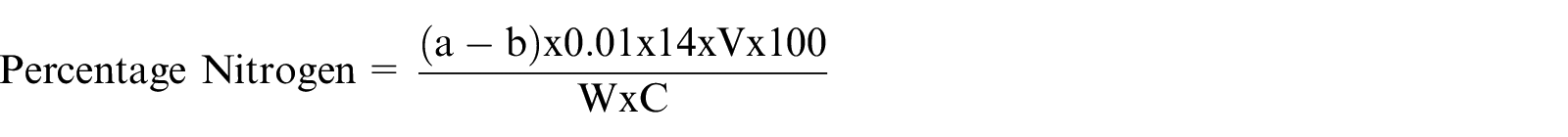Abstract
The aim was to evaluate the amino acid compositions of three commonly consumed leafy vegetables (Solanum aethiopicum, Amaranthus hybridus, and Telfairia occidentalis) in Abakaliki, Ebonyi State. Leafy vegetables are important protective foods and beneficial for the maintenance of healthy living and prevention of diseases. The fresh leaves of A. hybridus, T. occidentalis, and S. aethiopicum were air-dried under room temperature for 1 week. The dried samples were further milled into a fine powder using a mechanical grinder and were stored in an air-tight plastic container. Amino acid content was determined using an applied Bio-system (phenylthiohydantoin, PTH) amino acid analyzer. Among amino acids determined in the vegetables, glutamic acid had the highest value with 12.59, 11.20, and 11.96 g/100 g protein, which was followed closely by leucine with 9.81, 7.94, 9.28 g/100 g protein, and aspartic acid with 8.99, 8.62, and 9.74 g/100 g protein in S. aethiopicum, A. hybridus, and T. occidentalis, respectively on dry weight bases. The leaf that contained the highest total amino acid (TAA) was S. aethiopicum with 88.69 g/100 g protein followed by T. occidentalis with 80.39 g/100 g protein while A. hybridus being the lowest, had 73.38 g/100 g protein. The limiting essential amino acid was tryptophan with 1.98 g/100 g protein while leucine with 9.0 g/100 g protein was the most abundant TAA. The percentage concentration of different groups of amino acid in vegetables revealed that total essential amino acid (TEAA) had 54.85%, total non-essential amino acid (TNEAA) had 48.27%, total neutral amino acid (TNAA) had 22.24%, total acidic amino acid (TAAA) had 32.48%, total basic amino acid (TBAA) had 11.53%, total aromatic amino acid (TArAA) had 11.89% while total sulfur amino acid (TSAA) had 3.94%. The results indicate that the vegetables studied are rich in essential amino acids and could serve as a good source of quality protein. Therefore, they could be recommended as food supplements, especially when animal proteins become more expensive as a source of protein.
Introduction
Vegetables are the edible part of plants consumed wholly or in parts, raw or cooked as part of the main dish or salad. Indeed, they are the cheapest and most readily available sources of protein, vitamins, minerals, and essential amino acids. 1 The varieties of leafy vegetables utilized are diverse, ranging from leaves of an annual shrub to leaves of the tree. 2 Leafy vegetables are the key sources of natural antioxidants such as different types of pigments like chlorophylls,3,4 betacyanins, 5 betaxanthins, 6 betalains, 7 carotenoids, 8 beta-carotene, 9 xanthophylls, 10 phenolic acids, including different hydroxybenzoic acids and hydroxycinnamic acids,11–13 and flavonoids, including flavonols, flavones, flavanols, flavanones.14–16 These are also essential sources of minerals, such as microelements including, K, Ca, Mg, P, and S 17 and microelements including Fe, Cu, Mn, Zn, Na, Mo, B, 18 protein, 19 carbohydrates, 20 dietary fiber, 21 and vitamins,22,23 for human nutrition. Leafy vegetables are important protective food, highly beneficial for the maintenance of health and prevention of diseases. They contain valuable food ingredients that can be utilized to build up and repair body tissues. The fight against malnutrition and under-nourishment continues to be a basic goal of development, and a variety of strategies are applied. 24 Strategies based on nutrient-rich food like vegetables are considered essential. 25 Studies have repeatedly shown that increasing colon and stomach cancer correlate with low vegetable meals, 26 suggesting that vegetables may help resist them. 26 There is also increasing epidemiological evidence in favor of an association between nutrition and susceptibility to infection. Health disorders such as appendicitis, hemorrhoid, gall stones, heart disease, obesity, and constipation can be corrected or treated by copious consumption of vegetables. 27 Therefore, consuming leafy vegetables may be the basic strategies for fighting against poverty, hunger, malnutrition, and undernourishment. 28
The present study analyzed amino acid profiles of three vegetable leaves commonly consume in Ebonyi State. Amaranthus hybridus, known as “Inene” in the Eastern part of Nigeria, belongs to the family of Amaranthaceae. 29 Fluted pumpkin (Telfairia occidentalis), known as “Ugu” in the Eastern part of Nigeria, is a known West Africa leafy vegetable, and it has edible seeds and belongs to the family of Cucurbitaceae. 30 Solanum aethiopicum (Garden egg leaf) is commonly known as “Akwukwo anara” in the Eastern part of Nigeria. 31 Despite the high availability of varieties of indigenous, wild, or semi-wild, neglected, and underutilized vegetables in Nigeria, 32 which are good sources of protein and essential amino acids needed in the body, many of such plants have been unidentified. The inadequate data on the chemical composition of leafy vegetables in Nigeria has limited the prospects and their utilization in food and nutrition for dietary counseling. This study, therefore, was carried out to generate information on protein content and amino acid composition of important underutilized and neglected vegetable food in Nigeria, including S. aethiopicum, A. hybridus, and T. occidentalis leaves.
Aim and objectives
This research was aimed at determining the amino acid profiles of S. aethiopicum, A. hybridus, and T. occidentalis leaves: commonly consumed vegetables in Ebonyi State, Nigeria.
Materials and methods
Biological material
S. aethiopicum, A. hybridus, and T. occidentalis leaves were used for this study. They were purchased from Abakpa market in Abakaliki, Ebonyi State, Nigeria, and identified by Prof. S. C. Onyekwelu of Applied Biology Department, Ebonyi State University, Abakaliki, Nigeria.
Solvents and reagents
All the solvents and reagents used were of analytical grades. They include: chloroform, methanol mixture, concentrated sulfuric acid, sodium sulfate (Na2SO4), copper sulfate (CuSO4), selenium oxide (SeO2), 2% boric acid, bromocresol green, methyl red indicator, 6 N HCl, acetate buffer (pH 2.0), and 0.01 N hydrochloric acid.
Preparation of plant materials
The fresh leaves of A. hybridus, T. occidentalis, and S. aethiopicum were air-dried under room temperature for 1 week. The dried samples were further milled into a fine powder using a mechanical grinder and stored in an air-tight plastic container for analysis.
Determination of amino acid profile
The amino acid profiles of the vegetable leaves were determined using the method of Benitez. 33 The vegetable leaves were dried to constant weight and defatted using chloroform and methanol of ratio 2:1. They were evaporated in a rotary evaporator and loaded into the Applied Bio-Systems, PTH Amino Acid Analyzer. About 300 mg of the vegetables were put in an extraction thimble and extracted for 15 h in a Soxhlet extraction apparatus. 34 The tryptophan in the samples was hydrolyze using 4.2 N sodium hydroxide. 35
Determination of protein content
Principle
The Kjeldahl method, developed in the late 1800s, consists of three basic steps: digestion of the sample with a catalyst, which results in conversion of nitrogen to ammonia; a distillation of the ammonia into a trapping solution and quantification of the ammonia by titrating with a standard solution.
Procedure
One gram of the ground sample was put in a Kjeldahl flask, followed by 2 ml of digestion mixture. The flask and its content were subjected to heat in a fume cupboard until the solution became clear. The colorless solution formed was made up to 20 ml mark with distilled water. Five milliliter of the clear solution was added to two separate test tubes, followed by 0.5 ml Nessler’s reagent. Finally, the absorbance was read immediately at a wavelength of 470 nm against a reagent blank. The blank was prepared with 5 ml distilled water and 0.5 ml of Nessler’s reagent. This procedure was repeated for the remaining two vegetable samples.
Calculation:
Where: A = vol (ml) of 0.2 of solvent used sample titration
B = vol (ml) of 0.2 of blank titration
N = normality of solvent used
W = weight of sample (g)
1.4007 = atomic weight of sample
6.25 = protein nitrogen conversion factor.
Nitrogen determination
A small amount (115 mg) of ground sample was weighed, wrapped in Whatman filter paper, and put in the Kjeldahl digestion flask followed by 10 ml concentrated sulfuric acid. 0.5 g of the catalyst mixture containing sodium sulphate (Na2SO4), copper sulphate (CuSO4), and selenium oxide (SeO2) in the ratio of 10:5:1 was added into the flask to facilitate digestion before six pieces of anti-bumping granules were added to the mixture.
The flask was then put in the Kjeldahl digestion apparatus for 3 h until the liquid turned light green. The digested sample was cooled and diluted with distilled water to 100 ml in a standard volumetric flask. An aliquot (10 ml) of the diluted solution with 10 ml of 45% sodium hydroxide was put into the Markham distillation apparatus and distilled into 10 ml of 2% boric acid containing four drops of bromocresol green/methyl red indicator until about 70 ml of distillate was collected. Then the distillate was titrated with standardizing 0.01 N hydrochloric acid to the gray-colored endpoint.
Where:
a = titre value of the digested sample
b = titre value of blank sample
v = volume after dilution (100 ml)
W = weight of dried sample (mg)
C = aliquot of the sample used (10 ml)
14 = nitrogen constant in mg.
Hydrolysis of the sample
The defatted sample was weighed into a glass ampoule containing 7 ml of 6 N HCl and was ejected by passing nitrogen into the ampoule (this is to avoid possible oxidation of some amino acids during hydrolysis, e.g. methionine and cystine). The glass ampoule was then sealed with Bunsen burner flame and put in an oven preset at 105°C ± 5°C for 22 h. The ampoule was allowed to cool before broken it open at the tip, and the content filtered to remove the humins. 6 N HCl was used to destroy the tryptophan during the hydrolysis. The filtrate was then evaporated to dryness using a rotary evaporator. The residue was dissolved with 5 ml to acetate buffer (pH 2.0) and stored in plastic specimen bottles kept in the freezer.
Loading of the hydrolysate into the analyzer
Sixty microliter of the hydrolysate was loaded into the analyzer and then dispensed into its cartridge. The analyzer separates and analyzes free acidic, neutral, and basic amino acids of the hydrolysate.
Method of calculating amino acid values
An integrator attached to the Analyzer calculates the peak area proportional to the concentration of each of the amino acids.
Statistical analyses
The mean, standard deviation, and coefficient of variation were carried out using SPSS statistical software, version 17.0 (SPSS Inc., Chicago, Illinois, USA).
Results
Table 1 shows the amino acid (AA) composition for each sample. Glutamic acid had the highest concentration of 12.57, 11.20, and 11.96 g/100 g protein in S. aethiopicum, A. hybridus, and T. occidentalis leave, respectively. On the other hand, while leucine an essential amino acids were the second-highest concentrated amino acid in S. aethiopicum and T. occidentalis leaves with the values of 9.8 and 9.28 g/100 g protein respectively, aspartic acid was the third-highest amino acid in S. aethiopicum, A. hybridus, and T. occidentalis leaves followed by arginine. Generally, the essential amino acid levels are high relative to nonessential amino acids, as shown in Table 1. The results revealed the concentrations of the essential amino acids in the three vegetables analyzed in the following order: leucine > valine > arginine > lysine > isoleucine > threonine and they are higher than nonessential amino acids except glutamic and aspartic acids that were found to be high as shown in Table 1. The percentage coefficient of variation (CV%) of the amino acid values was generally low except methionine with a CV% value of 80.18, while the rest of the CV% values ranged from 54.5 to 15.44, indicating the closeness of the amino acid values in the three vegetables to each other. The TAA values of S. aethiopicum, A. hybridus, and T. occidentalis leaves were 88.69, 73.38, and 80.39, respectively. Tables 2 and 3 show the concentrations of a total amino acid (TAA), the total essential amino acid (TEAA), the total acidic amino acid (TAAA), the total neutral amino acid (TNAA), the total sulfur amino acid (TSAA), the total aromatic amino acid (TArAA), and their percentage levels.
Amino acid composition of dry weight of A. hybridus, T. Occidentalis, and S. aethiopicum in g/100 g protein.

Essential amino acid.
Concentrations of essential, non-essential, acidic, neutral, sulfur, and aromatic amino acid composition of dry weight of A. hybridus, T. occidentalis, and S. aethiopicum in g/100 g protein.
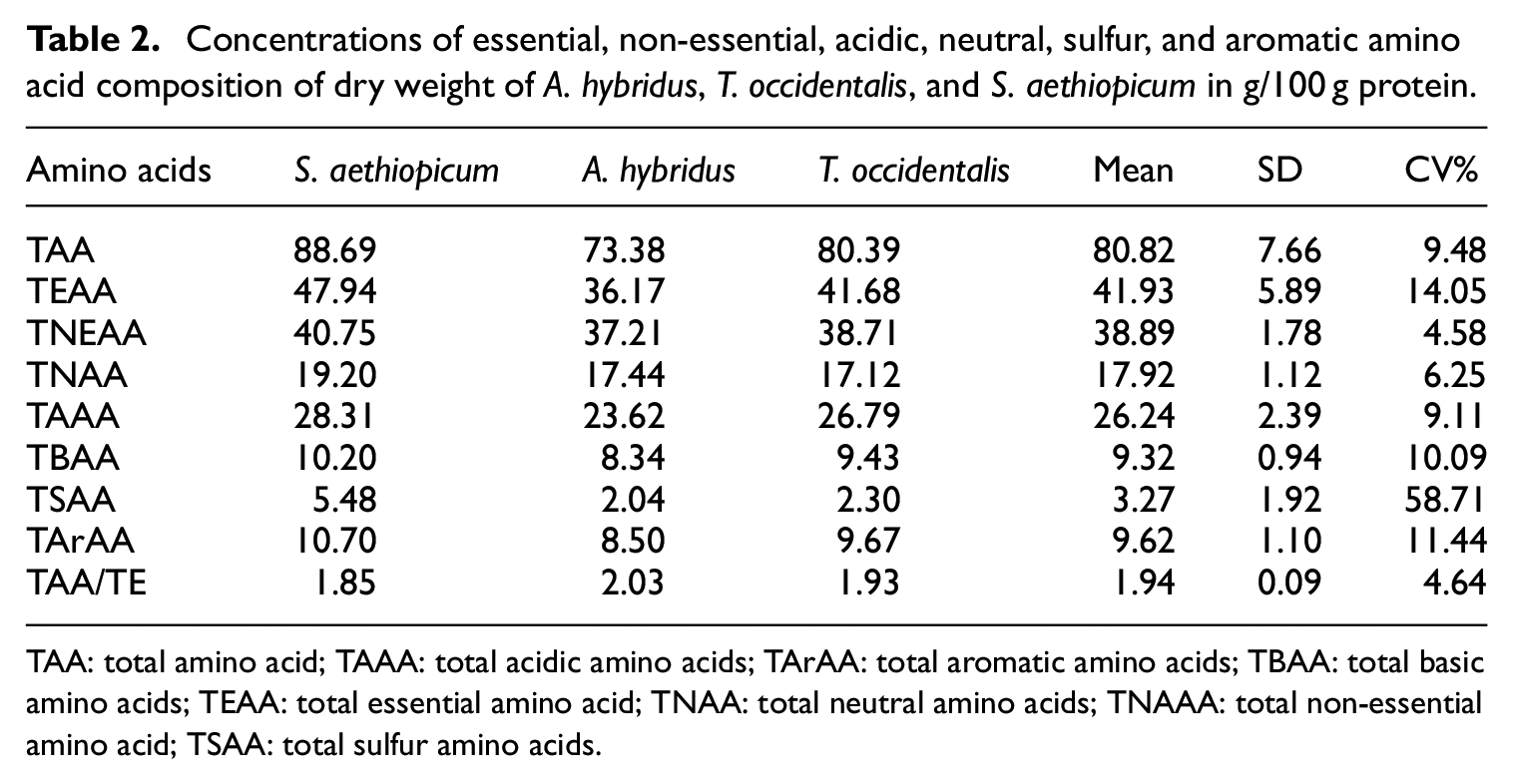
TAA: total amino acid; TAAA: total acidic amino acids; TArAA: total aromatic amino acids; TBAA: total basic amino acids; TEAA: total essential amino acid; TNAA: total neutral amino acids; TNAAA: total non-essential amino acid; TSAA: total sulfur amino acids.
Percentage concentrations of essential, non-essential, acidic, neutral, sulfur, aromatic, and cysteine in total sulfur amino acid in g/100 g crude protein of A. hybridus, T. occidentalis, and S. aethiopicum.

%: percentage; TAA: total amino acid; TAAA: total acidic amino acids; TArAA: total aromatic amino acids; TBAA: total basic amino acids; TEAA: total essential amino acid; TNAAA: total non-essential amino acid; TNAA: total neutral amino acids; TSAA: total sulfur amino acids.
Discussion
This study showed that S. aethiopicum, A. hybridus, and T. occidentalis leaves have more concentration of glutamic acid with 12.57, 11.96, and 11.20 g/100 g protein followed by leucine and aspartic acid, respectively. Aspartic acid is non-essential and acidic amino acid; 36 leucine is an essential amino acid and is a neutral amino acid. 36 The results also revealed a high concentration of other essential amino acids. The concentration of the essential amino acids in the three vegetables analyzed is in the order: leucine > valine > arginine > lysine > isoleucine > threonine. Although there were high concentrations of essential amino acids in the samples, the concentration of glutamic and aspartic acids was higher, as shown in Table 1. The highest essential amino acid (EAA) was leucine (9.81, 7.94, and 9.28 g/100 g) in S. aethiopicum, A. hybridus, and T. occidentalis leaves, respectively, as shown in Table 2.
Again, the result indicated that total amino acid (TAA) values of S. aethiopicum, A. hybridus, and T. occidentalis leaves were 88.69, 73.38, and 80.39, respectively, as shown in Table 2. Interestingly, this is in line with the report of Olorunfemi et al. 24 that reported the same in Moringa oleifera leaves (76.4 g/100 g), root (70.9 g/100 g), and stem (65.4 g/100 g). The result is also in agreement with the report of Omoyeni et al., 38 which revealed a high value of glutamic acid, aspartic acid, and leucine and low level of cysteine, histidine, methionine, and serine in dried leaves of Melanthera scandens. Amino acid compositions of Luffa cylindrica seed, as reported by Oyetayo and Ojo, 39 showed that Luffa cylindrica seed contains a high proportion of essential amino acids and non-essential amino acid. Aremu et al. 40 made a similar observation in L. cylindrical seed kernel.
Písaříková et al. 41 reported high content of Lys and Arg in both heat-treated and untreated grains, as well as a satisfactory amount of Cys and lower levels of Met, Val, Ile, and Leu in a grain of six selected amaranthus varieties and four species: Amaranthus cruentus, A. hypochondriacus, A. caudatus, and A. hybridus, cultivated in the Czech Republic. This result was followed by an essential amino acid index (EAAI) value of 90.4%, indicating the high nutritional quality of amaranthus protein, which is almost comparable with egg protein. Heat treatment by popping at 170°C–190°C for the 30s resulted in decreased EAAI to 85.4%. 41 Hamad and Idres 42 reported that amino acid contents in Thyme, Rosemary, and Salvia showed a different level from plants to another, but, generally, the high level was (methionine) (11,263,834.14 µM) found in salvia stems while the lower value was in thyme. Moran-Palacio1 et al. 43 reported 15 amino acids in plant samples of some medicinal plants from Southern Sonora, Mexico, with aspartic acid (Asp), glutamic acid (Glu), serine (Ser), glycine (Gly), alanine (Ala), and leucine (Leu) (43.55, 44.84, 29.60, 58.17, 43.05, and 38.73 nM, respectively). A similar study by Akubugwo et al. 44 reported seventeen amino acids (isoleucine, leucine, lysine, methionine, cysteine, phenylalanine, tyrosine, threonine, valine, alanine, arginine, aspartic acid, glutamic acid, glycine, histidine, proline, and serine) in Amaranthus hybridus leaves. Furthermore, Rita et al. 45 reported that the weedy species (A. viridis, A. blitum L., and A. dubius) have the highest leaf protein and TAA values. The leaf protein and TAA in most of the weedy types range from 12 to 29 g/100 g DM and 84 to 93 g/100 g DW protein, respectively. 45
Robust evidence confirms the level of Arginine (1.77–8.22 g/100 g) crude protein that is essential for children, 46 and reasonable levels were present in the two samples (Figure 1). The lysine contents of the vegetables (2.17–3.53 g/100 g cp) were about one-half to the content of the reference egg protein (6.3 g/100 g). 46 Therefore, any of the samples will serve as an average source for the amino acid. The study further revealed that TEAA in S. aethiopicum, A. hybridus, and T. occidentalis leaves were 47.94, 36.17, and 41.68 g/100 g protein, respectively. This result is comparable to some literature values of nonconventional meat sources (g/100 g protein): 38.6 for heart and 42.2 for the liver of African giant pouched rat (Cricetomys gambianus). 40 The contents of TSAA of the three vegetables were generally lower than the 5.8 g/100 g cp recommended for infants. 47 The ideal protein range of TArAA is 6.8–11.8 g/100 g, 31 whereas S. aethiopicum, A. hybridus, and T. occidentalis leaves have values 10.70, 8.50, and 9.67 g cp, respectively, indicating that these vegetable TArAA values fall within the ideal protein value. The aromatic amino acid such as tyrosine is a precursor of epinephrine and thyroxine. 48

The protein contents of Solanum aethiopicum, Amaranthus hybridus, and Telfairia occidentalis leaves were 6.96 ± 0.8, 17.96 ± 0.96, and 4.64 ± 0.55% respectively. Data are shown as mean ± SD (n = 3).
The percentage ratios of TEAA to the TAA were 53.11, 49.29, and 51.85 in S. aethiopicum, A. hybridus, and T. occidentalis leaves, respectively. These are strongly comparable, relative to that of an egg (50%), 47 43.6% reported for pigeon pea flour, 48 43.8%–44.4% (beach pea protein isolate), 49 46.2% (liver), and 46.3% (heart) reported for African giant pouched rat (Cricetomys gambianus). 46 The percentage ratios of TEAA to the TAA in the samples were well above the 39% considered to be adequate for ideal protein food for infants,50,51 26% for children, and 11% for adults. 47
Conclusion
From the results obtained from this study, Solanum aethiopicum, Amaranthus hybridus, and Telfairia occidentalis leaves, vegetables commonly consumed in Ebonyi State, Eastern Nigeria, are richly endowed with numerous essential amino acids. Therefore, they could be recommended as food supplements, especially when animal proteins become more expensive as a source of protein.
Footnotes
Author contributions
P. M. Aja, E. U. Ekpono, and N. C. Asouzu conceived and designed the study; P. M. Aja, B. A. Ale, L. Aja, E. U. Ekpono, I. Nwite, and A. Njoku carried out the laboratory experimental work; P. M. Aja, E. U. Ekpono, L. Aja, and Nwabumma C. Asouzu were involved in data analysis; P. M. Aja wrote the first draft of the manuscript while author B. A. Ale revised the second draft and carried out the editing of the manuscript for final submission. Prepared by the corresponding author (B. A. Ale) and approved by all the authors involved in this work. Thank you.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.