Abstract
Diffuse large B-cell lymphoma is the most prevalent subtype of non-Hodgkin lymphoma (NHL), which makes up the majority of malignant lymphomas of the head and neck region. Lymphoma of the salivary gland is extremely rare. Most patients present with a unilateral, hard, non-tender mass that gradually enlarges over time. A thorough evaluation is necessary for a precise diagnosis because its clinical presentations are nonspecific, increasing the risk of misdiagnosis, leading to an inappropriate treatment plan, and ultimately delaying the optimal treatment of the disease. Imaging techniques like computed tomography scans and magnetic resonance imagings are part of the initial workup. Prior to biopsy, it can be challenging to differentiate NHL of the parotid gland from other tumors. Histological analysis and subsequent immunohistochemistry are essential for the definitive diagnosis. Systemic chemotherapy is the mainstay of treatment, while surgery and radiation may be necessary in some circumstances. Herein, we describe a 60-year-old man who had a steadily growing mass in his right parotid region. The pathology results confirmed it to be NHL, diffuse large B-cell lymphoma subtype. No similar lesions were found on clinical examination and subsequent radiological imaging. The patient received R-CHOP chemotherapy with impressive treatment outcomes. To date, at least 12 months of regular follow-up have passed since the patient completed oncological care; he remains free from signs and symptoms of the disease. This case points out the importance of considering lymphoma in the differential diagnoses when examining parotid neoplasms.
Introduction
Parotid gland malignancies are rather uncommon, and the majority of parotid gland tumors are benign. 1 The three most common malignant neoplasms are mucoepidermoid carcinoma, acinic cell carcinoma, and adenoid cystic carcinoma. 1 Primary salivary gland lymphoma is an extremely uncommon condition that makes up 1% to 2% of all salivary gland neoplasms. 2 The diagnosis of the primary parotid gland lymphoma can be challenging both clinically and histopathologically, especially in small biopsies, since both malignant and benign conditions can mimic lymphoma. 3 The etiology for parotid lymphoma is not clearly understood.1,2 While most benign and malignant parotid tumors are treated surgically, lymphoma requires chemotherapy and/or radiation therapy. Therefore, a delayed diagnosis may result in a poor prognosis.
Herein, the authors describe a case of a primary non-Hodgkin lymphoma (NHL) of the parotid gland in a 60-year-old male patient and discuss imaging findings and possible treatment strategies for salivary gland lymphoma. This case study contributes to the body of knowledge and can be used to provide treatment recommendations in environments with limited resources. This work has been reported as being in line with the CARE guidelines. 4
Case presentation
A male patient in the 60s age range presented to Kilimanjaro Christian Medical Centre (KCMC), a zonal tertiary hospital located in Moshi, Kilimanjaro, Tanzania, in January 2024 with the complaint of a right-sided parotid mass for 6 months. The mass started as a small nodule on the right parotid gland initially. It kept on increasing in size over time, occupying the whole of the parotid region. His past medical history revealed that he was HIV-infected and on regular medications. He reported a history of slight weight loss, periodic fevers, and cough. His past medical history was essentially unremarkable. He denied a history of autoimmune conditions, including Sjögren's syndrome. The laboratory blood work results indicated a CD4 cell count of 500 cells/mm3. The tuberculosis and hepatitis test results were negative. Immunoglobulin profile and autoimmune markers including Rheumatoid factor results were unremarkable. His vital signs, including blood pressure, body temperature, and perfusion, were within normal limits.
On general physical examination, he was alert and well oriented to time, place, and persons. On local examination, a firm, irregular, and tender mass of about 10 × 7 cm in greatest dimension was appreciated over the right parotid gland area (Figure 1A,B). The mass was extending inferiorly to the mid-neck, and it was associated with pain and episodes of fainting. There were no nasal symptoms reported. Similarly, his voice was normal, and he could swallow normally. The facial nerve function was intact. The review of other systems was essentially unremarkable.
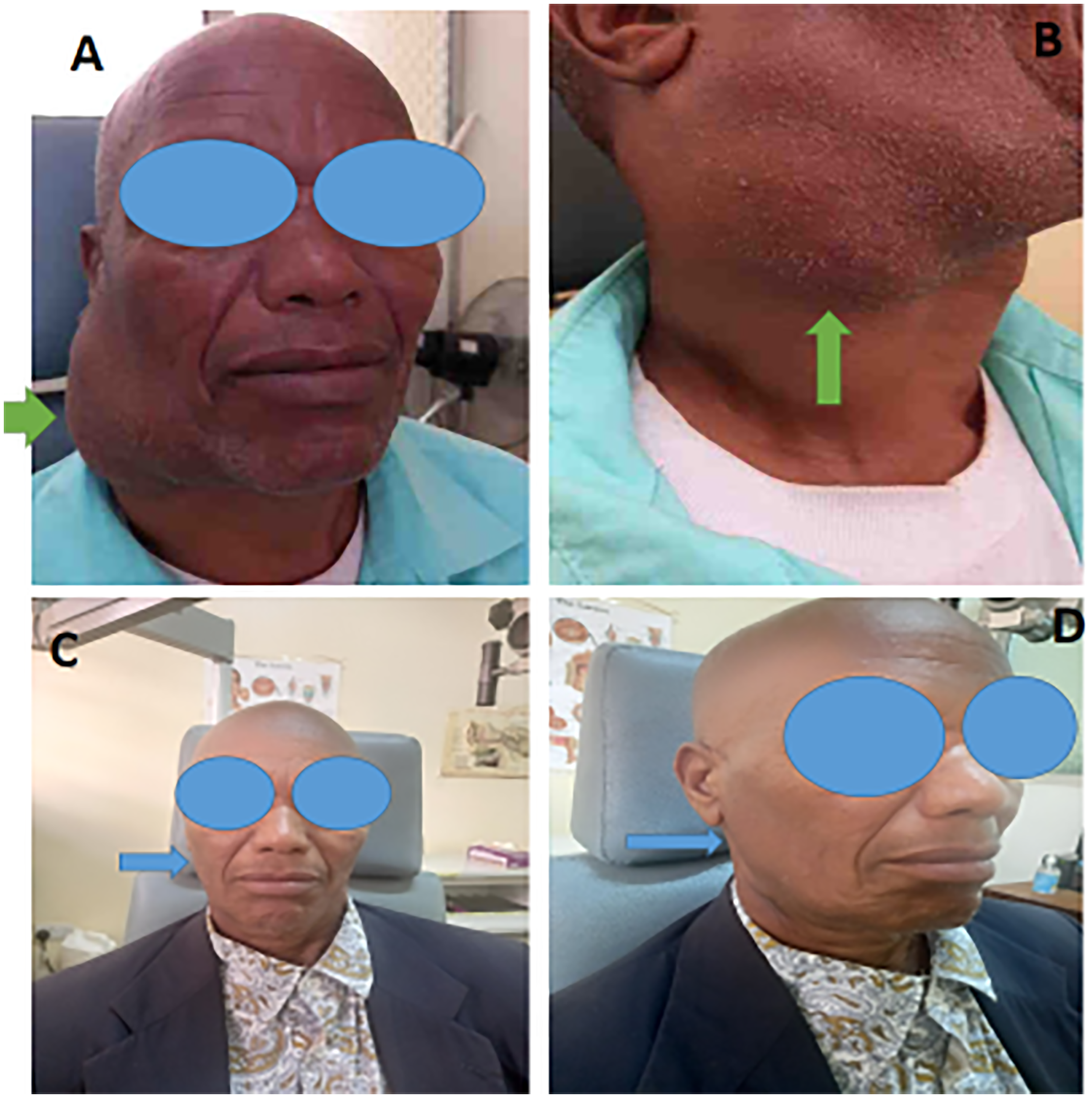
Photographs of the patient showing a huge right parotid mass before treatment (green arrow) (A), the mass extends inferiorly to the upper neck (green arrow) (B), presentations of the patient after oncological care (blue arrows) (C,D).
The head and neck computed tomography (CT) scans demonstrated a huge lobulated heterogeneous enhancing soft tissue lesion with areas of necrosis centered on the right parotid gland. The mass was arising from the superficial lobe of the right parotid gland, and it was extending to the ipsilateral carotid space and paravertebral space, with encasement of the jugular vein. It was abutting the adjacent sternocleidomastoid, scalene, and trapezius muscles. Neither neuroforamina extension nor bone remodeling was associated. The mass measured 10.2 × 7.6 × 10.5 cm in size (Figure 2A,B). The clinical diagnosis of parotid gland malignancy was entertained.

The head and neck CT scans highlight a huge lobulated heterogeneous enhancing soft tissue tumor centered on the right parotid gland, extending to neck inferiorly (A). The huge mass is abutting the adjacent sternocleidomastoid, scalene and trapezius muscles giving the clinical impression of parotid gland malignancy (B). CT: computed tomography.
Fine-needle aspiration cytopathology results were in favor of a malignant lymphoid neoplasm (Figure 3A). Wedge biopsy histopathology report confirmed the presence of an infiltrating tumor comprised of a diffuse population of medium to large atypical lymphoid cells replacing the parotid gland epithelial tissue (Figure 3B,C). The tumor cells were strongly positive for CD20 (Figure 3D), with high Ki67 expression, but negative for CD3, EBER, TdT, and cytokeratin

Photomicroscopy of a fine-needle aspiration cytology smears from the parotid mass demonstrate scattered large atypical lymphoid cells with vesicular chromatin and prominent nucleoli, consistent with a large cell lymphoma, hematoxylin and eosin (H&E) staining at 40 x original magnification (A), histopathology of the biopsy from the parotid mass shows atypical lymphoid cells invading salivary gland tubules, H&E staining at 20 x original magnification (B); complete effacement and replacement of the salivary gland tissue by diffuse population of large atypical lymphocytes with vesicular nuclei, H&E staining at 20 x original magnification (C), diffuse strong membrane immunostaining of the tumor by CD20, IHC at 10 x original magnification (D).
Discussion
After squamous cell carcinoma, lymphoma is the second most prevalent tumor in the head and neck region. 1 Salivary gland tissue lymphomas are extremely uncommon; they account for about 0.3% of all head and neck malignancies, 1% to 2% of all salivary gland tumors, and about 5% of extranodal lymphomas. 5 Most of the published data in the English literature are on case reports.6–18 When referring to malignant lymphomas of the parotid gland, the terms primary and secondary, as well as nodal and extranodal, are used with some ambiguity in the literature. 5 The presence of intraglandular lymph nodes in the parotid gland is a significant issue with the definition. It can be challenging to identify the lymphoma's origin because the gland parenchyma is frequently secondary. Nodal NHL should be used to describe cases of NHL that are restricted to these lymph nodes and do not include glandular parenchyma. 1 Making this distinction is often not simple, though, as patients with significant nodal and parenchymal involvement may nonetheless have started intraglandular lymph nodes. 6 It can be challenging to identify the lymphoma's origin because the gland parenchyma is frequently secondary. The way these individuals are treated is unaffected by this controversy.5,7 However, most scholars agree that for a case to be regarded as a primary parotid lymphoma, the glandular parenchyma must be affected, and the bulk of the disease must occur in the parotid gland.3,8,9,15,18
Among salivary gland lymphomas, the parotid gland is the most frequently affected site (79%), with the submandibular gland coming in second (20%). 3 Although lymphomas of the parotid gland are usually NHL of B-cell origin, other histopathological lymphoma subtypes such as Hodgkin lymphoma and, rarely, T-cell lymphomas have also been documented.2,6,8–10,12,14–19 Salivary glands may also be affected by extranodal NK/T-cell lymphoma of the nasal type, peripheral T-cell lymphoma nonspecific, and anaplastic large cell lymphoma. 19 However, the most common subtypes are mucosa-associated lymphoid tissue (MALT) lymphoma and marginal zone lymphoma.16,17 Diffuse large B-cell lymphoma, which is one of the most common NHL subtypes seen in our patient, is an infiltrative tumor associated with effacement of the salivary gland tissue.6,10,14
The actual cause of primary parotid NHL is unknown. It typically develops on the background of benign conditions such as Sjögren's syndrome, lymphoepithelial lesions, or myoepithelial sialadenitis 1 ; although, this was not the case in our patient. The majority of these lesions are likely to be either extranodal marginal zone B-cell lymphoma of MALT type with substantial follicular colonization, or they are nodal lymphomas originating from intraglandular lymph nodes with later glandular parenchyma penetration. 14 Parotid diffuse large B-cell lymphoma can develop from an underlying extranodal marginal zone B-cell lymphoma of the MALT type in certain cases. 15 There have been isolated reports of NHL developing in the lymphoid stroma of Warthin tumors.6,10 It is possible that the lymphoma found in the Warthin tumor is the initial sign of a more widespread illness.
Primary lymphoma of the parotid gland is rarely suspected preoperatively. Due to its nonspecific clinical presentation, parotid lymphoma is usually overlooked and thus is diagnosed and treated later than it should be. 3 Among salivary gland lymphoma cases, the parotid (70%), followed by the submandibular gland (20%), are the most affected areas. 3 As was mirrored in our patient, the average age of detection is 61 years, and generally there is a relative tendency more toward males than females, at a ratio of about 2:1.5,18 In some cases, the tumor was bilateral; that is, it was involving both parotid glands. 20 Typically, most patients demonstrate a palpable mass, and tenderness is reported in some cases. As observed in our patient, most NHLs occurring in salivary glands are B-cell lymphomas. 10 The lymphoma discovered in the Warthin tumors may be the presenting feature of more generalized disease. Approximately 15% of all NHLs of the salivary glands are caused by diffuse large B-cell lymphoma of the parotid gland.10,14 The tumor causes partial or complete effacement of normal salivary gland tissue architecture, replacing it with a diffuse infiltrate of large atypical B lymphoid cells, as mirrored in the index case.3,5
Malignancies, in particular lymphoma, are typically not likely to be regarded as important differential diagnoses because 80% of parotid gland tumors are mostly benign. 5 When diagnosing parotid lymphoma, it is crucial to keep in mind the important differential diagnoses, such as benign conditions that may mimic NHL histologically, especially in small biopsies. These include lymphoepithelial sialadenitis, Rosai-Dorfman disease, and Kimura disease, to name a few.21,22 In our case, we were able to exclude potential differential diagnoses through thorough clinicopathological correlation and careful histopathology evaluation that was complemented by a panel of immunohistochemistry tests. Radiological imaging plays a crucial role in identifying parotid lymphomas from the lymphoid tissue within the parotid gland or its surrounding structures. Imaging can help differentiate between benign and malignant lesions, determine the extent of disease, guide biopsy, and plan treatment. 23 The main imaging modalities used include ultrasound, CT, and magnetic resonance imaging, each with its own advantages and pitfalls.24,25 However, despite the critical role of imaging tests in the diagnosis, staging, and management of parotid lymphomas, a definitive diagnosis of lymphoma still requires histopathological examination. 3
While most benign and malignant tumors are treated surgically with procedures including lumpectomy and parotidectomy,20,26 lymphoma typically requires chemotherapy and/or radiation therapy. 5 Postoperative radiation therapy is typically used to treat patients with localized disease, whereas chemotherapy is recommended for those with multifocal or systemic disease.7,8 As mirrored in the index case, diffuse large B-cell lymphomas GCB subtype generally have a better prognosis and respond better to standard chemoimmunotherapy (like R-CHOP) when compared to activated B-cell-like or non-GCB diffuse large B-cell lymphomas. 27 The clinical stage and histologic type of salivary gland lymphomas determine the prognosis. A delayed diagnosis could make this disease worse. 8 A worse prognosis is typically linked to T cell lymphomas and extranodal NK/T cell lymphomas.19,26
A potential caveat for our case study is the absence of control CT scans after therapy due to the patient's financial constraints. Such imaging would have been instrumental in guiding decisions regarding consolidation radiotherapy, considering the fact that a bulky mass was present at initial diagnosis.
Conclusion
In conclusion, because it is extremely uncommon to encounter lymphoma in a parotid gland, this case study is quite interesting to physicians. Timely pathology diagnosis and prompt treatment are critical to improving treatment outcomes. This case highlights the importance of keeping this diagnosis in mind when dealing with patients with parotid masses and underscores the role of pathological diagnosis prior to invasive surgical treatment of salivary gland tumors.
Footnotes
Acknowledgments
The authors wish to thank the patient for allowing us to use his medical information for academic purposes. We are equally thankful to staff in the departments of ENT, Pathology and Oncology as well as the KCMC hospital management for supporting this study.
Ethical considerations
The patient provided written informed consent to allow his de-identified medical details to be used in this publication. A waiver for ethical approval was obtained from the authors’ institution review board (IRB) committee since case reports do not need IRB approval.
Author contributions
MM contributed to conceptualization, data curation, methodology, supervision, and writing—review and editing. AM contributed to conceptualization, data curation, investigation, methodology, and writing—original draft. MK contributed to data curation, methodology, and writing—review and editing. AJ contributed to data curation, methodology, and writing—review and editing. FK contributed to data curation, methodology, resources, and writing—review and editing. EA contributed to data curation, investigation, methodology, supervision, and writing—review and editing. KM contributed to data curation, investigation; methodology, supervision, and writing—review and editing. DC contributed to data curation, investigation, methodology, and writing—review and editing. AM contributed to conceptualization, data curation, investigation, methodology, and writing—review and editing.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was in part funded by the Science for Africa Foundation for the Developing Excellence in Leadership, Training, and Science in Africa (DELTAS Africa) program (Del-22-008) with support from Wellcome Trust and the UK Foreign, Commonwealth, & Development Office, and is part of the EDCPT2 program supported by the European Union.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
No data were generated from this study.
