Abstract
Bone scintigraphy with combined single-photon emission computed tomography (SPECT) and computed tomography (CT) has become widely used for the detection of bone metastases. However, calculation of the semi-quantitative standardized uptake value (SUV) requires measurement of the pre- and post-injection radioactivity of the radiopharmaceutical. This study aimed to compare measured and fixed input radioactivity values for quantitative SPECT/CT bone imaging to examine whether the fixed measurement method of radiopharmaceutical radioactivity could be used as an alternative method. Four different methods were used to quantify the Tc-99m hydroxymethylene diphosphonate input radioactivity: (A) measured pre- and post-injection radioactivity values; (B) measured pre-injection and fixed post-injection radioactivity values; (C) fixed pre-injection and measured post-injection radioactivity values; (D) fixed pre- and post-injection radioactivity values. All SPECT/CT acquisitions were analyzed using bone SPECT analysis software, and the semi-quantitative parameters (SUVpeak and SUVmean) were recorded and compared for each analytical method. Two semi-quantitative parameters showed significant differences between analytical methods A and B, A and D, and C and D. However, an additional subgroup analysis performed on patients whose median post-injection measured radioactivity value was <1.5 MBq showed no significant differences in parameters between all analytical methods. Measurement of the radiopharmaceutical radioactivity can be an alternative method because it reduces the volume of radioactivity post-injection. The simplified fixed measurement method of radiopharmaceutical radioactivity can be used as an alternative method in cases when measuring the radioactivity in quantitative bone SPECT/CT imaging is missed.
Introduction
The relative incidence of bone metastases in different carcinomas has been reported to be 65%–75% in breast cancer, 65%–75% in prostate cancer, 60% in thyroid cancer, 30%–40% in lung cancer, 40% in bladder cancer, 20%–25% in renal cell carcinoma, and 14%–45% in melanoma. 1 Pathological conditions caused by bone metastases include severe pain, impaired mobility, pathologic fractures, spinal cord compression, bone marrow aplasia, and hypercalcemia. 1 Poor control of bone metastases results in impaired quality of life for cancer patients. The detection of bone metastases via bone scintigraphy has high sensitivity but low specificity. However, it has been reported that the addition of single-photon emission computed tomography (SPECT) imaging could improve the specificity.2,3
Bone scintigraphy with combined SPECT and computed tomography (SPECT/CT), which has been widely used in recent years, is superior to both SPECT and planar imaging.4–8 The utility of quantitative SPECT/CT imaging for the evaluation of bone has been reported.9,10 However, to calculate the semi-quantitative standardized uptake value (SUV), it is necessary to measure the radioactivity of the radiopharmaceutical before and after the injection. In cases such as when the dose calibrator is crushed or when measuring the radioactivity is missed, the semi-quantitative values are not calculated.
In this study, we compared the measured and fixed input radioactivity values for calculating semi-quantitative parameters of bone SPECT/CT imaging to investigate whether the fixed measurement method of radiopharmaceutical radioactivity could be used as an alternative method.
Materials and methods
Patients
This study was fully evaluated and approved by the institutional ethics committee of Hyogo Cancer Center, and all research was performed in accordance with the relevant guidelines and regulations. The institutional ethics committee of Hyogo Cancer Center waived the need for patients’ written informed consent for this retrospective analysis of clinically acquired data.
The study population for this retrospective study consisted of 70 Japanese patients (48 men (68.6%) and 22 women (31.4%); mean ± standard deviation (SD) aged 71.8 ± 7.8 years; lung cancer n = 43, prostate cancer n = 21, other types of cancer n = 6). Each patient underwent bone scintigraphy between March and April 2019.
Imaging protocol
In all cases, whole-body scintigraphy and bone SPECT/CT using Tc-99m hydroxymethylene diphosphonate (HMDP; Nihon Medi-Physics Co., Ltd., Tokyo, Japan) were performed as consecutive acquisitions. The median radioactivity value of the injected HMDP was 916.8 MBq (first quartile–third quartile, 902.4–943.2 MBq). The image acquisition start time was 3.4 h after administration of the tracer (first quartile–third quartile, 3.0–4.0 h). The bone SPECT/CT was acquired using three-bed SPECT/CT imaging (Figure 1). SPECT acquisitions were performed using a dual-head SPECT/CT system (NM/CT 850, GE Healthcare Japan, Co., Ltd., Tokyo, Japan) with low-energy high-resolution and sensitivity (LEHRS) collimators. Radioactivity measurements were obtained using a NaI dose calibrator (CRC127R, Capintec Inc., NJ, USA).
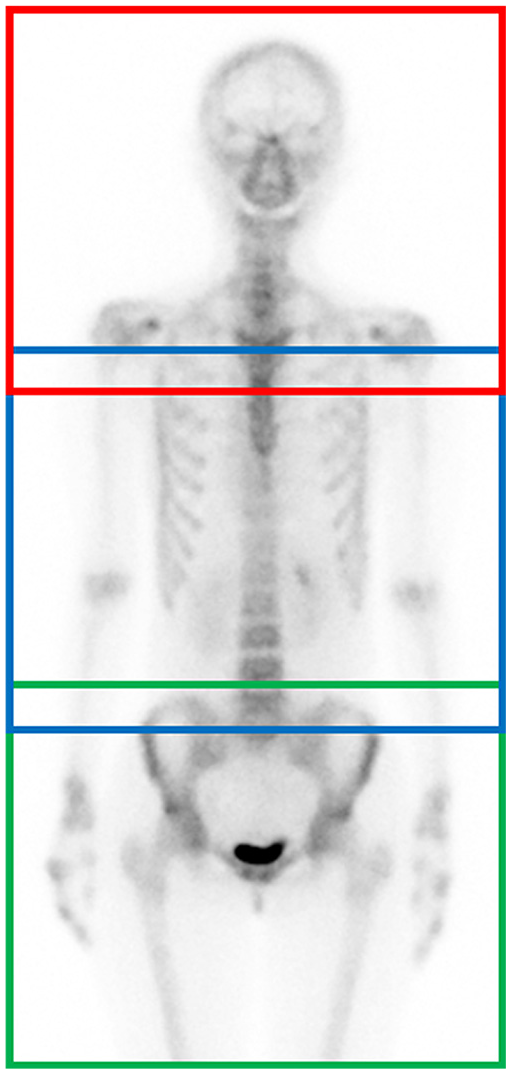
An example of a planar bone scintigraphy image. All images were acquired using three-bed SPECT imaging. First-bed SPECT imaging: red line box, second-bed SPECT imaging: blue line box, third-bed SPECT imaging: green line box.
SPECT acquisitions were performed using a 128 × 128 matrix set and 1.0 zoom, with 60 projections in a noncircular orbit (auto-contouring) in the step-and-shoot mode with SwiftScan SPECT, 11 a pixel size of 4.42 mm, and a slice thickness of 4.42 mm. The acquisition time was 8 s/step, with a total acquisition time of approximately 18 min (three-bed SPECT acquisition and moving table time). The SPECT images were reconstructed using ordered subset expectation maximization, incorporating CT attenuation correction, scatter correction with a dual-energy window,12,13 and resolution recovery correction. The photo-peak and scatter windows of SPECT acquisition were set to 140.5 keV ± 10% and 120.0 keV ± 5%. The subset and iteration parameters of the ordered subset expectation maximization were 10 and 4, respectively. A Gaussian filter was used for smoothing (full width at half maximum = 8.84 mm). CT acquisitions were performed using a hybrid-dedicated low-dose 8-slice CT subsystem (CT acquisition and reconstruction parameters: 120 kV, 30 mA, 512 × 512 matrix, 3.75 mm slice thickness, and pitch 1.675:1). The volume computed tomography dose index was 1.58 mGy and the dose length product was 179.32 mGy·cm, which is the same value in all cases.
Examples of SPECT/CT images are shown in Figure 2.

Examples of SPECT/CT images. (a) trans-axial CT image, (b) trans-axial SPECT image, and (c) trans-axial SPECT/CT fused image.
Analysis of the semi-quantitative parameters
All SPECT/CT acquisitions were analyzed using the bone SPECT analysis software (GI-BONE; AZE Co., Ltd., Tokyo, Japan), and the automatic SUV calculation was performed using the same formula as reported previously.14–16 The SUV is calculated as follows: SUV = becquerel calibration factor × (SPECT count density/scan duration) × (body weight/injected radioactivity). The Becquerel calibration factor was used to convert voxel counts of SPECT image value into the radioactivity concentration. The system was set to automatically extract the volume of interest (VOI) where the SUV was ≥8.0, and the SUVpeak and SUVmean values were calculated and recorded. The SUV threshold of ≥8.0 was determined with reference to normal vertebral bodies.9,17 The SUVpeak was defined as the average SUV of the voxel in a 1 cm3 spherical VOI centered on the maximum voxel value within the VOI. SUVmean was defined as the mean voxel value within the VOI.
Examples of GI-BONE images are shown in Figure 3.

Examples of GI-BONE images. In GI-BONE, SPECT (128 × 128 matrix) and CT (512 × 512 matrix) images are converted into 256 × 256 matrix. (a) VOI image showing the SPECT image indicating the calculated VOI regions; the system was set to automatically extract the VOI where the SUV was ≥8.0, and (b) SPECT and CT fused image.
For SUV calculation in GI-BONE software, it is necessary to input the radioactivity values of Tc-99m HMDP and the measurement time of the radioactivity pre- and post-injection. To investigate whether a fixed value (does not require the measurement of the amount of radioactivity) could be used, four different methods for quantifying the input radioactivity values of Tc-99m HMDP were evaluated:
A: Pre- and post-injection radioactivity values were measured using a NaI dose calibrator (gold standard);
B: Pre-injection radioactivity values were measured using a NaI dose calibrator, while post-injection radioactivity values were fixed values determined with reference to the measured radioactivity values of all 70 cases;
C: pre-injection radioactivity values were fixed values determined with reference to the measured radioactivity values of all 70 cases (fixed pre-injection values were calculated by correcting the radioactivity values measured at 8:00 am), while post-injection radioactivity values were measured using a NaI dose calibrator;
D: pre- and post-injection radioactivity values were fixed values determined with reference to the measured radioactivity values of all 70 cases (fixed pre-injection values were calculated by correcting the radioactivity values measured at 8:00 am).
Differences between the four analytical methods were compared for all patients.
An additional subgroup comparative analysis was performed on patients whose median post-injection radioactivity value was lesser or greater than the median radioactivity value.
Statistical analysis
Statistical analyses were performed using commercial statistical software (Bell Curve for Excel version 3.1; Social Survey Research Information Co., Ltd., Tokyo, Japan, and StatView version 5.0; SAS Institute, Inc., Cary, NC, USA). All continuous variables were expressed as mean ± SD or median (first quartile–third quartile). Normality was checked using the Shapiro-Wilk test. Values of p < 0.05 were considered statistically significant. All differences between the four analytical methods were assessed by applying Bonferroni’s correction to the Wilcoxon signed ranks test for multiple comparisons.
Results
Pre- and post-injection radioactivity values
The pre-injection radioactivity values were normally distributed (p = 0.13), with a mean value of 1214.7 ± 8.3 MBq. The pre-injection radioactivity fixed value was therefore set to 1215 MBq.
The post-injection radioactivity values were not normally distributed (p < 0.01) and showed median post-injection radioactivity value of 1.5 MBq (0.90–2.15 MBq). The fixed value for post-injection radioactivity was therefore set to 0 MBq, as more than half of the measured radioactivity values after the administration were <1.5 MBq. Since the input time can vary, it would be better to set the fixed value of radioactivity to 0 MBq after injection.
Semi-quantitative parameters
The semi-quantitative parameters are listed in Tables 1 and 2. The VOIs were set on 192 segments in 48 cases. All semi-quantitative parameters were non-normally distributed across all four analytical methods (all p < 0.01).
Results obtained from SUVpeak measurements in 192 segments of 48 cases.

A: measured pre- and post-injection radioactivity values, B: measured pre-injection and fixed post-injection radioactivity values, C: fixed pre-injection and measured post-injection radioactivity values, D: fixed pre- and post-injection radioactivity values.
SD: standard deviation.
Results obtained from SUVmean measurements in 192 segments of 48 cases.

A: measured pre- and post-injection radioactivity values, B: measured pre-injection and fixed post-injection radioactivity values, C: fixed pre-injection and measured post-injection radioactivity values, D: fixed pre- and post-injection radioactivity values.
SD: standard deviation.
Significant differences were found between the following analytical methods: A and B (p<0.01), A and D (p<0.01), and C and D (p<0.01), as shown in Table 3.
Comparison of the two semi-quantitative parameters of radioactivity measurement methods in 192 segments of 48 cases.

Data were collected from all the VOIs automatically extracted with SUV ≥8.0.
A: measured pre- and post-injection radioactivity values, B: measured pre-injection and fixed post-injection radioactivity values, C: fixed pre-injection and measured post-injection radioactivity values, D: fixed pre- and post-injection radioactivity values.
SUV: standardized uptake value.
p < 0.05.
For patients with median post-injection radioactivity <1.5 MBq, the results for 114 segments from 27 cases are shown in Table 4. In this subgroup, there were no significant differences in parameters between all analytical methods. For patients with median post-injection radioactivity ≥1.5 MBq, the results for 78 segments from 21 cases are shown in Table 5. This subgroup showed the significant differences found between the following analytical methods: A and B (SUVpeak, p < 0.01; SUVmean, p < 0.01), A and D (SUVpeak, p < 0.01; SUVmean, p = 0.01), B and C (SUVpeak, p = 0.04; SUVmean, p = 0.01), and C and D (SUVpeak, p < 0.01; SUVmean, p < 0.01).
Comparison of the two semi-quantitative parameters of radioactivity measurement methods in 114 segments of 27 cases.
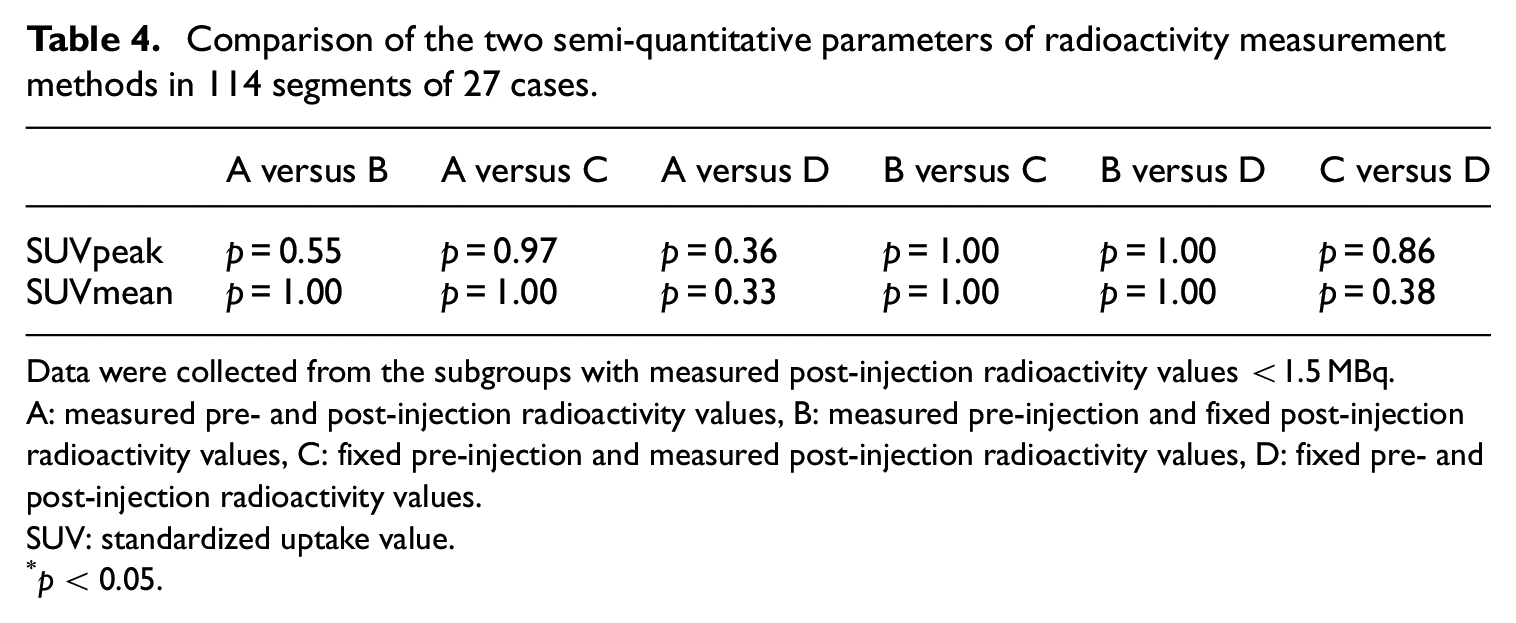
Data were collected from the subgroups with measured post-injection radioactivity values <1.5 MBq.
A: measured pre- and post-injection radioactivity values, B: measured pre-injection and fixed post-injection radioactivity values, C: fixed pre-injection and measured post-injection radioactivity values, D: fixed pre- and post-injection radioactivity values.
SUV: standardized uptake value.
p < 0.05.
Comparison of the two semi-quantitative parameters of radioactivity measurement methods in 78 segments of 21 cases.

Data were collected from the subgroups with measured post-injection radioactivity values ≥1.5 MBq.
A: measured pre- and post-injection radioactivity values, B: measured pre-injection and fixed post-injection radioactivity values, C: fixed pre-injection and measured post-injection radioactivity values, D: fixed pre- and post-injection radioactivity values.
SUV: standardized uptake value.
p < 0.05.
Examples of the patient images are shown in Figures 4 and 5.

Bone metastasis in the 10th thoracic vertebra (arrow) of an 83-year-old man with prostate cancer. (a) VOI image showing the SPECT image indicating the calculated VOI regions; the system was set to automatically extract the VOI where the SUV was ≥8.0, and (b) SPECT and CT fused image. The post-injection radioactivity value was 1.20 MBq. The results of the comparison of methods A and D (measured pre- and post-injection radioactivity values vs. fixed pre- and post-injection radioactivity values) SUVpeak, 34.54 versus 34.57; SUVmean, 15.36 versus 15.34.

Bone metastasis in the left proximal humerus (arrow) of a 75-year-old man with prostate cancer. (a) VOI image showing the SPECT image indicating the calculated VOI regions; the system was set to automatically extract the VOI where the SUV was ≥8.0, and (b) SPECT and CT fused image. The post-injection radioactivity value was 1.65 MBq. The results of the comparison of methods A and D (measured pre- and post-injection radioactivity values vs. fixed pre- and post-injection radioactivity values) SUVpeak, 13.93 versus 13.54; SUVmean, 11.22 versus 11.01.
Discussion
Bone scintigraphy is a highly sensitive imaging technique that is used to evaluate bone metastases in various types of cancer, while in recent years bone SPECT/CT imaging has also become popular.18,19 SPECT/CT using CT attenuation correction, scatter correction, and resolution recovery correction can provide semi-quantitative parameters such as SUVs.20,21 Beck et al. 22 showed that changes in tracer uptake in individual lesions should be measured by quantifying the tracer uptake, rather than by visual assessment alone. However, to obtain quantitative parameter values, it is necessary to measure the radioactivity. Measurement of radiopharmaceutical radioactivity involves an increase in radiation exposure to the operator (the majority of radiation exposure is pre-injection). Moreover, in cases such as when the dose calibrator is crushed or when measuring the radioactivity is missed, radioactivity cannot be measured before and after injection. In such cases, semi-quantitative values are not calculated. Previous reports examining the method of simplification of the radioactivity measurement concluded that it was possible to omit the actual measurement of radioactivity by using the proposed method. 23 However, measurement SUVs values were taken only at the normal fourth lumbar vertebra and the abnormal regions were not measured. We considered this to be necessary to examine the simplified method of radiopharmaceutical radioactivity in the abnormal regions.
In this study, we compared four analytical methods for measuring semi-quantitative parameters (SUVpeak and SUVmean) calculated from bone SPECT/CT imaging in the abnormal regions. SUVmax of the typical SUV measure in positron emission tomography (PET) was excluded from the analysis because SUVmax was subjected to noise and statistical variations in data.24,25 For some of the comparisons between the analytical methods A and B (measured pre- and post-injection values vs. measured pre-injection and fixed post-injection values), A and D (measured pre- and post-injection values vs. fixed pre- and post-injection values), and C and D (fixed pre-injection and measured post-injection values vs. fixed pre- and post-injection values), significant differences in the two semi-quantitative parameters were found. From our analyses of 192 segments in 48 cases, we considered that it was necessary to measure post-injection radioactivity values. The fixed post-injection radioactivity values were set to 0 MBq, and we considered that the difference between the true values and 0 MBq increased in cases with high post-injection radioactivity.
We performed an additional subgroup analysis on patients, splitting them according to the median post-injection radioactivity. For patients with post-injection radioactivity <1.5 MBq, no significant differences were found between all analytical methods. In contrast, for patients with median post-injection radioactivity ≥1.5 MBq, a significant differences were found between methods A and B (measured pre- and post-injection values vs. measured pre-injection and fixed post-injection values), A and D (measured pre- and post- injection values vs. fixed pre- and post-injection values), B and C (measured pre-injection and fixed post-injection values vs. fixed pre-injection and measured post-injection values), and C and D (fixed pre-injection and measured post-injection values vs. fixed pre- and post-injection values). Measurement of the radioactivity of the radiopharmaceutical in quantitative bone SPECT/CT imaging can be simplified by reducing the volume of radioactivity post-injection. To reduce post-injection radioactivity, it is recommended to use physiological saline while injecting the radiopharmaceutical, flush the injection port, and rinse the radiopharmaceutical syringe multiple times.
Conclusion
The simplified fixed measurement method of radiopharmaceutical radioactivity can be used as an alternative method for cases such as when the dose calibrator is crushed or when measuring the radioactivity in quantitative bone SPECT/CT imaging is missed.
Footnotes
Acknowledgements
Author contributions
M.I. and Y.K. conceived the study and performed the research/study. M.I., M.O. and T.S. conceived the report. M.I. wrote the first draft of the article. M.I., Y.K., M.O. and T.S. reviewed and edited the article and approved the final version of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Compliance with ethical standards
This study was fully evaluated and approved by the institutional ethics committee of Hyogo Cancer Center, and all research was performed in accordance with the relevant guidelines and regulations.
Ethical approval
The institutional ethics committee of Hyogo Cancer Center waived the need for patients’ written informed consent for this retrospective analysis of clinically acquired data.
