more accurate diagnosis.13,19,21–23
In light of these previous studies, we aimed to explore the potential of US as a tool to predict oncological outcomes in patients with parotid cancer. Various parameters used for US evaluation including shape, margin, echogenicity, echotexture, acoustic artifact, and vascularity were included in our study, with an addition of several length parameters such as diameters and orientation of the tumor.
As described in Table 2, most US features showed a slight or heavy deviation toward one side of the spectrum. Features with higher proportions, such as irregular shape, ill-defined margins, and heterogeneous echotexture are generally considered—if not proven—possible predictors of malignancy.13,24–26 Conversely, features such as acoustic artifact (posterior enhancement) and vascularity demonstrated more balanced distributions across subtypes.
Using these US features, we investigated their influence on the oncological outcomes of patients with parotid cancer. The results of univariate and multivariate analysis, performed using the log-rank test and Cox regression analysis, are summarized in Table 3. Clinical features such as age (p = .007, .001), clinical nodal status (p < .001, p < .001), and tumor grade (p < .001, p < .001) were significantly associated with both 5Y DFS and 5Y OS in univariate analysis. Multivariate analysis further identified age and clinical nodal status as significant variables. Age, cervical nodal metastasis, tumor size, and tumor grade are well-described prognostic factors which is consistent with our survival analysis results.27,28 It is noticeable that among the various US features evaluated, the presence of posterior acoustic enhancement emerged as a significant predictor of better oncological outcomes in multivariate analysis (p = .019, p = .037).
To the best of our knowledge, no previous research has investigated the potential of US as a prognostic tool in parotid cancer. We hereby analyzed the correlation between US findings and known prognostic factors such as occult nodal metastasis, EPE, LVI, and PNI.29–33 The results of Fisher's exact test and chi-square test are described in Table 4. Although occult nodal metastasis and tumor grade did not show any significant relationship with US findings, EPE, LVI, and PNI exhibited strong correlations with several features, such as tumor shape, margin, and posterior enhancement. Although no previous studies have evaluated the relationship between US findings and EPE in parotid cancer, similar research has been conducted on extracapsular extension in thyroid cancer and extranodal extension in thyroid and breast cancer. These studies reported that several US findings, including discontinuous capsules and unclear margins, are significant associated with extrathyroidal and extranodal extension.34–37 Consistent with these findings, our results suggest that the presence of an irregular tumor shape or ill-defined margin can serve as a reliable predictor of histological EPE in parotid cancer.
The presence of posterior acoustic enhancement also exhibited a strong correlation with EPE, LVI, and PNI (all p < .001). Posterior acoustic enhancement is an ultrasound artifact that usually occurs when examining lesions or anatomical structures largely composed of liquid. When ultrasound pulses travel through the body, they are reflected back from surfaces possessing different sound transmissions, creating a gray-scale spatial image. As a result of less attenuation in cystic lesions, more signals can penetrate deeper to the lesion, eventually emitting an enhanced signal posterior to the lesion. It can also be seen occasionally in tissues with larger blood content or low sound attenuation—not necessarily a fluid filled structure—in relative with surrounding tissues.38–42 This type of US artifact has been discussed in relation to various types of tumors in numerous studies. In hepatocellular carcinoma, posterior acoustic enhancement has been correlated with central necrosis, and therefore, associated with more advanced tumors. 39 In breast cancer, posterior enhancement is known to be indicative of high-grade tumors such as triple-negative breast cancer, which exhibit higher mitotic rates that also cause internal necrosis, reducing attenuation and resulting in posterior acoustic enhancement.43,44 In contrast, our study found that posterior enhancement was associated with a better outcome in parotid cancer, supposedly reflecting a less aggressive nature. It is well known that the tumor grade in MEC—the most common pathological type of parotid cancer—is determined by the proportion of cystic formation with predominantly cystic tumors classified as low-grade MEC.45,46 However, in our study setting, there was no significant difference in the composition of common pathological types between groups. Moreover, as parotid cancer incorporates a wide variety of pathological types and each demonstrates distinct pathophysiological background, we further attempted to analyze factors under different types of pathologies, 47 and the two most common types in our group—MEC and SDC—were targeted for statistical analysis. However, due to their small sample size we were unable to extract any meaningful results, and as a result, the exact histological mechanism underlying the appearance of posterior enhancement in tumors with better prognoses remains unclear and warrants further investigation in future studies.
As posterior acoustic enhancement was also significantly associated with oncological outcomes, we consequently extended our research to incorporate US image evaluation into the clinical decision-making process. The Kaplan–Meier survival curves for 5Y DFS and 5Y OS are illustrated in Figure 3. Survival curves are stratified based on the existence of posterior enhancement in Figure 3(a) and (b), with significant differences observed for both DFS and OS (both p = .004), consistent with our previous results from the log-rank test.
Next, we further evaluated patients with positive posterior enhancement and investigated the eventual outcomes under different surgical approaches. First, in patients with parotid cancer with early clinical T stage tumors confined to the superficial lobe, we compared outcomes between those who underwent partial parotidectomy and those who underwent total parotidectomy (Figure 3(c) and (d)). The results showed no significant differences in 5Y DFS (80.8% ± 7.7% vs 85.7% ± 9.4%, p = .506) or 5Y OS (86.3% ± 7.4% vs 84.4% ± 10.2%, p = .814), implying that the extent of primary lesion resection does not affect oncological outcomes in early-stage, superficial lobe parotid cancer. On a nodal metastasis basis, we analyzed patients with posterior enhancement and clinically negative nodal status to evaluate the effect of concurrent elective neck dissection (Figure 3(e) and (f)). Empirical neck dissection also had no significant impact on 5Y DFS (88.2% ± 7.8% vs 94.1% ± 3.3%, p = .441) and 5Y OS (100% vs 88.3% ± 5.0%, p = .278). These findings suggest that in patients with parotid cancer with posterior enhancement on US evaluation, minimal-extent surgery does not adversely affect oncological outcomes in terms of recurrence or death.
In practice, there were 13 patients who were provided with nonmalignant results from preoperative CNBs. The majority were absent of malignant US or radiological features and received partial resection under the assumption of a benign tumor. However, all 13 patients had superior five-year outcomes with no deaths or recurrences, and posterior acoustic enhancement was present in 10 patients, including all four “nondiagnostic” patients. We could assume from these results that although preoperative CNB may occasionally provide nondiagnostic results in parotid cancer patients, posterior acoustic enhancement can assist in estimating the potential outcomes even in situations of an unexpected pathological diagnosis of parotid malignancy. 48
Our study has a few limitations. First, its single-center, retrospective design may limit the statistical value of our results. Second, the heterogeneity of the study cohort, which includes various tumor pathological types and cancer stages, may introduce statistical variability. Finally, the relatively small size underscores the need for further research involving a larger number of subjects to validate our results.
Conclusion
US features of parotid malignancies, such as the absence of posterior acoustic enhancement and irregular shape, are associated with adverse pathological indicators, namely EPE, LVI, and PNI. Conversely, the presence of posterior acoustic enhancement on US evaluation is correlated with better outcomes in terms of recurrence and death, and offers a statistical rationale for reducing the intensity of surgical management in patients with early-stage parotid cancer.
Supplemental Material
sj-docx-2-sci-10.1177_00368504251356179 - Supplemental material for Ultrasonographic findings as a prognostic factor in parotid cancer
Supplemental material, sj-docx-2-sci-10.1177_00368504251356179 for Ultrasonographic findings as a prognostic factor in parotid cancer by Jeong Heon Kim, Jeong Ho Woo, Minsu Kwon, Young Ho Jung, Seung-Ho Choi and Yoon Se Lee in Science Progress
Supplemental Material
sj-docx-3-sci-10.1177_00368504251356179 - Supplemental material for Ultrasonographic findings as a prognostic factor in parotid cancer
Supplemental material, sj-docx-3-sci-10.1177_00368504251356179 for Ultrasonographic findings as a prognostic factor in parotid cancer by Jeong Heon Kim, Jeong Ho Woo, Minsu Kwon, Young Ho Jung, Seung-Ho Choi and Yoon Se Lee in Science Progress
Footnotes
Acknowledgments
The authors would like to express their gratitude to Scientific Publications Team for the English language review.
Ethical considerations
The study protocol was approved by the institutional review board of Asan Medical Center (Approval No. 2024-1417).
Consent to participate
Owing to the retrospective nature of this study, the requirement for obtaining patients’ informed consent was waived by the institutional review board of Asan Medical Center.
Author contributions
Jeong Heon Kim: conceptualization, data curation, formal analysis, investigation, methodology, and writing—original draft, review, and editing; Jeong Ho Woo: data curation, investigation, methodology, and writing—review and editing; Minsu Kwon: data curation and writing—review and editing; Young Ho Jung: data curation, and writing—review and editing; Seung-Ho Choi: data curation, and writing—review and editing; Yoon Se Lee: conceptualization, data curation, funding acquisition, and writing—review and editing; this manuscript has been approved for submission by all the authors above.
Funding
This study was supported by a grant (Grant No. 2023IP0094-1) from the Asan Institute for Life sciences Fund from Asan Cancer Institute of Asan Medical Center, Seoul, Korea and a grant (Grant No. 23C-023-0100) SNUH Lee Kun-hee Child Cancer and Rare Disease Project, Republic of Korea.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
