Abstract
Thermophilic strains of Bacillus can express enzymes of higher thermal stability, which allows carrying out industrial fermentations under higher temperatures. This lowers the contamination potential, accelerates mixing rates and facilitates the recovery of fermentation end products. The present study was thus designed to isolate and characterize thermophilic Bacillus cultures from soil and compost samples. Forty-two thermophilic Bacillus isolates could be identified employing morphological, physiological and the 16S rRNA gene sequencing analyses. The isolates showed a high degree of biological diversity involving 13 Bacillus species and 1 subspecies but were dominated by Bacillus licheniformis. Phylogenetic analysis of B. licheniformis isolates based on the DNA sequencing of gyrA and rpoB genes presented them in two main genetic groups. Isolates of five thermophilic species including B. licheniformis, Bacillus altitudinis, Bacillus paralicheniformis, Bacillus subtilis and Bacillus thermoamylovorans showed multiple activities to degrade all of cellulose, hemicellulose and lignin. Those multifunctional thermophilic Bacillus isolates can be harnessed in the degradation of plant wastes for the production of biofuels and compost.
Keywords
Introduction
The Bacillus genus involves a diversity of endospore-forming, rod-shaped species that have important biotechnological implementations in health, agriculture and industry. The biological diversity of Bacillus has been broadened by the application of molecular methodologies that allowed the description of a new Bacillus species every week during 2004–2006. 1
The ability of Bacillus species to express significant amounts of different enzymes has presented them as highly relevant industrial workhorse organisms. 2 Various Bacillus species have been already exploited in the production of antibiotics, vitamins and enzymes, and this has been even extended to healing concrete cracks. 3 Bacillus species have been also shown to promote plant growth and provide protection against pests and plant diseases. 4 Several commercial bio-pesticide, fungicide or fertilizer preparations produced by Bacillus have been already marketed. 5
Much focus has been given to the mesophilic Bacillus subtilis as a key species for biological studies and industrial applications. 1 However, this interest has been also extended to the thermophilic species of Bacillus during the last few decades.6–8 This is due to the higher thermal stability of the enzymes produced by thermophilic Bacillaea, which allows carrying out industrial fermentations under higher temperatures. The application of those elevated temperatures lowers the contamination potential, accelerates mixing rates and facilitates the recovery of fermentation end products. 9
Among other potential implementations, thermophilic Bacillus strains have been considered for the biodegradation of plant lignocellulosic materials.6,10 Those materials consist of cellulose, hemicellulose and lignin. They act as a supporting tissue of plant cell wall and present 30%–50% of the total dry weight of plants. 11 Given the huge abundance of plant materials being the largest biomass on Earth, 12 lignocellulosic materials present a sustainable, cost-effective carbon source feedstock for industrial fermentations. However, those materials require extensive physical, chemical and biological pre-treatments to allow their degradation into fermentable substrates. Characterizing thermophilic Bacillus strains that can enzymatically degrade lignocellulosic materials is therefore highly relevant to the exploitation of plant biomass under more hygienic and more efficient fermentation conditions.
The present study was thus designed to examine the diversity and biotechnological properties of thermophilic Bacillus cultures isolated from Egyptian soil and compost samples. We report a diversity of thermophilic Bacillus strains with a highly relevant potential application to the degradation of plant lignocellulosic materials.
Materials and methods
Isolation of thermophilic
Bacillus
A total of 10 soil and 6 compost samples were aseptically collected from various locations in the Nile Delta and commercial compost suppliers in Egypt. A standard plate count procedure was used for the isolation of thermophilic Bacillus cultures from soil and compost samples. An amount of 10 g of each sample was homogenizing in 90 ml of sterile saline solution (0.85% NaCl) followed by heating at 80°C for 10 min. 13 Serial dilutions were prepared and plated onto the standard plate count agar (Oxoid, Basingstoke, UK), followed by incubation at 50°C for 48 h. Suspect colonies were picked up from plates and subjected to morphological screening and biochemical and molecular identification.
Morphological and biochemical identification of thermophilic
Bacillus
Isolates were examined for Gram-staining, morphological shape, spore formation and catalase production. Spore forming, Gram-positive rods that showed a catalase-positive reaction were further biochemically identified using the simplified scheme of Deak and Timar. 14
Molecular speciation of
Bacillus
isolates
Potential Bacillus isolates were further identified by DNA sequencing of the 16S rRNA gene. 6 Purified genomic DNA was prepared from 1 ml of a 24 h culture of each isolate using the GeneJET Genomic DNA Purification Kit (Thermo Fisher Scientific, Waltham, MA, USA). A polymerase chain reaction (PCR) reaction mixture of a total volume of 50 μl was formulated using 25 μl ready-to-use PCR master mix (Thermo Fisher Scientific), 0.2 μM of each of the forward primer (27F), and reverse primer (1492 R), 2.5 μl of 1:10 diluted DNA template and 20.5 μl nuclease-free water. The 27F forward primer (5′-AGAGTTTGATCATGGCTCAG-3′) and 1492R reverse primer (5′-GGTTACCTTGTTACGACTT-3′) were designed to target a 16S rRNA gene sequence of 1465 bp. 6 PCR reactions were conducted at 95°C for 3 min, followed by 35 cycles of 95°C for 30 s, 55°C for 30 s and 72°C for 45 s and 72°C for 5 min using the Veriti PCR thermal cycler (Applied Biosystems, Thermo Fisher Scientific). PCR products were analyzed onto 1.5% agarose in TAE buffer including SYBR safe stain (1 μl/10 μl agarose solution) (Invitrogen, Thermo Fisher Scientific). DNA bands were visualized using the BioDocAnalyzer gel documentation system (Biometra, Goettinge, Germany). Positive samples showing DNA bands of the 1465 bp expected size were further examined for the nucleotide sequence of PCR amplicons after purification using the GeneJET gel extraction kit (Thermo Fisher Scientific). Sanger DNA sequencing was carried out by using the big dye terminator v3.1 cycle sequencing kit (Applied Biosystems) and the 3500 Genetic Analyzer (Applied Biosystems).
Bacillus isolates identified as Bacillus licheniformis were further examined by DNA sequencing of the gyrA and rpoB genes. 15 PCR reactions of a total volume of 50 μl were formulated as described above. For gyrA, a forward primer (5′ CAG TCA GGA AAT GCG TAC GTC CTT 3′) and reverse primer (5′ CAA GGT AAT GCT CCA GGC ATT GCT 3′) were used to target a DNA sequence of 1024 bp. Primers for the rpoB gene involved a forward primer (5′ AGG TCA ACT AGT TCA GTA TGG ACG 3′) and a reverse primer (5′ GTC CTA CAT TGG CAA GAT CGT ATC 3′) targeting a DNA sequence of 808 bp. 15 PCR reactions were conducted at 95°C for 3 min, followed by 35 cycles of 94°C for 30 s, 55°C (for gyrA) or 50°C (for rpoB) for 30 s and 72°C for 2 min and 72°C for 5 min. PCR products were analyzed onto 1.8% agarose in TAE buffer. Purified amplicons were further sequenced employing the Sanger method as described above.
Assessment of the degradation of cellulose, hemicellulose and lignin
Cellulolytic and hemicellulolytic activities of the thermophilic Bacillus isolates were examined by using a defined medium supplemented with avicel (microcrystalline cellulose) or carboxymethyl cellulose (CMC) as cellulose substrates or xylan as a hemicellulose substrate. Isolates were grown from −70°C stock cultures onto the standard plate count agar (Oxoid) at 50°C for 24 h. They were then successively transferred twice into a defined broth medium, designated mT (modified Tansey) broth that was supplemented with 0.5% of avicel (Sigma-Aldrich, St Louis, Missouri, USA), CMC (Sigma-Aldrich) or xylan (TCI, Tokyo, Japan) and incubated at 50°C for 24 h for each transfer. For the analysis of cellulolytic activity, 10 µl from the second transfer was used to inoculate 6 ml of molten mT soft agar containing 6 g agar/l and 0.5% avicel or CMC to be poured onto the surface of hard agar of the mT medium to form a soft-agar overlay. For the examination of hemicellulolytic activity, 10 µl from the second transfer was directly spread onto the surface of a hard agar of the mT medium supplemented with xylan (0.5%). Plates were incubated at 50°C for 48–96 h. Degradation of avicel was indicated by the development of pores within the soft-agar overlay. To examine CMC and xylan degradation, plates were stained with Congo red as described by Sanders and Miller 16 and Teather and Wood. 17 In brief, plates were flooded with Congo red (Sigma-Aldrich) (0.1% wt/vol) for 15 min and the stain was then poured off, followed by the addition of NaCl (1 M) for 15 min. Plates were finally examined for degradation zones around the colonies or color change. The mT defined medium was formulated according to Tansey 18 and Kauri and Kushner 19 with modifications. It consisted of (NH4)2 SO4 (1.5 g/l), K2HPO4 (0.4 g/l), KH2PO4 (0.6 g/l), MgSO4.7H2O (0.2 g /l), KCL (0.5 g/l), NH4Cl (0.268 g/l), yeast extract (0.1 g/l), water (1 l) and pH = 7.0 ± 0.2.
Ligninolytic activities of the isolates were assessed by examining their ability to decolorize methylene blue (MB) and express peroxidase. To examine MB decolorization, a loopful from a 24 h culture grown on standard plate count agar at 50°C was streaked onto potato dextrose agar containing MB (25 mg/l).20,21 Plates were incubated at 50°C for 24–72 h and examined for dye decolorization. Peroxidase expression was assessed by growing isolates on nutrient agar (Oxoid) for 24–48 h at 50°C, followed by the addition of 30 μl of hydrogen peroxide (0.4%, vol/vol) (H2O2) and 1% pyrogallol (1%, vol/vol).22,23 Peroxidase-positive isolates developed yellow-brown colonies on the plates.
Characterization of proteolytic activity
Cultures were grown on standard plate count agar for 24 h at 50°C and then streaked onto the same agar supplemented with 1% sodium caseinate (Sigma-Aldrich) followed by incubation at 50°C for 24–48 h. 24 Plates were examined for the development of degradation zones around colonies.
Results
Diversity of thermophilic
Bacillus
associated with compost and soil
Thermophilic Bacillus isolates could be recovered from soil and compost samples and were identified to the species level by using morphological, biochemical and DNA sequencing of the 16S rRNA gene. The isolates showed a high degree of biological diversity involving 13 Bacillus species and 1 subspecies (Table 1). Two Bacillus species namely B. licheniformis and Bacillus altitudinis dominated this pool of isolates presenting approximately 38% and 14% of the total number of isolates, respectively. The other 12 Bacillus species and subspecies existed in approx. 2%–7% of the isolates (Table 1). The 16S rRNA gene sequencing of the isolates has been successfully submitted online and designated GenBank accession numbers OK189148 through OK189186.
Diversity of thermophilic

*Percent identity is presented as a range of all the identified isolates within the same species.
The prevalence of Bacillus species in soil and compost samples differed from one species to another (Figure 1). Although the isolates of B. licheniformis, Bacillus smithii, Bacillus subtitlis and B. subtitlis subsp. spizizenii equally existed in both soil and compost samples, those of B. altitudinis, Bacillus paralicheniformis and Bacillus amyloliquefaciens were more associated with compost samples. The remaining species of Bacillus aerophilus, Bacillus rugosus, Bacillus aerius, Bacillus thermoamylovorans, Bacillus vallismortis, Bacillus alkalilacus and Bacillus mojavensis were however more frequently isolated from soil samples.

Distribution of thermophilic Bacillus species in the soil (black column) and compost (open column) samples.
Isolates of the most frequently isolated species B. licheniformis were further characterized for their gyrA and rpoB gene sequences. The phylogenetic analysis of those sequences using the maximum likelihood algorithm is presented in Figure 2. It could be seen that the isolates were diverse in their DNA sequences of the gyrA and rpoB genes. Two main groups could be observed for each gene, each of which further diverged into two secondary subgroups involving 5 and 11 isolates, and 13 and 3 isolates based on the gyrA and rpoB sequencing alignment, respectively (Figure 2). It is also noted that three out of five isolates in the first main group of the gyrA sequencing namely 16, 15 and 10 also existed in the first main group of the rpoB sequencing. However, other isolates clustered differently in the two groups of the two genes. This suggests that differences in nucleotide sequence and gene length of gyrA and rpoB resulted in different effects of the evolutionary events on those genes.
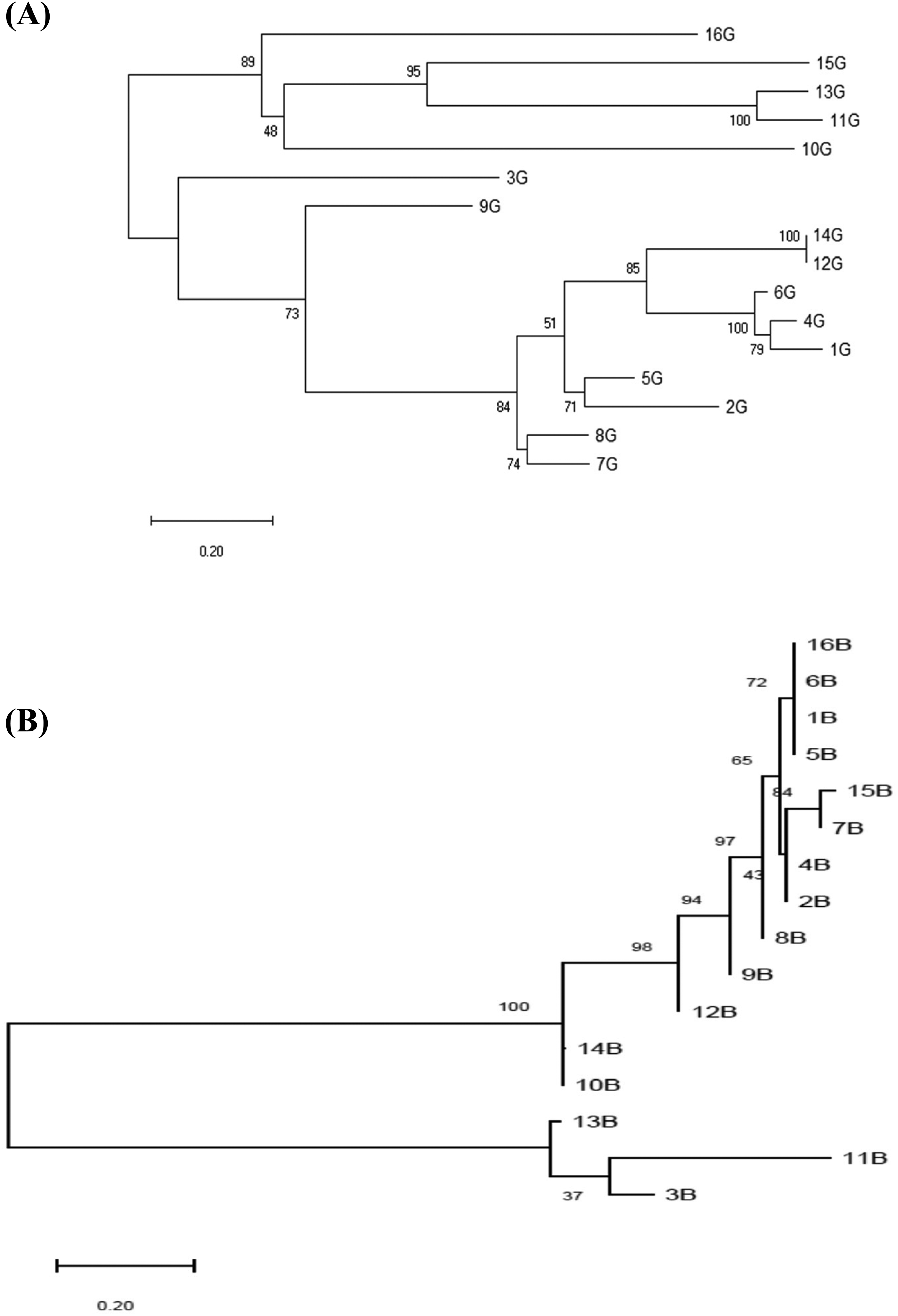
Maximum likelihood-phylogenetic trees of thermophilic Bacillus licheniformis isolates based on DNA sequencing of the gyrA (A) and rpoB (B)genes.
Characterization of the cellulolytic, hemicellulolytic, proteolytic and ligninolytic activities of thermophilic
Bacillus
isolates
All identified thermophilic Bacillus isolates recovered from soil and compost samples were further characterized for their ability to degrade bio-organic materials including cellulose, hemicellulose, proteins and lignin. Those activities were examined by measuring the ability of each culture to degrade relevant substrates or express particular enzymes. Cellulolytic activity was examined by using avicel (microcrystalline cellulose) and CMC as two separate cellulose substrates. Xylan and sodium caseinate were used as substrates for the assessment of hemicellulolytic and proteolytic activities, respectively. Ligninolytic activity was assessed by examining the isolates’ ability to de-colorize MB and express peroxidase.
Proteolytic activity was the most frequently observed in the thermophilic Bacillus isolates compared to cellulolytic and hemicellulolytic activities (Table 2). Although approx. 90% of the isolates showed proteolytic activity, lower numbers of approx. 38%, 52% and 48% were able to degrade avicel, CMC and xylan, respectively. The higher ability of the isolates to hydrolyze CMC and xylan could be attributed to the higher solubility of those substrates in water compared to avicel, which makes them more readily available to the cells in the growth medium. Six of the thermophilic Bacillus species could hydrolyze all of the three substrates avicel, CMC and xylan (Table 2). Those involved B. licheniformis, B. altitudinis, B. paralicheniformis, B. subtilis subsp. spizizenii, B. subtilis and B. thermoamylovorans.
Cellulolytic, hemicellulolytic and proteolytic activities of thermophilic

CMC: carboxymethyl cellulose.
The thermophilic Bacillus isolates variably showed positive reactions in the MB reduction and peroxidase expression examinations used to assess the ligninolytic activity (Table 3). Positive results in both assays could be observed with B. licheniformis, B. altitudinis, B. paralicheniformis, B. aerophilus, B. smithii, B. subtilis, B. aerius and B. thermoamylovorans cultures, which presented them as more potentially ligninolytic cultures compared to other isolates. As described above, isolates of five of those species including B. licheniformis, B. altitudinis, B. paralicheniformis, B. subtilis and B. thermoamylovorans could also express cellulolytic and hemicellulolytic activities (Table 2). This extends the potential of using those multifunctional isolates in various biotechnological applications.
Ligninolytic activities of thermophilic

Discussion
The association of Bacillus with soil and compost has been very frequently reported in previous studies. Bacillus was reported as the most prevailing bacterial genus to be isolated from compost feedstock. 25 This is due to its ability to form endospores that allow its survival under harsh environmental conditions. A number of the Bacillus species that were isolated from the Egyptian soil and compost samples in the present study have been also previously detected in compost and composting materials in different countries including Spain and China. Those involved B. licheniformis, B. altitudinis, B. aerius, B. aerophilus, B. thermoamylovornas, B. amyloliquefaciens, B. smithii and B. subtilis.6,10 However, other species including B. subtitlis subsp. spizizenii, B. paralicheniformis, B. rugosus, B. vallismortis, B. alkalilacus and B. mojavensis were not reported in those previous studies. In consistent with the present work, those studies also showed that B. licheniformis was the most dominant species among other thermophilic Bacillus isolates detected in compost and composting materials. However, B. licheniformis could not be isolated from compost samples prepared in the Netherlands, where B. smithii and B. coagulans were the only thermophilic bacilli to be isolated from those samples. 8 This could be attributed to various environmental conditions and the use of different ingredients in the preparation of compost. Employing different culture media and conditions for the isolation of Bacillus could also contribute to the isolation of different species in those studies. 8
The phylogenetic analysis of the gyrA and rpoB genes of B. licheniformis isolates which was, the most frequently isolated thermophilic Bacillus species in the present study separated those cultures into two main phylogenetic groups. This was consistent with a previous report describing two gyrA and rpoB genotypically distinguished lineages within B. licheniformis strains collected from diverse sources including soil, milk, dairy products, gelatine, flour, infected animals, etc. 26 This could suggest a universal phylogenetic pattern of B. licheniformis based on the gyrA and rpoB gene sequences.
The ability of the thermophilic Bacillus isolates characterized in the present work to degrade lignocellulosic components has been reported in previous studies.6,10,21,23 However, the multifunctionality of several isolates in the present work being able to degrade all of cellulose, hemicellulose and lignin has not been reported before. For instance, while Jurado et al. 6 observed hemicellulolytic activity in isolates of B. licheniformis, B. subtilis and B. altitudinis, none of those isolates showed a cellulolytic activity. The same study did not also report a ligninolytic activity to be associated with isolates of B. licheniformis.
These differences in the degradation capabilities of isolates could be strain-dependent and could be also attributed to the use of different assessment protocols and culture media. The use of rich media for the assessment of the ability of the isolates to degrade cellulose and hemicellulose could lead to false-negative results since cells may favor utilizing other more metabolizable components in those media. The use of two to three subcultures in a defined medium containing cellulose or hemicellulose substrates also improves the ability of cells to degrade those components. We were able to observe the degradation of avicel by the thermophilic Bacillus isolates in the present study only when two successive subcultures were made in a defined broth medium containing avicel followed by plating on the same defined medium (data not shown). The inclusion of Congo red in the agar medium did not allow recognizing the degradation of hemicellulose, which was possible with the addition of this stain into the plates after incubation and the development of Bacillus colonies (data not shown).
Conclusion
The present work shows that soil and compost can serve as a rich source of diverse thermophilic Bacillus strains. The use of those strains improves the performance of biotechnological processes since it allows the use of elevated temperatures, which in turn lower the contamination risk, improve the mixing rates and facilitate the separation of end products. The thermophilic strains characterized in the current study showed multiple degrative activities being able to degrade all of cellulose, hemicellulose and lignin. This can be exploited in the degradation of plant wastes into fermentable sugars to be further converted into biofuels and compost.
Footnotes
Acknowledgements
This paper is based upon work supported by Science, Technology & Innovation Funding Authority (STDF) under grant no. 33557. The authors are grateful to Dr Scott Hamilton-Brehm for his technical help and advice during the realization of this work.
Authors note
WME conceived and designed the study, conducted lab experiments and wrote the manuscript. SAZ conducted lab experiments. Both authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/ or publication of this article.
Funding
The authors received funding from the Egyptian Science, Technology & Innovation Funding Authority (STDF) under grant no. 33557.
