Abstract
This study examined the histological aberrations in the gill and liver tissues and behavioural changes of Tilapia guineensis fingerlings exposed to lethal concentrations of used Oilfield-based emulsifiers for 96 h. Various concentrations of the surfactants were tested, ranging from 0.0 to 15.0 ml/L. The behaviour of the fish was observed throughout the experiment, and the results showed that increasing concentrations of the surfactants led to progressively abnormal behaviour, including hyperventilation and altered opercular beat frequency. These behavioural changes indicated respiratory distress and neurotoxic effects. Histological analysis revealed structural aberrations in the gill and liver tissues, with higher concentrations causing more severe damage, such as lesions, necrosis, inflammation, and cellular degeneration. This implies that surfactants released even at low concentrations are capable of inducing changes in the tissues of aquatic organisms. These findings highlight the toxic effects of the surfactants on fish health and provide biomarkers of toxicity. Future research should focus on understanding the specific mechanisms and long-term consequences of surfactant toxicity on fish genetic composition, populations, and ecosystems to implement effective conservation measures.
Introduction
Water pollution is one of the major problems in the world today due to rapid industrialization and unplanned urbanization. 1 Due to these factors, many rivers in Nigeria are experiencing complex pollution problems. 2 Because of the high concentration of chemicals in the environment, aquatic sources are highly susceptible to pollution. 3 These water bodies are frequently stored for a wide variety of xenobiotics that cause biochemical and histopathological differences in fish because they alter the physicochemical parameters of the water. 4 Monitoring the chemical composition of water makes it possible to determine how contaminated it is. 5 The study of biomarkers’ response to pollution can be used to assess and evaluate the effects of pollution on various organisms, including fish. 6 Industrial activities have significantly increased the production and use of various chemical compounds, including emulsifiers or surfactants. 7 These emulsifiers are extensively employed in industries such as food processing, cosmetics, agriculture, and pharmaceuticals. 8 While emulsifiers are crucial in enhancing product stability and performance, their potential adverse effects on aquatic ecosystems and organisms cannot be overlooked. In particular, the ecotoxicity of commonly used industrial emulsifiers on aquatic organisms has become a matter of concern due to their widespread environmental distribution and persistence. 9
Tilapia guineensis, commonly known as the Guinean tilapia, is a widely distributed freshwater fish species found in various aquatic habitats.10,11 Due to its ecological and economic importance, it serves as an excellent indicator species for assessing the impact of environmental contaminants on aquatic ecosystems. 12 As emulsifiers can be discharged into aquatic environments through industrial effluents or accidental spills, understanding their effects on T. guineensis is essential for evaluating potential risks and implementing effective mitigation measures. Histopathology, however, has a broader scope of expression than most biomarkers. Changing histopathology in animals’ tissues is a reliable indicator of environmental stress. 13 Moreover, it is the best method for assessing the effects of a substance both in the short and long term. 14 Toxicant impact assessment uses histopathological evaluation to indicate the effects of toxicants on fish health, as well as to detect early warning signs of diseases or injuries. 15 The structure of fish has also been studied as a biomarker by many researchers in various species of fish.16,17 To determine toxicological effects, histopathology is the gold standard; however, it is time-consuming, expensive, and invasive. According to Drishya et al., 18 the use of biomarkers with known histological distribution could also be useful in localizing toxic injury throughout distinct organs or tissues, since histological testing may be impractical in human subjects. To assess the adverse effects of xenobiotics on the whole organism, histopathological evaluation remains essential. 17 A review of the most important histocytopathological changes in various fish organs, which have been used as biomarkers during various pollution monitoring programs, is presented in the present study. 19
Emulsifiers used in oilfield operations can significantly impact fish species in affected waters, as they are sensitive to environmental changes and are key indicators of aquatic ecosystem health. 3 These emulsifiers, designed to break down oil-water interfaces, can introduce chemical compounds into aquatic environments, leading are crucial on fish behaviour, physiology, and overall performance. 8 Exposure to emulsifiers can disrupt fish's sensory perception, impacting their ability to locate prey and navigate their habitats. These changes can have cascading effects on the entire food web, influencing predator-prey dynamics and ecosystem stability. 20 The physiological responses of fish to oilfield chemicals extend beyond behavioural changes. Substances released during oil recovery processes can enter the bloodstream of fish, affecting organs and vital functions. 7 Exposure to emulsifiers can lead to oxidative stress, immune system suppression, and reproductive abnormalities, which can compromise the fitness and resilience of fish populations and the long-term sustainability of affected ecosystems. 21 Different fish species have varying degrees of vulnerability to oilfield-based emulsifiers, with some demonstrating higher tolerance while others may face severe consequences even at lower concentrations. 10
Histopathology, the study of tissue changes or abnormalities, provides valuable insights into the sublethal effects of chemical exposure on organisms. 22 By examining histological alterations in fish tissues, researchers can assess the impact of emulsifiers on organs such as the liver, gills, and kidneys. 23 Histopathological changes can serve as reliable indicators of toxicity and provide valuable information regarding the mechanisms of action and potential long-term effects on fish health and population dynamics.16,24 In addition to histopathology, behavioural alterations can provide early warning signs of chemical toxicity and serve as sensitive indicators of sublethal effects on fish. Emulsifiers may interfere with the normal behaviour patterns of T. guineensis, such as feeding, swimming, shoaling, and reproductive behaviour. 25 By carefully observing and quantifying behavioural changes in response to emulsifier exposure, researchers can gain insights into the impacts of these chemicals on fish ecology, population dynamics, and overall ecosystem functioning. 26
T. guineensis fingerlings were included as the study subjects due to their relevance to the aquatic ecosystem under investigation. Only fingerlings of a specific age range were included to maintain uniformity and minimize developmental variations. Fish displaying normal health indicators, without pre-existing illnesses or abnormalities, were included in the study to ensure baseline health before exposure to emulsifiers. Individuals belonging to fish species other than T. guineensis were excluded from the study to maintain a species-specific focus. Fish exhibiting signs of illness, malformations, or abnormalities during the pre-experimental assessment were excluded to prevent confounding factors. Adult fish in reproductive stages were excluded to avoid potential influences on behaviour and histopathological responses related to breeding activities. Fish with a history of exposure to similar emulsifiers or contaminants were excluded to ensure a clean slate for observing the effects of the oilfield-based emulsifiers. Fish outside the specified size range were excluded to maintain consistency in the experimental setup. Individuals with visible physical deformities that could affect normal behaviour or physiological responses were excluded to enhance the clarity of observed effects.
By adhering to these inclusion and exclusion criteria, the study aimed to create a controlled and standardized experimental environment, ensuring that observed responses could be attributed specifically to the exposure of T. guineensis fingerlings to varying concentrations of oilfield-based emulsifiers. Thereby investigate the ecotoxicity of commonly used industrial emulsifiers (surfactants) on T. guineensis (juveniles) by employing histopathological analysis and behavioural alterations as assessment protocols. In tests of toxicity, it is considered one of the most suitable organisms. 27 The objectives of the study include identifying histological changes in fish tissues and correlating them with the different concentrations of the emulsifier exposure, quantifying behavioural alterations in response to different concentrations of emulsifiers, and evaluating the potential risks posed by emulsifiers to the health and ecology of T. guineensis populations.
Materials and methods
Ethical approval
The present study has been ethically approved by the 59th Meeting of Research Ethics Committee with references number of UPH/R&D/REC/04, as the main application of the ethics was based on the first author doctorate study.
Fish collection and acclimation
Two hundred and ten (210) healthy juvenile Guinean tilapia (T. guineensis) were collected from the Nigeria Institute for Oceanography and Marine Research (NIOMR), Buguma, Rivers State (Latitude 4°74'03.48827″N; Longitude 6°85'89.7601″E), Nigeria and were transported in plastic containers to the Department of fisheries wet laboratory at the University of Port Harcourt. The fish had a mean length of 15.2 ± 1.2 cm and a mean weight of 14.1 ± 5.2 g. They were acclimated in an aerator 150 l capacity glass aquarium tank for 14 days for continuous oxygenation at a laboratory room temperature of 27.8 ± 3.1°C. They were fed twice daily with commercial fish feed (Coppens fish feed) purchased from a commercial shop named Animal Afears and the water in each glass tank was replaced every 48 h with tap water from the laboratory. Feeding was suspended 24 h before and during the static exposure period that lasted for 96 h.
Test chemical
The commercial oilfield-based emulsifier (Surfactant) was collected in a 2.5 l container from an industrial chemical laboratory in Port Harcourt, Rivers State (Joe-chem Nigeria limited) and was stored under ambient conditions before use. The chemicals were available in liquid form and were treated directly in the test medium after producing a working stock solution.
Preparation of a working stock solution for Xylene & Diesel
The working stock solution used for this assay was prepared from the chemical following the method of King et al.
28
The test chemical was prepared, using the equation:
This was prepared using the method described in previous studies by Davies et al. 29 Six test concentrations of the toxicants were prepared, each test concentration of (0.0 ml/L as control), 2.5, 5.0, 7.5, 10.0, and 15.0 ml/L were introduced into a plastic aquarium tank of 15 l and each concentration was in replicates. The tanks were filled to 10 marks, and 10 healthy fish samples were randomly selected and put in each test concentration. The different groups of fish were exposed for 96 h during which the behavioural response of the fish samples was observed by closely monitoring the movement of the fishes from 24 to 96 h.
Behavioural responses
Each treatment group of fish was exposed for 96 h during which the behavioural changes of the fish samples were assessed by closely monitoring the movement of the fish. Parameters such as respiratory movement (operculum beat) and the tail fin beat frequency, loss of reflex, hyperventilation, erratic swimming suffocation or spiralling were recorded from 24 to 96 h and dead fishes were removed immediately to avoid contamination of the experimental water. These were conducted using a standard method as described by Davies and Uedeme-Naa. 20
Determination of the histopathological effect on Guinean Tilapia (Tilapia guineensis I)
After 96 h of exposure, two active juvenile T. guineensis from each test concentration were euthanized for dissection and the liver and gill of each fish were harvested and prepared for histopathological sections. The organs were fixed in Bouin's fluid and subsequently dehydrated through a graded series of ethanol 30 (Oladokun et al., 2020). They were embedded in paraffin and manually sectioned with a microtome at 4 to 5 µm. The sections were dewaxed and stained with hematoxylin and eosin (H&E) and examined using a digital light microscope (Leica DM500). 31
Statistical method
The results were subjected to a one-way Analysis of variance (ANOVA) using the Statistical Package for the Social Sciences (SPSS Version 23) to determine significant differences between various treatments and controls. The Multiple Range Test was used to separate differences among means. Differences were considered significant at (P < 0.05). 32
Results
Behavioural changes
The behavioural changes observed in T. guineensis exposed to different concentrations of aqueous surfactant for 24 to 96 h (Table 1). Normal behaviour was observed in the controlled group. Fish exposed to 0.0 ml/L had normal behaviour from 24 to 96 h but afterwards, the fish that were alert stopped swimming and remained static for a while in response to the sudden changes in the surrounding environment. Generally, fish exposed to higher concentrations such as 2.5, 5.0, 7.5, 10.0, and 15.0 ml/L, respectively showed the chemicals showed progressive hyperventilation and abnormal behaviour such as very fast swimming, and erratic swimming movement, gulping of air, jumping, and displaying vigorous jerky movement suffocation and loss of reflex. A faster operculum and tail beat movement was also observed with spiralling. The behavioural responses varied significantly (P < 0.05) with an increase in concentration per time as compared to the control group of fish.
Behavioural response after 24, 48, 72 and 96 h of exposure to an aqueous surfactant.

Note: + = Positive response and − = Negative response.
Opercular beat frequency (OBF)
The result of Opercular Beat Frequency (Figure 1) of T. guineensis exposed to an aqueous surfactant for 96 h showed significant variation (P < 0.05) in the treatment means and the control group (Table 2). The least value (52.0 ± 0.6 beats per minute) of the opercula beat frequency was observed in the control (0 ml/L) and the maximum opercula beat frequency value (81.6 ± 0.9 beats per minute) was observed at the 15.0 ml/L exposed concentration of the toxicant. The test fish showed initial erratic movements, rapid opercula beat and increased mucus secretions on their skins and gills. There was also an increase in the opercula beat frequency per minute from 24 to 72 h and a decrease in the time of exposure at 96 h before death. The OBF rate decreased significantly (P < 0.05) with an increase in time as compared to the control group of fish.
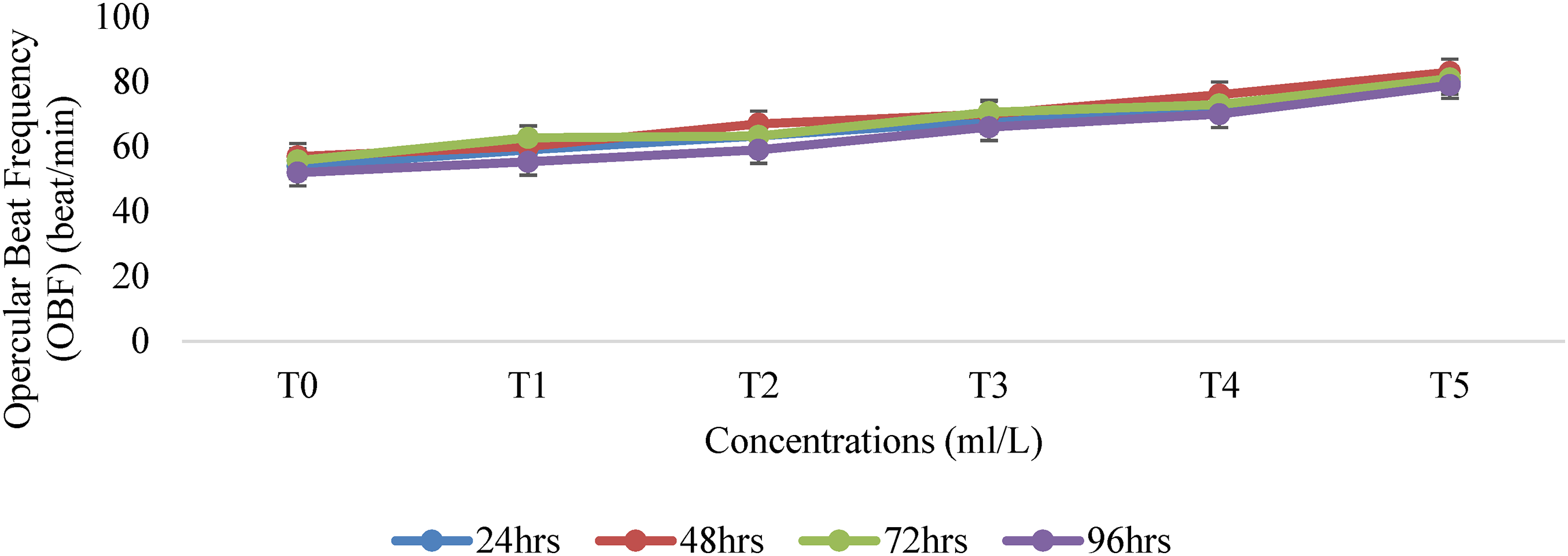
Opercular Beat Frequency (OBF) of Guinean tilapia (T. guineensis) exposed to Aqu. Surfactant for 24–96 h.
Effect of different concentrations of aqueous surfactant on Opercular Beat Frequency (OBF) of Guinean tilapia (T. guineensis) during 96 h of the exposure period.
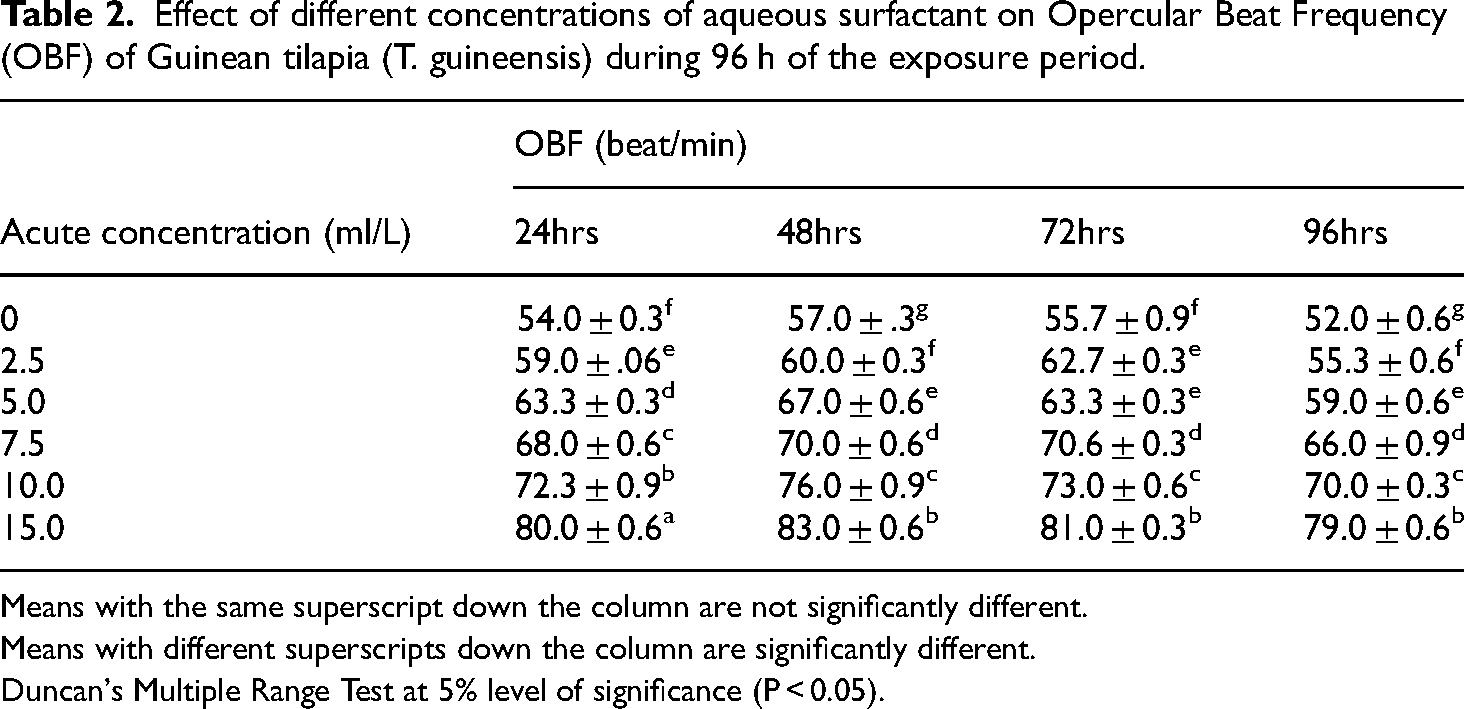
Means with the same superscript down the column are not significantly different.
Means with different superscripts down the column are significantly different.
Duncan's Multiple Range Test at 5% level of significance (P < 0.05).
Tail fin beat frequency (TBF)
The result in Table 3 showed that the tail beat frequency of T. guineensis was 34.0 ± 0.6, 31.0 ± 0.6, 39.0 ± 0.6, 40.7 ± 0.3, 43.6 ± 1.2, 53.7 ± 0.3 and 52.3 ± 0.9 beats per minute for the different groups exposed to 0 ml/L (control), 2.5, 5.0, 7.5, 10.0, and 15.0 ml/L of aqueous surfactant for 96 h respectively (Figure 2). These values showed statistical significance (P < 0.05) between the control group and the fish exposed to the toxicant. The TBF per minute of T. guineensis decreased with an increase in time and increased with an increase in the concentration of the chemical. There was a progressive indication of stress observed before death. There was also an increased tail beat frequency per minute from 24 to 48 h and a decline in the frequency from 72 to 96 h before death. There was a significant difference (P < 0.05) between the TBF rate for the exposed group of T. guineensis and those in the control.

Tail Beat Frequency (TBF) of Guinean tilapia (T. guineensis) exposed to Aqueous Surfactant for 24–96 h.
Effect of different concentrations of aqueous surfactant on Tail Beat Frequency (TBF) of Guinean tilapia (T. guineensis) during 96 h of the exposure period.
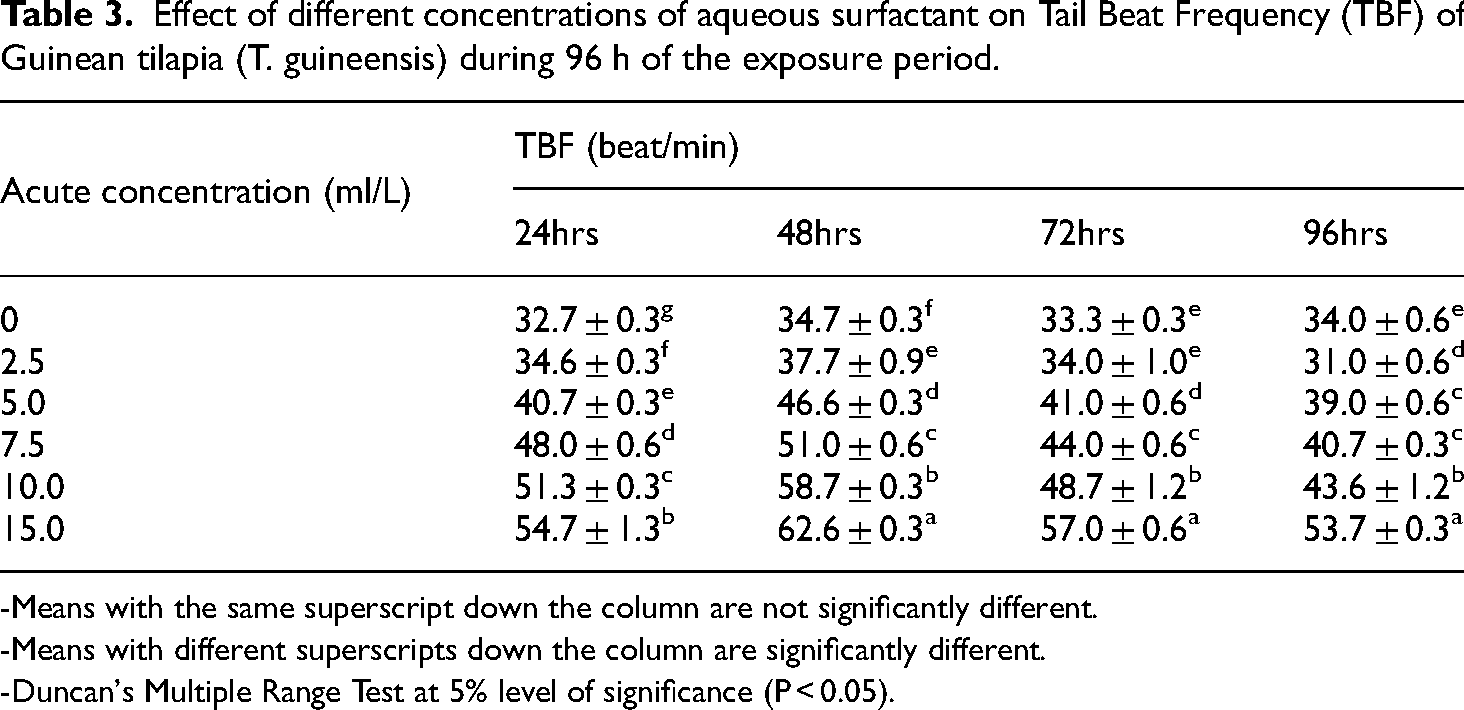
-Means with the same superscript down the column are not significantly different.
-Means with different superscripts down the column are significantly different.
-Duncan's Multiple Range Test at 5% level of significance (P < 0.05).
The structural aberrations observed in the sections of livers of Tilapia guineensis
Figure 3 represents the liver sections of Guinean tilapia (T. guineensis) exposed to different concentrations of aqueous surfactant for 96 h. The control group (0.0 ml/L) depicted in Plate A exhibits a normal cellular pattern, a normal central vein, and a space of disse. No lesions, necrosis, pigments, malignancy, inflammation, or inclusion bodies are observed. Plate B shows the presence of a lesion (L), inflammation, and necrosis (IN). Plate C exhibits a lesion and necrosis (LN). Plate D displays necrosis (N), inflammation, and cellular degeneration (ICD). Plate E demonstrates cellular degeneration, necrosis, inflammation, and a lesion. Finally, Plate F shows necrosis, pigment deposition, and inflammation. In summary, the liver sections of the Guinean tilapia exposed to various concentrations of aqueous surfactant exhibit different pathological findings, including lesions, necrosis, inflammation, cellular degeneration, and pigment deposition, depending on the concentration of the surfactant.
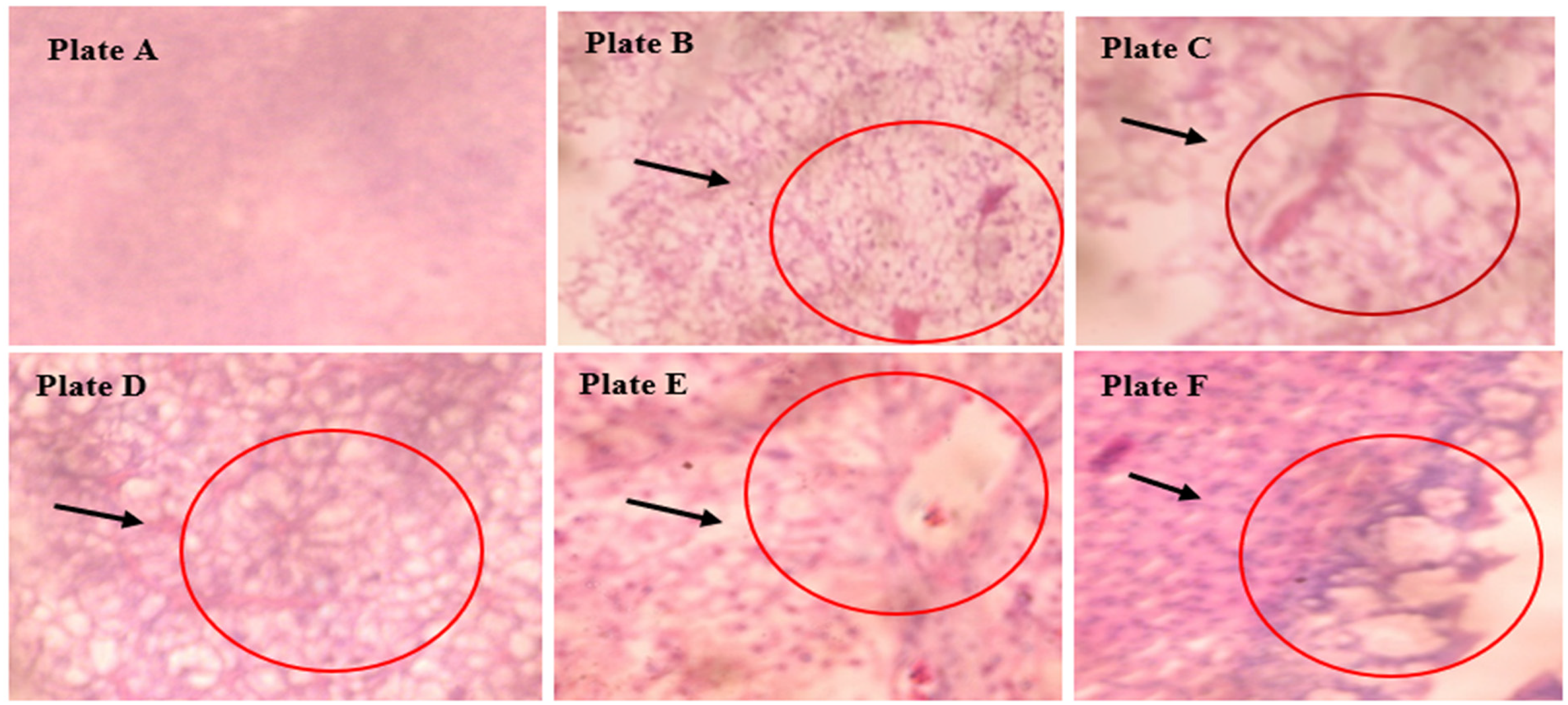
Photomicrography of the liver tissues of the control and exposed T. guineensis at X400 magnification.
The structural aberrations observed in the gills of Tilapia guineensis
The gill sections of T. guineensis exposed to different concentrations of aqueous surfactant for 96 h are depicted in Figure 4. The control group (0.0 ml/L), represented by Plate A, exhibits a normal cellular pattern, specifically in the pseudo-branchial lamella. Plate B displays various abnormalities, including lesions (L), pigment deposition (P), necrosis (N), cellular degeneration (CD), malignancy, and inflammation (MI). Plate C shows lesions, pigment deposition, malignancy, and inflammation. Plate D exhibits lesions, pigment deposition, necrosis, and inflammation. Plate E showcases inflammation, pigment deposition, malignancy, and cellular degeneration, while Plate F shows pigment deposition, malignancy, and cellular degeneration. In summary, the gill sections of the Guinean tilapia exposed to different concentrations of aqueous surfactant demonstrate a range of pathological findings, including lesions, pigment deposition, necrosis, cellular degeneration, malignancy, and inflammation, depending on the concentration of the surfactant.

Photomicrography of the gill of the control and exposed T. guineensis at X400 magnification.
Discussion
Behavioural response and changes
The observed differences in the behavioural responses of Guinean tilapia (T. guineensis) exposed to different concentrations of aqueous surfactant can be attributed to the toxic effects of the surfactant on the fish's nervous system and overall physiological well-being. Behavioural changes in fish can serve as indicators of stress, discomfort, or toxicological effects caused by exposure to chemical substances as suggested by. 20 The fish in the control group exhibited normal behaviour throughout the observation period. This indicates that the absence of surfactant exposure did not cause any significant disruption to their normal swimming patterns or responses to the environment. 33 Initially, the fish exposed to 0.0 ml/L surfactant concentration showed normal behaviour for 24 to 96 h. However, after this period, they exhibited a response to sudden changes in the surrounding environment by becoming static for a while. This behaviour suggests heightened alertness or a cautious response to potential threats or disturbances. It is important to note that this behavioural change is not directly associated with the presence of surfactants but rather a reaction to external stimuli. 34
The fish exposed to increasing concentrations of surfactants displayed progressively abnormal behaviour. Hyperventilation, characterized by an increased breathing rate, was observed, indicating respiratory distress and potential oxygen deprivation. 35 The abnormal behaviour included very fast and erratic swimming movements, gulping of air, jumping, and vigorous jerky movements, suggesting a state of panic or an attempt to escape from the toxic environment. 36 The fish also exhibited suffocation-like behaviours and a loss of reflex, indicating a compromised physiological state. Davies et al. 29 state that the behaviour of organisms is the outcome of various biochemical and physiological processes. When exposed to aqueous surfactants, there is a possibility that the organism's physiological and biochemical functions could be disrupted, resulting in noticeable alterations in behaviour and, ultimately, dose-dependent mortality. 37 However, Kunz et al. 38 stated that faster operculum and tail beat movements, along with spiralling behaviour, were observed. These abnormal swimming patterns can be attributed to the effect of the surfactants on the fish's neuromuscular coordination and motor control.
The observed behavioral responses varied significantly (P < 0.05) with an increase in surfactant concentration and exposure duration, compared to the control group. This demonstrates a dose-dependent relationship, indicating that higher concentrations of surfactants have a more pronounced impact on fish behaviour. 39 Overall, the observed behavioural changes in Guinean tilapia exposed to different concentrations of aqueous surfactants indicate a progressive deterioration of normal swimming patterns, increased stress levels, and compromised physiological functioning. According to Davies and Uedeme-Naa, 20 These behavioural responses serve as indicators of the toxic effects of surfactants on fish and highlight the need to assess and minimize the environmental impacts of surfactant exposure on aquatic organisms.
Opercular beat frequency (OBF)
The observed variations in opercular beat frequency (OBF) at different concentrations of surfactant exposure in T. guineensis can be attributed to the toxic effects of the surfactants on the fish's respiratory and physiological systems. The significant variation (P < 0.05) in treatment means compared to the control group suggests that the surfactants have a dose-dependent impact on the fish's opercular beat frequency. The control group, which was not exposed to any surfactant, exhibited a relatively stable OBF of 52.0 ± 0.6 beats per minute. This value represents the baseline opercular beat frequency for T. guineensis under normal conditions. However, when exposed to increasing concentrations of surfactants, the fish exhibited a significant increase in opercular beat frequency. The maximum OBF value of 81.6 ± 0.9 beats per minute was observed at the highest concentration of 15.0 ml/L. The initial erratic movements, rapid opercular beat, and increased mucus secretions observed in the test fish indicate a stress response to the presence of surfactants. 40 Daniels et al. 41 reported that surfactants can disrupt the normal functioning of fish gills, which are responsible for gas exchange and respiration.
The increased opercular beat frequency is often a compensatory mechanism in fish to overcome impaired oxygen uptake caused by chemical stressors. 42 The fish may increase their respiratory rate in an attempt to obtain sufficient oxygen to meet their metabolic demands. The observed increase in opercular beat frequency per minute from 24 to 72 h of exposure suggests an adaptation response of T. guineensis to the presence of surfactants. However, this adaptive mechanism seems to be short-lived as the opercular beat frequency decreased with prolonged exposure at 96 h before eventual death. This agrees with43,44 who stated that the decrease in OBF rate with increasing exposure time indicates that the fish's respiratory system becomes progressively compromised, leading to reduced oxygen uptake and ultimately mortality. The significant decrease (P < 0.05) in OBF rate with increasing exposure time compared to the control group further supports the toxic effects of the surfactants on T. guineensis. The declining opercular beat frequency indicates a deteriorating respiratory function, which can be attributed to the accumulation of surfactant-induced damage to the gills and associated respiratory structures. 33
Overall, the observed variations in opercular beat frequency at different surfactant concentrations and exposure times provide evidence of the detrimental effects of surfactants on T. guineensis. The increased opercular beat frequency initially indicates a compensatory response to maintain adequate oxygen uptake, while the subsequent decrease reflects the fish's inability to withstand prolonged exposure to the toxicants.14,45 These findings highlight the sensitivity of T. guineensis to surfactant-induced respiratory stress and emphasize the importance of considering behavioural and physiological alterations as indicators of ecotoxicological impacts on aquatic organisms.
Tail beat frequency (TBF)
The observed differences in tail beat frequency (TBF) at different concentrations of surfactant exposure in T. guineensis can be attributed to the toxic effects of the surfactants on the fish's neurological and physiological functions. The statistical significance (P < 0.05) between the control group and the groups exposed to different concentrations of surfactants suggests that the surfactants have a dose-dependent impact on the fish's tail beat frequency. The control group, which was not exposed to any surfactant, exhibited a TBF of 34.0 ± 0.6 beats per minute. This value represents the baseline tail beat frequency for T. guineensis under normal conditions. However, exposure to increasing concentrations of surfactants resulted in a significant increase in tail beat frequency. The highest TBF value of 53.7 ± 0.3 beats per minute was observed at the concentration of 10.0 ml/l, while the concentration of 15.0 ml/l exhibited a TBF of 52.3 ± 0.9 beats per minute. The progressive indication of stress observed before death, such as increased tail beat frequency and subsequent decline, indicates the fish's response to the presence of surfactants. These alterations in TBF can be attributed to the neurotoxic effects of the surfactants, which may affect the fish's central nervous system and disrupt motor coordination. 20 The initial increase in TBF from 24 to 48 h suggests a compensatory response of T. guineensis to the chemical stressor. 2 The fish may exhibit hyperactivity or increased swimming behaviour as an adaptive strategy to cope with the adverse effects of the surfactants. 40
However, as exposure time progresses, the fish's ability to maintain the increased tail beat frequency diminishes. The decline in TBF from 72 to 96 h before death indicates a compromised neurological function and a progressive loss of motor control. 46 According to Athira and Jaya 6 stated that a decline in tail beat frequency is likely associated with the cumulative effects of surfactant exposure on the fish's nervous system, leading to impairment of muscular coordination and ultimately mortality. The significant difference (P < 0.05) between the TBF rates of the exposed groups and the control group further supports the toxic effects of surfactants on T. guineensis. The alterations in tail beat frequency reflect the fish's sensitivity to the neurotoxic properties of the surfactants, which can disrupt normal physiological processes and compromise locomotor activity. 46
Histological changes in gill
The structural aberrations observed in the gills of T. guineensis exposed to different concentrations of surfactants can be attributed to the toxic effects of the surfactants on the gill tissue. Surfactants are known to disrupt the integrity of cell membranes and interfere with cellular processes, which can result in various pathological changes in the gills.46,47 Exposure to a 2.5 ml/L concentration of surfactants resulted in lesions, pigment deposition, necrosis, cellular degeneration, malignancy, and inflammation. 45 Hinton et al., 24 reported that the lesions indicate damage to the gill tissue, while pigment deposition may be a response to the presence of toxic substances. 48 Necrosis refers to the death of cells, which could be caused by the toxicity of surfactants. 16 Cellular degeneration implies that the cells in the gills are undergoing structural changes due to the toxic insult. Malignancy and inflammation suggest the presence of abnormal cell growth and inflammatory response, respectively. 49
The 5.0 ml/L concentration exposure led to similar observations of lesions, pigment deposition, malignancy and inflammation. However, necrosis is not absent in the gill. The absence of necrosis may indicate that the concentration of surfactants at 5.0 ml/L was not sufficient to cause cell death in the gills. 50 The 7.5 ml/L concentration exposure showed lesions, pigment deposition, necrosis, and inflammation. According to Reddy and Rawat, 17 reported that necrosis and inflammation suggest a more severe toxic effect on the gill tissue compared to the previous plate. The higher concentration of surfactants likely increased the damage to the cells, resulting in necrosis and a stronger inflammatory response.
However, the gills exposed to 10.0 ml/L concentration exhibited inflammation, pigment deposition, malignancy, and cellular degeneration. The absence of lesions and necrosis in this plate suggests that the toxic effects primarily affected the cellular level, leading to abnormal cell growth (malignancy) and structural changes such as cellular degeneration. 51 Inflammation indicates an immune response triggered by the presence of toxic substances. The 15.0 ml/L concentration exposure resulted in pigment deposition, malignancy, and cellular degeneration. This agrees with Sadeghi et al. 51 who reported that the absence of lesions suggests that the damage may be more focused on the cellular level, causing abnormal cell growth and structural changes.
In several regions of the gill, the cartilaginous rod at the core of the primary lamellae showed disruption. The observed changes included mild to severe epithelial proliferation, hyperplasia, lamellae fusion, and gill filament malignancy, which align with the findings of Oladunjoye et al. 48 in their study on petroleum wastewater exposure. The present study revealed more severe damage to the gills compared to the earlier report by Doherty et al., 52 who investigated the effects of Paraquat Dichloride on Clarias gariepinus tissues. These results are consistent with the observations made by Hassan et al. 45 Similarly, Yaji et al. 53 documented significant effects of Aronil on the gills of Oreochromis niloticus, such as epithelial lifting, edema, a fusion of secondary lamellae, and thickening of the primary lamellar epithelium.
Mucous cells contain glycoprotein-based polyanions called mucins, which can effectively trap toxic substances and aid in preventing their entry into the gill epithelium, as described by Ogueji et al. 54 Due to the epithelial lifting and hyperplasia impeding the rate of gaseous exchange, the natural mechanism to increase the pollutant-blood diffusion distance, excessive mucus secretion, and necrosis occurred, leading to oxygen deficiency, as explained by Okey et al. 55 Muñoz et al. 56 previously reported filament cell proliferation, lamellar fusion, lamellar cell hyperplasia, and epithelial lifting in juvenile silver catfish (Rhamdia quelen) exposed to sublethal lead concentrations. Abalaka et al. 57 also observed similar mucous cell proliferation and hypertrophy, widening of the epidermal and dermal layers, and erosion of the epithelial lining in C. gariepinus exposed to Adenium obesum extract.
The gill surfaces come into direct contact with toxic substances present in the water. 58 Exposure to these substances has been shown to cause various structural changes in the respiratory epithelial surface of the gills. 59 The gill lesions observed in this study were similar to those reported by Simonato et al. 58 and Akaishi et al. 60 in fish exposed to acute concentrations of crude oil. In this study, the fish exposed to the highest concentration of the chemical exhibited severe degenerative lesions such as vacuolization, lifting of the epithelia, and shedding of the epithelial lining. There was also a proliferative response in the form of hypertrophy. A similar dose-response relationship with degenerative lesions was observed by Olufayo and Alade, 49 in Heterobranchus bidorsalis exposed to acute toxicity of Cypermethrin. These changes in the gill structure are adaptive and necessary to reduce the absorption rate of toxic substances. Generally, the observed structural aberrations in the gills of T. guineensis exposed to different concentrations of surfactants indicate varying degrees of toxic effects on the gill tissue. Higher concentrations of surfactants generally led to more severe damage, including lesions, necrosis, and stronger inflammatory responses. The specific effects observed, such as pigment deposition, malignancy, and cellular degeneration, may vary depending on the concentration and nature of the surfactants used.
Histological changes in liver
The liver serves as a crucial organ for various metabolic pathways and serves as an indicator of the toxic effects of chemicals, providing valuable insights into their toxicity and mode of action, as noted by Sauer et al. 61 It is also responsible for detoxification processes, and the observed changes in the liver in this study may be attributed to the degenerative necrotic condition commonly associated with toxicity effects, as explained by Li et al. 62 As stated by Grynevych et al., 19 the liver, owing to its role, position, and blood circulation, is particularly vulnerable to waterborne contaminants, resulting in it being the organ most impacted by these substances. The liver is an important organ responsible for various metabolic processes, and its sensitivity to toxic substances makes it a common target for assessing the effects of chemical exposure. 25
The control group (0.0 ml/L) exhibited a normal cellular pattern, including a normal central vein and the space of Disse. No lesions, necrosis, pigments, malignancy, inflammation, or inclusion bodies were seen, indicating that the liver tissue was healthy and unaffected. 24 Exposure to a 2.5 ml/L concentration of surfactants resulted in lesions, inflammation, and necrosis. Lesions indicate damage or abnormality in the liver tissue, while inflammation suggests an immune response to the presence of surfactants. 63 Necrosis refers to the death of liver cells, which may have occurred due to the toxic effects of the surfactants. The fish exposure to 5.0 ml/L concentration showed lesions and necrosis in the liver tissue. Similar to the 2.5 ml/L concentration, the presence of lesions indicates damage or abnormality, while necrosis suggests cell death caused by the toxic effects of the surfactants 64 (Roth et al., 2021). While the liver of T. guineensis exposed to 7.5 ml/L concentration exhibited necrosis, inflammation, and cellular degeneration. Roth et al. 64 suggest that the presence of Necrosis indicates cell death, inflammation indicates an immune response and cellular degeneration implies structural changes and damage to liver cells. The higher concentration of surfactants likely intensified the toxic effects on the liver tissue, resulting in more severe structural aberrations. 65
On the other hand, the liver exposed to 10.0 ml/L concentration showed cellular degeneration, necrosis, inflammation, and lesions. Cellular degeneration refers to structural changes in liver cells, necrosis indicates cell death, inflammation suggests an immune response, and the presence of lesions further indicates damage or abnormality in the liver tissue.66,67 The higher concentration of surfactants likely intensified the toxic effects, leading to multiple structural aberrations.46,67 While, the 15.0 ml/L concentration exposure resulted in necrosis, pigments, and inflammation. Necrosis indicates cell death, pigments may be a response to the presence of toxic substances, and inflammation suggests an immune response,.65,66 The high concentration of surfactants likely caused severe damage to liver cells, leading to cell death, the deposition of pigments, and inflammatory response.
The structural aberrations observed in the liver of T. guineensis exposed to different concentrations of surfactants can be attributed to the toxic effects of the surfactants on the liver tissue. Ladipo et al. 68 observed similar results when C. gariepinus was exposed to the acute toxicity of paraquat dichloride for 96 h. Ferguson, 59 suggested that osmoregulatory dysfunction resulting from toxin-mediated diseases could be responsible for hepatocyte necrosis. The duration of exposure to a toxic substance determines the severity of organ damage in fish. 37
During exposure to toxicants in aquatic organisms, the liver often accumulates lesions and undergoes histopathological alterations, as reported by Kolawole and Iyiola. 69 Several researchers have also observed similar effects and have utilized liver histopathology as reliable biomarkers in their studies on contaminants, as documented by Sadeghi et al. 51 and Stoyanova et al. 70 This is because the liver plays a significant role in fish physiology, contributing to anabolism (protein, lipid, carbohydrate synthesis) and catabolism (glycogenolysis, detoxification), and also serves as a storage site for various substances, particularly glycogen, as highlighted by Padrilah et al. 50 Such studies have been incorporated into national surveys and monitoring programs concerning aquatic environments and biological impacts in many developed countries, as described by Sousa et al. 71 In their study on copper accumulation, Padrilah et al. 50 investigated the toxicity effects of fish liver histopathology and observed the degeneration of numerous hepatocytes. Inflammatory changes in liver alterations are considered significant indicators of pollutant exposure, providing valuable information on the overall health and condition of fish, as emphasized by Agbohessi et al. 72
In summary, the observed structural aberrations in the liver of T. guineensis exposed to different concentrations of surfactants indicate varying degrees of toxic effects on the liver tissue. Higher concentrations of surfactants generally led to more severe damage, including lesions, necrosis, inflammation, and cellular degeneration. The specific effects observed, such as pigments and inclusion bodies, may vary depending on the concentration and nature of the surfactants used. One notable limitation of this study is the absence of sample size and power analysis. Future research endeavours should prioritize the incorporation of rigorous sample size and power calculations to strengthen the validity of conclusions drawn from experimental data.
Conclusion
This study evaluates the ecotoxicity of oilfield-based emulsifiers on T. guineensis using histopathology and behavioural alterations. The research reveals behavioural differences in exposed fish, indicating toxic effects on the nervous system and overall physiological well-being. The behavioural responses varied with concentration and exposure duration, demonstrating a dose-dependent relationship. The study also found significant effects on the fish's respiratory and physiological systems, as reflected in variations in opercular beat frequency. Exposure initially increased opercular beat frequency, suggesting a compensatory response, but prolonged exposure led to respiratory deterioration. Neurotoxic effects were observed through changes in tail beat frequency, indicating disrupted motor coordination and compromised neurological function. The toxic effects of the chemical on gill and liver tissues resulted in lesions, necrosis, inflammation, and degeneration, with severity increasing with higher concentrations. Future research should focus on investigating the specific mechanisms and long-term consequences of surfactant toxicity on fish genetic composition, populations, and ecosystems.
Footnotes
Acknowledgements
Research funding and Article Processing Charge (APC) for this article were supported in part by funds provided by USDA-NIFA Sustainable Agriculture Systems, Grant No. 2019-69012-29905 to GTI and ZAK. Title of the project: Empowering US Broiler Production for Transformation and Sustainability USDA-NIFA (Sustainable Agriculture Systems): No. 2019-69012-29905. MNA also partly supported the APC.
Author contribution statement
All authors contributed to the study's conception and design. Material preparation, data collection and analysis were performed by Davies Ibienebo Chris, Okechukwu Kenneth Wokeh, Guillermo Téllez-Isaías, Zulhisyam Abdul Kari, Mohamad Nor Azra. The first draft of the manuscript was written by Davies Ibienebo Chris, Okechukwu Kenneth Wokeh and Guillermo Téllez-Isaías, Zulhisyam Abdul Kari, Mohamad Nor Azra revised the subsequent version of the manuscript and the revision. All authors read and approved the final manuscript
Consent to participate
Not applicable
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript. Ethical Number: UPH/R&D/REC/04.
Ethical approval
Ethical approval was obtained from the Office of Research and Development (Research Ethics Committee) at the University of Port Harcourt after due deliberation and my research proposal was approved, according to the references number of UPH/R&D/REC/04, generated by the 59th meeting of research committee from University of Port Harcourt, Nigeria.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship,and/or publication of this article: The APC for this article was supported by funds provided by USDA-NIFA Sustainable Agriculture Systems (Grant No. 2019-69012-29905; title of project: Empowering US Broiler Production for Transformation and Sustainability USDA-NIFA (Sustainable Agriculture Systems): No. 2019-69012-29905) to Prof. Dr. Guillermo Téllez-Isaías (GTI) at Department of Poultry science, University of Arkansas, USA.
Author biographies
Davies Ibienebo Chris is an Aquatic Ecotoxicologist with a PhD in Fisheries and Hydrobiology, an MSc in fisheries from the University of Port Harcourt. His expertise lies in genotoxicity, biomonitoring environmental chemistry in the aquatic environment, and environmental sustainability. With six years of experience at the Department of Fisheries, he has experience in designing and carrying out scientific research and writing technical reports. He has personal and written communication skills as well as oral presentation skills with more than 60 senior authored and 48 co-authored fisheries research articles in scientific journals indexing in Scopus/WOS. Davies is a scholar of the World Bank African Center of Excellence, Center for Oilfield Chemical Research, University of Port Harcourt, and a Certified Environmental Scientist with the National Registry of Environmental Professionals (NREP) in the USA. He is currently the Secretary of the Fisheries Society of Nigeria (FISON) Rivers State Chapter. Davies is passionate about lecturing, career counselling, research programs planning, public health importance, environmental pollutants, and the health impact of industrial, agricultural, and domestic waste on the aquatic environment. His recent research focused on the environmental impacts of artisanal crude oil refining effluence on the aquatic environment and the human health risk associated with it. He has attended several national and international conferences and has conducted interdisciplinary and transdisciplinary studies in areas such as genotoxicity of aquatic organisms, aquaculture, environmental impacts like industrial wastes and plastic waste in landfills and waterways in the Niger Delta. Furthermore, he believes that an informed, open, just, and reporting culture, combined with comprehensive planning and implementation of SMART objectives, is crucial for achieving excellence in the field of aquatic ecotoxicology.
Okechukwu Kenneth Wokeh holds an MSc and a PhD in hydrobiology and fisheries from the University of Port Harcourt, Nigeria. He is a research assistant at the Department of Animal and Environmental Biology of the same university, with a lecturing appointment in the Department of Fisheries and Aquaculture, Federal University of Technology Owerri, Nigeria. He has broad research experience in water quality management, pollution control, fish nutrition and aquaculture. He is a professional member of International Society of Comparative Education, Science and Technology, Association of Environmental Impact Assessment of Nigeria.
Guillermo Téllez-Isaías was born in Mexico City, in 1963. He received his Doctor in Veterinary Medicine degree in 1986 and his Master in Science degree in Veterinary Sciences in 1989 from the National Autonomous University of Mexico (UNAM), and his PhD from Texas A & M University. He worked as a Full Professor at UNAM for 16 years, 8 as head of the Avian Medicine Department at the College of Veterinary Medicine. Tellez was President of the National Poultry Science Association or Mexico during 1997–1998 and is a member of the Mexican Veterinary Academy and the Mexican National Research System. In 2001, Tellez worked during his sabbatical as Visiting Scientist at the Center of Excellence in Poultry Science of the University of Arkansas, where he is still working as a Research Professor. His research is focused on the advantages of the poultry gastrointestinal model to evaluate the beneficial effects of functional foods to enhance intestinal health and disease resistance.
Zulhisyam Abdul Kari is a senior lecturer at the Faculty of Agro-Based Industry, Universiti Malaysia Kelantan. At the moment, he is the head of Advanced Livestock and Aquaculture Research Group – ALAReG under the Faculty of Agro-Based Industry. His research interest is on animal nutrition and molecular study. He has extensive experience in writing and publishing in high-impact journals. He has published over 100 high-impact articles indexing in Scopus/WOS. As a senior lecturer, he has more than five innovation products that have been patented. He also has won and got recognition for the 40 products that he innovated during the national and international exhibition. His work has become a point of interest for the community as well as a reference for other researchers.
Mohamad Nor Azra was born in Port Dickson, Negri Sembilan, in 1987. Azra received his Diploma, BSc, MSc and PhD degrees in fisheries and aquaculture environment from the Universiti Malaysia Terengganu, Malaysia. He was sponsored by the Malaysian Public Service Department Scholarship, Scholarship Talent Attraction and Retention (STAR), and MyBrain15 (MyPhD & MyMaster) Postgraduate Scholarship during his journey of study from 2011 to 2018. His current research interest include the adaptation of aquatic animals towards changing climate, focusing on ecophysiological response and behavioural changes. He is currently the inaugural fellow for Leadership for Climate Resilient Fisheries Fellowship, USA, and Visiting Researcher at National Research and Innovation Agency, Indonesia.

