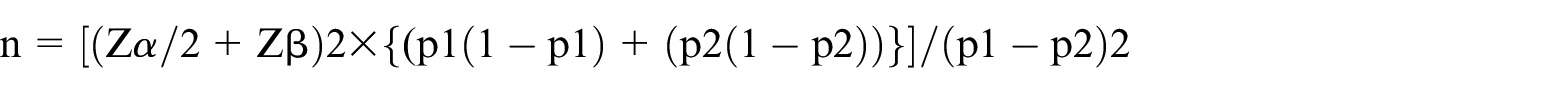Abstract
The objective of this study was to compare the silk suture with a cyanoacrylate adhesive to stabilize the free gingival graft in conjunction with Er: YAG laser-assisted recipient site preparation to augment the keratinized tissue in gingival recession cases. This randomized trial comprised of 300 recession defects patients. All the included patients were diagnosed using Miller class I and II gingival recession defects classification. Group I sites were treated with a free gingival graft (FGG) harvested using an Er: YAG laser and further sutured with silk. Group II sites were stabilized with isoamyl 2 cyanoacrylate bio-adhesive material. Clinical parameters, such as gingival recession depth, clinical attachment level, gain in gingival tissue thickness, and width of keratinized gingiva were recorded at baseline, and at third month, sixth month, and 12th month postoperatively. The mean changes in gingival recession from months 3 to 6 and months and 6 to 12 were significant (p < 0.05) in both groups. However, the improvement in recession depth was better in group II than in group I. The mean change in clinical attachment level did not differ significantly between the groups at the different time intervals. However, values tended to be higher in group II than in group I. The width of the keratinized gingiva tended to be higher from baseline to 3 months, baseline to 6 months, baseline to 12 months, 3 to 6 months, and from 6 to 12 months in group II as compared with group I (p > 0.05). Cyanoacrylate could be used as a substitute to silk sutures to stabilize FGGs. Cyanoacrylate was easy to apply, consumed less operating time, and was considered equally efficacious for stabilizing FGGs.
Introduction
Treatment of recession defects is a topic of interest amongst dental professionals, as the number of cases has been on the rising, particularly in China. Gingival recession affects almost all middle-aged and older people to some degree in all parts of China. 1 Gingival recession exposes the root, which leads to clinical problems such as hypersensitivity, root caries, cervical abrasions, difficulty in removing the plaque, and unpleasant aesthetics.2,3 Gingival recession usually creates aesthetic problems for the patient, particularly when it affects the anterior teeth. 4 Shape, size, color, symmetry of teeth, contour of the gingiva, papillary shape, and papillary height are the main factors in maintaining the esthetics. Maxillary anterior teeth play a dominant and important role in maintaining the aesthetics, as they create a facial form by supporting the upper lip.
The main focus of these periodontal procedures is to complete the root coverage and further leading to good aesthetic outcomes. 5
A variety of gingival grafting techniques have been advocated to treat the gingival recession, such as lateral pedicle flaps, coronally positioned flaps, free gingival grafts, connective tissue grafts, and guided tissue regeneration.6,7 Recent studies investigated the clinical outcomes of many grafts. The root coverage comparing a soft tissue collagen matrix derived from porcine dermis (XDM) with connective tissue graft (CTG) using a modified coronally advanced tunnel technique (MCAT). Authors have taken similar clinical parameters for both treatment modalities, but a better healing index for XDM compared with CTG sites.8,9 So, authors concluded that the results achieved with a porcine-derived dermal collagen matrix were satisfactory and could be considered as a CTG substitute in the treatment of multiple adjacent gingival recessions when used in conjunction with MCAT.8,9 Free gingival graft (FGG) continues to be a reliable procedure for increasing the width of keratinized gingiva and stopping the progression of gingival recession. At present, even though the free gingival grafts have lost their race to subepithelial connective tissue grafts as far as root coverage is concerned, however, they still hold an edge in considerations like being simple, multiple teeth can be treated at one time, easy tissue handling, and can be performed when keratinized gingiva adjacent to involved is insufficient.10,11The basic disadvantages of FGG are total lack of predictability in regard to esthetics especially in anterior region. The studies concluded that the mandible anterior teeth face even more challenges because some of the factors such as gingival biotype, shallow vestibular depth and higher attachment of the frenum. However, still FGG could be a good procedure in the management of recession defects as reported by the recent iterature.12,13 But in contrast to this some authors have also revealed that the FGG may be associated with esthetic problems due to discrepancies of color and texture between the healed graft and surrounding mucosa as well as a bulky appearance. 14
Silk suturing is the conventional and gold standard technique to stabilize the free gingival graft. However, the graft must be correctly positioned and should be immobile. Improper immobilization may delay the tissue revascularization and eventually lead to tissue necrosis. The number of sutures must be minimized to prevent damage to the graft. 15 Other than silk sutures, there are several techniques that help to achieve the hemostasis such as tissue adhesives and adhesive bandages. Coover et al. 16 assessed the properties of cyanoacrylate adhesives and proposed their application in surgical procedures in 1959. Hoxter et al. 17 introduced the sutureless free gingival graft technique in 1978 and concluded that even without the use of sutures, it is possible to achieve immobilization, asepsis, and hemostasis.
Cyanoacrylates have been used in the closure of organs, skin, mucosa grafts, closure of lacerations, incisions and post-extraction dressings in dentistry. The most favorable qualities of Cyanoacrylates includes strong bonding in the presence of moisture to the tissues, workable polymerization time, biodegradability and bacteriostatic ability. 18 Cyanoacrylates have been used in many treatment modalities in the dental field, such as for pulp capping, endodontic surgeries, during tooth bleaching, and as an adjunct to sutures. Many clinical studies have revealed the feasibility and clinical application of fibrin adhesive systems also.
Many authors have assessed the role of lasers in periodontal therapy. The Er: YAG laser, which emits at a wavelength of 2.94 µ, has been demonstrated to be useful for both hard and soft tissues. 19 This type of laser provides the most effective treatment strategy for periodontal diseases, including ablation of granulation tissues, achieving favorable root conditions, and making incisions for lifting the flap. 20 Clinical trials have demonstrated that periodontal pocket therapy using an Er: YAG laser produces equivalent or better results than the conventional mechanical therapy with Gracey-type curettes. 19 Therefore, the clinical safety and effectiveness of Er: YAG lasers have been demonstrated for both non-surgical and surgical periodontal therapy. 19
Nevertheless, very few studies have compared the stabilization of a free gingival graft with sutures and with tissue adhesives. Thus, this study compared 5-0 silk suture with a cyanoacrylate adhesive to stabilize the free gingival graft in conjunction with Er: YAG laser-assisted recipient site to augment the keratinized tissue in gingival recession cases.
Material and methodology
This research protocol was approved by the institutional ethics committee and review board (approval no. SH/dental/23-12/2013). All participants were informed verbally about the study protocol and provided written informed consent.
The total sample size was 300 subjects, who attended the Shanghai Huangpu District 2nd Dental Disease Prevention and Treatment Institute. The design of the study was a randomized, prospective, double-blinded, and controlled clinical trial with a 12-month follow-up. Of the 300 subjects, 150 were male and 150 were female, with a mean age of 32.1 years. All patients were diagnosed on the basis of Miller class I and II gingival recession defects classification. The first study sample was enrolled in January 2014 and the last sample was enrolled in December 2017. Subjects who reported with Miller’s Class I and Class II and there was a need to increase the width of attached gingiva were included in the study population.21,22
Patients with a history of any systemic disease; those taking medication that could affect periodontal therapy outcomes; smokers; pregnant and lactating women; and those with poor oral hygiene, mobile teeth, or cervical caries were excluded from the study.
Full-mouth supra- and subgingival scaling and root planning procedures were performed under local anesthesia in all the patients. A periodontal evaluation was performed at sixth week after the phase I therapy to confirm the desired sites and to record the baseline measurements. After the inflammation had completely resolved, the subjects underwent the surgical procedures. In those patients when the vertical bone loss of more than 50% was noted then Bone Allografts has been used in the surgical resection to further manage the cases. As we are not focusing on the management of the severer periodontitis with bone loss more than 50%, so we have skip these parameters. The flow chart of the study has been explained in Figure 1.

Flow chart of the study.
Surgical procedure
A short case history, including specific clinical parameters such as the PI (Turesky–Gilmore–Glickman modification of the Quigley–Hein index)25,26 and periodontal probing depth, was recorded on the day of surgery. Local anesthesia using 2% lidocaine with 1:100,000 epinephrine was administered to the appropriate chosen site. An Er: YAG laser with a frequency of 10 Hz, at 300 mj, and a pulse speed of 500 μs was used to prepare the recipient area for the free gingival graft. With Er: Yag Laser, beveled vertical incisions were given to prepare the recipient bed, which removed the surfaces of interdental papillae and extended apically mesial to the convexities of adjacent teeth, 3–4 mm beyond the mucogingival line. A horizontal incision was given at the level of the mucogingival junction (MGJ).
A graft thickness of approximately 1 mm was obtained from the palate, which extended to the interdental papillae. The graft, thus, obtained was contoured, adapted, and sutured on to the recipient bed. The measurements for harvesting the graft were outlined on the palate using the Er: YAG laser. A no. 21 blade was used when the Er: YAG caused too much of tissue ablation. The harvested graft was immediately placed on the recipient area after excision.
In group I, the free gingival graft was sutured using 5-0 silk; one suture was inserted in the distal and mesial edges, and a suspensory recipient suture was used to stabilize the graft, without penetrating the graft. The graft was held firmly in place using digital pressure for 5 min using gauze moistened with saline (Figure 2).
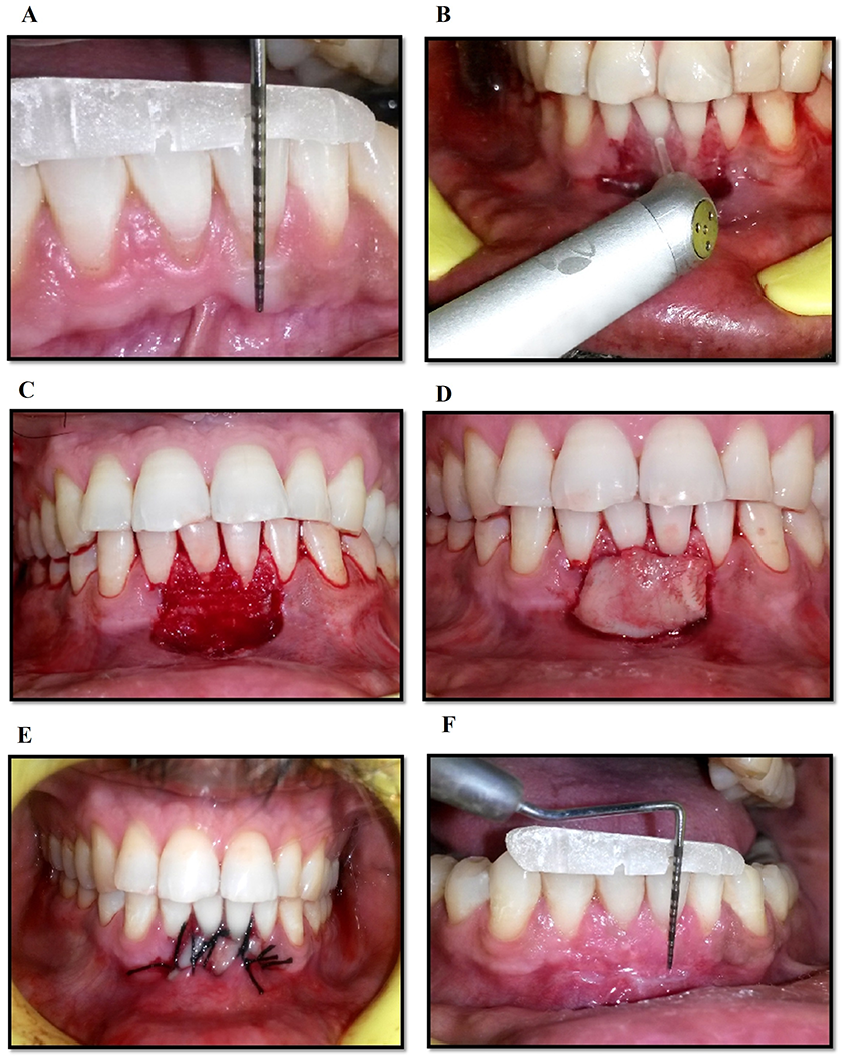
Preoperatively and postoperative clinical pictures in group I.
A: Measurement of Gingival Recession (Preoperative), B. Preparation of Recipient Site using Er:YAG Laser, C. Placement of Free Gingival Graft, D 5-0 Placement of Silk Suture for Stabilization, E. Post-Operative picture after 7 days, F. Measurement of Width of Keratinized Gingiva at sixth month Postoperatively.
In group II, the free gingival graft was stabilized using isoamyl 2 cyanoacrylate. Gentle pressure was applied to the graft for 5 min and then the cyanoacrylate was applied using a special 0.25-ml pipette to achieve an approximately 2-mm-wide line of adhesive along the graft borders. Excess adhesive was removed using gauze moistened with saline. Then 1–2 min was allowed for the graft to adhere to the recipient site before a periodontal dressing was applied to protect the surgical area for 14 days (Figure 3).
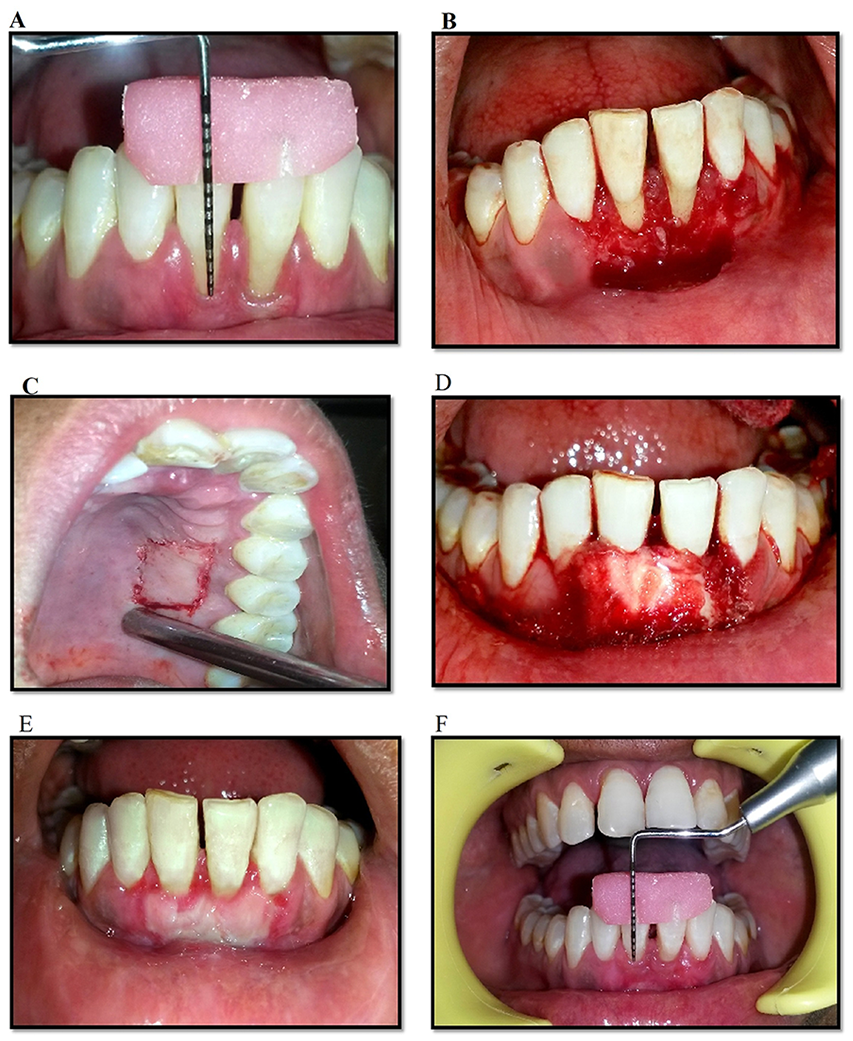
Preoperatively and postoperative clinical pictures in group II.
Measurement of Gingival Recession (Preoperative), B: Preparation of Recipient Site, C: Donor site, D: Graft stabilization with Iso-Butyl-2-Cyanoacrylate, E: Post-Operative picture after 7 days, F: Measurement of Width of Keratinized Gingiva at sixth month Postoperatively.
Pre-surgical clinical evaluation
The following clinical parameters were checked before the surgical procedure: plaque index (PI), gingival index (GI), gingival recession depth (RD), CAL, width of keratinized gingiva (WK), and gingival tissue thickness. All clinical parameters were measured at baseline and at 3, 6, and 12 months postoperatively.
Measurement of the gingival index, plaque index, and clinical attachment level
The GI and PI were determined and recorded at four gingival sites per tooth. Pocket depth (PD) measurements were obtained using a Williams periodontal probe. The PD was measured from the free gingival margin to the base of the pocket. PD and CAL were measured for each tooth at six sites, namely the mesiobuccal, midbuccal, distobuccal, mesiolingual, midlingual, and distolingual.
Assessment of keratinized gingiva width
The width of keratinized gingiva was measured as the distance from the gingival margin to the mucogingival junction. Sulcus depth was measured as the distance from the gingival margin to the base of the sulcus. The width of the attached gingiva was calculated as the difference between sulcus depth and the width of the keratinized tissue.
Assessment of gingival tissue thickness
Gingival tissue thickness was assessed in each patient by a single examiner. For assessment of gingival morphology, the following parameters were recorded: Crown width/crown length ratio (CW/CL) of both central incisors and canines was determined according to Olsson and Lindhe. 18 The CL was measured between the incisal edge of the crown and the free gingival margin. CW is the border between the middle and the cervical thirds. 23
Gingival thickness was evaluated and categorized into thick or thin on site. This evaluation was based on the transparency of the periodontal probe through the gingival margin while probing the sulcus at the midfacial aspect of both central maxillary incisors and canines to the full depth. If the outline of the underlying periodontal probe could be seen at the whole subgingival part, it was categorized as thin (score: 1); if not, it was categorized as thick (score: 2) 23 The following operative clinical parameters were also assessed.
Operative clinical parameters 24
The time needed to reconstitute the fibrin adhesive system
The time needed to fix tissues
Tissue stability
Time taken for suture removal
Chair-side time 24
Preparation time
Time taken to fix tissues
Time taken to remove sutures
Other clinical parameters 24
Tissue stability (score)
0—Stable at 5 min
1—Unstable at 5 min
Postoperative bleeding 15
0—No bleeding at 1 min
1—Bleeding at 1 min
5—Bleeding at 5 min
Postoperative treatment modality
Antibiotics and analgesics (500 mg amoxicillin, four times daily for 5 days, and 800 mg ibuprofen, three times daily) were prescribed, along with chlorhexidine digluconate rinses (0.12%) twice daily for 2 weeks). The periodontal dressing and sutures were removed at 2 weeks postoperatively. Surgical wounds were gently cleansed with 0.12% chlorhexidine digluconate. Each patient was reinstructed on proper oral hygiene measures.
Statistical analysis
The data were analyzed using SPSS software (version 23.0 for Windows; SPSS Inc., Chicago, IL, USA). The gingival recession width of the keratinized gingiva, gingival tissue thickness, and CAL were tested for normality using the Kolmogorov test. Gingival recession was compared between groups using the Mann–Whitney U-test and the mean change in gingival recession within groups was compared using the Wilcoxon rank-sum test. The width of keratinized gingiva, gingival tissue thickness, and CAL were compared between the groups using an unpaired t-test and the mean change within groups was compared using a paired t-test. A p-value <0.05 was considered significant. Measurements were recorded by a single examiner, and the surgical procedures were performed by the same operator. All patients completed the study and fully complied with the recall program.
Estimation of sample size
The level of confidence in this study was taken 95% and by assuming that 30% of the population is exposed to a risk factor and assuming an equal number of cases and controls in matched study design hypothesized odds ratio of 2.0. Level of significance = 5%, Power = 80%, Type of test = two-sided
Level of Significance: This is typically assumed as 5%. Type I error is inversely proportional to sample size.
Power: Power was taken as 80% and Type II error is directly proportional to sample size.
Formula of calculating sample size is
where
n = sample size required in each group,
p1 = proportion of subject treated by Group I = 0.50,
p2 = proportion of subject treated by Group II = 0.34,
p1–p2 = clinically significant difference = 0.16,
Zα/2: This depends on level of significance, for 5% this is 1.96,
Zβ: This depends on power, for 80% this is 0.84.
Based on above formula the sample size required per group was 125. Hence total sample size required was calculated as 250. However, this study has been designed to follow strict inclusion criteria and follow up plan. If n is the sample size required as per formula and if d is the dropout rate then adjusted sample size N1 is obtained as. N1 = n/(1−d) and Maximum drop out of 20% per group, So, 25 subjects per group has been added more. Based on the calculations, the total sample size was taken as 300.
Results
Of the total sample, 150 subjects were in group I and 150 subjects were in Group II. Preoperative and postoperative clinical parameters were assessed in both the groups at 3, 6, and 12 months.
Gingival index
The GI was assessed at 3, 6, and 12 months in both groups. The results revealed a non-significant difference in the GI at 3, 6, and 12 months within group for the both the groups (p > 0.05 for all). The mean change in the GI score did not differ significantly between the two groups (p > 0.05) also. However, GI score tended to be better in group II than that of group I post-operatively (Table 1).
Gingival index scores in groups I and II and comparisons thereof at different time intervals (mean change ± standard deviation (SD)).

NS means Non Significant.
Plaque index
The PI was assessed at 3, 6, and 12 months in both groups. The results revealed a non-significant difference in the PI score at 3, 6, and 12 months within group for the both groups (p > 0.05 for all). The mean change in the PI score did not differ significantly between the two groups (p > 0.05). However, the PI score of group II tended to be better than that of group I (Table 2).
Plaque index scores in groups I and II and comparisons thereof at different time intervals (mean change ± standard deviation (SD)).

NS means Non Significant.
Gingival recession depth
The mean changes in gingival recession depth at different time intervals for both groups are shown in Table 3. The mean changes in gingival recession from baseline to 3, 6, and 12 months were significant with respect to baseline in both groups (p < 0.05 for all). The mean changes in gingival recession from 3 to 6 months and 6 to 12 months were also significant (p < 0.05) in both groups. However, recession depth improved more in group II than in group I at significant level.
Gingival recession depths in groups I and II and comparisons thereof at different time intervals (mean change ± standard deviation (SD)).

NS means Non Significant.
The percentage root coverage of recession depth in groups I and II at different time intervals is shown in Table 4. A significant reduction in gingival recession depth was detected between baseline and at the 3, 6, and 12-month results for both groups (p < 0.05). The results also showed that the percentage of root coverage values tended to be higher in group II than in group I (p > 0.05) (Figures 2 and 3).
Percentage root coverage in groups I and II and comparisons thereof at different time intervals.

Statistically significant.
Clinical attachment level
The gain in mean CAL from baseline to 3 months, from baseline to 6 months, from baseline to 12 months, from 3 to 6 months, and from 6 to 12 months was significantly higher in group I than in group II (p < 0.05). In group II, mean CAL increased 2.79 ± 0.61 mm from baseline to 3 months (p < 0.05). The mean CAL gain from baseline to 6 months was 3.83 ± 0.81(p < 0.05), and the mean CAL gain from baseline to 12 months was 4.18 ± 0.11 (p < 0.05). The mean CAL gain from 3 to 6 months was 0.81 ± 0.76 (p < 0.05) and the mean CAL gain was 0.98 ± 0.11 from 6 to 12 months (p < 0.05). No differences in the mean changes in CAL were observed at different time intervals between the groups (Table 5). However, group II values tended to be higher than those in group I (Figures 2 and 3).
Clinical attachment levels (mm) in groups I and II and comparisons thereof at different time intervals (mean change ± standard deviation (SD)).

NS means Non Significant. S means Significant.
Width of keratinized gingiva
The gain in the mean width of the keratinized gingiva from baseline to 3 months in group I was 1.18 ± 0.13 mm (p < 0.05), that from baseline to 6 months was 1.39 ± 0.11 mm (p < 0.05), that from baseline to 12 months was 1.66 ± 0.15 mm (p < 0.05), that from 3 to 6 months was 0.27 ± 0.12 mm (p < 0.05), and that from 6 to 12 months was 0.57 ± 0.32 mm. The gain in the mean width of the keratinized gingiva in group II was 1.83 ± 0.32 mm from baseline to 3 months (p < 0.05), that from baseline to 6 months was 2.11 ± 0.12 mm (p < 0.05), that from baseline to 12 months was 2.76 ± 0.15 mm (p < 0.05), that from 3 to 6 months was 0.28 ± 0.21 mm (p < 0.05), and that from 6 to 12 months was 0.78 ± 0.82 mm. The values from baseline to 3 months, baseline to 6 months, baseline to 12 months, 3 to 6 months, and 6 to 12 months tended to be higher in group II than in group I (p > 0.05; Table 6) (Figures 2 and 3).
Width of keratinized gingiva (mm) in groups I and II and comparisons thereof at different time intervals (mean change ± standard deviation (SD)).

NS means Non Significant. S means Significant
Gingival tissue thickness
Changes in gingival tissue thickness at different time intervals in the groups are shown in Table 7. The gain in mean gingival thickness in group I was 1.32 ± 0.01 mm from baseline to 3 months (p < 0.05), that from baseline to 6 months was 1.65 ± 0.04 mm (p < 0.05), that from baseline to 12 months was 1.78 ± 0.11 mm (p < 0.05), that from 3 to 6 months was 0.19 ± 0.03 mm (p < 0.05) and that from 6 to 12 months was 0.45 ± 0.12 mm (p < 0.05). The gain in mean gingival thickness in group II was 1.47 ± 0.04 mm from baseline to 3 months (p < 0.05), that from baseline to 6 months was 1.83 ± 0.18 mm (p < 0.05), that from baseline to 12 months was 2.16 ± 0.12mm (p < 0.05), that from 3 to 6 months was 0.24 ± 0.01 mm (p < 0.05), and that from 6 to 12 months was 0.87 ± 0.12 mm (p < 0.05). However, the mean changes in gingival tissue thickness at different time intervals did not differ significantly between the groups. The mean increase in gingival thickness tended to be higher in group II than in group I (Figures 2 and 3).
Mean change in gingival tissue thickness (mm) in groups I and II at different time intervals (mean change ± standard deviation (SD)).

NS means Non Significant. S means Significant
Other clinical parameters
The time required to fix the tissues with cyanoacrylate averaged 30 s, which was far less than the time required for suturing (5–15 min). Furthermore, the time needed to remove the sutures ranged from 1 to 5 min. Tissues stabilized with cyanoacrylate exhibited less postoperative bleeding than tissues stabilized with sutures. The tissues stabilized with cyanoacrylate were very stable, as they attached to underlying tissues within 4 min, whereas tissues stabilized with sutures were mobile and slightly unstable even after 5 min. The preparation time for cyanoacrylate was also less compared to sutures (Table 8).
Evaluation of other clinical parameters in groups I and II.

Discussion
Recent studies have reported that cyanoacrylates are bacteriostatic, biodegradable, and hemostatic with a long half-life and good tissue compatibility. 27 Saquib et al. concluded that the cyanoacrylate material has got good bonding properties and bond strength to hold the tissue margins together. It also has good working properties like the flow and fast setting that is, within 5 to 10 s. 20 The results of this study was also in coordination with these study results. Our study also revealed that cyanoacrylate was easy to apply, required less operating time, and resulted in no adverse reactions during graft healing.
Patients are concerned about sensitivity and poor esthetics in cases of gingival recession. So, dentists and periodontists required a treatment protocol that provides a good relief from sensitivity and maintain the good aesthetics.26–29 Free gingival grafts are widely used to increase the dimensions of the attached gingiva. 11 Free gingival grafts have become the gold standard to increase the dimensions of attached gingiva due to their autogenous character, maintenance of keratinization, predictability of surgical outcomes, ease of technique and the possibility of implementation for a group of teeth. 29 So, with these kind of outcomes reported by the various authors we have decided to use the FGG for this study.
Laser irradiation has recently been investigated for use in periodontal therapy. The advantages of a laser include surface sterilization, good hemostasis, reduced intraoperative bleeding, increased comfort, and less postoperative pain. Studies have demonstrated that the Er: YAG laser has considerable ability to remove lipopolysaccharides from root surfaces and facilitate removal of the smear layer after root planing.30–32 In coordination with the conducted studies we have chosen Er: YAG as it can be used on soft tissue for de-epithelization and on hard tissues to modify the root surface.
Some researchers have suggested that sutureless stabilization techniques could help to decrease or even prevent the graft shrinkage, and provides better adhesives.27,33–35 Thus, in this study, we compared 5-0 silk with a cyanoacrylate adhesive to stabilize free gingival grafts harvested with the Er: YAG laser to augment keratinized tissue for managing class I and II gingival recession.
The mean changes in the GI and PI at the different time intervals did not differ significantly between the groups (p > 0.05). The GI and PI did not improve in either group at 3, 6, or 12 months. The results of the present study in coordination with those of Hangorsky and Bissada 36 and Dorfman et al. 37 and imply that neither silk suture nor cyanoacrylate enhanced gingival inflammation and both were well tolerated by the host tissue.
A significant reduction in mean gingival recession was observed from baseline to 3 months, baseline to 6 months, and from baseline to 12 months in both groups, which was further in coordination with studies conducted by Paknejad et al., 15 Deeplakshmi and Balu 38 and Gumus and Buduneli. 27 However, greater reduction in gingival recession was detected in group II than in group I, which was a new finding in this study. These findings may have been due to creeping attachment. Creeping attachment is postoperative migration of the gingival marginal tissue in the coronal direction, covering partially or denuded root. The gingival tissue becomes firmly attached to the root surface and probing does not reveal sulcular depth. The study conducted by Saquib et al. also revealed the similar results. 20
Significant gains in CAL were observed in both groups from baseline to 3 months, from baseline to 6 months, and from baseline to 12 months. Group II tended to have a greater gain in CAL compared to group I (p > 0.05). Similar results were reported by Barbosa et al. 40 A gain in CAL may imply new attachment, reattachment, or periodontal regeneration.
Significant increases in the width of the keratinized gingiva were observed in groups I and II from baseline to 3 months, baseline to 6 months, and baseline to 12 months has been noticed in our study. These results were parallel to studies conducted by Popova and Boyarova 41 and Anand et al. 42 In both groups, the significant gain in the width of keratinized gingiva could be an outcome of increased recession coverage. The width of the keratinized gingiva did not differ significantly between the groups (p > 0.05) from baseline to 3 months, baseline to 6 months, and baseline to 12 months. The results also revealed a more attached keratin layer in group II than in group I.
Gingival tissue thickness is a factor that may be relevant in an increased risk of gingival recession and could affect treatment outcomes. A significant increase in gingival tissue thickness was observed from baseline to the end of the study period in both groups. Similar results were reported by Silva et al. 43 and Aroca et al. 44
In the current study, immediate hemostasis was observed after applying the cyanoacrylate, which further helped in positioning the graft precisely. However, bleeding was still detected at 1 min after suturing. We also observed that cyanoacrylate produced less trauma and minimal swelling and postoperative bleeding. There was marginal end-to-end fixation of the graft with the underlying tissue during suturing; however, the graft fully adhered to the underlying surface after use of cyanoacrylate. Zenóbio et al. 45 reported similar findings.
Many limitations of this study should be discussed. First, the cost of the treatment procedure using bioadhesive material and the Er: YAG laser was high. Second, the sample size was small and the follow-up should have been longer. A longitudinal study may be needed. Another limitation of the study was that no histological sectioning was done to determine the type of attachment. We also realized that while the clinical parameters were assessed by two experienced periodontologists, inter-examiner agreement was not determined.
Conclusion
Cyanoacrylate was easy to apply, required less operating time, and resulted in no adverse reactions during graft healing. The 5-0 silk sutures and isoamyl 2 cyanoacrylate were equally efficacious for stabilizing the free gingival grafts harvested with the Er: YAG laser. However, the study also concluded that Clinical attachment Level, Width of keratinized gingiva, Gingival tissue thickness and the percentage root coverage values tended to be higher with cyanoacrylate as compared with silk suture.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from *This research protocol was approved by the Shanghai Huangpu Disease Prevention and Treatment Ethical and Review board with approval no. SH/dental/23-12/2013.
Informed consent
Written informed consent was obtained from all subjects before the study. Reply: Written informed consent was obtained from each and every participant of the study.
Trial registration
*NAME OF TRIAL REGISTRY: TRIAL REGISTRATION NUMBER: researchregistry5609