Abstract
Ganoderma multipileum, a wood decay mushroom, was initially discovered and classified in Taiwan through the analysis of its morphology and the internal transcribed spacer (ITS) region of nuclear ribosomal DNA. In this study, we identified a mushroom associated with the dieback of Delonix regia (Boj. ex Hook.) Raf., a woody ornamental street tree in Vietnam, as Ganoderma multipileum. This classification was based on phylogenetic analysis of ITS, RPB2, and TEF1 sequences, as well as morphology assessment and scanning electron microscope observation of basidiospores. The phylogenetic analysis revealed that the specimens collected in Vietnam formed a monophyletic group of Ganoderma multipileum with a high bootstrap value and posterior probability (100%/1.00). Furthermore, the morphological features consistent with laccate Ganoderma, including a thin pileipellis composed of enlarged and bulbous hyphae, and the basidiomes exhibited two different phenotypes. Notably, scanning electron microscopy of the basidiospores revealed ovoid spores with numerous echinules, providing the first documented evidence of this characteristic for Ganoderma multipileum. This research represents the first recorded instance of Ganoderma multipileum in Vietnam associated with the dieback of Delonix regia.
Keywords
Introduction
The genus Ganoderma belongs to the family Ganodermataceae and has more than 418 species (www.indexfungorum.org, accessed July 30, 2022). Ganoderma is found in many regions around the world in diverse ecosystems and many species have been used as a medicinal herb for thousands of years in many Asian countries. Species in the genus Ganoderma are white-rot fungi that mostly grow on substrates of decayed wood or fibrous materials, where they degrade the lignocellulose in wood. Basal stem rot on oil palm is caused by Ganoderma, resulting in a yield loss of 23.8 fresh fruit brunches/t ha. 1 This has affected millions of farmers in Malaysia, Indonesia, and other countries, which annually produce 17.8 million tons of oil palms worldwide. Ganoderma species have also been documented attacking Acacia species in India, with many Acacia species serving important economic roles, such as growing it for pulp and wood. G. applanatum and G. lucidum have also been reported in Pakistan 2 and India on 144 crop perennials hosts such as Acacia, Arnacardium, Artocarpus, Bamboo, Cocos, Coffea, Dalbergia, Pinus, Quercus, and other species. 3 Stem, butt, and root rot diseases are typical results of Ganoderma attacks. Before a stressed tree dies from such an infection, its symptoms may include branch dieback and the loss of foliage. The main entry points for diseases may be through soil roots, vegetative spread, or introduction by spores through wounds. 4 Among the Ganoderma species, G. multipileum was discovered on a stump of decaying wood for the first time in Taiwan. 5 Since then, G. multipileum has been described based on morphology and phylogeny in subtropical and tropical countries such as China,6–8 India, 9 Pakistan, 10 and Thailand. 11 In previous reports, G. multipileum was described as a white-rot fungus that mainly decays dead softwood such as Sterculia nobilis, Delonix regia, 5 and Pinus merkusii, 11 as well as hardwood plants such as Dalbergia sissoo and Vachellia nilotica. 10
Delonix regia is a flowering plant of the Fabaceae family and is widely grown in Vietnam and other tropical countries. Various parts of this plant have been used in folk medicine.12,13 A decoction of the bark is used to treat malaria, bloating, and rheumatism, 13 while the leaves can treat lower blood pressure. 14 In addition, phenolic compounds effectively increase antioxidant and antimicrobial activities, which hold potential for functional food and medicinal therapies. 15 In Vietnam, the tree is also cultivated as an ornamental avenue tree along streets or near schools and offices. It is considered a summer symbol and is deeply rooted in Vietnamese culture. Notably, a recent document from Vietnam showed that this broadleaf tree host had Ganoderma colossum reported on a stump as a wood decay mushroom. 16 In addition, tree protection in urban areas is significant because most plants that grow in metropolitan areas are woody. 17 If urbanized trees are damaged by attacking pathogens, they can potentially harm citizens. Therefore, understanding the harmful pathogens of woody plants is necessary to determine and conduct control measures.
In this study, a species of Ganoderma was found to cause dieback of the ornamental plant D. regia in Ho Chi Minh City, Vietnam. Phylogenic and morphological analyses of the mushroom resulted in its classification as G. multipileum. The relation of this mushroom with the dieback of the host plant D. regia was noted.
Materials and methods
Host plant observation
The host plant, D. regia, was a healthy 27-year-old tree in 2018, standing ∼ 17 m tall. It was located at coordinates 10o45'47.28″N and 106o40'55.85″E in District 5, Ho Chi Minh City, Vietnam. The observation of disease symptoms began after the fruiting bodies had emerged on this tree, spanning a period of 40 months from the rainy season of (August–December) 2018 to December 2021.
Mushroom collection and mycelium isolation
Fresh specimens of the mushroom were collected in November 2018 by Nguyen, T.T.T. from the Faculty of Biology and Biotechnology, University of Science. These specimens were designated as voucher specimens and were deposited at the Laboratory of Microbiology, Faculty of Biology and Biotechnology, University of Science, Vietnam National University Ho Chi Minh City, Vietnam. They were given the code VNHCM1805 for identification and reference purposes. To cultivate the mycelium, tissue samples were taken from the fresh fruiting bodies and transferred to potato dextrose agar (PDA) in sterile Petri dishes (manufactured by STRIPLAN, Germany). The dishes were then incubated at a temperature of 25 ± 2 °C. For further studies, the mycelium was stored in 13 × 100 mm tubes (Pyrex, USA) at a temperature of 4 °C.
Morphological characterization
Macroscopic features were observed on field notes and digital images (Canon EOS 7D, made in Japan). Kornerup and Wanscher's color standards
18
were used to aid in identification. Mushroom samples were dried at 50 °C and stored in resealable plastic bags. The samples were cut into small pieces, inserted into a 5% KOH solution, stained with 1% cotton blue and Melzer reagent solution, and observed using a stereomicroscope (Meiji EMZ 127212, Saitama, Japan). Fruiting bodies were observed at 1000 × magnification using an optical microscope (S/N EU1611034 microscope and CMEX-10 PRO USB 3.0 microscope camera, Euromex, Arnhem, the Netherlands). The description of the basidiomata of Ganoderma followed Karst.
19
Dimensions for basidiospores are given using the notation of the form (a) b–c (d). The b–c range contains a minimum of 90% of the measured values. Extreme values (a) and (d) are provided in parentheses.
DNA extraction, PCR, and sequencing
The mycelia were extracted using the phenol:chloroform: isoamyl alcohol technique (Merck, Darmstadt, Germany) with TE buffer (Tris HCl pH 8.0, 1.0 mM EDTA). 20 To obtain only DNA, RNAse A (60 μg) was added after extraction. The total DNA content was determined using NanoVue Plus (GE Healthcare, Gillingham, UK) and stored at −20 °C after purification and concentration. For amplification of the nuclear ribosomal internal transcribed spacers (ITSs; ITS1, 5.8S, ITS2), the primers ITS1F and ITS4 were utilized. Additionally, two protein-coding genes, translation factor 1α (TEF1) and RNA polymerase II's second-largest subunit (RPB2), were amplified using the primer pairs EF1-983F/EF1-2218R and fRPB2-5F/bRPB2-7R2, respectively (Table 1). The PCR components, with a total volume of 25 μL, consisted of 4 μL of sample DNA, 2.5 μL of Taq buffer, 2.5 μL of 2 mM dNTP, 0.125 μL of primers, 0.25 μL of Taq DNA polymerase (Thermo Fisher Scientific, Waltham, MA, USA), 1.68 μL of MgCl2 with optimized buffer, and 13.95 μL of sterile Milli-Q water. The PCR cycling conditions involved an initial denaturation step of 5 min, followed by 35 cycles of denaturation at 95 °C for 30 s, annealing at 55 °C for 30 s, and extension at 72 °C for 1 min for ITS amplification and 2 min for TEF1 and RPB2 amplification. The final elongation step was performed at 72 °C for 5 min. PCR products (2 μL) were electrophoresed on a 2% agarose gel stained with EtBr 21 in Tris-acetate EDTA buffer, and then visualized and photographed under UV light. Subsequently, Sanger sequencing was conducted by Macrogen (Seoul, Korea).
Primers were used in this study.

ITS: internal transcribed spacer; RPB2: RNA polymerase II's second-largest subunit; TEF1: translation factor 1α.
Phylogenetic tree construction
According to the corresponding Sanger sequencing chromatograms, misleading data from the ends of raw sequencing fragments were manually trimmed and assembled into consensus sequences using BioEdit v7.2. All newly obtained sequences have been submitted to GenBank. 27 Results for related genera on studies or searched and downloaded from the GenBank (Table 2) were acquired using BLAST via MAFFT v7.487 28 to align the obtained sequences. The alignments were edited and manually corrected using AliView. 29 The best-fit model was determined by jModelTest v2.1.10 30 based on the corrected Akaike information criterion (AICc). Maximum likelihood (ML) analyses were performed using IQ-TREE v2.0.3 on CIPRES Science Gateway v3.3 31 with 1000 ultrafast bootstrap replicates. For Bayesian analysis, the best-fit nucleotide model, HKY + I + G, was selected by jModelTest v2.1.10, and posterior probabilities (PPs) were determined by Markov chain Monte Carlo (MCMC) using MrBayes v3.2.7a. 32 Three different runs with 1 million generations and four chains were executed until the split deviation frequency value was < 0.01 and sampled every 100th generation. The initial 25% of sample trees were discarded as burn-in. The MCMC runs were checked to reach convergence with all ESS values above 200 by using Tracer v1.7.2. 33 The ML bootstrap values (BSs) ≥ 70% and PP ≥ 0.95 were presented on topologies from ML analyses, respectively. Tomophagus colossus TC-02 was selected as the outgroup for both ML and Bayesian analyses. Phylogenetic trees were visualized and modified using FigTree v.1.4.4.
Genbank accession numbers of Ganoderma species sequences used in the phylogenetic analysis.

*This species is presented in this study.
ITS: internal transcribed spacer; RPB2: RNA polymerase II's second-largest subunit; TEF1: translation factor 1α.
Results
Disease symptoms on the host plant
Ganoderma multipileum VNHCM1805 was initially noticed on the host tree, D. regia, during the rainy season of 2018. The tree exhibited blooming in the summer of May 2019, but it did not flower again in May 2020. Over time, the leaves of the host plant were lost, and the branches began to die back, leading up to May 2021 (Figure 1(a) to (c)). The basidiocarps of the mushroom persisted and were observed on the roots and trunk of the host tree during each rainy season from 2018 to 2021 (Figure 1(d) and (f)). No infections caused by pets or insects were observed on the plant. Furthermore, there was visible damage to the phloem on the side where the basidiocarps were growing, resulting in abnormal bark. The bark displayed signs of sap spillage, which then reversed its flow (Figure 1(g)). Considering the potential risks of extensive damage to nearby houses and residents, the decision was made to cut down the dying tree before 2022.

Delonix regian disease symptoms and the macroscopic structures of G. multipileum VNHCM1805 basidiomes: (a) D. regia tree in May 2019, (b) the same tree in May 2020, (c) the same tree in May 2021, (d) basidiocarps growing at the base of the same living tree, (e) lower surface of a basidiome, (f) young basidiocarp on tree roots, (g) cutting part of the bark from the same living tree where the young basidiocarp was growing, and (h) section of basidiome. Scale bars: d: 20 cm; e to h: 2 cm.
Phylogeny
In this experiment, we conducted phylogenetic analyses using a combined dataset comprising ITS, RPB2, and TEF1 sequences from 36 taxa. The results indicated that these sequences were attributed to the Ganoderma genus and were classified into six distinct clades (Figure 2). Reference sequences from GenBank, obtained from samples collected in the USA, the UK, Czech Republic, France, Italy, Korea, China, Japan, the Netherlands, Thailand, and Vietnam, were included, and they encompassed holotypes of G. lingzhi, G. leucocontextum, and G. multipileum. Tomophagus colossus (Fr.) Murill 1905 (TC-02, Vietnam) served as the outgroup in the taxonomic tree. The dataset consisted of 2176 sites, including gap regions (558 sites for ITS, 946 sites for RPB2, and 672 sites for TEF1). The tree topologies resulting from the maximum likelihood (ML) analysis were consistent with the Bayesian analysis. The best-scoring ML tree and Bayesian analysis are depicted in Figure 2 (and supporting information). Notably, the specimens collected from Vietnam formed a significantly distinct, monophyletic group with the G. multipileum taxon. This group included the holotype (CWN 04670) and other sequences from China (Cui 14373, Dai 9447) and Thailand (MLU 19-2166). The strong support for this clustering was reflected by high ML/PP values of 100% and 1.00, respectively.

Phylogeny of Ganoderma spp. based on data from ITS, RPB2, and TEF1 sequences. Bootstrap values were obtained from maximum likelihood above 70% and posterior probabilities equal to or greater than 0.95 (given above branches). The tree is rooted in Tomophagus colossus TC-02 (Vietnam). New species were collected in Vietnam in bold (black).
Taxonomy
Basidiome: annual, stipitate, laccate, woody. Pileus 15–25 × 7–10 cm up to 2.2 cm thick at the base, flabelliform, upper surface brownish orange (6C8), orange-rooted to reddish brown (8D8) at maturity, weakly to strongly laccate, reniform, dimidiate, pilei expanding, sometimes with pilei growing from the lower pilei or growing together, up to 36 cm, and 54 cm in overall width from the lower pilei, curly and wavy edges; lower surface yellowish white (4A2) to brownish gray at maturity. The young basidiocarp's upper surface is brownish orange (6C8), orange-rooted to reddish brown (8D8), somewhat lacquered, obviously sulcate, rugged and non-rough, margin obtuse or not, white to orange-yellow. Context 0.1–1.8 cm thick, light brown (6D6) to dark brown (6F8), occasionally melanoid in KOH, corky. Hymenophore whitish cream on the surface while young, straw, or light brown (6D6) at maturity. Pores, subcircular to circular, 25–110 μm thickness, 60–80 μm per mm, 60–220 μm in diameter, and 3–5 spores/mm. Stipe 1.5–3.5 cm in length, up to 0.7–3.0 cm in width, sub-cylindrical to cylindrical, strong laccate, reddish brown (9D7), and brownish red (10D6) at maturity (Figure 3).

Microscopic structures of the basidiome G. multipileum VNHCM1805: (a) section of pileus (2 ×), (b) pores in lower surface (4 ×), (c) a pore (40 ×), (d) section of Pileipellis (100 ×), (e and f) hyphae of context (100 ×), (f) basidiospores (100 ×); (g) chlamydospores (100 ×), (h) culture after incubation at 25 ± 2 °C for 7 days. Scale bars: a: 1 cm, b: 1 mm, c: 50 µm, d: 20 µm, and e to g: 10 µm.
Basidiome: The basidiome of the mushroom is annual, stipitate (having a stem), laccate (shiny), and woody in texture. The pileus (cap) measures 15–25 cm in width and 7–10 cm in thickness at the base. It has a flabelliform (fan-shaped) appearance, with the upper surface exhibiting shades of brownish orange (6C8) and becoming reddish brown (8D8) as it matures. The pileus is weakly to strongly laccate (lacquer-like), reniform (kidney-shaped), and dimidiate (having two symmetrical halves). The pilei may expand and sometimes have additional pilei growing from the lower portion, resulting in an overall width of up to 36 and 54 cm. The edges of the pilei are curly and wavy. The lower surface is yellowish-white (4A2) and turns brownish-gray with maturity. When the basidiocarp is young, the upper surface of the pileus is brownish orange (6C8) and reddish brown (8D8), displaying a lacquered appearance. It is also visibly sulcate and has grooves or furrows, and has a rugged, non-rough texture. The margin of the pileus may be obtuse (blunt) or not, and it can range in color from white to orange-yellow.
Context: The context (inner flesh) of the mushroom is 0.1–1.8 cm thick and varies in color from light brown (6D6) to dark brown (6F8), occasionally becoming melanoid (darkening) when treated with potassium hydroxide (KOH). The context has a corky texture.
Hymenophore: The hymenophore (the spore-bearing surface) appears whitish cream when young and turns straw or light brown (6D6) as it matures. The pores on the hymenophore are subcircular to circular in shape, with a thickness of 25–110 μm, 60–80 pores per millimeter, and a diameter of 60–220 μm. The mushroom produces 3–5 spores per millimeter. The stipe (stem) measures 1.5–3.5 cm in length and 0.7–3.0 cm in width. It is sub-cylindrical to cylindrical in shape and has a strong laccate appearance. The stipe color ranges from reddish brown (9D7) to brownish red (10D6) as it matures (Figure 3).
Hyphal structure: The hyphal structure of the mushroom consists of generative hyphae, which are trimetric in shape, colored, and measure 2.0–5.2 μm in diameter (n = 30). These hyphae have thin walls. The skeletal hyphae, on the other hand, are broader, measuring 4.3–7.1 μm (n = 30). They are yellowish brown to reddish brown in potassium hydroxide (KOH) solution. The binding hyphae are few in number and measure 1.2–2.0 μm in diameter (n = 30). They have thick walls and a highly branched structure.
Basidiospores: The basidiospores of the mushroom are ovoid in shape, mostly truncated, and possess double walls. They range in color from brownish orange (7C4) to brown (7E5). The spores measure (7.3) 8.0–11.5 (12.2) × (5.3) 5.5–7.8 (8.3) μm, with an average length (Lm) of 10.6 μm and an average width (Wm) of 6.8 μm (n = 50). The spores have an average Q-value (Qavg) of 1.79 ± 0.28. In the absence of a myxosporium, the spores measure (6.6) 7.3–10.9 (11.3) × (4.9) 5.1–7.2 (7.7) μm, with an average length (Lm) of 8.7 μm and an average width (Wm) of 6.2 μm (n = 50). These spores have an average Q-value (Qavg) of 1.47 ± 0.34. The exosporium (outer wall) is hyaline, while the endosporium (inter wall) displays conspicuous echinulae and has a fine dark brown color (7F). The eursporium is also present (Figure 4).
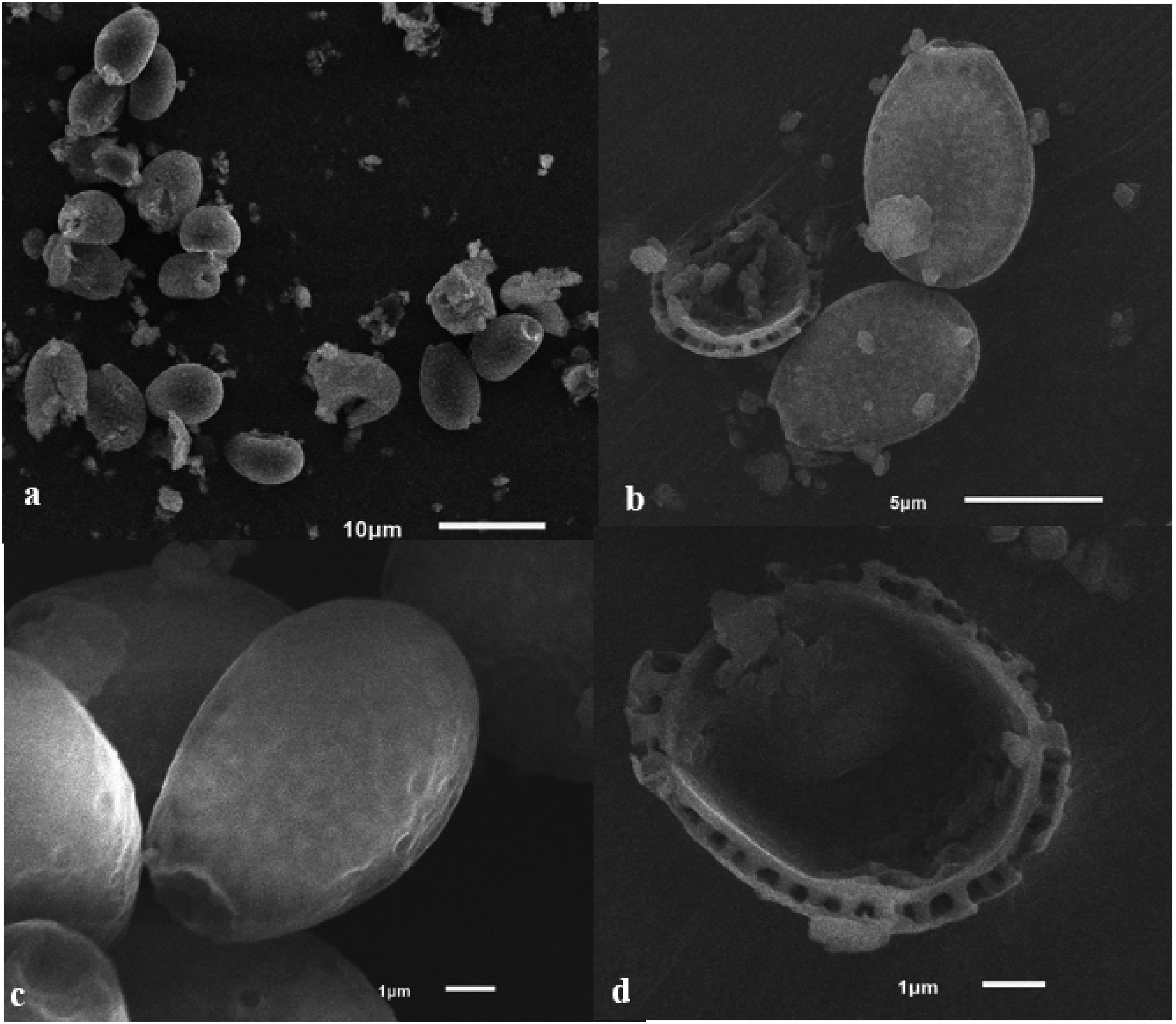
Basidiospores of Ganoderma multipileum VNHCM1805 observed via a scanning electron microscope (SEM): (a) view at 1500 ×, (b) view of exosporium and endosporium at 5000 ×, (c) view of exosporium at 10,000 ×, and (d) detail in endospore ornamentation at 10,000 ×.
Pileipellis: The pileipellis (surface layer of the cap) is cutis in structure and consists of clavate cells measuring 13–42 μm in length and 6.0–13.6 μm in width. These cells exhibit dextrinoid to mildly or severely amyloid characteristics.
Colonies: The colonies of the mushroom exhibit the mycelia are white (4A1) when young and turn pale yellowish white (4A2) to grayish yellow (4B6) after seven days of growth on the PDA medium at a temperature of 25 ± 2 °C. The colonies are round in shape and display growth in a radiating pattern, with the center being thicker and gradually thinning outward. The hyphae are hyaline (transparent) and possess clamp connections, indicating a specific structure at the point of hyphal fusion. The hyphae are also branched. After 5–7 days of growth, chlamydospores become visible. These chlamydospores are club-shaped and have thick walls. They stain with 1% cotton blue and measure 5.5–8.5 × 9.0 × 11.0 μm in size.
Discussion
The genus Ganoderma and the species G. lucidum were initially described in 1881 by Finnish mycologist Karsten. 19 However, the taxonomy of the G. lucidum complex has been challenging due to its wide distribution across various regions, leading to morphological variability. Recent advancements in DNA sequence analysis have provided effective tools for taxonomists to combine data. In 1950, Ding Hou et al. discovered G. multipileum in Taiwan and classified it as a new species within the G. lucidum complex. The nrDNA of this species was subsequently sequenced by Wang et al. 5 In addition, Zhou et al. 8 sequenced the RPB2 and TEF1 genes of this strain. Phylogenetic analysis based on these sequenced genes revealed that they clustered together with the holotype and other strains of G. multipileum. The phylogenetic tree also demonstrated that the relationship between G. multipileum and G. tropicum was closer than with the G. lucidum complex, supporting that G. multipileum belongs to the laccate species of Ganoderma. Wang et al. 5 further suggested that G. multipileum exhibits two phenotypes of fruiting bodies based on morphological features, including those growing from the stem with some stipes and mushroom caps growing together, as well as from lower pilei. The specimens collected in Vietnam displayed both phenotypes of sporocarps when growing on a living royal poinciana tree. In contrast, specimens from Thailand and Pakistan exhibited multiple-pileate sporocarps and were found on decayed hardwoods.10,11 The basidiospores of Vietnamese G. multipileum had typical oval shapes similar to the holotype of G. multipileum, as confirmed by SEM photos for the first time. The basidiospores displayed numerous echinules, consistent with the descriptions by Wang et al. in their drawings. G. multipileum has been described and illustrated in four countries and territories, including Taiwan, China, Thailand, and Pakistan. Previous research has shown its association with the decay of various wood types, including gymnosperms (Merkus pine, Pinus merkusii) and angiosperms (Sterculia nobilis—Chinese chestnut, Dalbergia sissoo—North Indian rosewood, Vachellia nilotica—gum arabic tree). Interestingly, most host plants of G. multipileum from the angiosperm group belong to the Fabaceae family, including D. regia, D. sissoo, and V. nilotica. According to the literature, Ganoderma spp. are predominantly found in the vicinity of tree stumps and roots, and this genus is known as one of the most prevalent plant pathogens.42–45 Species within this genus are categorized as white-rot fungi and possess enzymes capable of degrading lignin and cellulose. These enzymatic properties enable Ganoderma spp. to decompose as much as 99% of lignin in various types of wood. 46 Consequently, Ganoderma species typically parasitize older trees and rarely affect younger ones. The infection of trees occurs when airborne fungal spores enter the bark through open wounds in the trunk and roots. 43
As a result of infection, the xylem cells in the affected tree become necrotic, impeding the transport of water and minerals to the leaves and other parts of the plant. 47 Among the Ganoderma species, G. adspersum, G. applanatum, G. resinaceum, and G. pfeifferi are known to be highly detrimental fungi in urban landscapes in Europe, 48 while G. boninense primarily affects oil palm plantations in Southeast Asia.1,49 In the United States, pathogens affecting landscape trees include G. applanatum, G. boninense, the G. lucidum complex, G. sessile, and G. zonatum.4,42,50 In this study, G. multipileum was observed and collected from D. regia, an old tree aged 27 years, which served as a street ornamental plant in the metropolitan area of Ho Chi Minh City, Vietnam. The region has a dense and high-density population. 51 The symptoms of tree dieback included the lack of flowering, the absence of new branches after the spread of fungal growth on the base and root, leaf shedding, and gradual withering of branches. The process of decline began with the drying of twigs, followed by branches, limbs, and eventually the trunk. At the site where the fungus grew, there was bleeding sap that turned black, leading to symptoms of root rot, root dryness, and tree death. No signs of pest or insect attacks were observed on the plant, while neighboring trees thrived and grew well. This suggests that factors such as soil layers or nutrient supply did not significantly impact the plant's health.
In the limitation of this study, it should be noted that experiments following Koch's postulates, which involve infecting mycelia or basidiospores on another royal poinciana of the same age, observing the infection, and obtaining the mycelia, were not conducted. These experiments usually require a minimum of three years to complete. However, in this study, the growth of the fungus was observed concurrently with the plant's dieback symptoms over a period of 40 months. It is also important to consider previous studies that have reported on the harmful effects of Ganoderma spp. on landscape plants.
Conclusion
In conclusion, this study presents a comprehensive examination of the morphology, phylogenetic analysis based on ITS, RPB2, and TEF1 sequences, and assessment of basidiospores using scanning electron microscopy for G. multipileum. The collected specimen significantly clustered together and formed a monophyletic group with other G. multipileum taxon, including the holotype, with strong support from ML/PP values. These findings also highlight the distinctiveness of G. multipileum compared to related species worldwide. Importantly, this research unveils the first documented occurrence of G. multipileum in Vietnam. The study establishes a clear association between the presence of these fungi and disease symptoms observed in the host plant Delonix regia.
Footnotes
Acknowledgements
The authors give thanks to the Center for Bioscience and Biotechnology, University of Science, Vietnam National University Ho Chi Minh City for technical support. The preprint of the manuscript was submitted in Research Square with DOI:https://doi.org/10.21203/rs.3.rs-2421198/v1.
Authors contribution
All authors contributed to the study’s conception and design. Material preparation and description were performed by TTT Nguyen, HD Nguyen, and AT Bui. Fungal remains were analyzed and interpreted by TTT Nguyen, KHT Pham, and KTP Van. The manuscript draft prepared by TTT Nguyen was revised and finalized by MH Tran and LT Tran. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Trang Thi Thu Nguyen was funded by the Vingroup Joint Stock Company and supported by the PhD Scholarship Program of the Vingroup Innovation Foundation (VINIF), Vingroup Big Data Institute (VINBIGDATA) (grant number: code VINIF.2020.TS.68).
Data availability
The data generated during the current study are available from the corresponding author upon reasonable request.
