Abstract
Lightweight cementitious composite (LCC) produced by incorporating lightweight silica aerogel was explored in this study. Silica aerogel was incorporated as 60% replacement of fine aggregate (sand/crushed glass) in producing the LCC. The effect of aerogel on the drying shrinkage and alkali-silica expansion of LCC was evaluated and compared with those of lightweight expanded perlite aggregate. At the density of 1600 ± 100 kg/m3, the aerogel/ expanded perlite LCC had attained compressive strength of about 17/24 MPa and 22/26 MPa in mixtures with sand and crushed glass as a fine aggregate, respectively. The inclusion of aerogel and expanded perlite increased the drying shrinkage. The drying shrinkage of aerogel LCC was up to about 3 times of the control mixtures. Although the presence of aerogel and expanded perlite could reduce the alkali-silica expansion when partially replacing crushed glass, the aerogel-glass LCC still recorded expansion exceeding the maximum limit of 0.10% at 14 days. However, when 15% cement was replaced with fly ash and granulated blast furnace slag, the alkali-silica expansion was reduced to 0.03% and 0.10%, respectively. Microstructural observations also revealed that the aerogel with fly ash can help in reducing the alkali-silica expansion in mixes containing the reactive crushed glass aggregate.
Keywords
Introduction
Lightweight cementitious composite (LCC) is a type of cement-based material produced with the introduction of lightweight fines. In contrast to conventional foamed concrete where the density is reduced by introduction of air voids, the density of LCC is lowered by using lightweight fines such as glass microsphere and cenosphere. These lightweight fines usually have high number of internal voids encapsulated by a strong outer shell, which imparts certain degree of crushing strength to the lightweight fines. As a result, LCC typically has good specific strength (strength-to-weight ratio) compared to lightweight concrete such as foamed concrete, especially at very low density.
A recent development in the utilization of lightweight fines in cement-based materials is the use of silica aerogel. Aerogel is a nano-porous solid having unique characteristics such as low density (3–350 kg/m3),1,2 low thermal conductivity (0.001–0.002 W/m.K),1,3 high porosity (80–99%),1,4 and high surface area (500–1200 m2/g).1,2 Silica aerogel is produced by sol–gel process, using silicon alkoxides precursors and a special drying process (supercritical or ambient pressure drying). It has been utilized in different available forms (powder/granules) as lightweight fines in mortar, 2 concrete, 5 and LCC.6–8 Encouraging results have been reported by researchers, particularly in terms of the insulation properties of the produced cement-based materials. The addition of highly porous aerogel can significantly improve the insulation properties,1,9 for instance,10,11 when 50 vol.% of cement was replaced by aerogel, the thermal conductivity reduced from 0.36–0.08 W/m.K. 12 Similarly, 85% reduction in the thermal conductivity was absorbed when 60% fine aggregates were replaced with aerogel.13,14 Even a small amount of aerogel as an additive can result in a significant reduction of thermal conductivity. The thermal conductivity was reduced by up to 35% when aerogel was incorporated up to 6 wt.% of the binder.15–17 Furthermore, the addition of aerogel up to 80% caused a reduction of thermal conductivity by up to 95% (0.8–0.04 W/m.K).6,18,19
Nevertheless, till date, the majority of the investigation focuses only on assessing the mechanical strength and insulating properties of the lightweight cement-based materials incorporating aerogel. Other characteristics such as drying shrinkage and alkali-silica reactivity (ASR), which may affect durability of the materials, have not been explored. For instance, the drying shrinkage of cement-based material containing aerogel requires attention, considering that lightweight cement-based materials typically experience higher shrinkage, which can be detrimental if excessive. The drying shrinkage of lightweight foamed concrete and lightweight aggregate concrete were reported to be about 0.1–0.35% of the total volume of the hardened concrete matrix, 20 and about 4–10 times higher than normal concrete.20,21
Another behaviour that is barely investigated is the expansion due to potential ASR when using lightweight fines in LCC. It was reported that conventional lightweight aggregates and lightweight fines such as microsphere are not potentially alkali-silica reactive and can instead reduce the expansion in the presence of other reactive aggregate. 22 However, such observation is unknown yet for the case of silica aerogel. In addition, silica aerogel has large interface with cement paste due to high surface area and may be reactive with the alkaline pore solution. 6 Hence, the ASR of the aerogel in LCC requires further investigation.
Therefore, this study aims to assess the dying shrinkage and alkali-silica expansion of LCC containing aerogel as the lightweight fines. To better understand the ASR of aerogel, another set of LCC was prepared using reactive crushed glass aggregate instead of normal mining sand as the fine aggregate. Additionally, to facilitate comparison with aerogel LCC, a separate series of LCC was prepared with a commonly and commercially used lightweight expanded perlite fine aggregate.
Materials and methods
Materials
For the binder, Type I ordinary Portland cement (specific gravity: 3.15) was used. Mining sand (specific gravity: 2.67) and crushed glass (specific gravity: 2.79) were used as a fine aggregate to prepare two separate sets of LCC. The crushed glass was processed from waste brown beverage glass bottle obtained from a local glass recycling facility. The maximum size of the mining sand and crushed glass was 4.75 mm, while the particle size distribution is given in Figure 1. Both mining sand and crushed glass had a similar fineness modulus of 2.99 and 2.97, respectively.

Particle size distribution of mining sand and crushed glass.
To produce the LCC, the fine aggregates were partially replaced with silica aerogel (specific gravity: 0.14) (Figure 2). A commercially available silica aerogel powder provided by JIOS Aerogel company was used, where the physical properties of silica aerogel (as provided by manufacturer) are given in Table 1. Lightweight expanded perlite (EP) aggregate (Figure 2) was also utilized partial fine aggregate replacement for comparison. For the EP, the particle size ranges from 0.1–2.5 mm, where the saturated surface-dry and bulk specific gravity was 0.7 and 0.35, respectively. The major oxide composition of EP is shown in Table 2. In addition, to control the potential ASR expansion, commercially available supplementary cementitious materials (SCM) such as class-F fly ash and ground granulated blast furnace slag (GGBS) were utilized in the aerogel-glass LCC. The chemical composition and physical properties of these SCMs are provided in Table 3.

Appearance of the silica aerogel and EP used in the study.
Physical properties of aerogel as provided by manufacturer.

Major chemical composition of EP.
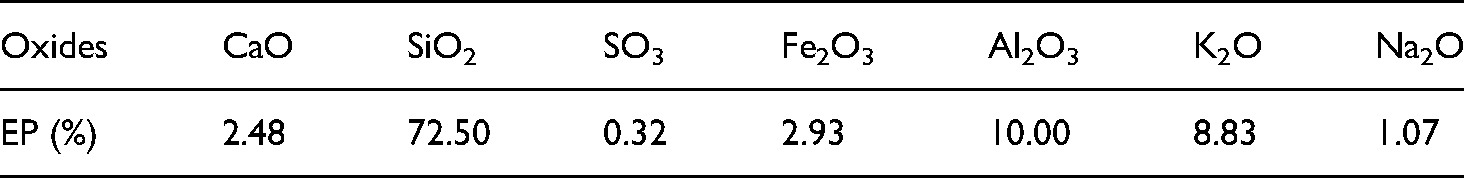
Chemical composition and physical properties of GGBS and fly ash.

Table 4 shows the mix proportion adopted in this investigation where a total of six mixtures were prepared. Two main variables were adopted, namely the different types of fine aggregate (sand and crushed glass), which were used at the same content, as well as the different types of fine aggregate replacement with lightweight materials (aerogel and EP). The lightweight mixtures (S-AG, S-EP, G-AG, and G-EP) were designed based on absolute volume method and established based on trials to achieve a density of 1600 ± 100 kg/m3 to facilitate comparison. The aerogel and EP were adopted as a partial fine aggregate replacement (65% by volume). On the other hand, the control mixtures (S-Control and G-Control) without lightweight materials were formulated to attain density of around 2200 ± 100 kg/m3. It should be noted that, in such case, the cement content and w/c ratio were varied in the mixtures with and without fine aggregate replacement. Laboratory tap water was used as mixing water, while superplasticizer was added in LCC mixes at a dosage of 0.3% of the binder content to facilitate workability. Furthermore, in the G-AG mix, fly ash and GGBS were adopted as partial cement replacement at 15% to limit the ASR expansion.
Mix proportions.
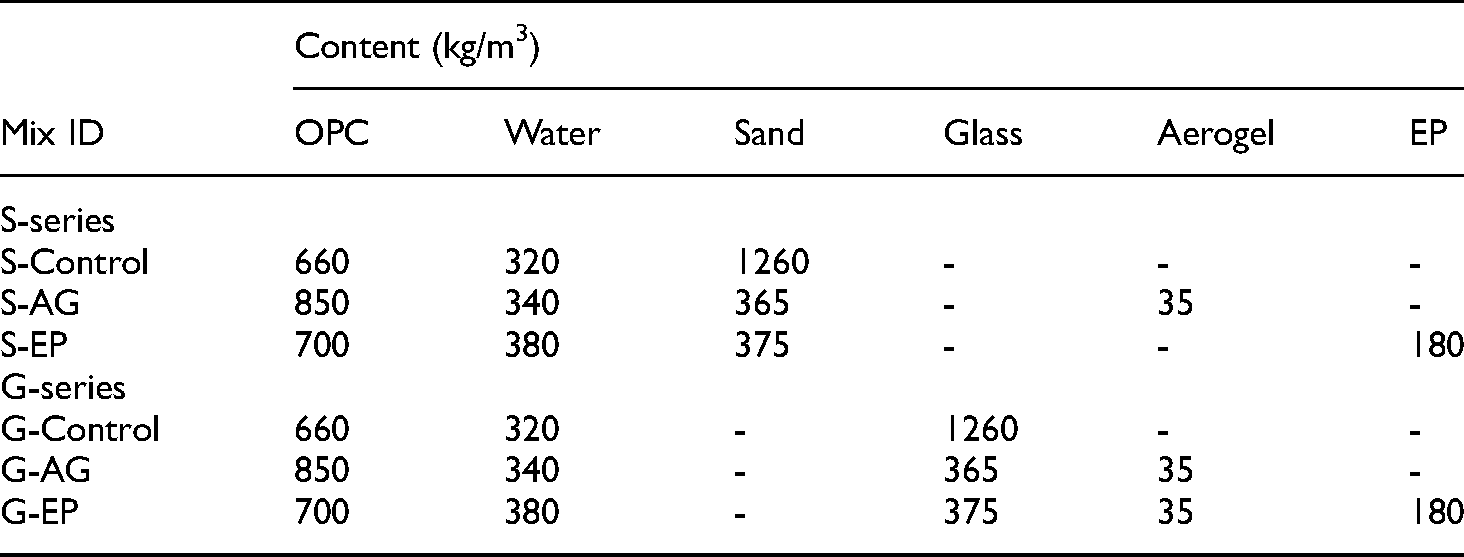
Mix proportions.

A standard mixing sequence was adopted for all mixes. The fine aggregate and cement were firstly dry-mixed, followed by addition of water and superplasticizer, and lastly the lightweight material (aerogel or EP) was added to complete the mix. Upon completion of mixing, the fresh mix was poured into oiled moulds and compacted on a vibration table. The specimens were demoulded after 24 h.
Test methods
Compressive strength
After demoulding, the 50 mm cube specimen was cured in water tank until the age of testing. The compressive strength test was carried out based on ASTM C109/C109 M. The compressive strength test was performed on specimen at the age of 28 days. The weight of the cube specimen was also taken prior to the compressive strength test to determine the density of the specimen.
Drying shrinkage
Mortar bar specimens measuring 25 × 25 × 285 mm were used for the drying shrinkage test in accordance with ASTM C596. The demoulded mortar bar specimen was cured in water for 72 h. After removing the specimen from water, the surface water was wiped off and the initial length of the specimen was measured with a length comparator. The specimen was then kept in a room with temperature of 23 ± 2°C and relative humidity of 50 ± 4% up to 28 days. At the age of 7 days, 14 days, and 28 days, the length comparator measurement was taken, and the drying shrinkage was computed based on the change in length of the mortar bar specimen.
Alkali-silica expansion
The alkali-silica expansion of the mixtures was assessed based on the test procedure outlined in ASTM C1260. Mortar bar specimens measuring 25 × 25 × 285 mm were used in this test After demoulding, the specimen was kept fully immersed in water in a sealed container, and then kept in oven at 80°C for a period of 24 h. After that, the specimen was removed, dried and the initial length of the specimen was measured with a length comparator. Then, the specimen was immersed in 1 M NaOH solution in sealed container and kept in oven at 80°C. Subsequently, the change in length of the mortar bar specimen was measured up until 14 days to evaluate the expansion.
Microstructural investigation
Microstructural investigations were also carried for selected specimens which were exposed to ASR testing. Field emission scanning electron microscope (FESEM) with an accelerating voltage of 15 kV was used to study microstructure of LCC specimen with crushed glass, aerogel, and fly ash, whereas powder X-ray diffraction (XRD) patterns were obtained with a CuKa radiation source within range of 5–80°. To evaluate the calcium hydroxide content, thermogravimetric analysis (TGA) was performed, and the sample was heated up to 950°C at a heating rate of 10°C/min. Fourier transform infrared spectroscopy (FTIR) analysis of the LCC was also carried out.
Results and discussion
Density and compressive strength
Table 5 presents the compressive strength of the aerogel LCC in comparison with the other mixtures and the corresponding density. Generally, the lightweight mixtures containing aerogel and EP achieved the targeted density of 1600 ± 100 kg/m3. This represents a substantial decrease of about 30% in the density compared to the control mixtures. Due to lighter nature as well as finer particle size of aerogel compared to EP, there is higher requirement of cement content in the mix design to produce the aerogel LCC. Also, because of the extremely lightweight nature of aerogel, the w/c ratio adopted had to be lowered to 0.40 to prevent the mix from being too fluid and hence floating of the aerogel. Floating of aerogel would result in a mix with lesser homogeneity.
Density and compressive strength of aerogel LCC compared to other mixtures.

Density and compressive strength of aerogel LCC compared to other mixtures.

*values in the parenthesis represent standard deviation.
Despite adopting better quality cement paste with higher cement content and lower w/c ratio, the aerogel LCC still exhibited lower compressive strength compared to the lightweight mixture containing EP at a similar density. This is likely due to the very high porosity of aerogel particle, causing it to possess lower crushing strength than the EP particle. Furthermore, the highly hydrophobic nature of aerogel can further weaken the bonding between the cement paste and the aerogel, resulting in the reduced compressive strength of the aerogel LCC. 9 Compared to the corresponding control mixture (S-series and G-series), the LCC produced by substituting 65% sand with the aerogel experienced about 50% strength decrease, which is within the range of strength reduction in aerogel LCC reported previously by Shah et al. 9
It is also apparent that utilizing crushed glass instead of mining sand as the fine aggregate improved the strength of all mixes. Since the particle size of the crushed glass is similar as the sand, it is believed that the finer portion of the crushed glass could exhibit certain degree of pozzolanic reaction 23 and hence could improve the 28-day compressive strength of the mixtures. The angular shape of crushed glass could also contribute to the strength enhancement. 24 Therefore, despite the strength reduction in the LCC due to the aerogel, adopting crushed glass could partially compensate for the reduction and attained an improvement by about 29%. Nevertheless, since crushed glass typically exhibits ASR,25,26 the use as aggregate and the resulting alkali-silica expansion of the aerogel LCC should be carefully considered.
The 7-day, 14-day, and 28-day drying shrinkage of all mixes are presented in Figure 3. As drying shrinkage is a time-dependent property, the shrinkage of all mixtures increased expectedly with age due to gradual loss of water through drying.
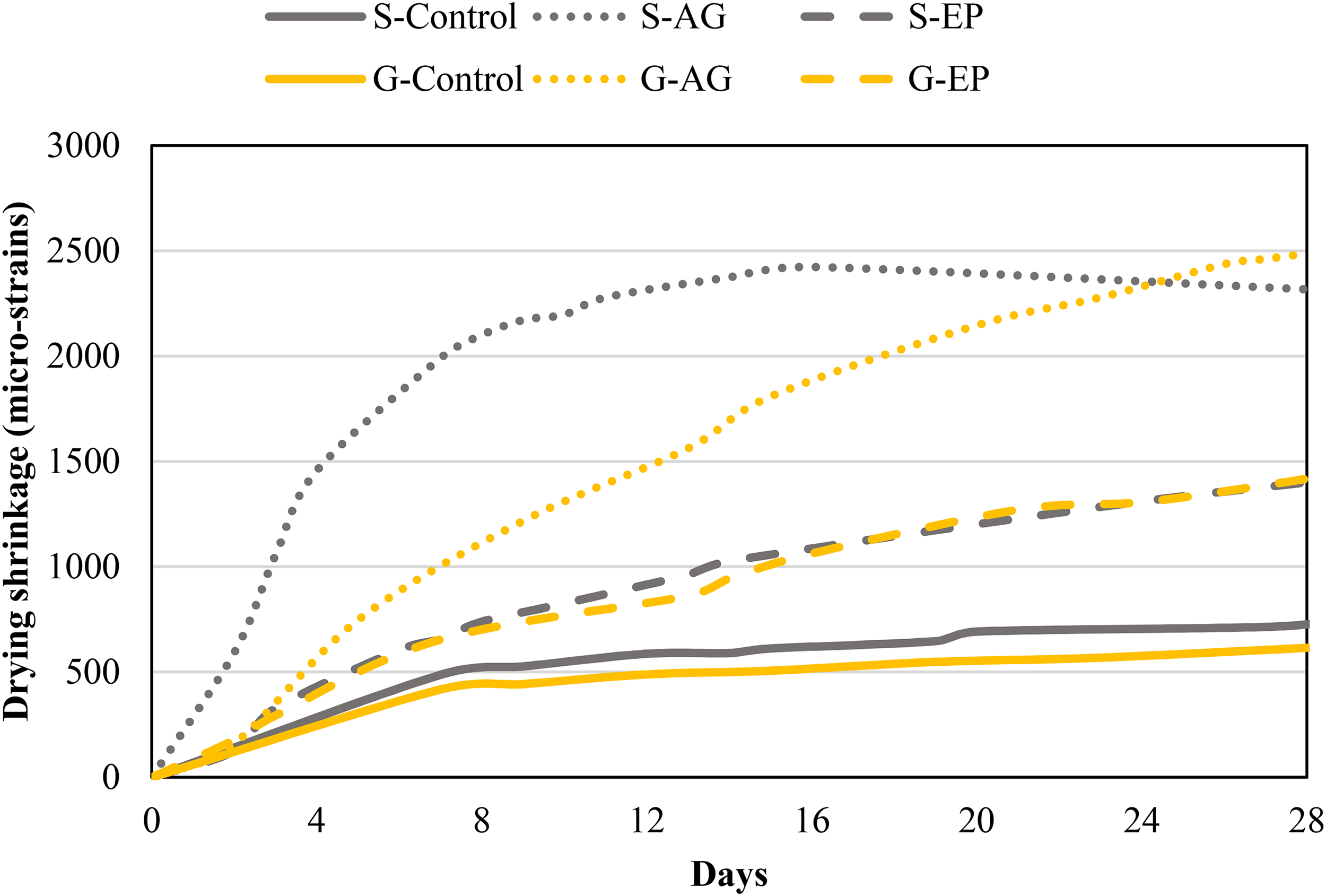
Drying shrinkage of aerogel LCC compared to other mixtures.
Comparing the effect of aerogel, the aerogel LCC had distinctively higher drying shrinkage than the corresponding EP and control specimens. At 28 days, the drying shrinkage of aerogel LCC was about 2.5–3 times higher than the control specimen (S-Control and G-Control), whereas the drying shrinkage at this age was about 75% higher than the EP lightweight mixtures (S-EP and G-EP). There are few explanations for the higher drying shrinkage found for the aerogel LCC. Firstly, as the extremely lightweight aerogel has low strength and low stiffness, replacing stiffer fine aggregate like mining sand and crushed glass reduced the resistance against the shrinkage movement. 27 This also explains the lower shrinkage of EP mixture compared to the aerogel LCC due to the better resistance of the EP against shrinkage movement. Secondly, the finer particle size of aerogel compared to the mining sand, crushed glass and EP is another contributing factor for the higher drying shrinkage of aerogel LCC. The very fine particle size of aerogel functions less as ‘aggregate’ in providing resistance towards shrinkage movement, and finer particle was reported to cause higher shrinkage than larger aggregates. 28 Lastly, the high cement content adopted in the mix design of aerogel LCC could cause the high shrinkage of the cement paste. With regards to this, the utilization of SCM to partially replace cement in the binder of the aerogel LCC can be considered.
Expansion of aerogel LCC compared to other mixtures
The expansion of specimens based on the exposure condition in the ASTM C1260 AMBT method is presented in Figure 4. The ASTM C1260 method specifies that expansion exceeding 0.10% at 16 days after casting (or 14 days under accelerated exposure condition) is considered to indicate potential deleterious expansion. Therefore, in the present investigation, only the mixes G-Control and G-AG had exceeded the limit of 0.10%.

Alkali-silica expansion of specimens.
None of the mixes using mining sand as fine aggregate exhibit potential deleterious expansion. In this S-series mix (with mining sand as fine aggregate), the expansions were below 0.05% and have low risk of expansion. This shows that the mining sand used as fine aggregate, as well as the aerogel and EP as partial mining sand substitute, were not alkali-silica reactive. However, it is noticed that when the aerogel and EP were employed to partially replace mining sand, the expansion of these mixes were slightly increased compared to the S-Control. This could be attributed to the presence of slightly reactive silica in the aerogel and EP. It was reported that the use of EP may be partially reactive, but does not cause deleterious expansion.29,30 Furthermore, the mix designs of S-AG and S-EP consist of higher cement content than the S-Control, and this could also provide a more alkaline environment for some alkali-silica reaction.
In the G-series, as expected, the G-Control mix experienced the highest expansion. The recorded expansion was 0.23% after 14 days in accelerated exposure condition, due to the reactive crushed glass aggregate, as reported widely in past research.25,26,31 The reactive silica in the crushed glass can react with the hydroxyl ions in the pore solution of cement paste, forming expansive alkali-silica gel which causes volume expansion. In the aerogel-glass LCC mix G-AG where the aerogel was incorporated as partial substitute of the crushed glass, the expansion still exceeded the limit of 0.10% even though it was reduced compared to the G-Control. As aerogel was utilized to partially replace the reactive crushed glass, the alkali-silica reaction could be reduced due to the lesser amount of reactive silica as the crushed glass content was lowered.
In view of the 14-day expansion of G-AG aerogel LCC which exceeded 0.10%, additional investigation was carried out to utilize commercially available SCMs such as fly ash and GGBS as a partial cement replacement. It is known that presence of SCM can aid to reduce the expansion in cement-based materials containing reactive aggregate.32,33 Table 6 gives the 14-day expansion values of the aerogel-glass LCC containing the SCMs.
14-day expansion of aerogel-glass LCC.

14-day expansion of aerogel-glass LCC.

* values in the parenthesis represent standard deviation.
The SCMs were able to reduce the expansion of the aerogel-glass LCC to below the maximum expansion limit of 0.10% at 14 days. The 15% fly ash was found to be more effective than the use of same amount of GGBS, where the expansion of the aerogel-glass LCC can be reduced to the same level as aerogel LCC using non-reactive sand as fine aggregate (S-AG). The fly ash could effectively reduce the alkali-silica expansion in the aerogel-glass LCC due to the following reasons: (i) reducing the alkalinity of pore solution when used to partially replace cement; (ii) consuming more alkali in producing CSH through pozzolanic reaction; and (iii) providing large silicate surface area for reaction with hydroxyl ions instead of reaction between the hydroxyl ions with reactive crushed glass. Class F fly ash has been reported to be more effective in reducing the concentration of alkalis compared to GGBS.32,34 The more effective reduction in expansion due to the use of fly ash can also be attributed to the higher silica content in the fly ash compared to GGBS. Higher replacement levels of GGBS are normally required as it is less effective in reducing alkali-silica expansion. 32
After the expansion test, the G-AG and G-AG-15% fly ash specimens were extracted for FESEM, XRD, FTIR and TGA for further microstructural investigations and comparison. Figure 5 shows the FESEM images of G-Control, G-AG, and G-AG-15% fly ash specimens. The G-Control specimen (Figure 5 a and b) with highest expansion, had cracks passing through the glass particles possibly due to pressure exerted by ASR product. The addition of aerogel in the G-AG seemed to have compensated the expansion force exerted by ASR product. Due to the weak bonding between aerogel and cement paste, there was available space around aerogel where the ASR product can be formed (Figure 5 c and d). However, in the aerogel-glass LCC with fly ash as cement replacement, the ASR product was observed to be less both around the crushed glass and aerogel particles, which resulted in almost no crack in the glass particle (Figure 5 e and f).

FESEM images of specimens: (a-b) G-control; (c-d) G-AG; (e-f) G-AG-%15 fly ash. (a) Cracks in glass particle. (b) Reacted glass particle. (c) Aerogel particle in the void. (d) Reacted surface of aerogel particle. (e) Glass particle G-AG-15% fly ash. (f) Aerogel particle in G-AG-15% fly ash.
Calcium hydroxide (CH) content is an important factor in formation of ASR gel. It serves as a cation exchanging agent that forces the alkali from the ASR gel media back into solution to recycle the attack on reactive silica. 35 Therefore, fly ash is a suitable option as SCM for ASR mitigation due to its low alkali and calcium oxide content. The CH content in G-AG and G-AG-15% fly ash specimens was evaluated by TGA. TGA samples were heated from room temperature to 950°C and weight losses of samples due to heating are presented in Figure 6. The weight loss in the range of 440°C to 530°C was evaluated as it is attributed to the decomposition of CH in the hydrated LCC.36,37 The CH content was about 3% in the G-AG and reduced to 2% with 15% fly ash (G-AG-15% fly ash specimen), respectively. The reduction in CH content could be attributed to pozzolanic activity and low calcium oxide content in the fly ash. Thus, reducing the CH content from the LCC matrix could decrease the ASR and expansion of the specimen.

TGA results of G-AG and G-AG-15% fly ash.
The reduction in CH was also corroborated in the XRD analysis. The major CH peaks in both G-AG and G-AG-15% fly ash specimens were identified through Xpert Highscore Plus software. The peak intensity of CH at 2° Theta of 18°, 34°, and 47° decreased in sample with fly ash as cement replacement (Figure 7).

Powder XRD patterns of G-AG and G-AG-15% fly ash.
The FTIR spectrum of both G-AG and G-AG-15% fly ash specimens are presented in Figure 8. The bands appearing in the FTIR spectra were similar for both specimens. The four major bands were marked as B1-B4 and their possible assignment from the literature are listed in Table 7. Despite the high degree of similarity, the band B1 assigned to CH around 3640 cm−1 almost disappeared when cement was replaced by fly ash in the G-AG-15% fly ash specimen, and this is consistent with the TGA and XRD observations.

FTIR spectra of G-AG and G-AG-15% fly ash.
Major FTIR bands assignment.
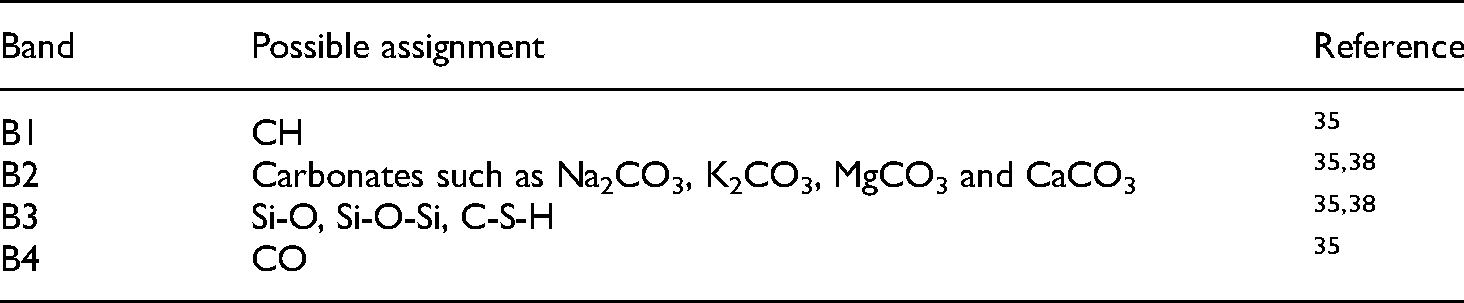
The present study investigated the effect of aerogel on drying shrinkage and alkali-silica expansion of LCC specimens with sand and crushed glass as fine aggregate. In addition, the results were compared with the lightweight mixtures with the use of EP. The key findings obtained are summarized below:
At the density of 1600 ± 100 kg/m3, the LCC with aerogel experienced about 55% compressive strength reduction, whereas the mixes with EP had lower compressive strength loss. Nevertheless, the compressive strength of aerogel LCC was higher when utilizing crushed glass as the fine aggregate. Due to its fine particle size and utilized in the form of fine aggregate replacement, the drying shrinkage of aerogel LCC was increased by about 2.5–3 times and 75–80% higher than the control and EP mixtures, respectively. In the presence of reactive crushed glass as fine aggregate, the alkali-silica expansion exceeded the limit of 0.10%; although the expansion can be reduced in the presence of aerogel and EP, the aerogel-glass LCC still exhibited expansion exceeding the limit. The use of 15% SCMs can significantly decrease the alkali-silica expansion in the aerogel-glass LCC, with fly ash outperforming GGBS. FESEM images show that aerogel and fly ash together can compensate the ASR expansion caused by the reactive crushed glass, while the XRD, TGA, and FTIR concurred that fly ash can reduce the CH, hence decreasing the alkali-silica activity and expansion.
Footnotes
Acknowledgements
The authors wish to acknowledge the financial support provided by the Ministry of Higher Education Malaysia under the grant FP056-2018A (FRGS/1/2018/TK06/UM/02/1) and Higher Education Commission of Pakistan under the HRDI-UESTPS-UETS Phase-1, batch VI program.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Ministry of Higher Education Malaysia under the grant FP056-2018A (FRGS/1/2018/TK06/UM/02/1).
