Abstract
In the field of regenerative medicine, cell therapy is often used to regenerate organs or tissue that can be widely applied to several diseases. In recent years, the Taiwanese government has listed the biotechnology industry as a key developmental objective and has enacted laws to regulate cell therapy. This has led to substantial change and has necessitated a reliable direction of management in the cell therapy industry. Therefore, this study aims to develop a new method to explore the key success factors in three dimensions of relevant biotechnology company management. It adopts a qualitative method combined with a quantitative analysis method. The study uses Gwo Xi Stem Cell Company, a qualified cell therapy company in Taiwan, as a case study. It adopts a literature analysis method and conducts interviews with cell therapy experts to develop an innovative assessment framework. The analytic hierarchy process is combined with decision-making trial and evaluation laboratory to establish the key success factors in the management of the cell therapy industry. Thus, the study offers strategic recommendations for the development of domestic stem-cell-related industries. The results demonstrate technical characteristics as the most important direction for successful management. Moreover, due to the substantial change in Taiwan's cell therapy industry, technical characteristic-related key success factors, including product development strategy (technical characteristic 1) and technological mastery (technical characteristic 2), are currently considered to be crucial key success factors in decision making.
Keywords
Introduction
In recent years, the biotechnology industry has developed rapidly, due to a saturated electronic industry and active investment by national governments worldwide. In 1982, the Taiwanese government listed biotechnology as a key technology in its development program. However, due to the rapid growth in Taiwan's biotechnology industry, especially the emerging advanced-therapy sector (e.g. the cell therapy and gene-therapy industries), strategic management has always been an urgent concern.
In the late 1990s, rapid breakthroughs in medical technologies occurred in the global biotechnology industry, including the cell therapy market that was driven by stem- and immune-cell technology. 1 Cell therapy is an emerging industry within the biotechnology industry. In Taiwan, the cell therapy industrial chain comprises upstream, midstream, and downstream divisions. In the upstream division, raw materials are collected and stored, while the midstream division includes purification procedures, cell culture, and inspection and storage. The cells are stored in pharmaceutical-grade cell banks for future clinical use. The downstream division conducts the research on and clinical application of cell therapy and genetic testing. 2 However, although cell therapy has gradually become available in recent years, its practice and evaluation process remains without standards. Therefore, in July 2018, Taiwan adopted the dual-tract regulatory framework from Japan and initiated its own cell therapy regulation through which patients in Taiwan can receive safe and effective treatments. Based on the Japanese dual-tract regulatory framework, Taiwan has built a new framework that comprises two approaches. One is called Special Regulation for Cell Therapy (SRCT Act), which governs the contract manufacturing organization of designated autologous cell therapy as a routine medical practice that is executed in hospitals or clinics; this is similar to the Japanese Act on the Safety of Regenerative Medicine (ASRM Act). The other one is called the Act on the Management of Regenerative Medical Preparations (MRMP Act), and promotes the license approval of cell therapy products (CTPs) in Taiwan. Unapproved CTPs with enough clinical trial-related data can apply the 5-year short-term marketing authorization (MA) to sell in hospitals and clinics, which is similar to the Japanese Pharmaceuticals, Medical Devices, and Other Therapeutic Products Act (PMD Act). 3 Based on the SRCT Act, Taiwan has approved 86 cell therapy technologies in 19 cell therapy companies (until May 2021). Besides the cell therapies that are exempted from the SRCT Act, according to the Taiwan Research-based Biopharmaceutical Manufacturers Association, there are 83 ongoing clinical trials related to cell therapy in Taiwan; however, none of them have obtained MA in Taiwan. 4 The MRMP Act encourages the Taiwanese cell therapy industry to accelerate these non-commercial clinical research studies to reach commercialization and selling, and further boost economic growth in Taiwan. Moreover, Taiwan is famous for its powerful health care system worldwide; thus, although the Taiwanese cell therapy industry was established relatively late compared to other advanced countries (e.g. Japan, the United States, and Europe), the country retains the great potential to increase competitiveness through the new regulation, combined with its own intrinsically powerful health care system.
Regarding the cell therapy market, according to the Alliance for Regenerative Medicine's Annual Report of 2020, total global regenerative medicine financing reached US$19.9 billion in 2020, which broke previous records. Additionally, 1220 clinical trials relevant to regenerative medicine aim to enroll more than 90,000 patients globally, indicating the great potential value in the global cell therapy industry. 5 In Taiwan's cell therapy market, there were 26 cell therapy-related companies in 2016, while the total production value of the regenerative medicine industry reached NT$1.99 billion, registering a compound annual growth rate of 1.9% from 2012. Of this amount, the cell-storage service segment dominated the market and accounted for the largest value share of NT$1.23 billion, which could be attributed to the fact that all of the CTPs in Taiwan were still in the pre-clinical and clinical trial phases. 2 However, increasingly more CTPs have been approved and used clinically in regions including America (the US and Canada), Asia (Japan, Korea, and Singapore), Europe (European Medicines Agency, EMA), and Oceania (Australia and New Zealand). 6 Under these circumstances, flexible regulation is an advantage and encourages the cell therapy industry to accelerate product development. Thus, Taiwan's cell therapy industry is expected to grow rapidly.
Considering this macro trend and shifting in Taiwan's cell therapy industry, many financial agencies have started to invest in this field, while cell therapy companies with abundant funds have sought appropriate strategies to increase its competitiveness, for example, through partnership, technology transfer, and joint development. Thus, strategic management will be crucial in differentiating companies from rivals in the field in the near future. However, we have not found any relevant management studies in the cell therapy industry. Therefore, this study aims to investigate the key success factors (KSFs) in innovative management by integrating the research background and motives and adopting both a qualitative method and a quantitative analysis method. The study uses Gwo Xi Stem Cell Company as a case study. In contrast to a general stem-cell company that employs a single operating strategy (e.g. cell preparation, cell storage, and cell-product test), Gwo Xi Stem Cell Company's operations span the entire cell therapy industrial chain in Taiwan, with four innovative CTPs in human clinical trials. Data collection is through a literature review and a conceptual framework that is mainly based on meeting the three dimensions and 14 KSFs of development in the cell therapy industry, each with different goals. Then, through analysis and a discussion of the cell therapy industry and case study company, suggestions are offered for the strategic development of the cell therapy industry.
Review of related literature
Enterprise internal resource (EIR) factors
Wernerfelt (1984) first proposed the resource-based theory, which holds that an entity's EIR and capability will guide the direction of its management strategy, and will become a major source of its profits. 7
R&D resource (EIR1)
According to Wernerfelt (1984), an internal resource is critical for an enterprise; this view has changed how strategies are formulated in practice. 7 According to Barney (1986), an enterprise can obtain a long-term competitive advantage through the accumulation of its own resources, allowing it to execute its strategies under situations of incomplete market information. 8 According to Aaker (1989), the essence of strategic management is the development and maintenance of assets and skills that can directly affect a firm's performance. 9 Thus, possession of sufficient R&D resources is important for an enterprise's strategic management, especially in the high-tech industry.
Supervisors’ support (EIR2)
Sheng-Wei Luo (2010) proposed that the success and failure of an enterprise's product development lay not in whether the supporting resources were sufficient, but in whether the full support of senior managerial personnel had been won; full support of senior managerial personnel, together with good synergy, would ensure new product R&D success. 10 Thus, supervisors’ support is a crucial factor in the development direction of a company's R&D.
Project management capability (EIR3)
The fields of network analysis and planning techniques were the first signs of project management for facilitating knowledge sharing between practitioners. However, there was a gradual movement toward the concept of project management as an organizational capability from the mid-1990s. 11 In the last decade, due to technological development, an increasing number of emerging industries, including the cell therapy industry, brought complexity to the whole industrial environment, thus causing considerable project variation. According to Aslami (2019), most non-profit organizations manage projects in a very complex environment. Project management practices significantly impact organizational performance in achieving competitive advantages in the market; project scope management and cost management should especially be prioritized due to the greater impact on organizational performance. 12
Talent management mode (EIR4)
Taiwan has no large pharmaceutical companies, while most cell therapy companies are new and small. Therefore, most cell therapy talents remain scattered in academic research and industrial circles, which has led to a lack of interdisciplinary integration of personnel. Bagne (1969) proposed the “push and pull theory,” according to which the purpose of population flow is to improve living conditions; conditions in favor of inflow are “pull,” while outflow conditions are “push.” 13 According to Becker (1964), personal professional knowledge and production capacity can be improved through education and training. 14 The accumulation of manpower capital is crucial for the cell therapy industry; proper talent management and attracting personnel with skills in cell therapy are critical issues in the development of a cell therapy company.
Technical characteristics (TC) factors
Over time, technological development will go through the product life cycle, which is divided into the introduction, growth, mature, and decline stages. 15 According to Tarek M. Khalil (2000), optioning and supporting crucial technologies have a great impact on organizations in enhancing their competitive advantages in the international market. 16
Product development strategy (TC1)
Enterprises must always develop new products to satisfy market customers’ demands. Takeuchi and Nonaka (1986) proposed the great benefit of new product development in the operation of an enterprise. 17 According to Porter (1990), a country's competitiveness lies in the innovative capability of its industry. 18 In addition, the product life cycle is a key factor in determining an enterprise's market competitiveness. 15 In the pharmaceutical industry, an enterprise suffers considerable losses when a new product fails. Therefore, the successful development of new products has always been a concern.
Technological mastery (TC2)
Scientific publications and patents, which represent the value of proprietary technologies for enterprises, are seen as an output measure of investments in technological capability. 19 According to Carl J. Dahlman and Larry E. Westphal (1981), technological mastery has important implications for gaining advantages by making effective use of technological knowledge. 20 Products in the biotechnology industry (i.e. new drugs) typically have long durations, require heavy investment, and are highly technical in nature. Hence, the level of a technical mastery of a biotechnology company will impact the direction of its strategy.
Level of technological complexity (TC3)
Tushman and Rosenkopt (1992) propose that technological uncertainty depends on the level of technological complexity and technological discontinuity; the higher the technological complexity, the higher the uncertainty associated with the technology, indicating that such technology is rare, with unpredictable potential, which may significantly impact an enterprise. 21 According to Teece (1996), the fundamental characteristics of technological development include uncertainty, path dependency, cumulative nature, irreversibility, technological interrelatedness, tacitness, and appropriability. 22 Regarding implicit and explicit degrees of knowledge, Patrick Sullivan (1996) defines them as follows: Implicit knowledge means that it is difficult to describe such knowledge in specific terms; for effective technological knowledge transfer, it requires one person to pass it on to another through in-service training or mentorship. Explicit knowledge means that it can be written down as texts for sharing; such knowledge can be protected through legal intellectual property rights; however, since it is easy to learn, it is also easily transferred or embezzled.23,24
Technological cooperation mode (TC4)
According to Chesnais (1988), an enterprise's means of capturing new technology mainly include internal and external sources. The internal source refers to voluntary R&D achievements, while the external source refers to the methods that involve purchasing machinery equipment, patent licensing, R&D alliances, market transactions, and so on. 25 According to Grand (2002), a strategic alliance is a cooperation strategy that can reduce the risks that must be borne by an enterprise, and can integrate the resources and capabilities of multiple organizations, which reduces the R&D cost. 26 Biotechnology is a strategic industry in Taiwan; however, due to the exorbitant costs of R&D and the rapid changes in technology, biotechnology companies frequently choose an appropriate collaboration strategy (e.g. technology transfer and strategic alliances) to spread the risks of development and unsuccessful commercialization.27,28 Thus, the technological cooperation mode is a critical consideration in decision making in the biotechnology industry.
Patent portfolio deployment (TC5)
As mentioned in the TC2 section, patents are seen as an output measure of investments in technological capability. 19 According to Niosi and Bas (2001), most previous scholars in knowledge circulation used the patent quantity as a variable for measuring the circulation of intellectual property rights. 29 According to Holger Ernst (2003), patents can be classified into two major functions, patent protection and patent information, which can be effectively used for technology management with powerful strategic value, for example, patent sale and cross-licensing. 30
Clinical promotion manner (TC6)
According to Fishman and Porter (2005), a drug's R&D schedule should take at least 8 years, from finding a target to a clinical trial. However, after the new drug has been launched into the market, it will have high profitability, long product life cycle, while the acquisition of patent protection can seize market exclusivity. 31 According to Gail A. Van Norman (2016), the development of a new drug takes between 10 and 15 years, on average, and costs millions of dollars. In the United States, ∼0.01% of potential candidates for new drugs obtain approval from the laboratory (∼1/1000) to the pharmacy (∼1/10). 32 Due to the difficulty of conducting clinical trials, Barbara Farrell and colleagues suggested that an effective and standard clinical trial management method was crucial for future research and health care. 33 Thus, the manner of clinical promotion is crucial to the biotechnology industry.
Enterprise external environment (EEE) factors
The crucial successful external factors in the biotechnology industry include internal and external factors. External factors include cluster effect, infrastructure construction, national policy, and so on. According to Arojärvi (2001), the biotechnology field is highly diversified, while several financial issues entail a variety of factors from an enterprise's external environment. 34
Level of government support (EEE1)
According to Tanja Rautiainen (2000), external KSFs in the biotechnology industry include clustering, infrastructure, and national policies. Additionally, she observes that the Finnish biopharmaceutical companies she interviewed value the essence of a company (i.e. products, resources, and national policies). Arojärvi (2001) refers to these KSFs too and adopts them for study.34,35 Thus, government policy is one of the KSFs with an impact on the successful development of the biotechnology industry.
Degree of regulatory rigor (EEE2)
Since medical products are eventually applied to humans, they are under rigorous control by the government, with close monitoring for safety and efficacy; therefore, it takes a long time and costs millions of dollars to obtain MA. 32 According to Laínez (2012), the heavy regulatory burden, with many national and/or regional variations, is one of the important characteristics of the pharmaceutical industry, while it is important to harmonize regulatory standards among different countries. 36 Thus, the degree of regulatory rigor in each country influences enterprises’ strategic management, especially regarding transnational strategy.
Industry development situation (EEE3)
Hofer and Schendel (1978) observe that the measurement of the industrial environment dimension can be divided into capital intensity, degree of vertical integration, intra-industry competition degree, entry and quit barriers, seller concentration and a relative scale of competitors, industrial added value, economic scale and learning curve, degree of product differentiation, and so on. 37 J. Stuart Evans (1991) propose that strategic flexibility is an essential conceptualization for a firm to dynamically understand the current situation in high-technology related industries. 38 Taiwan's cell therapy industry, which is a high-technology industry, is currently in a rapid-growth phase. Hence, we need to refer to the development and cooperation models of other advanced countries (e.g. Japan, the United States, and Europe) that are suitable for our cell therapy industry.
Competitive pressure (EEE4)
Michael. E. Porter (1980) proposed that an industry's profitability was not determined by product appearance or the depth of technology applied by it, but by the competitive structure of the industry; additionally, to analyze the situation of industrial circles, he proposed Michael Porter's Five Forces analysis: horizontal competition, potential competitor, substitute, supplier, and customer. Moreover, he could find the KSFs among them. 39 Murdick Moor and Eckhouse (1989) point out that it is only when a competitor's status is understood that an enterprise can formulate a set of strategies to exert its advantages and recompense its disadvantages. 40
Study method
Research flowchart establishment
To clarify the purpose of this study, we first establish the research objective and scope and collect relevant studies and secondary data on the case study company to formulate the structure of the study and conceptual framework. Thereafter, we design the questionnaire based on the conceptual framework to establish the crucial KSFs.
Conceptual framework
To develop an assessment framework for the innovative management of the cell therapy company, this study extracted a preliminary dimension based on the relevant literature (Wernerfelt, 1984; Khalil, 2000; Arojarvi, 2001);7,16,34 furthermore, we selected the cell therapy company management's KSFs through an expert interview. This study has generalized three major dimensions: TC, EIR, and EEE. The TC dimension is divided into six KSFs (i.e. TC1, TC2, TC3, TC4, TC5, and TC6); the EIR dimension is divided into four KSFs (i.e. EIR1, EIR2, EIR3, and EIR4), while the EEE dimension is divided into four KSFs, (i.e. EEE1, EEE2, EEE3, and EEE4) (Figure 1).

Conceptual framework.
Questionnaires in the analytic hierarchy process (AHP) and decision-making trial and evaluation laboratory (DEMATEL)
Through the relevant literature review and by means of an expert interview and discussion, we developed the dimensions and assessment indicators for the innovative management of the stem-cell company. Three major dimensions and 14 KSFs that are used as assessment indicators are clearly defined and established to further develop the fundamental structure of the DEMATEL-AHP approach questionnaire. The questionnaire has two parts: the AHP and the DEMATEL parts. In the first part, the AHP questionnaire scheme is shown in Figure 2; for each pair of the factors (i, j) of dimension and KSF, the respondents were asked to select each KSF on the left and to compare it to the KSF factor on the right. According to Saaty's theory, the options include 1 (equally), 3 (important), 5 (more important), 7 (much more important), and 9 (extremely important), which are on each factor's side, while the 2, 4, 6, and 8 can also be selected by selecting the two options containing a number, that is, selecting 1 and 3 is equal to 2.41,42 Thereafter, the questions were repeated until all the options were exhausted. For the second part, the DEMATEL scheme is shown in Figure 3; for each pair of KSFs (i, j), the respondents were asked to select each KSF's influence on others; the options include the scale from 0 (No influence), 1 (low influence), 2 (medium influence), 3 (high influence), to 4 (very high influence). The questions were repeated until all judgments were exhausted. At the end of this questionnaire, the respondents were asked to provide their basic information, including gender, age, educational stage, job, and seniority.

Scheme of analytic hierarchy process (AHP) questionnaire.

Scheme of decision-making trial and evaluation laboratory (DEMATEL) questionnaire.
Integrated methods combining DEMATEL and AHP
To investigate the causal relationship and relative importance of each KSF shown in Table 1, the DEMATEL-AHP approach is used. From an industrial viewpoint, the DEMATEL-AHP approach may be a tool to help management in terms of providing reliable suggestions to the cell therapy company. The details regarding the DEMATEL and AHP methods are individually provided as follows.
Significance of scores in analytic hierarchy process (AHP). 41

The AHP method
The AHP, a decision-making tool, is developed to quantify managerial judgment of the relative importance of each of several conflicting criteria used in the process of making a decision. The AHP uses a hierarchical structure and enables decision makers to measure the relative importance weight of projects (i.e. KSFs) by scale; the score can be equally divided into nine rating degrees. The AHP method allows for a slight inconsistency in judgment because humans are not always consistent. Having obtained a comparison matrix to compute a priority vector, which is a normalized Eigen vector of the matrix, the AHP can be used to convert ordinal scale to ratio scale, and even check its consistency.
41
In the AHP method, the goal and primary problem is broken down into several components/elements. The ratio of the scales is then used to compare their relative importance and to identify the most important factor. Therefore, it is quite useful in identifying the critical factors required for solving problems containing uncertainty or multi-attribute decision making. The AHP method and its calculated process can be summarized in the following steps:41,43 (1) To list and define the overall goal, criteria, decision alternatives, and questionnaire design by reference to the literature review and expert interview; (2) To establish a judgment matrix (represented as “A”). A1, A2, …, An are the set of stimuli. The computed judgments on a pair of stimuli, Ai, Aj, are
Table of random consistency index. 41

The DEMATEL method
DEMATEL, a multicriterial decision-making (MCDM) method, can effectively understand the complicated causal relationships by reviewing the level of influence among factors and utilizes matrix operations to determine the causal relationships and influence level among KSFs. It helps to visualize the degree of influence through an impact diagraph map.44,45 The DEMATEL method has three basic assumptions: (1) The question needs to be clear at the stage of question formation and planning; clear knowledge about the question being studied is required to set the question correctly; (2) The degree of relevance between and among KSFs needs to be clear, and on a scale from 0 to 4 (0—“No influence”, 1—“Low influence”, 2—“Medium influence”, 3—“High influence”, and 4—“Very high influence”) to indicate the relevant level thereof; (3) The essential characteristics of every KSF in a questionnaire need to be understood. DEMATEL determines the interdependence and relationships among variables according to specific features of objective matters, hence reflects the characteristics and evolution trend of a system. The DEMATEL method and its calculated process can be summarized in the following steps: (1) Designing the questionnaire based on the established overall goal, criteria, and decision alternative; (2) Computing the average matrix (M). The respondents were asked to evaluate the direct influence between two KSFs on the scale mentioned above. If k is the number of respondents and n is the number of KSFs, with
Data collection
Through the literature review and in-depth interviews with senior experts in the regenerative field, we designed the AHP-DEMATEL approach questionnaire to examine the development factors in the cell therapy company and to further determine the causal relationship and impact degree of each KSF. However, to obtain a diversified and objective insight, based on the DEMATEL and AHP questionnaire, we interviewed 25 respondents (i.e. experts), divided into three groups of interview objects: cell therapy industry experts, biotechnological experts (non-cell therapy expertise), and physicians (Table 3). Of these, 9 respondents are experts working in the cell therapy company (average work experience: 7.22 years), 10 respondents have worked in the biotechnology industry for more than 9 years, representing the biotechnological experts (average work experience: 17.6 years), and 6 respondents are physicians who are involved or interested in regenerative medicine clinical trials, representing the medical experts (average work experience: 21.83 years).
Experts background information.

Experts do not have relevant experience in the field of cell therapy.
Study results
The TC-related factors are comparatively important in AHP analysis
Due to the considerable change in Taiwan's cell therapy industry in recent years, this study aims to investigate the crucial KSFs to provide reliable suggestions to help decision makers involved in the management of cell therapy industry operations. To obtain objective suggestions, we interviewed three expert groups who filled out the questionnaire. In the AHP analysis, the results are slightly different between the three expert groups, indicating that experts from different fields provide unique perspectives to the cell therapy industry. The AHP result demonstrates that the cell therapy expert group and the physician group have similar perspectives, of which the dimension ranking order is: TC > EIR > EEE; however, the dimension ranking order for the biotechnological expert group is: TC > EIR > EEE (Table 4). Additionally, the KSFs with comparatively high importance weights are mostly TC-related factors in the biotechnological expert and physician groups, of which the ranking orders are: TC2 > TC1 > TC5 > TC6 > TC4 > EEE3 > EIR1 > EIR2 > EIR3 > TC3 > EIR4 > EEE1 > EEE2 > EEE4 and TC2 > TC5 > TC6 > EEE1 > EEE3 > TC1 > TC4 > EIR3 > TC3 > EEE4 > EEE2 > EIR4 > EIR1 > EIR2, respectively. However, the ranking order for the cell therapy expert group is different from that for the biotechnological expert and physician groups, which is: TC2 > EEE3 > EIR2 > EEE1 > TC5 > EEE2 > EIR 4 > TC6 > EIR3 > EEE4 > TC1 > EIR1 > TC4 > TC3, indicating that, although the cell therapy expert group considers TC2 to be the most important KSF, the EEE-related and EIR-related factors also have an important impact on the cell therapy industry (Table 4). For the overall group, it is concluded that the top half of the most important KSFs at the moment of providing reliable suggestions for the decision maker involved in the cell therapy industry comprises seven factors, of which the ranking order is: TC2 > TC5 > TC6 > EEE3 > TC1 > EEE1 > EIR3. Notably, three of the six TC-related factors (i.e. TC2, TC5, and TC6) are located in the top three; of these, TC2 and TC5 are also in the top half of the ranking order in the three expert groups. Especially, TC2 is among the highest relative importance weights in the three expert groups. On the one hand, it can be observed that EEE3 is the most important KSF in the EEE dimension, which is also in the top half of the ranking order in the three expert groups. On the other hand, EIR3 is the most important KSF in the EIR dimension (Table 5). In the AHP results, all the
Statistical table of the three dimensions’ importance weights in the analytic hierarchy process (AHP) analysis (

Statistical table of key success factors’ (KSFs’) importance weights in the analytic hierarchy process (AHP) analysis (

The causal relationship between the 14 KSFs in the DEMATEL analysis
To identify the causal relationships between the KSFs of each dimension at the time of decision making, the r − c value indicates the degree of factor influence or being influenced by others, while the r + c value indicates the relation degree between each factor. Notably, seven KSFs’ r − c values are positive in the overall group, indicating that these KSFs belong to the cause group, which can affect other KSFs, while the ranking order is: EIR3 > TC2 > TC1 > EIR1 > EIR2 > TC3 > EIR4 (Table 6). Thus, carrying out strategic management according to these KSFs with positive r − c values may bring about a comparatively high benefit to the company; this is recommended to the decision makers in the cell therapy industry for inputting resources for formulating strategy. However, the KSFs with positive r − c values are slightly different among the three expert groups. The ranking order for the cell therapy expert group is: TC2 > EIR3 > TC3 > TC1 > EIR2 > EIR1 > EIR4, while the ranking order for the biotechnological expert group is: TC2 > EIR3 > EIR1 > EEE3 > TC1 > EIR2 > TC3 > EEE4, and the ranking order for the physician group is: EIR3 > EIR4 > TC1 > TC2 > EIR2 > EIR1 > TC3 (Table 6). Notably, all these TC-related KSFs and EIR-related KSFs (i.e. TC1, TC2, TC3, EIR1, EIR2, EIR3, and EIR4) show high influence in the three expert groups; especially the TC2 and EIR3 possess extremely high r − c values in the three expert groups, while only EIR4 turns negative in the biotechnological expert group, replaced by EEE4, which turns positive. To review the company management performance through the r + c value, which represents prominence, it is noted that the ranking order of the top three KSFs by r + c value is the same in the three expert groups (i.e. EIR1, TC1, and TC2), whereas the ranking order for the overall group is: EIR1 > TC1 > TC2 (Table 6). However, the results of the top 3 ranking orders by r + c value are slightly different in three groups: the ranking order of the top three KSFs for the cell therapy expert group is: TC2 > TC1 > EIR1, while that for the top three KSFs for the biotechnological expert group is: EIR1 > TC2 > TC1, and that for the top three KSFs for the physician group is: TC1 > EIR1 > TC2 (Table 6). Additionally, EIR1, TC1, and TC2 simultaneously belong to the cause group, which means that these three KSFs have a significantly important influence in strategic management. Altogether, it is necessary to highlight that only TC2 simultaneously possesses high r + c and r − c values in the three expert groups, indicating that it plays an extremely important role in strategic management in the cell therapy industry.
(r + c) and (r − c) values of each key success factor (KSF) in the decision-making trial and evaluation laboratory (DEMATEL) analysis.
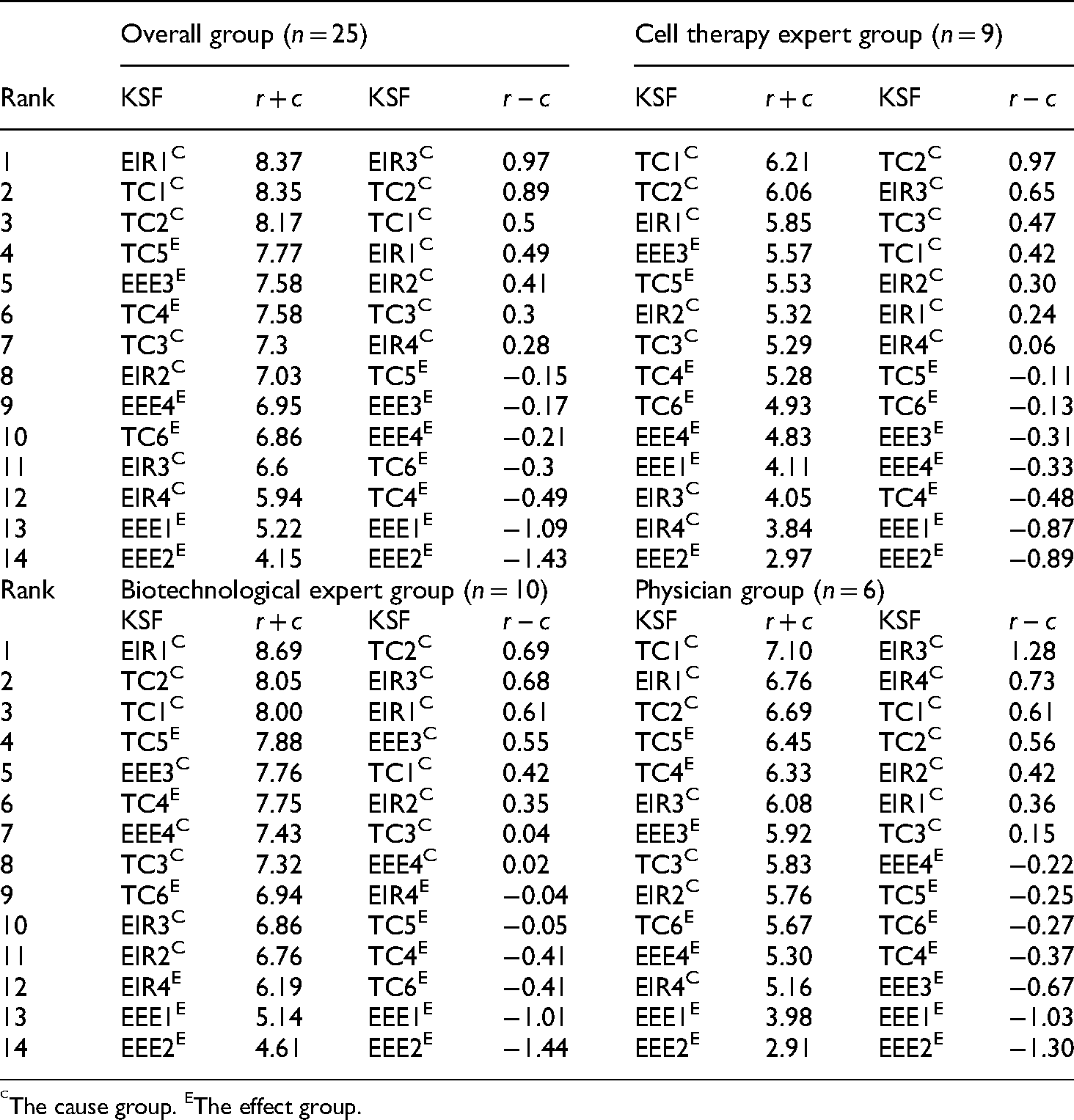
The cause group. EThe effect group.
Conclusions and discussions
To provide reliable suggestions for strategic management in the cell therapy industry, this study develops an assessment framework through a literature review and an in-depth discussion with each respondent using the AHP-DEMATEL method. The reason for integrating the DEMATEL approach with the AHP method is that the AHP does not allow for an evaluation of the interrelations and influences between the factors in the decision-making process. Although Saaty developed the analytic network process (ANP) in 2013, it is generally used to explore important KSFs that are already known in the interrelations and influences between each KSF in the decision-making process. 47 However, in some cases, the decision maker has difficulty understanding the influence and interdependence; thus, DEMATEL is frequently used to identify cause-and-effect relationships based on graphs theory, which can help decision makers to obtain a better understanding in some complex situations. Therefore, this study adopts an integrated method to establish the crucial KSFs using DEMATEL and the hierarchical process.
In the AHP result, we establish that the most important dimension affecting decision making in the three expert groups is TC, suggesting that strategic management that focuses on TC-related KSFs may enhance the competitiveness of the cell therapy industry. Among the dimensions, TC2, TC5, and TC6 are relatively important in the AHP result. On the other hand, TC1 and TC2 also show comparatively high r − c and r + c values in the DEMATEL analysis. Altogether, this demonstrates that TC2 has the most impact on company management performance in the cell therapy industry. Regarding the legislative aspect, despite the substantial change in the cell therapy regulation in Taiwan, EEE-related factors show relatively low impact in both results in the three expert groups, while only EEE3 shows the comparatively high influence. Hence, we speculate that mature regulation would enhance the competitiveness of the entire industrial environment and, consequently, the TC-related factors would become more important. In conclusion, TC1 and TC2 are the most important KSFs for strategic management, while the decision makers in the cell therapy industry can currently focus on TC-related factors for formulating strategy in Taiwan.
However, this study has the following study limitations. First, since we have only used one cell therapy company as a reference, while the cell therapy industry is currently small in Taiwan, some crucial KSFs may have been omitted. Second, only a few respondents were involved in this study. Third, the respondents were only domestic experts, who might lack global insights. In the future, this study hopes to further include a new group of cell therapy regulatory experts, to provide useful insights into the adoption of reliable KSFs in the cell therapy industry.
Footnotes
Acknowledgments
We wish to thank Gwo Xi Stem Cell Applied Technology Co., Ltd, for their constructive suggestions. We thank them and their experts for suggesting directions for improving the paper. We are also thankful for the support for the in-depth interview provided by the Industrial Technology Research Institute (ITRI), Taiwan, and Hualien Tzu-Chi Hospital, Taiwan.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
