Abstract
The objective of this study is to quantitatively evaluate inhibitory action of halogen-free fire retardants (HFR) on combustion properties and volatile emission of such bituminous components as saturates, aromatics, resins, and asphaltenes (SARA). Thermogravimetry-Fourier transform infrared spectroscopy (TG-FTIR) tests were performed on SARA fractions containing matched fire retardants, respectively, and thermal kinetics parameters based on TG curves and functional and structural indices from FTIR spectra were calculated, respectively. The selected fire retardants have not affected the combustion process of SARA fractions, but the combustion temperature intervals are elevated and combustion progresses are retarded. Also, the char yields of SARA fractions are obviously increased by the matched fire retardants, improving their heat stability. The activation energy is elevated because of the added fire retardants, indicating combustion resistance of SARA fractions become larger. Additionally, the matched fire retardants inhibit the toxic gas emission in the combustion process of SARA fractions, but have few effects on gaseous product constituents. H2O and CO2 are identified as two typical released gases in various combustion phases of each SARA fraction. Finally, the added hydroxide play a role of fire retardants through cooling, dilution, adsorption, and neutralization, and the generated active oxide facilitates the expandable graphite (EG) and matrix to form densified and thick carbon layer. These suppress the volatile emission, and hinder the heat conduction and oxygen supply. Fire retardant composite exhibits the synergistic effect of fire retardancy and smoke inhibition in the combustion process of SARA fractions.
Introduction
As one of important materials of pavement in China, bitumen is extensively applied in the pavement engineering. 1 Bituminous pavement is also utilized in highway tunnels because of better skid resistance, lower noise, shorter construction cycle, easier maintenance, etc. 2 Nevertheless, fire safety problem caused by traffic accidents should be considered because of the tunnel space limitation. When the fire event happens in a road tunnel, the hazardness is more destructive and dangerous than that in open space because the smoke are difficult to quickly dissipate in the relatively confined space. 3 Bituminous pavement was found to be ignitable at high temperature, leading to dumping heat, smokes, and harmful gases which greatly impeded people to escape and rescue. 4
Thus, many researches were the improvement in the fire retardation of bituminous pavement by adding different fire retardants. 5 The first attempts were based on halogen-based fire retardants which played fire retardant effect of gas phase, retarding bitumen combustion. 6 However, halogen-based fire retardants had some obvious drawbacks, for example, smoky, toxic, and corrosive products were released although they showed satisfactory fire retardancy. 7 As a result, the HFR received widespread attention, such as hydrated lime (HL), magnesium hydroxide (MH), aluminium hydroxide (ATH), expandable graphite (EG), calcium carbonate (CaCO3), etc. 8
Some researchers discussed the impacts of HFR on combustion properties of bitumen. Qin et al. 9 provided the useful way to evaluate fire retardancy of self-developed composite fire retardants on bituminous mortars and mixtures. Cong et al. 10 paid attention to the impacts of fire retardants on rheological behavior and long-term aging of modified bitumen. Ren et al. 11 discussed the effects of content and particle size of MH on fire retardancy of bitumen, and found 3000 mesh MH exhibited a satisfactory fire retardancy. Moreover, a series of rapid, cost-effective, and environmentally-friendly approaches were developed for the production of high-performance thin films to reduce the flammability of materials. Sohbatzadeh et al. 12 used the new version of dielectric barrier discharge to deposit the HFR on firwood surfaces.
Additionally, bitumen is identified as a complex mixture of heteroatoms and hydrocarbons. Due to the difference in crude oil source and processing, molecular weight and chemical composition of bitumen are also different. 13 Four generic fractions of SARA are generally extracted from bitumen for the further study of bitumen properties. 14 At the same time, this method is also utilized to investigate the chemical composition of heavy crude oil. 15
Currently, the TG-FTIR combined technique was often conducted to monitor and identify the volatilization law of various gases during the material thermolysis. 16 Stankovikj and Garciaperez 17 developed a methodology to identify the thermolysis process and chemical compositions of oil vapors by TG-FTIR technique. Xu et al. 18 conducted TG-FTIR tests to investigate the inhibiting effect of fire retardant on the volatilization processes of pyrolytic volatile volatilized from bitumen. Mouazen et al. 19 discussed the influence of gamma irradiation on the rheological, structural, and chemical changes of bitumen materials using TG-FTIR method.
At the same time, some kinetic models were also developed to explain the solid and viscoelastic material thermolysis. Elbeyli 20 reported the asphaltite decomposition steps through the kinetic analysis based on thermal experiments. Kissinger 21 developed the kinetics model of bitumen combustion, and calculated kinetics parameters to quantitatively evaluate the fire retardancy of MH on bitumen.
It is noted that the impacts of different HFR on bitumen thermolysis were discussed during the past decades. However, bitumen was considered as the homogeneous entirety. Actually, bitumen is a multi-composition polymer and each SARA fraction shows different chemical structures, pyrolysis, and combustion properties. 22 Few studies involve the impacts of fire retardants on the combustion performance and volatile emission of SARA fractions. Moreover, the inhibitory behaviors of HFR were seldom reported on the basic of combustion behaviors of SARA fractions using TG-FITR technique.
In our previous study, 23 it was demonstrated by experiments that the combustion temperature interval of each SARA fraction exhibited prominent temperature graded distribution characteristics, and the combustion of each SARA fractions was suppressed by selecting the corresponding flame retardant. Through comparing the combustion temperature ranges of bituminous four SARA components with the temperature ranges of thermal decomposition of EG, ATH, MH, and HL, 23 as well as referring to optimum ratio in our previous study, 24 EG/ATH (1/2), EG/MH (1/4), HL and MRP/MH (1/4) were matched for saturates, aromatics, resins, and asphaltenes, respectively. The objective of this study is to quantitatively estimate inhibitory action of HFR on combustion properties and volatile emission of bitumen SARA fractions. Thus combustion behaviors of bitumen are further investigated at the component level, and suitable fire retardants are selected for each SARA fraction to produce a fire retardant composite for bitumen materials.
In this research, the thermolysis temperature intervals of SARA fractions were firstly chosen to match various HFRs. After that, TG-FTIR was utilized to discuss the impacts of selected fire retardants on heat stability, kinetic characteristics, and volatile emission of each SARA fraction in various combustion phases before and after adding fire retardants, further revealing an inhibitory performance of fire retardants on the combustion behaviors of SARA fraction.
Materials and methods
Materials
Each SARA fraction was extracted from bitumen in accordance with ASTM D4124-09, including 10.2% saturates, 36.0% aromatics, 36.1% resins, and 17.1% asphaltenes. Preparation process of SARA fractions, their thermolysis temperature intervals, physical properties of FRs, selection of matched FRs for each component, dosage selection of different FRs, and preparation of component samples containing FRs were described elsewhere in details. 25 Here quantitative evaluation of inhibitory action of HFR on combustion properties and patterns of volatile emission of SARA fraction were investigated in this study.
Characterization method
The TG tester (NETZSCH STA 409 PC/PG) is combined with FTIR tester (TGA/FT-IR Nicolet-IZ10). The 21% O2 and 79% N2 were input into the apparatus at the flow rate of 120 mL/min. The sample of the 10 mg was put into alumina crucibles for TG analysis. Then, the sample was heated in the range of 40°C_800°C at the heating rate of 5°C/min, 10°C/min, and 15°C/min, respectively. The N2 as the purge gas was input to apparatus at a flow rate of 40 mL/min. The volatiles emitted from SARA fractions were simultaneously imported into FTIR tester and the FTIR spectra were collected, and composition changes of released gases were recorded by FTIR spectrometry. The resolution of FTIR is 4 cm−1. Finally, the thermal kinetic parameter (activation energy) during each SARA fraction combustion was calculated using Flynn-Wall-Ozawa method. 21
For the further quantificationally study of the impacts of different fire retardants on the volatile emission, the chemical structure of each SARA fraction can be obtained through analyzing FTIR spectrum at the representative heating rate of 10°C/min. 26 Moreover, the functional and structural indices of main volatiles at the specific bands are calculated in relation to the total area of the FTIR spectrum. 27
Results and discussion
Influences of fire retardants on heat stability of SARA fractions
It is found that TG-DTG curves of each SARA fraction before and after adding different fire retardants are similar at the three heating rates, so the influences of fire retardants on the heat stability of SARA fractions are analyzed at the representative heating rate of 10°C/min. Figure 1 exhibits TG-DTG curves of each SARA fraction before and after adding different fire retardants at the heating rate of 10°C/min. Table 1 depicted the combustion temperature intervals, characteristic temperature at maximum mass loss rate (Tm) and char yields in the combustion phase I and II of each SARA fraction before and after adding fire retardants at the heating rate of 10°C/min.

TG-DTG curves of each SARA fraction before and after adding different fire retardants at the heating rate of 10°C/min.
Combustion temperature intervals, Tms and char yields during each SARA fraction combustion phases before and after adding fire retardants at the heating rate of 10°C/min.

As shown in Figure 1 and Table 1, after the matched fire retardants are added into each SARA fraction, respectively, the combustion temperature intervals (phase I and II) and Tms of each SARA fraction move toward high temperature. The char yields of each SARA fraction are also obviously increased. This phenomenon indicates that the heat stabilities of SARA fractions are elevated dramatically due to the added fire retardants although the increasing trend of heat stability of SARA fractions is not changed in each SARA fraction. Additionally, each constituent in the composite fire retardants is successively thermal decomposed as temperature further rises, showing a synergistic fire retardancy to SARA fractions. 28
Thermal kinetics analysis
The activation energy is selected to assess the impacts of fire retardants on the heat stabilities of SARA fractions (Figures 2 and 3).

TG curves of each SARA fraction at the three heating rates.
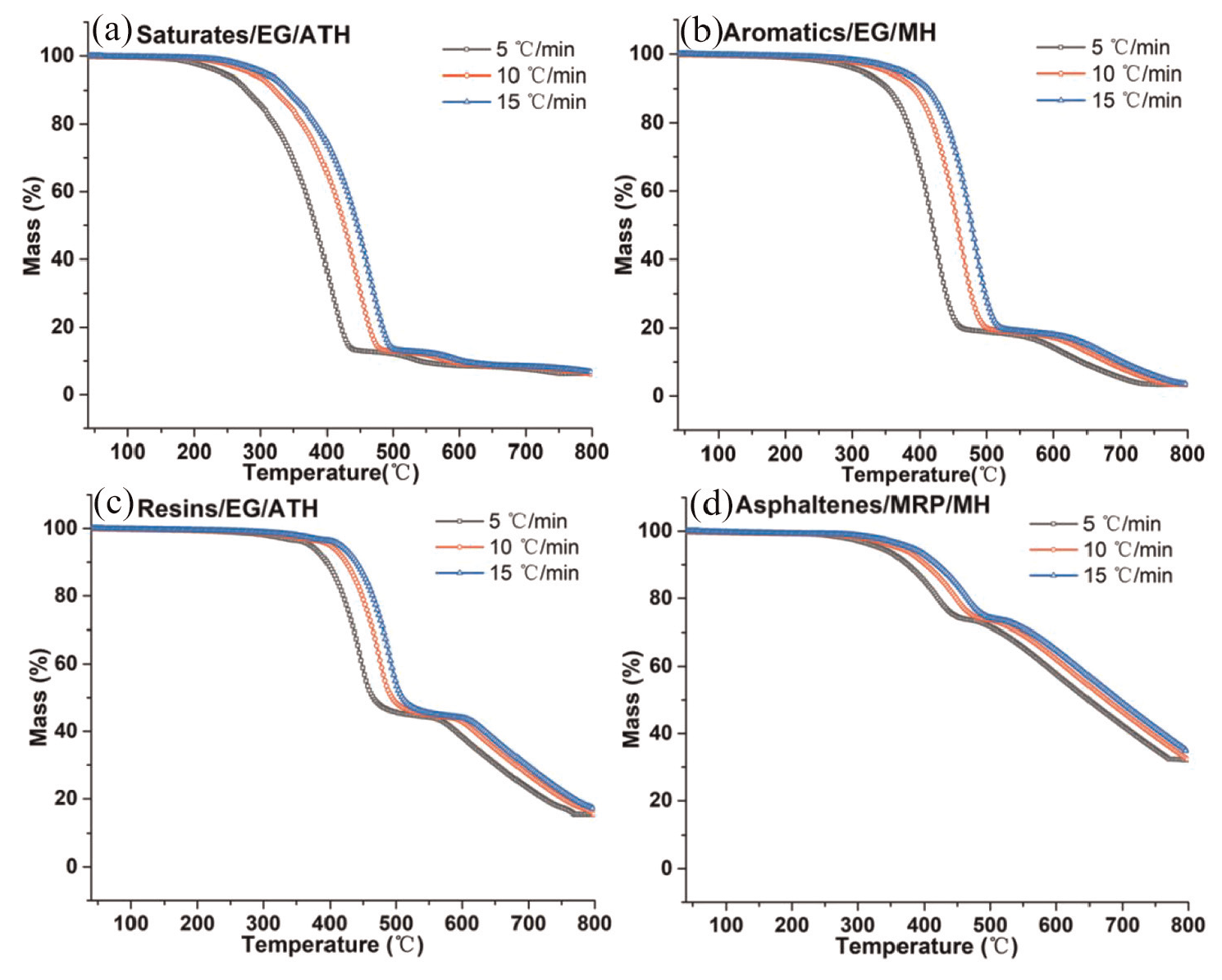
TG curves of each SARA fraction after adding different fire retardants at the three heating rates.
On the basic of TG test results, the activation energy of each SARA fraction combustion phase is calculated using the Flynn-Wall-Ozawa method before and after adding fire retardants. 21 Flynn-Wall-Ozawa can be expressed as equation (1).

Where, β = heating rate, °C/min; A = pre exponential factors, s−1; E = activation energy, kJ/mol; R = universal gas constant, 8.314 J/(mol K); T = peak temperature of TG curves, and G(α) = kinetic model functions.
The calculation results of activation energy values are given in Table 2.
Activation energy in the combustion phases of each SARA fraction before and after adding different fire retardants at the three heating rates of 5°C/min, 10°C/min, and 15°C/min.

Table 2 depicted that the values of activation energy in combustion phase I of each SARA fraction show an increasing trend from saturates to asphaltenes. This phenomenon shows that combustion resistance of SARA fractions increases from saturates to asphaltenes. 29 The heat stabilities of SARA fractions are elevated from saturates to asphaltenes since more heat is needed to activate the SARA fractions thermolysis.
This is because some active functional groups and heteroatoms are peeled off main chains and some unstable weak bonds are easily broken to generate gases in the initial decomposition phase of each SARA fraction, which needs less energy. These lead to a smaller value of activation energy in phase I of each SARA fraction. As temperature gradually increases, the macromolecular chain starts to break and the combustion reactions resistance is increased. This needs more thermal energy, leading to the increase in activation energy.
In phase II of each SARA fraction, it is found that the values of activation energy are larger than those in phase I, and show an increasing trend from saturates to resins. However, the values of activation energy of asphaltenes show no obvious increase when compared with that in phase I. This suggests that more energy is needed in initial combustion phase of asphaltenes, but excessive thermal energy is not needed to maintain the combustion process. Further, the phase state of asphaltenes shows no changes in the whole process of combustion reaction.
However, more energy is needed to maintain the combustion progresses of saturates, aromatics, and resins in phase II when compared with those in the phase I. This may be due to the rearrangement of molecular structures in saturates, aromatics, and resins through the crosslinking polymerization, generating the macromolecular structures and coke. The combustion reaction activity is lowered, thus more energy is needed to activate the thermal reactions.
After the adding different fire retardants into each SARA fraction, respectively, the changing trend of activation energy in both combustion phase I and II of each SARA fraction is not altered. However, the values of activation energy in both combustion phase I and II of each SARA fraction are larger than those before adding fire retardants. This is because the reaction activity of each SARA fraction is lowered, and their thermolysis behaviors are inhibited, and the combustion resistance becomes larger after adding fire retardants.
Additionally, the values of activation energy in the combustion phase I of saturates, aromatics and resins are still relatively smaller after adding fire retardants. The reasons for this are that some low molecular hydrocarbons are released due to unstable weak bond breakage in the combustion phase I although the addition of fire retardants effectively inhibits thermolysis behaviors of SARA fractions, and decreased the heat release amount and gases. This facilitates combustion to accelerate easily.
Finally, the added fire retardants promote the thick cyclization of aromatics and resins during their combustion phase II, generating macromolecular structures or coke due to the crosslinking polymerization. The C–C bonds are oxidized to C–O and C=O, and the combustion reaction activity is lowered so that the values of activation energy of aromatics and resins in their combustion phase II are larger than those of saturates and asphaltenes.
Effects of HFR on gaseous product release of SARA
To discuss the impacts of selected fire retardants on gaseous products from each SARA fraction, FTIR spectra of released gaseous products at the characteristic temperatures (Tms) in the combustion phase I and II of each SARA fraction are selected to study. FTIR curves at the three heating rates are similar, so we only analyze the effects of HFR on gaseous product release of SARA at the representative heating rate of 10°C/min.
Effects of EG/ATH on gaseous product release of saturates
FTIR spectra of released gaseous products in phase I and II of saturates at Tms before and after adding EG/ATH, and the assignment of characteristic FTIR spectrum wavenumbers, functional and structural indices of main volatiles at the specific bands at the heating rate of 10°C/min are depicted in Table 3.
Assignment of FTIR spectrum wavenumbers, functional and structural indices of main volatiles in the combustion process of saturates before and after adding EG/ATH at the heating rate of 10°C/min.

From Table 3, main released gases in the combustion process of saturates are H2O, CO2, CH4, CO, aldehydes, alcohols, olefins, a small number of ether, ester, ketone, etc. The gas release in phase I is more than that in the phase II. The band at 921 cm−1 is because of the out-of-plane bending vibration of O–H from the endothermic dehydration of ATH. This indicates that the ATH is decomposed to play a role of fire retardant. In addition, FTIR spectra are basically similar before and after adding EG/ATH, and no new bands are observed. This suggests that there is no chemical reaction between saturates and EG/ATH composites in the combustion process.
However, it is noted that the intensity of transmittance peaks after adding EG/ATH is larger than that before adding EG/ATH in saturates. This indicates that the concentration of released gaseous products becomes higher after adding EG/ATH. The reason for this is that EG/ATH can only elevate the initial decomposition temperature and postpone the release of volatile from saturates, but cannot obviously reduce the release amount of volatiles. 30 Another reason is the thermolysis of EG and ATH to increase the volatile concentration. However, the gaseous products from EG and ATH are nontoxic, and dilute flammable volatile concentration. 28
Especially, the addition of fire retardants EG/ATH in saturates leads to the increase in the content of hydrocarbon molecules in the released volatile compounds. This is because EG contains a lots of C radicals, and ATH contain a great deal of H radicals at high temperature, and these C and H radicals may react each other, or react with the C and H radicals in bitumen to produce CH molecules at high temperature.
Finally, the characteristic band of the out-of-plane bending vibration of O–H at 921 cm−1 is still found because of the endothermic dehydration of ATH in the combustion phase II. This indicates that partial ATH continues to decompose in the phase II, which is conductive to form a densified and thick superposed carbon layer through the synergistic effects of ATH decomposition and EG expansion. 30 The carbon layer prevents the volatile emission and improves the heat stability of saturates.
Effects of EG/MH on gaseous product release of aromatics
FTIR spectra of released gaseous products in phase I and II of aromatics at Tms before and after adding EG/MH, and the assignment of characteristic FTIR spectrum wavenumbers, functional and structural indices of main volatiles at the specific bands at the heating rate of 10°C/min are summarized in Table 4.
Assignment of FTIR spectrum wavenumbers, functional, and structural indices of main volatiles in the combustion process of aromatics before and after adding EG/MH at the heating rate of 10°C/min.

As shown in Table 4, the main volatiles of aromatics in the phase I are H2O, CO2, Olefin, CO, methane, SO2, aldehydes, alcohols, phenols, and a small amount of ether, ester, ketone, etc. Compared with that in the phase I, the release amount of main products is less in the phase II, which are H2O, CO2, CO, and small molecular hydrocarbons, alcohols, phenols, etc. This is because the active MgO from MH thermolysis accelerates the EG intumescent layer to carbonize, generating a superposed and thicker carbon layer. 31 The carbon layer effectively hinders to volatile releasing and increases heat stabilities of aromatics, revealing a good fire retardancy, and smoke suppression.
From Table 4, it is found that the release amount of CO2 and SO2 decreases after adding EG/MH. This is due to the fact that the generated MgO adsorbs some CO2 and SO2, and the generated H2O increases the functional index at the bands of 4000–3500 cm−1 in the combustion phase II of aromatics after adding EG/MH. Additionally, from Table 4, the characteristic band at 921 cm−1 due to MH dehydration is observed in phase I of aromatics, but this band disappears in phase II. This suggests that MH is decomposed completely in the combustion phase I. Finally, the content of small CH molecules in the gas phase is increased after the addition of EG/MH. This is because EG contains a lots of C radicals, and MH includes many H radicals at high temperature, these C and H radicals may react each other, or react with the C and H radicals in bitumen to produce CH molecules at high temperature.
Effects of HL on gaseous product release of resins
FTIR spectra of released gaseous products in phase I and II of resins at Tms before and after adding HL, and the assignment of characteristic FTIR spectrum wavenumbers, functional and structural indices of main volatiles at the specific bands at the heating rate of 10°C/min are summarized in Table 5.
Assignment of FTIR spectrum wavenumbers, functional, and structural indices of main volatiles in the combustion process of resins before and after adding HL at the heating rate of 10°C/min.
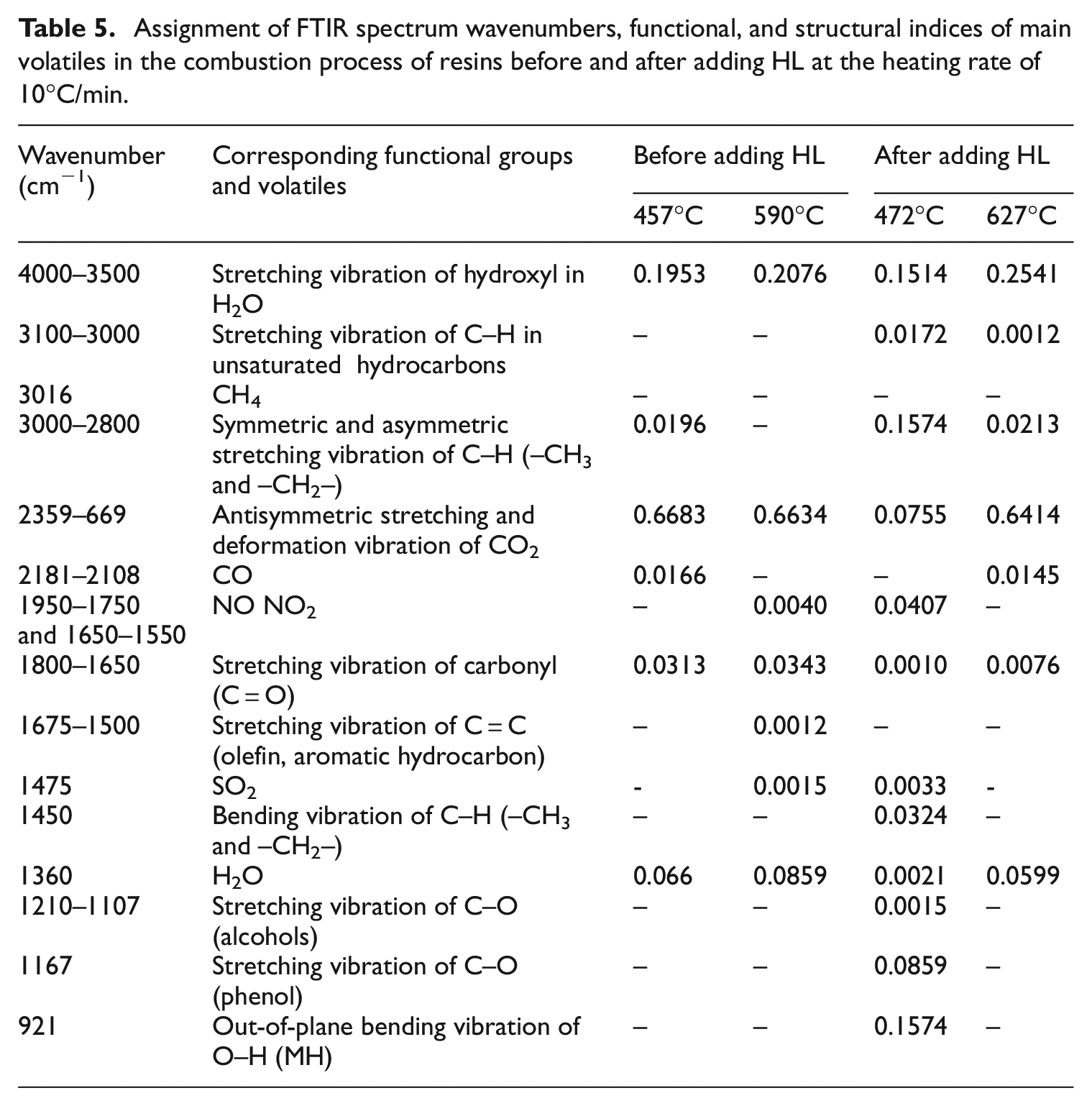
It is found from Table 5 that there are almost no NO, SO2, NO2, aldehydes, phenols, alcohols, and other gases and smoke in the volatiles although the gas release in phase I is more after adding HL than that before adding HL. It indicates that HL inhibits the combustion of resins in the phase I. It is due to the fact that the CaO from HL decomposition promotes the resins to carbonize. 32 The formed carbon layer effectively prevents the release of gases, and improves the fire retardancy and smoke inhibition and heat stability of resins. 33
As shown in Table 5, the release amount of H2O is increased in the combustion phase II after adding HL. This is because H2O is not only derived from HL decomposition, but also from resins combustion. More H2O can dilute the concentration of flammable volatiles and lower toxicity. However, the release amount of CO2 is decreased in the same phase because of the adsorption of generated CaO.
After adding HL into resins, the main gaseous products are H2O and CO2 in addition to a small number of small molecules such as CO and alkanes in phase II. Further, the characteristic band at 921 cm−1 of out-of-plane bending vibration of O-H is not observed in the phase II, reflecting HL is completely broke down to form the carbon layer. Particularly, the content of small CH molecules in the gas phase is increased after the addition of HL. The reason is many C radicals are produces during the bituminous combustion, and HL contains a lot of H radicals at high temperature. These C and H radicals may react to produce CH molecules at high temperature.
Effects of MRP/MH on gaseous product release of asphaltenes
FTIR spectra of released gaseous products in phase I and II of asphaltenes at Tms before and after adding MRP/MH, and the assignment of characteristic FTIR spectrum wavenumbers, functional and structural indices (I) of main volatiles at the specific bands at the heating rate of 10°C/min are depicted in Table 6.
Assignment of FTIR spectrum wavenumbers, functional, and structural indices of main volatiles in the combustion process of asphaltenes before and after adding MRP/MH at the heating rate of 10°C/min.

From Table 6, the main volatiles in the combustion phase I of asphaltenes are H2O, CO2, olefins, aromatics, methane, CO, and trace amounts of SO2, carboxylic acid, etc. However, it is found that H2O in the combustion phase I is obviously reduced after adding MRP/MH. This is due to the fact that MRP is broke down to generate P2O5 which reacts with H2O to generate the dehydrated metaphosphoric acid. 34 As a result, H2O is consumed during the reaction, and the densified carbon layer is formed on top of asphaltenes, which prevents the heat to transfer and inhibits flammable volatiles to release.
As shown in Table 6, H2O, CO2, CO, and trace amounts of low molecular alkanes, olefins, aromatics, etc. are typical released products in the combustion phase II of asphaltenes. The toxic volatile release is reduced after the addition of MRP/MH, for example, SO2, NO, NO2, etc. This is because MH is decomposed to absorb the heat from the asphaltenes and to form a carbon layer on top of asphaltenes which hinders the volatiles to release. MH and MRP show synergetic effects of fire retardancy and smoke inhibition during the asphaltenes combustion.
Finally, the content of small CH molecules in the gas phase is increased after the addition of MRP/MH. This is because MRP accelerates bitumen carbonization and a lot of C radicals are simultaneously produced, as well as MH contains a great deal of H radicals at high temperature. These C and H radicals may react each other, or react with the C and H radicals in bitumen to produce CH molecules at high temperature.
Conclusions
Five HFR were chosen to match the combustion temperature interval of each SARA fraction. The TG-FTIR was utilized to investigate the impacts of various fire retardants on combustion behaviors and volatile emission of each SARA fraction, and their thermal kinetic parameters were also calculated. The following conclusions are obtained as follows.
The added fire retardants have not affected the combustion process of each SARA fraction, still including two typical combustion phases. Whereas, the combustion temperature intervals (phase I and II) of each SARA fraction are increased due to their matched fire retardants, respectively, and the combustion progress of each SARA fraction is also retarded.
The char yield of each SARA fraction is obviously increased by its matched fire retardants. Fire retardant composite show the synergistic effects of both fire retardancy and smoke inhibition in the combustion process of SARA fractions. The heat stabilities of SARA fractions are improved dramatically due to the addition of selected fire retardants.
The matched fire retardants increase the activation energies in both combustion phase I and II of SARA fractions. The combustion resistance of each SARA fraction becomes larger after adding the fire retardants. However, the changing trend of activation energy in both combustion phase I and II of each SARA fraction is not changed after adding fire retardants.
The selected HFR inhibit the toxic gases release in the combustion process of each SARA fraction, but have little impact on the volatile emission of each SARA fraction. H2O and CO2 are the two main volatiles in various combustion phases of SARA fractions before and after adding matched fire retardants.
The added hydroxide plays a role of fire retardants through the impacts of cooling, dilution, adsorption, and neutralization, and the generated active oxide accelerates the matrix and EG intumescent layer to carbonize. These inhibit volatile emissions, hinder the heat conduction, and lower the combustion rate, and elevate the combustion temperature intervals of SARA fractions.
Supplemental Material
sj-docx-1-sci-10.1177_00368504211035215 – Supplemental material for Inhibitory action ofhalogen-free fire retardants on combustion and volatile emission of bituminous components
Supplemental material, sj-docx-1-sci-10.1177_00368504211035215 for Inhibitory action ofhalogen-free fire retardants on combustion and volatile emission of bituminous components by Wenjing Xia, Suying Fan and Tao Xu in Science Progress
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: Authors would like to thank the financial support from Provincial Natural Science Foundation of Jiangsu (no. SBK2021045525).
Author biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
