Abstract
Allergic rhinitis (AR) and chronic rhinosinusitis (CRS) are chronic inflammatory disorders of the upper airways with rising global prevalence, contributing significantly to health and socioeconomic burdens. Conventional treatments—including glucocorticoids, antibiotics, antihistamines, and leukotriene inhibitors—remain insufficient in targeting the underlying pathophysiology. Bacterial lysates (BLs), functioning as immunomodulators, activate both innate, and adaptive immune pathways, reestablish Th1/Th2 equilibrium, lower immunoglobulin E concentrations, and attenuate Th2-mediated inflammation. Emerging evidence indicates that BLs (e.g. OM-85, Broncho-Vaxom) markedly improve total nasal symptom scores in AR, suppress Th2 cytokine (IL-4 and IL-13) secretion, and reduce recurrence rates and surgical interventions in CRS. In conjunction with standard therapies, BLs enhance mucosal immune defense and contribute to sustained clinical efficacy. Following the Scale for the Assessment of Narrative Review Articles guidelines, this review critically evaluates the composition, manufacturing processes, immunoregulatory mechanisms, and clinical progress of BLs in AR and CRS, providing a conceptual framework for their clinical application.
Introduction
Allergic rhinitis (AR) and chronic rhinosinusitis (CRS) are prevalent chronic inflammatory conditions of the upper airways frequently encountered in clinical settings. Over recent decades, global prevalence rates have risen markedly. European data reveal an increase in AR prevalence among Danish adults from 19% to 32% over the past 30 years. 1 In China, a 2018 cross-sectional study in Wuhan reported a 28.6% AR prevalence among children aged 6–12 years, 2 nearly triple the 9.8% reported in 2005. 3 Notably, self-reported rates of pollen-induced AR in northern grassland regions reached 32.4%. 4 In Japan, three national surveys conducted between 2005 and 2015 documented a continuous rise in pediatric AR prevalence. 5 Similarly, the burden of CRS is significant worldwide: prevalence estimates include 10.9% in Europe, 6 12–14% in the United States, 7 6.95% in South Korea, 8 5.51% in São Paulo, Brazil, 9 and 8.0%—equating to 107 million individuals—in China. 10 These widespread prevalence rates exert profound public health and economic impacts. For instance, annual treatment costs per patient with AR in the Netherlands amount to €4,827, 11 while Chinese patients with CRS demonstrate a 4.5-fold increase in healthcare utilization and an average of 11.7 days of work absenteeism annually compared to non-CRS individuals, 12 emphasizing the urgency of targeted interventions. 13
Pathophysiologically, AR is characterized by chronic, non-infectious inflammation of the nasal mucosa, predominantly driven by allergen-specific immunoglobulin E (IgE) responses in atopic individuals following allergen exposure. CRS represents a multifactorial and heterogeneous disease influenced by anatomical anomalies, genetic predisposition, and environmental exposures. Although conventional therapeutic approaches can mitigate symptoms, they seldom address the underlying mechanisms.
Recent interest has centered on bacterial lysates (BLs) for their immunomodulatory potential. By mimicking pathogen-associated molecular patterns (PAMPs), BLs activate mucosal immunity and restore immune homeostasis, offering promising avenues for the treatment of nasal inflammatory disorders. Accumulating evidence supports their efficacy across in vitro, in vivo, and clinical contexts. Emerging research has identified variability in receptor expression profiles, patterns of eosinophil infiltration, and modulatory roles of organic salts and short-chain fatty acids (SCFAs). In accordance with the Scale for the Assessment of Narrative Review Articles, 14 this narrative review synthesizes recent developments in BL research, constructs a theoretical framework to bridge basic science with clinical application, and outlines future directions to advance innovation in this domain. A comprehensive literature search was conducted through MEDLINE (PubMed) using the Boolean query (“bacterial lysates” OR “OM-85” OR “BV”) AND (“allergic rhinitis” OR “chronic rhinosinusitis”), covering publications from 1 January 1988, to April 1, 2025. Findings were presented in a narrative format aligned with established methodological standards to ensure clarity and coherence.
Composition and preparation of bacterial lysates
BLs are immunomodulatory agents derived from inactivated antigens of common respiratory pathogens,15,16 including Staphylococcus aureus, Moraxella catarrhalis, Klebsiella pneumoniae, Streptococcus pneumoniae, and Haemophilus influenzae. 17 Designed for the prevention of respiratory tract infections (RTIs) and allergic conditions, BLs are produced through two primary methodologies. Chemical lysis (polyvalent chemical bacterial lysates, PCBLs), which disrupts bacterial membranes under alkaline conditions (pH 11.5–12.5), may compromise protein structure and reduce antigenicity. In contrast, mechanical lysis—used to produce polyvalent mechanical BLs (PMBLs)—employs ultrasonication or high-pressure homogenization to preserve antigenic integrity and enhance immunogenic potential.18–20 Commercial formulations such as OM-85 BV(Broncho-Vaxom®) and Liuvac (LW-50020) are chemically lysed preparations. OM-85 comprises 21 bacterial strains from five genera and promotes both cellular and humoral immunity, reinforcing systemic defenses and mucosal barriers while modulating immune responses to deliver synergistic prophylactic and therapeutic effects.19,21 Liuvac, a composite of BLs, 22 stimulates dendritic cell (DC) activation, T and B cell proliferation, IgA secretion, and production of antigen-specific antibodies, while also enhancing cross-protective immunity against unrelated pathogens. 23 Oral administration remains the most common route, typically in the form of sublingual drops, tablets, or capsules. 18 Sublingual delivery is especially effective due to the dense presence of DCs in the sublingual mucosa, ensuring robust immunogenicity and a favorable safety profile. 24 BL-induced secretory immunoglobulins form a protective mucosal barrier across the respiratory tract, inhibiting microbial adhesion and neutralizing toxins to strengthen respiratory immunity. 25 Evidence supports that sublingual administration elicits durable systemic and mucosal antigen-specific humoral and cellular responses. Notably, it facilitates the generation of antigen-specific memory CD4+ T cells, which promote Th1-biased immunity and bolster antimicrobial defense mechanisms. 26 Compared to probiotics, BLs offer advantages such as reduced storage requirements, shorter treatment durations, and direct immune stimulation. Unlike vitamin D, which enhances antimicrobial peptide synthesis and inhibits pathogen colonization indirectly,27,28 BLs exert their effects through active immunostimulation, promoting comprehensive immune balance. Additionally, BLs represent a cost-effective alternative to biologics by combining therapeutic and preventive benefits with a long-term safety profile.2,29
Immunomodulatory mechanisms of bacterial lysates
Innate immunity (non-specific immunity)
The innate immune response constitutes the primary defense barrier against exogenous insults, involving both hematopoietic and non-hematopoietic tissues. Central effector cells include DCs, neutrophils, eosinophils, macrophages, mast cells, and natural killer (NK) cells, which express a range of pattern recognition receptors (PRRs) to detect pathogens and initiate immune responses. 30 BLs potentiate antiviral cytokine secretion, activate macrophages and NK cells, and attenuate eosinophilic infiltration.31,32 The immune response operates through coordinated multicellular cascades targeting invading pathogens.
Within BLs, lipopolysaccharide binds toll-like receptor 4 (TLR4) on NK cells, inducing IFN-γ release. Human NK cells transcribe multiple TLRs, notably TLR2 and TLR4.33–37 IFN-γ, via paracrine signaling, activates neighboring macrophages and promotes TNF synthesis. In turn, macrophages engage in autocrine feedback through TNF receptors while simultaneously producing and secreting interleukin-12 (IL-12). The IFN-γ/IL-12 axis provides dual regulation of NK cells by maintaining activation and amplifying IFN-γ output through positive feedback, ultimately enhancing macrophage phagocytic capacity and recruiting neutrophils to infection foci for bactericidal action.38–42 Notably, BL administration increases circulating NKT and CD4+ NKT cell counts relative to baseline, with post-treatment profiles characterized by elevated IFN-γ/IL-4 ratios, increased IL-10 levels, and significantly reduced IL-4 concentrations compared to corticosteroid-treated cohorts. 31
DCs are indispensable to innate immunity but require maturation to achieve full immunological competency. Maturation pathways empower DCs to regulate innate responses via antiviral cytokine synthesis.
OM-85 BV stimulates bitter taste receptors (T2Rs) in the upper airway epithelium, inducing inducible nitric oxide synthase
Adaptive immunity (specific immunity)
Adaptive immunity confers broader and more antigen-specific recognition than innate immunity through tightly regulated interactions between antigen-presenting cells (APCs) and T/B lymphocytes, facilitating the development of pathogen-specific effector responses. 49 DCs, as pivotal APCs, function at the interface of innate and adaptive immunity by initiating antigen-specific activation of T and B cells.50,51
BLs induce DC activation, enabling antigen processing and presentation to naïve T cells and promoting their polarization into Th1, Th2, Th17, or Treg subsets.
51
Immunomodulatory effects of BLs encompass both innate activation and restoration of Th1/Th2 equilibrium.22,29 This balance is critical, as Th1 and Th2 lineages exert reciprocal inhibitory effects despite their distinct effector roles.
52
Th1 polarization is predominantly driven by DC-derived IL-12 and IFN-γ from NK cells, CD4+, and CD8+ T cells. In turn, Th1-secreted IFN-γ reinforces this axis by suppressing Th2 differentiation via cytokine-mediated feedback loops.
51
Th2 responses, characterized by IL-3, IL-4, IL-5, IL-9, and IL-13 secretion, facilitate eosinophil recruitment and type 2 immunity.
53
In murine bronchoalveolar lavage fluid, OM-85 administration increases IFN-γ levels and IFN-γ/IL-4 ratios while concurrently suppressing IL-4, indicating a Th1-skewing effect.
54
Bidirectional regulation by BLs preserves immune homeostasis in both in vitro and in vivo settings. Clinical data from Kamil Janeczek et al. demonstrate that patients with seasonal AR treated with BLs exhibit stable allergen-specific IgE levels, in contrast to significant IgE elevations in untreated controls.
55
These results suggest that BLs attenuate Th2-dominant immune responses in seasonal AR. Supporting animal studies further report reductions in Th2 cytokines (
Following oral administration of BLs, PAMPs are recognized by PRRs, including TLRs and other PRRs. 57 Hanif et al. demonstrated that TLR4 is essential in mice, whereas in humans, a synergistic activation of TLR2 and TLR4 is required. OM-85 activates NF-κB and MAPK signaling pathways via TLR2/4, inducing specific immunoregulatory genes, such as those involved in immune tolerance. 58 DCs, the most potent APCs, initiate naive T cell activation and proliferation into helper T cell subsets. These DCs migrate to cervical lymph nodes and enter the thoracic duct via lymphatic vessels, where they present antigens and deliver activation signals through TCR, CD28, and CD40L interactions on T cells. Subsequently, CD4+ T lymphocytes differentiate into Th1 and Treg subsets, contributing to B cell activation.53,57 Evidence indicates that PMBL enhances the proportion of activated B lymphocytes expressing surface CD25, suggesting modulation of the IL-2/IL-2R signaling axis—a pivotal pathway for lymphocyte survival, expansion, and differentiation in both humoral and cellular immunity. These findings align with prior in vitro data showing that 24-h PMBL exposure markedly upregulates IL-2Rα expression in human B cells. 59 Upon activation, B cells differentiate into antibody-secreting plasma cells, initially producing IgM and later undergoing isotype switching to IgA, IgG, or IgE. This class-switch recombination is influenced by the cytokine milieu; IL-4 and IL-13 promote IgE synthesis, whereas IL-10 and IFN-γ favor IgA and IgG production. Given that allergic disorders are associated with elevated IgE levels, the immunoglobulin switching trajectory may either ameliorate or exacerbate allergic manifestations. 60 Lee et al. reported that BV administration modulates SCFA production, mimicking probiotic activity. These findings reinforce the concept of a unified mucosal immune network, where SCFAs generated in the gut exert immunomodulatory effects on distal tissues via systemic circulation. 61 Additionally, while serum IgG and IgA levels exhibit minimal elevation following oral OM-85-BV intake, significant increases in salivary IgA and IgA within lymph node culture supernatants have been observed. Notably, IgA levels in saliva remained elevated even after immunization with unrelated antigens (e.g. tetanus toxoid), underscoring that OM-85-BV primarily stimulates mucosal immunity—particularly IgA secretion—with limited influence on systemic antibody responses. 48 Figure 1 summarizes the immune mechanisms of BLs in AR and CRS.
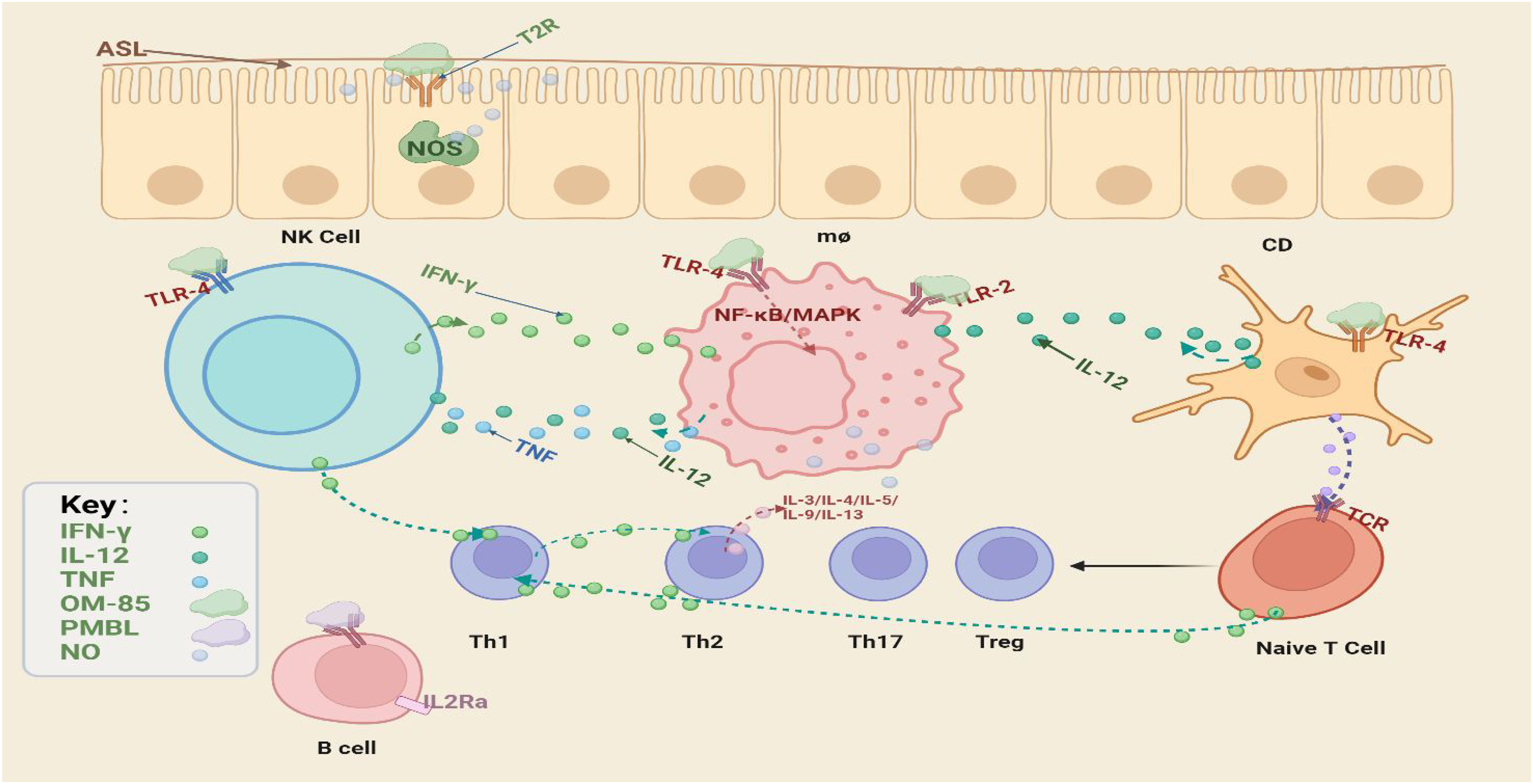
Main immune mechanisms of bacterial lysates (BLs) in AR and CRS. BLs activate T2R in the upper airways, triggering NOS to release NO, which plays a pivotal role in the innate immune response. BLs also stimulate DCs, which process and present antigens to naïve T cells, promoting their differentiation into Th1, Th2, Th17, or Treg subsets. Th1 differentiation is influenced by DC-derived IL-12 and IFN-γ produced by NK, CD4+, and CD8+ T cells. Mature Th1 cells secrete IFN-γ, which inhibits Th2 differentiation through cytokine cascades. Th2 cells, in turn, secrete IL-3, IL-4, IL-5, IL-9, and IL-13, orchestrating eosinophil recruitment and type 2 immune responses. Created with BioRender.com. AR: allergic rhinitis; CRS: chronic rhinosinusitis; BL: bacterial lysate; NK: natural killer; IL: interleukin; NO: nitric oxide.
Clinical applications of bacterial lysates (Table 1)
Clinical applications of bacterial lysates in AR
AR is a chronic, non-infectious inflammatory condition of the nasal mucosa, primarily mediated by IgE in individuals with atopy following allergen exposure. Allergen-activated T cells proliferate via the Th2 pathway, secreting cytokines that prompt plasma cells to produce allergen-specific IgE. These antibodies subsequently bind to mast cells and basophils in a process known as “sensitization.”62,63 Immunotherapies for AR are classified into allergen-specific and non-specific strategies. Due to the limitations of allergen-specific treatments, non-specific therapies—including biologics, microbial agents (e.g. probiotics, BLs), and vitamin D—have gained prominence. BLs, in particular, have garnered increasing attention as a potential treatment for AR. 64 Commercialized for decades under various brands, BLs exhibit both local and systemic immunomodulatory effects, though their clinical outcomes remain variable. 18
Clinical study of bacterial lysates in the treatment of allergic rhinitis and chronic rhinosinusitis.
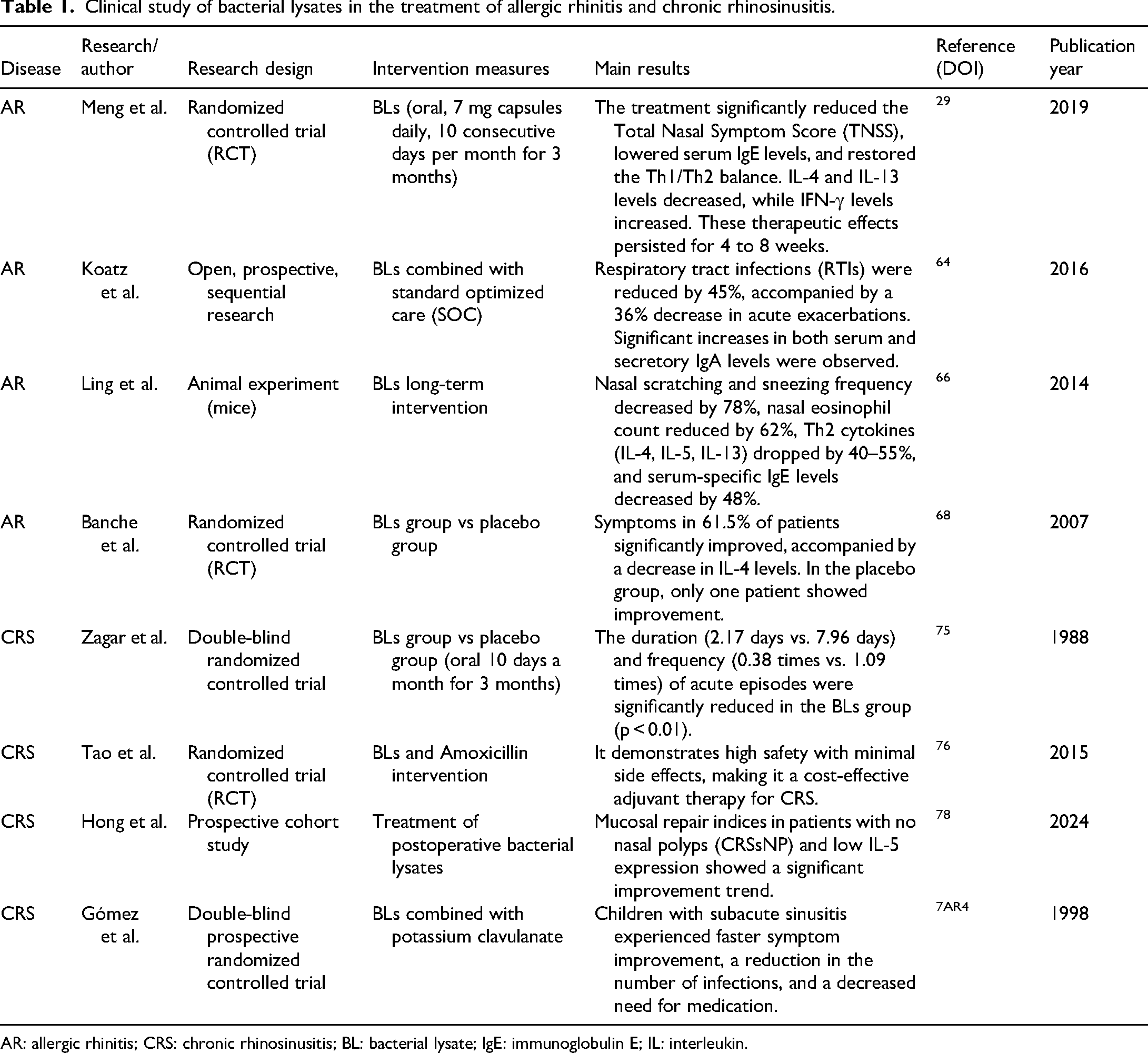
AR: allergic rhinitis; CRS: chronic rhinosinusitis; BL: bacterial lysate; IgE: immunoglobulin E; IL: interleukin.
Symptom alleviation and immunological biomarker modulation
Several randomized controlled trials (RCTs) have demonstrated that BLs (e.g. OM-85, Broncho-Vaxom) significantly reduce total nasal symptom scores (TNSS), lower serum IgE levels, and restore the Th1/Th2 cytokine balance in patients with AR. A 2019 study by Meng et al. found that OM-85 significantly reduced TNSS compared to placebo, effectively alleviating nasal itching and rhinorrhea. Nasal lavage fluid exhibited decreased IL-4 and IL-13 levels, an increase in IFN-γ, and a reduced IL-4/IFN-γ ratio. Eosinophil counts were also lower, with the benefits of OM-85 lasting 4–8 weeks post-treatment. These results suggest that OM-85 restores Th1/Th2 cytokine balance through cellular and signaling pathways, thereby enhancing mucosal immunity in AR. 29 In a separate study, Koatz et al. assessed the efficacy of OM-85 in preventing RTIs and modulating serum/secretory IgA levels in patients with AR, asthma, and COPD. This open-label, prospective sequential study revealed a 74% reduction in RTIs (from 266 to 69) with standard optimized care (SOC), and an additional 45% reduction (to 38) with OM-85 as an adjunctive therapy. Acute exacerbations of underlying diseases decreased by 36% (from 55 to 35) during OM-85 treatment. Immunological analyses revealed significant increases in serum and secretory IgA following OM-85 administration. These findings indicate that OM-85, when added to standard therapy, significantly reduces the incidence of respiratory infections and improves disease control, likely through enhanced mucosal immune responses. 64 Furthermore, sublingual PMBL tablets effectively alleviate seasonal AR (SAR) symptoms in grass pollen-sensitized children, with Th1-like γδ T cells serving as potential biomarkers for disease severity. 65
Long-term efficacy and preventive effects
Long-term use of BLs for ≥ 3 months has been shown to reduce the recurrence of AR, especially when used adjunctively with dust mite desensitization therapies. Ling et al. demonstrated that extended OM-85 treatment significantly alleviates AR symptoms, including a 78% reduction in nasal scratching and sneezing (p < 0.01), a 62% decrease in eosinophil infiltration, and a 40–55% reduction in Th2 cytokines (IL-4, IL-5, IL-13) in nasal lavage fluid. Additionally, serum-specific IgE and IgG1 levels were reduced by 48% and 37%, respectively. Histopathological analysis revealed decreased nasal mucosal inflammation (31% compared to controls) and improved goblet cell hyperplasia in OM-85-treated mice. Short-term OM-85 treatment also resulted in a 25–30% reduction in Th2 cytokines and a 22% reduction in IgE, although complete symptom suppression was not achieved. Mechanistically, OM-85 upregulates Foxp3+ regulatory T cells in nasal-associated lymphoid tissue and enhances IFN-γ secretion, restoring Th1/Th2 balance. Thus, OM-85 BV controls AR progression via dual, time-dependent immunomodulatory mechanisms, with long-term oral regimens holding promise as preventive strategies.66,67 Similarly, Banche et al. (2007) conducted an RCT involving 41 patients with AR (26 BL-treated, 15 placebo), assessing clinical and immunological outcomes at baseline (T0) and 3 months post-treatment (T3). BL-treated patients exhibited significant clinical improvements, including a 61.5% improvement in symptoms, a 53.8% reduction in nasal congestion and rhinorrhea, a 50% resolution of ocular symptoms, and a 38.4% improvement in asthma symptoms, with no adverse effects or exacerbations. Efficacy was sustained at the 3-month follow-up, matching T3 outcomes. Immunological analyses indicated selective effects of BL treatment, such as reduced IL-4 (Th2 cytokine) levels, without changes in total IgE, allergen-specific IgE, or IFN-γ levels. In contrast, the placebo group showed minimal improvement, with only one patient reporting partial symptom relief. These results provide mechanistic support for BLs as novel immunomodulatory therapies that can positively influence AR clinical trajectories. 68 A systematic review and meta-analysis by Li et al. concluded that BL therapy improves symptom control in patients with allergic diseases when combined with conventional treatments, without presenting any adverse risks. 69
Clinical application of bacterial lysates in the treatment of chronic rhinosinusitis
CRS is a long-term inflammatory condition of the sinus mucosa, persisting for over 12 weeks. CRS leads to physical symptoms such as a diminished sense of smell, rhinorrhea, nasal congestion, and facial/head pain, while also heightening the risk of anxiety and depression. Although not fatal, these persistent symptoms can significantly impact quality of life, work performance, and social function in affected individuals.70,71 The European consensus document recommends BLs, particularly OM-85BV, for the treatment of adult CRS. 72 OM-85BV has been used in various countries for decades, with applications in both adults and children for the treatment and prevention of recurrent respiratory infections, including COPD, asthma, and rhinosinusitis. However, it has not yet been FDA-approved in the United States.44,73
Reduce the number of attacks and surgical needs
BLs reduce the frequency of acute exacerbations in CRS by inhibiting bacterial biofilm formation and pro-inflammatory factors such as IL-6 and TNF-α, potentially allowing some patients to avoid surgical interventions. Gómez et al. conducted a 6-month, double-blind, prospective RCT to assess the safety and efficacy of OM-85 BV in treating subacute sinusitis in children. The experimental and control groups received clavulanate potassium (10 mg/kg daily for 21 days). In addition, the experimental group received OM-85 BV capsules (3.5 mg/day) for 10 days before each month, while the control group received a placebo for 3 months. Results indicated that the experimental group experienced faster symptom improvement and a shorter recovery period. After 6 months of follow-up, the experimental group showed a significant reduction in the number of infections and required less medication compared to the control group. These findings support OM-85 BV's role in accelerating the cure and improvement of subacute sinusitis. 74 In a separate study, Zagar et al. examined the preventive effect of BLs on CRS recurrence in children. Fifty-one children with acute CRS exacerbations were randomly assigned to receive either OM-85 BV or a placebo under double-blind conditions. Observations and follow-up revealed that BLs significantly reduced both the frequency and duration of CRS recurrences. 75 Furthermore, Tao et al. demonstrated a coordinated downregulation of SOCS1, SOCS3, TNF-α, and IFN-γ expression in both the OM-85 BV/amoxicillin combination and amoxicillin monotherapy groups compared to model controls, with significant reductions at both mRNA and protein levels. These mechanistic insights validate OM-85 BV's potential as a cost-effective adjunct for CRS management, aligning with previous therapeutic findings. 76
Combination therapy synergistic effect
Currently, drugs and surgery are the standard treatments for CRS. However, even with medical and surgical interventions, some patients remain prone to frequent relapses and may require secondary surgeries. 77 When combined with surgical treatment, BLs can offer additional benefits, particularly in improving postoperative endoscopic outcomes. The endotype of CRS has demonstrated clinical value in guiding treatment decisions. Hong et al. aimed to evaluate the efficacy of postoperative BL therapy in patients with different CRS endotypes, conducting a prospective cohort study involving patients who underwent standardized endoscopic sinus surgery. Patients with recurrent upper RTIs and no immune contraindications were included in the treatment group and received periodic BLs post-surgery, while the control group received standard postoperative care. Endoscopic mucosal repair, quality of life, and tissue inflammation were assessed through multiple follow-ups. Although no significant overall difference in efficacy was observed between the two groups, subgroup analysis revealed that mucosal repair in patients with CRS with no nasal polyps (CRSsNP) and low IL-5 expression showed significant improvement. 78
Conclusion and outlook
BLs exhibit broad immunomodulatory effects by activating both innate and adaptive immune responses. They enhance phagocytic and bactericidal functions through NK cell, macrophage, and neutrophil interactions, while also restoring Th1/Th2 balance through DC-mediated antigen presentation. This process helps reduce IgE levels and suppresses Th2-driven inflammation. Clinical studies indicate that BLs significantly improve the TNSS in patients with AR, reduce Th2 cytokine secretion (IL-4, IL-13), and decrease recurrence rates and the need for surgical interventions in CRS. However, despite substantial evidence supporting the efficacy of BLs, treatment effects can vary. Therefore, further research is essential to explore the underlying mechanisms, the immunogenicity and complexity of BLs, population differences, and more robust evidence on the long-term safety of these treatments. With the continued advancement of immunology and biotechnology, BLs hold the potential to overcome existing treatment limitations and become a key component of the “immune rebalancing” strategy for respiratory diseases, offering a new paradigm that moves beyond symptom control to address the underlying etiology.
Footnotes
Acknowledgment
We would like to thank Chunping Yang for reviewing and providing advice on the final search strategy. We thank Bullet Edits Limited for the linguistic editing and proofreading of the manuscript.
Author contributions
XH designed the literature search, collected the data, created the figures, and drafted the initial version of the manuscript with support from CY. XZ offered valuable feedback and revisions to the manuscript. ZZ and ML prepared original draft. DB and RY reviewed and edited the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of China (No. 82160211) and the Jiangxi Provincial Nature Science Foundation (No. 2021BAG70031).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
