Abstract
Objective:
Opioid use disorder (OUD) affects approximately 5.6 million people in the United States annually, yet rates of the use of effective medication for OUD (MOUD) treatment are low. We conducted an observational cohort study from August 2017 through May 2021, the MOUD Study, to better understand treatment engagement and factors that may influence treatment experiences and outcomes. In this article, we describe the study design, data collected, and treatment outcomes.
Methods:
We recruited adult patients receiving OUD treatment at US outpatient facilities for the MOUD Study. We collected patient-level data at 5 time points (baseline to 18 months) via self-administered questionnaires and health record data. We collected facility-level data via questionnaires administered to facility directors at 2 time points. Across 16 states, 62 OUD treatment facilities participated, and 1974 patients enrolled in the study. We summarized descriptive data on the characteristics of patients and OUD treatment facilities and selected treatment outcomes.
Results:
Approximately half of the 62 facilities were private, nonprofit organizations; 62% focused primarily on substance use treatment; and 20% also offered mental health services. Most participants were receiving methadone (61%) or buprenorphine (32%) and were predominately non-Hispanic White (68%), aged 25-44 years (62%), and female (54%). Compared with patient-reported estimates at baseline, 18-month estimates suggested that rates of abstinence increased (55% to 77%), and rates of opioid-related overdoses (7% to 2%), emergency department visits (9% to 4%), and arrests (15% to 7%) decreased.
Conclusions:
Our results demonstrated the benefits of treatment retention not only on abstinence from opioid use but also on other quality-of-life metrics, with data collected during an extended period. The MOUD Study produced rich, multilevel data that can lay the foundation for an evidence base to inform OUD treatment and support improvement of care and patient outcomes.
Approximately 5.6 million people aged 12 years or older in the United States have an opioid use disorder (OUD) according to estimates from the 2021 National Survey on Drug Use and Health. 1 The US Food and Drug Administration (FDA) 2 has approved 3 medications for OUD (MOUD) treatment: methadone (a full agonist opioid), buprenorphine (a partial agonist opioid), and naltrexone (an opioid antagonist). Despite an increasing number of patients receiving MOUD and the potential for lowering associated health care costs, 3 the percentage of people with OUD who receive MOUD treatment remains small, not even reaching 35% by some estimates.1,4 Furthermore, people who initiate MOUD treatment may face barriers, including financial constraints, limited social support, stigma, and limited treatment availability, that are associated with prescription opioid misuse and illicit opioid use.5-8 Reasons for the relatively low rates of OUD treatment initiation and high rates of recurrence of opioid use are not fully understood, but they may be multilevel (eg, patient, provider, organization, state regulations).9-12 Similarly, factors that influence adverse events while receiving OUD treatment—such as substance use–related hospitalization, overdose, and criminal justice involvement—are not well understood.
Prior longitudinal studies of OUD treatment assigned patients to treatment rather than employing a naturalistic design to capture information on how physicians and patients determine the choice of OUD treatment. In addition, prior studies of OUD did not collect data on treatment facility or data from patients on reasons for nontreatment (eg, financial constraints) that could provide insight into correlates for positive treatment outcomes. Consequently, CDC sought the collection of longitudinal observational data on adults receiving outpatient treatment for OUD and on treatment facilities servicing the patients to identify correlates of positive treatment outcomes. Thus, the objectives of this 18-month observational cohort study in outpatient settings without random assignment to OUD treatment 13 were to (1) identify factors that influence the type of OUD treatment offered to patients and the patient’s treatment choice, (2) better understand patient and outpatient treatment facility factors associated with key OUD treatment outcomes, (3) identify relevant antecedents or co-occurring conditions that could be addressed through primary prevention, and (4) inform evidence-based practices and OUD treatment policies.
This article describes the MOUD Study design, recruitment protocols, and characteristics of the participating patients and OUD treatment facilities. The information, including initial findings, can serve as a reference for future research. Detailed data are scheduled for release in 2024.
Methods
MOUD Study Design
We developed the MOUD Study with input from experts at several US federal health agencies. The final design was an observational cohort study of outpatient OUD treatment with data collected repeatedly during 18 months from 62 OUD treatment facilities across 16 states (eFigure 1 in the supplemental material) and from patients receiving OUD treatment in the participating facilities. We conducted MOUD Study activities from August 2017 through May 2021 (eTable 1 in the supplemental material) and developed a recruitment and data collection protocol (eFigure 2 and eTable 2 in the supplemental material). We also collected qualitative data from patients via focus groups and from OUD treatment providers via focus groups and one-on-one discussions.
The Advarra Institutional Review Board (protocol no. Pro00036325) and the US Office of Management and Budget reviewed and approved this study, and its conduct was consistent with applicable federal law and the Centers for Disease Control and Prevention (CDC) policy (eg, 45 CFR part 46). Before study enrollment, all patients signed an informed consent that contained information on human subjects protection and a Health Insurance Portability and Accountability Act (HIPAA) waiver and noted the study’s Certificate of Confidentiality.
Study Enrollment
Treatment facilities
We recruited OUD treatment facilities from cities purposively chosen from across the United States to include sizable, diverse populations and a range of OUD treatment practices and policies that influence treatment engagement and outcomes.14-18 To promote geographic diversity in the participant sample, we included cities identified as likely serving patients living in rural areas; rural facilities are few and have a low patient volume.19-22
We obtained an initial set of facilities from the 2016 National Survey of Substance Abuse Treatment Services (N-SSATS) directory. 23 We identified additional facilities through discussions with staff at the contacted facilities. Facility recruitment occurred on a rolling basis from August 2017 through June 2019. Facility inclusion criteria were the following: (1) provided at least 1 of 3 FDA-approved MOUD options—methadone, buprenorphine (oral, injected, or implanted), or naltrexone (oral or injected)—or counseling without MOUD (behavioral intervention) in an outpatient setting and (2) treated at least 15 adult patients per month. We excluded Veterans Health Administration facilities and facilities providing OUD treatment only to court-ordered adults. We used the facility’s physical address and US Census Bureau categories to define facility census region. 24
Patients with OUD
Patient enrollment occurred on a rolling basis from March 2018 through December 2019. Initial patient inclusion criteria were (1) being aged 18-64 years (minimum age 19 years in Alabama 25 ), (2) having begun a new treatment episode at a study treatment facility within the previous 30 days, (3) having received 1 of the 4 OUD treatments under study, and (4) being able to complete the self-administered questionnaires in either English or Spanish (eighth-grade reading level). We later removed the upper limits on age and length of time the patients had been receiving their current OUD treatment to better represent the patient population and to increase enrollment. Enrolled patients were retained in the MOUD Study for the full 18-month study period, whether they continued any type of OUD treatment or ended treatment for any reason.
We shared study information with potential participants via posters and brochures available at facilities and through conversations with trained facility and field staff. Facility staff entered the contact information for interested patients into a secure electronic system; patients could also provide their contact information through a toll-free telephone number available on the study brochure. Project staff administered an English or Spanish telephone screener to verify study eligibility, collect demographic information and zip code of residence, and record the patient’s current type of OUD treatment (hereinafter, “index treatment”). To determine patient-level urban–rural status, we used the patients’ zip code of residence and the National Center for Health Statistics’ urban–rural classification scheme. 26 Field staff met with study-eligible patients to obtain a signed consent form, including a HIPAA waiver; discuss procedures to maintain confidentiality; and assist with the self-administered (baseline) questionnaire. Of the 2337 patients providing contact information, 1974 patients (85%) signed the consent form and completed the baseline questionnaire.
Data Collection
Treatment facility questionnaires
We asked facility directors to complete a secure, web-based questionnaire at baseline and again 18 months after baseline. We selected many questions from the N-SSATS 23 and the National Treatment Center Study 27 ; we devised additional questions to gather information specific to the MOUD Study. Topics included facility ownership status, OUD treatments offered, patient capacity, health care focus, types of health insurance accepted, and other services provided (eg, medically supervised withdrawal, psychosocial treatments, pain management, peer navigation). In addition, the baseline questionnaire asked about the number of patients, patient demographic characteristics, accreditation, staffing, and ongoing staff training. Three new questions were included on the 18-month questionnaire on staff retention and policies for patients who experience a recurrence of use while in treatment.
Patients With OUD
Patient questionnaires
Study enrollees were asked to complete 5 web-based, self-administered questionnaires during an 18-month period: baseline and follow-up at 3, 6, 12, and 18 months after baseline. We sent automated invitations and reminders to complete each follow-up questionnaire to patients via email or text message based on their specified contact preference. Prior to March 2020 COVID-19 travel restrictions, patients could request an in-person visit from field staff to complete the self-administered follow-up questionnaire on our study laptop. After COVID-19 travel restrictions were issued, staff helped patients by telephone to access the questionnaire from an available device (eg, smartphone). Patients were retained in the study even if they missed a follow-up questionnaire unless they rescinded their study enrollment.
The 5 self-administered patient questionnaires covered numerous topics developed by the study team (eTable 2 in the supplemental material). All questionnaires asked if the patient was still receiving their index treatment; if the patient was not, the patient was asked to indicate any OUD treatment they were receiving. We added questions from sources such as the 2015 National Survey on Drug Use and Health, 28 CDC’s Healthy Days measures found in the Behavioral Risk Factor Surveillance System, 29 the EQ-5D, 30 the Multidimensional Scale of Perceived Social Support, 31 and the Substance Abuse Self-Stigma Scale item pool. 32 Measures collected in the baseline and 12- and 18-month questionnaires included, for example, recent and past substance use (eg, prescription opioids, illicit fentanyl, marijuana, alcohol), past opioid quit attempts, history with the judicial system, drug overdose episodes, comorbidities and current health status, primary care services, and employment status. Questionnaires also asked about sources of social support, childhood trauma, mental health disorders, and suicidal ideation.
The 3- and 6-month patient questionnaires were brief compared with the baseline, and 12- and 18-month questionnaires and limited data collection to index treatment status, treatment services currently being received, treatment episodes after ending index treatment (if applicable), recent substance use, drug overdoses, health care use, and current health status. The reporting period for the treatment outcomes covered the past 3 months for the 3- and 6-month questionnaires, the past 12 months for the 12-month questionnaire, and the past 6 months for the 18-month questionnaire.
Patient health records
Treatment providers supplied information about study patients to confirm information obtained from the patient questionnaires. We requested information on MOUD dosage and/or number of counseling sessions, urine drug screen dates and results, comorbidities, and comments on the patient’s progress with OUD treatment. For each patient, we asked treatment providers to submit responses to a web-based form 1, 2, 3, 6, 9, and 12 months after the patient’s enrollment into the study through the end of the patient’s index treatment at that facility; each patient had a unique study identification number. If a treatment facility provided forms for fewer than 50% of their study patients, we asked the facility to submit corresponding information from the patient’s electronic health record (EHR) via a secure file transfer protocol.
Treatment providers at 35 facilities (57%) submitted at least 1 health form for all of their study patients, and 10 facilities (16%) provided none. Across the 62 participating facilities, OUD treatment providers submitted at least 1 of the 6 requested health forms for 63% of the study patients. Three treatment facilities submitted corresponding information from the patient’s EHR; results from the urine drug screens were comparable to patient responses provided in the questionnaires. Discussions with contacts at the treatment facilities suggested that limited staff capacity and competing demands contributed to lower-than-desired health form submission rates.
Statistical Analysis
We do not report on all outcomes of the MOUD Study in this article. In this article, we summarize univariate statistics to characterize the 62 MOUD Study treatment facilities and the 1974 enrolled patients. We calculated univariate prevalence estimates for a subset of OUD treatment outcomes, without tests of significance, to give researchers a glimpse of the data. We classified the subset of OUD treatment outcomes into 4 groups: substance abuse outcomes (abstinence, overdose), health care utilization outcomes (emergency department visits, visits to primary care physician), health screenings (HIV, hepatitis C), and social outcomes (employed, arrested). In future publications, we will present comparisons by treatment across time with estimates produced via multivariable, multilevel models that adjust for key covariates.
Results
Treatment Facility Questionnaires
Of the 62 participating facilities, 61 (98%) responded to the baseline facility questionnaire and 37 (60%) responded to the 18-month follow-up questionnaire. The largest percentage (36%; 22 of 62) of facilities were in the western United States (Table 1). Of the 61 facilities responding to the question on their primary focus, most (62%; n = 38) focused primarily on substance use services, and one-fifth (20%; n = 11) offered both mental health and substance use services. Approximately half (51%; 31 of 61) were private nonprofit organizations, and one-third (33%; 20 of 61) were private for-profit organizations. Of 57 facilities, 47 (83%) accepted Medicaid and 24 (42%) accepted Medicare.
Characteristics of 62 study treatment facilities participating in the MOUD Study, United States, August 2017–May 2021 a
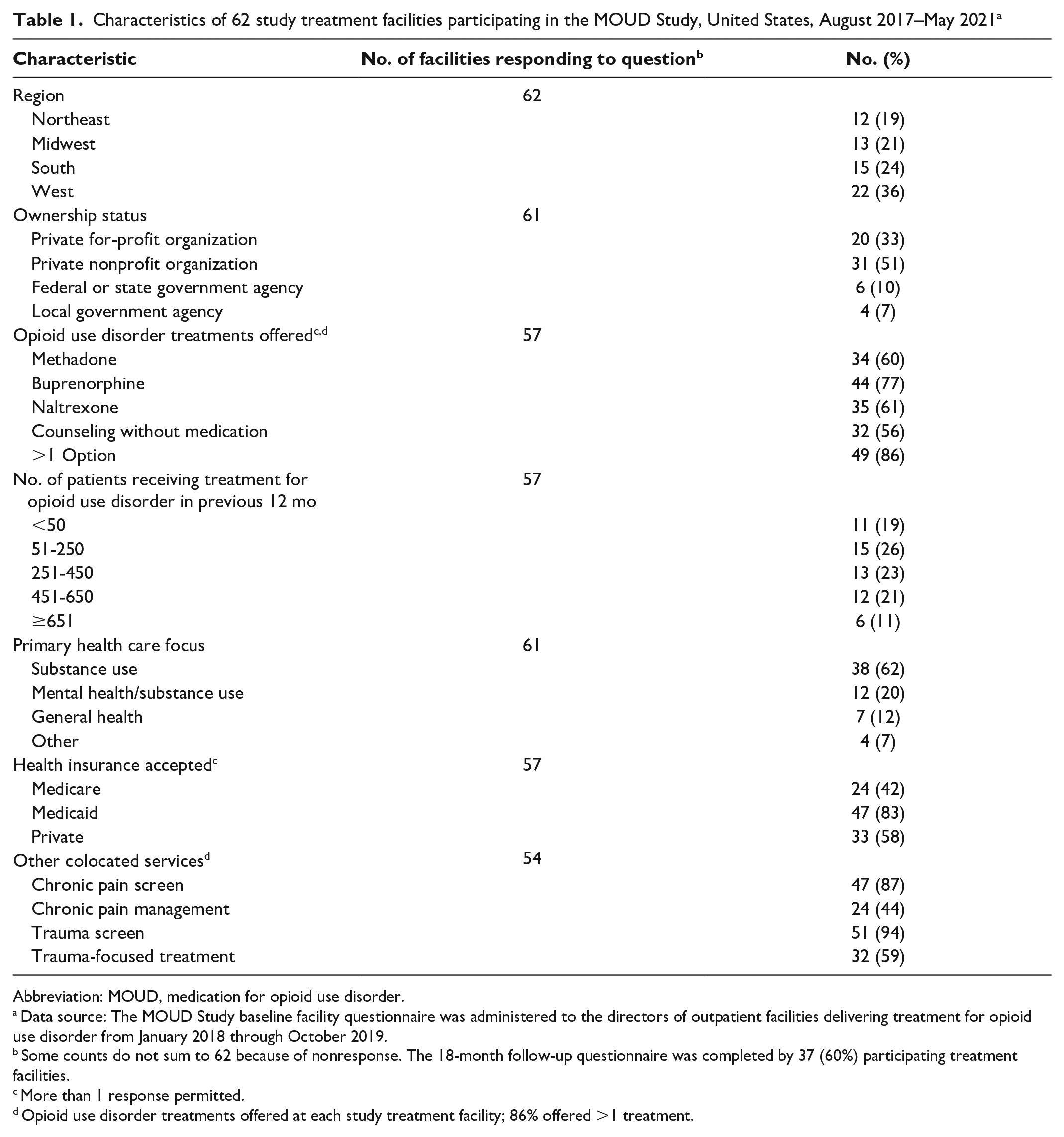
Abbreviation: MOUD, medication for opioid use disorder.
Data source: The MOUD Study baseline facility questionnaire was administered to the directors of outpatient facilities delivering treatment for opioid use disorder from January 2018 through October 2019.
Some counts do not sum to 62 because of nonresponse. The 18-month follow-up questionnaire was completed by 37 (60%) participating treatment facilities.
More than 1 response permitted.
Opioid use disorder treatments offered at each study treatment facility; 86% offered >1 treatment.
Of 57 facilities responding to the question on index treatments, 44 (77%) offered buprenorphine, 35 (61%) offered naltrexone, and 32 (56%) offered counseling without MOUD. Approximately 11% (6 of 57) of the facilities reported treating more than 650 patients for OUD in the previous 12 months. Facilities treating 51 to 250 patients were most prevalent (26%; 15 of 57).
In addition, many facilities screened for risk factors, including chronic pain (87%; 47 of 54) and trauma (94%; 51 of 54). However, only 44% (24 of 54) offered chronic pain management services, and 59% (32 of 54) offered trauma-focused treatments.
Patient Questionnaires
Of the 1974 patients who completed at least the baseline questionnaire, 72% (n = 1415) completed the 3-month questionnaire (Table 2). Participation rates for the subsequent questionnaires were lower, but all exceeded 50%. A total of 725 patients (37%) completed all 4 postbaseline questionnaires; 30 patients requested to withdraw from the MOUD Study after baseline.
Questionnaires completed by study patients (N = 1974), by index treatment, a in the MOUD Study, United States, August 2017–May 2021 b
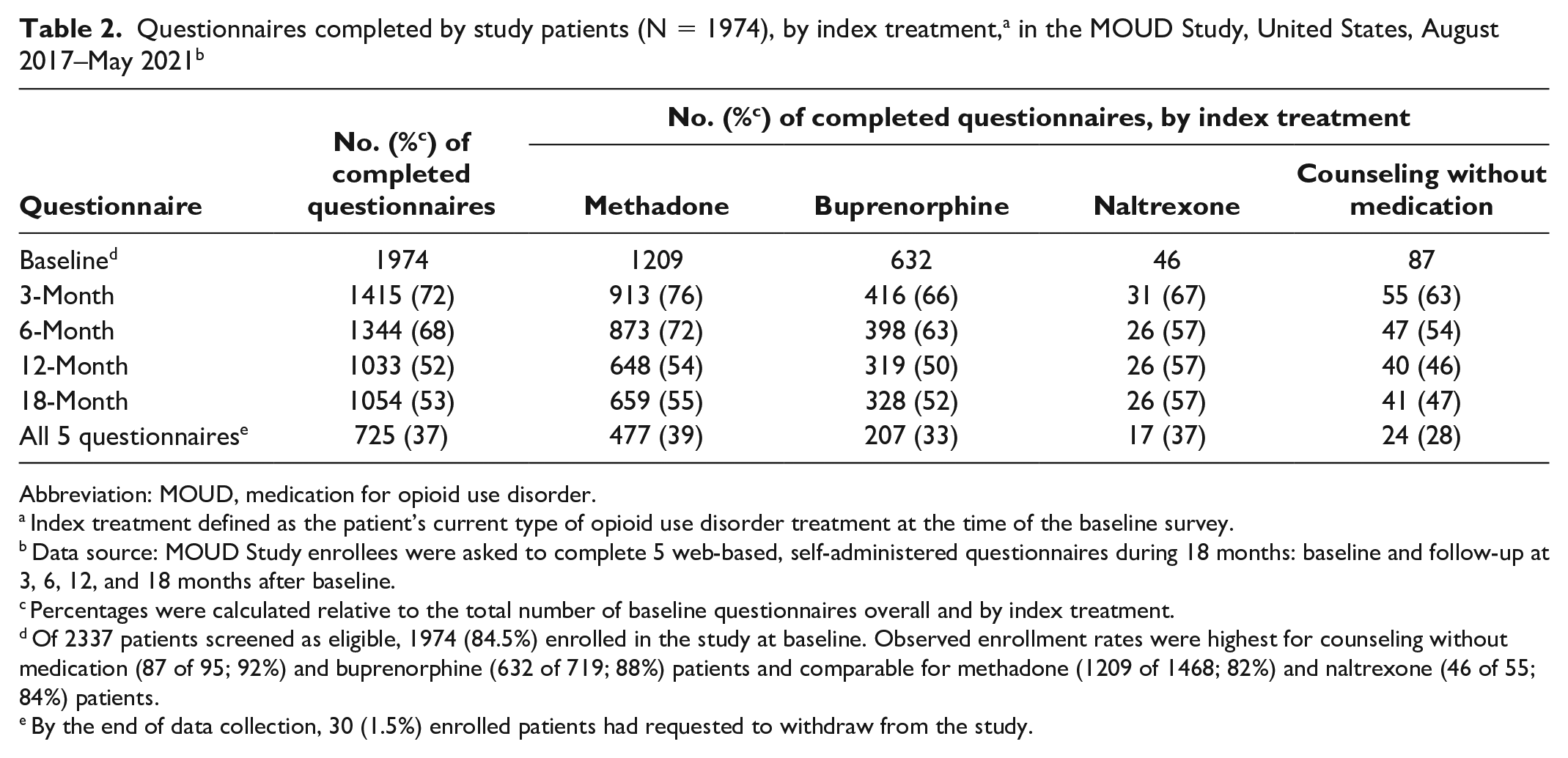
Abbreviation: MOUD, medication for opioid use disorder.
Index treatment defined as the patient’s current type of opioid use disorder treatment at the time of the baseline survey.
Data source: MOUD Study enrollees were asked to complete 5 web-based, self-administered questionnaires during 18 months: baseline and follow-up at 3, 6, 12, and 18 months after baseline.
Percentages were calculated relative to the total number of baseline questionnaires overall and by index treatment.
Of 2337 patients screened as eligible, 1974 (84.5%) enrolled in the study at baseline. Observed enrollment rates were highest for counseling without medication (87 of 95; 92%) and buprenorphine (632 of 719; 88%) patients and comparable for methadone (1209 of 1468; 82%) and naltrexone (46 of 55; 84%) patients.
By the end of data collection, 30 (1.5%) enrolled patients had requested to withdraw from the study.
Among the 1974 study patients, the most common index treatment was methadone (61%; n = 1209), followed by buprenorphine (32%; n = 632) (Table 3). Overall, the largest percentage of study patients (40%) were treated in facilities in the southern United States. More than half (54%) of the study patients were female. The sample was predominantly non-Hispanic White (68%). Only 28% of the study patients reported being employed at baseline.
Characteristics of study patients (N = 1974), overall and by index treatment, a in the MOUD Study, United States, August 2017–May 2021 b
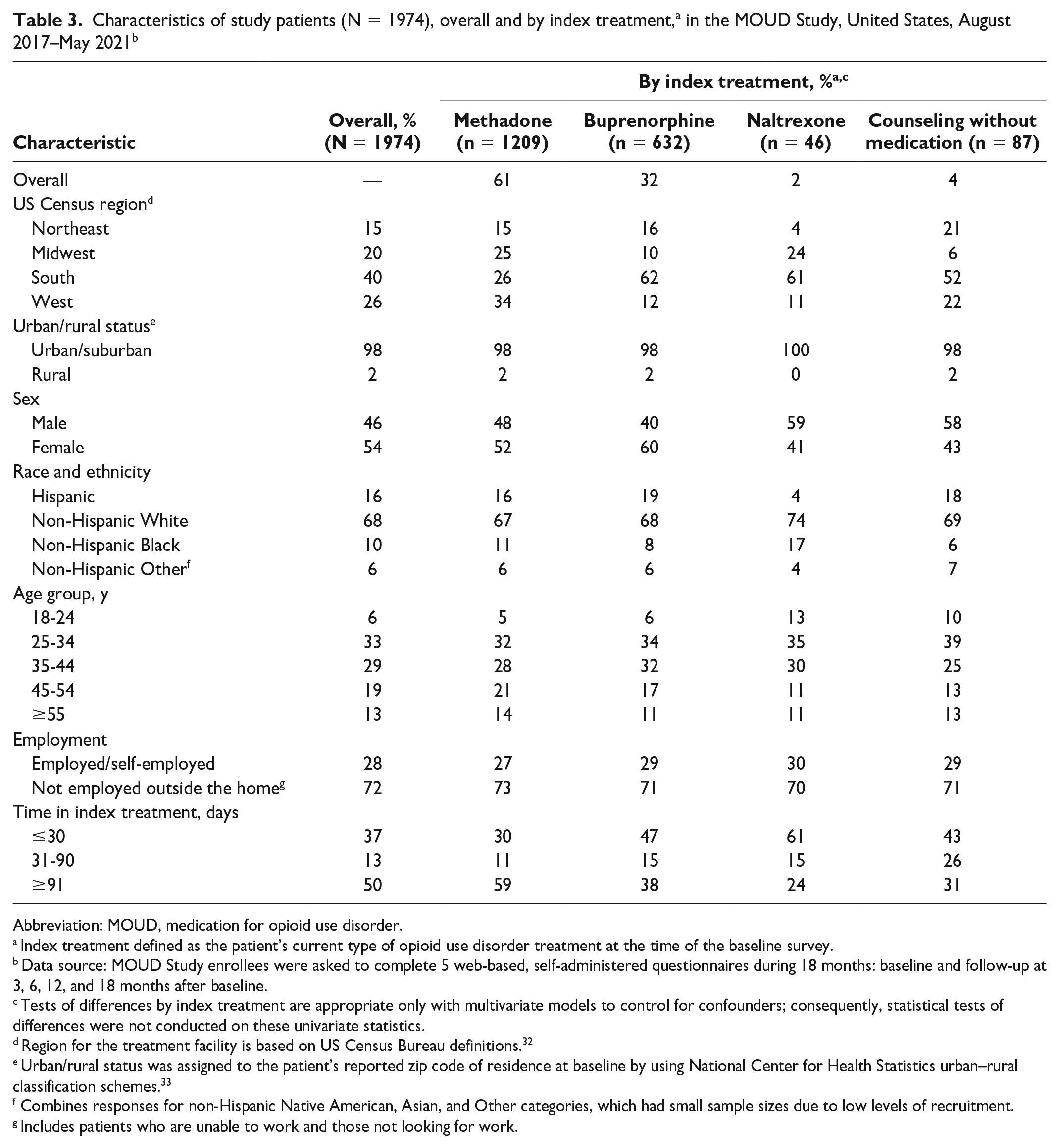
Abbreviation: MOUD, medication for opioid use disorder.
Index treatment defined as the patient’s current type of opioid use disorder treatment at the time of the baseline survey.
Data source: MOUD Study enrollees were asked to complete 5 web-based, self-administered questionnaires during 18 months: baseline and follow-up at 3, 6, 12, and 18 months after baseline.
Tests of differences by index treatment are appropriate only with multivariate models to control for confounders; consequently, statistical tests of differences were not conducted on these univariate statistics.
Region for the treatment facility is based on US Census Bureau definitions. 32
Urban/rural status was assigned to the patient’s reported zip code of residence at baseline by using National Center for Health Statistics urban–rural classification schemes. 33
Combines responses for non-Hispanic Native American, Asian, and Other categories, which had small sample sizes due to low levels of recruitment.
Includes patients who are unable to work and those not looking for work.
Most patients (63%) had been in their index treatment for more than 30 days when they enrolled in the MOUD Study. Overall, 35% (696 of 1974) of study patients had received any type of MOUD in the 12 months prior to study enrollment (Table 4). In the methadone index treatment group, for example, only 32% (382 of 1209) of patients reported recent prior experience with any MOUD at baseline. By the end of 18 months, 32% (342 of 1054) of study patients reported having ended their index treatment; at the 18-month interview, 83% (877 of 1054) reported receiving either their index treatment or some other OUD treatment.
Percentage of study patients (N = 1974) who reported at baseline having received MOUD in past 12 months, overall and by index treatment, a MOUD Study, United States, August 2017–May 2021 b
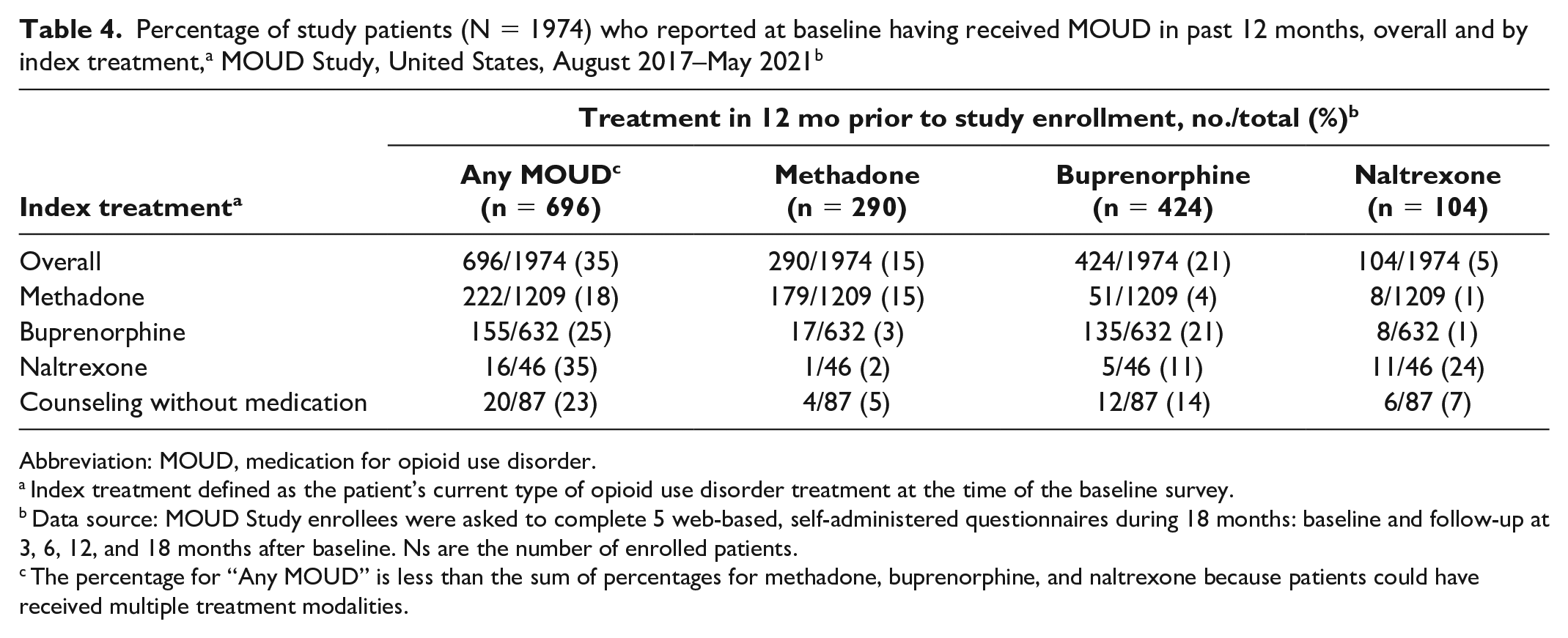
Abbreviation: MOUD, medication for opioid use disorder.
Index treatment defined as the patient’s current type of opioid use disorder treatment at the time of the baseline survey.
Data source: MOUD Study enrollees were asked to complete 5 web-based, self-administered questionnaires during 18 months: baseline and follow-up at 3, 6, 12, and 18 months after baseline. Ns are the number of enrolled patients.
The percentage for “Any MOUD” is less than the sum of percentages for methadone, buprenorphine, and naltrexone because patients could have received multiple treatment modalities.
Substance use outcomes
At baseline, 55% of study patients reported abstinence from prescription opioids, heroin, or illicit fentanyl in the previous 30 days; the percentage increased to 77% for study patients responding to the 12- and 18-month questionnaires (Table 5). At baseline, 7% of study patients reported an overdose related to opioid or substance use in the previous 90 days; rates of reported overdose decreased to 2% at 3 months and remained constant throughout the 18-month observation period.
Percentage of study patients who reported treatment outcomes, by patient questionnaire, MOUD Study, United States, August 2017–May 2021 a
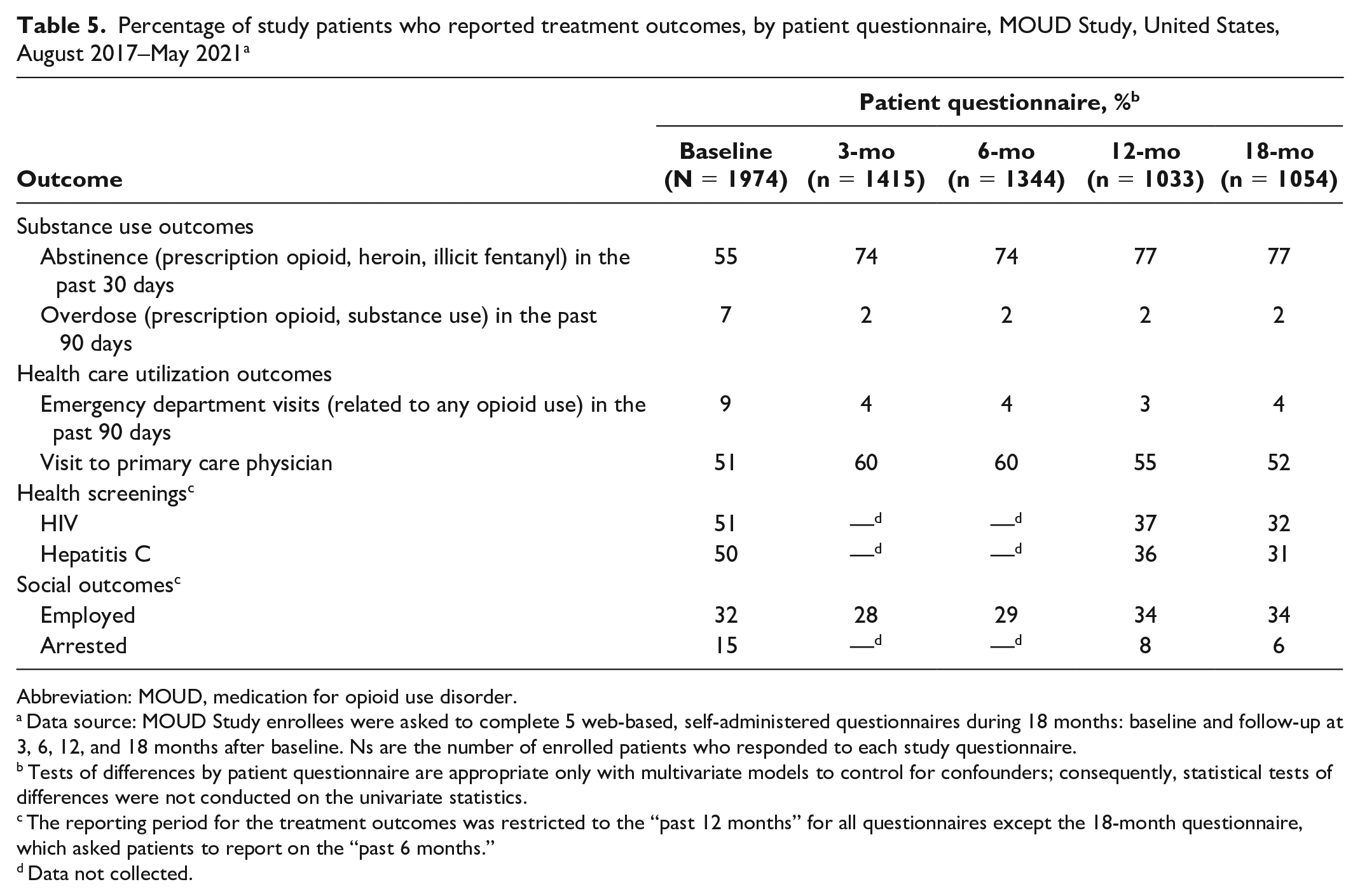
Abbreviation: MOUD, medication for opioid use disorder.
Data source: MOUD Study enrollees were asked to complete 5 web-based, self-administered questionnaires during 18 months: baseline and follow-up at 3, 6, 12, and 18 months after baseline. Ns are the number of enrolled patients who responded to each study questionnaire.
Tests of differences by patient questionnaire are appropriate only with multivariate models to control for confounders; consequently, statistical tests of differences were not conducted on the univariate statistics.
The reporting period for the treatment outcomes was restricted to the “past 12 months” for all questionnaires except the 18-month questionnaire, which asked patients to report on the “past 6 months.”
Data not collected.
Health care utilization outcomes
Approximately 9% of study patients reported an opioid-related emergency department visit in the 90 days before study enrollment; emergency department visits declined by half, to 4%, among study patients responding to the 18-month questionnaire (Table 5). Visits to a primary care physician at baseline (51%) increased slightly at 3 months and 6 months and declined thereafter to 52% at 18 months.
Health screenings
Half (51%) of patients reported having an HIV test in the 12 months prior to study enrollment; this rate dropped to 32% among patients who completed the 18-month questionnaire. Testing for hepatitis C was similar at baseline (50%) to HIV testing, with the rate dropping to 31% among respondents to the 18-month questionnaire.
Social outcomes
The employment rate at 18 months (34%) was slightly higher than reported at baseline (32%). At baseline, 15% of patients reported being arrested in the 12 months before study enrollment; at 18 months, 7% reported being arrested in the previous 6-month period.
Discussion
This observational cohort study during an 18-month period demonstrates the association of MOUD on key patient outcomes. Compared with estimates at baseline, patient-reported estimates at 18 months indicated that abstinence rates increased (55% to 77%); rates of opioid-related overdoses (7% to 2%), emergency department visits (9% to 4%), and arrests (15% to 7%) decreased; and rates of health care maintenance (51% to 52%) and employment (32% to 34%) improved slightly. In addition, results showed room for improvement in providing services for chronic pain management and trauma-informed care. Future investigations will control for selected correlates—such as length of index treatment, subsequent treatment episodes, demographic characteristics, and social support—to provide the basis for comparison with existing research and to test for significant changes over time.
The benefits of MOUD treatment for patients with OUD is well established, but the recommended length of treatment is not well defined. 34 Consequently, longitudinal studies, such as the MOUD Study, are needed to understand the interplay of various support systems (eg, MOUD, counseling, social programs, family dynamics) during an extended period. Other researchers have conducted longitudinal studies of patients with OUD to assess factors associated with positive treatment experience. For example, Hser et al 33 reported on a multiyear longitudinal study of 795 patients randomized to either methadone or buprenorphine, finding few differences between the treatments. Weiss et al 35 followed 375 patients receiving buprenorphine treatment during a 42-month period and found that those who misused prescription opioids had better outcomes than many who had used heroin. Although these studies obtained key information, neither captured OUD treatment data (eg, supportive services) from facilities or nontreatment data (eg, financial constraints) from patients that could provide insight into correlates for positive treatment outcomes such as abstinence from illicit opioid use.
Limitations
The MOUD Study data and results should be viewed in context of the study’s overall goals and several limitations. First, the MOUD Study was intended to provide information to assess correlates and outcomes by treatment instead of the relative efficacy of one treatment versus another. Consequently, results should not be interpreted as recommending one treatment modality over another. Second, the latest OUD practice guideline 36 emphasizes the use of MOUD in conjunction with other supportive services rather than treatment without MOUD. Potentially reflecting this shift, we experienced greater challenges than anticipated in enrolling patients who were receiving counseling without MOUD. Similarly, relatively few people who were receiving naltrexone were enrolled, perhaps because of its limited use in community-based settings due to higher cost compared with other MOUD and the additional services needed for medically supervised withdrawal.37,38 Consequently, estimates for patients receiving counseling or naltrexone are underpowered. Third, not every facet of outpatient treatment of adults with OUD is available for evaluation with the data collected. For example, study results may not pertain to patients with OUD being treated at rural facilities. 39 In addition, although we purposefully chose cities to capture data on a sizeable, diverse population, estimates may not be generalizable to all urban-based outpatient treatment conditions. As such, results from the MOUD Study may not reflect the full population of patients in treatment for OUD. Fourth, nonresponse is a common limitation of longitudinal studies. The patient questionnaires are the main source of data for this study, with participation rates ranging from 52% to 72% and almost 37% of participating patients completing all 5 questionnaires. Study participation decreased after March 2020, when COVID-19 pandemic–related travel restrictions were instituted, suggesting that in-person interactions between field staff and patients were beneficial in maintaining study engagement.40,41 Consequently, some analyses may be underpowered or unavailable with the MOUD Study data.
Fifth, during the COVID-19 pandemic, all communications between field staff and the study patients were made via telephone (voice or text) or email. Facility staff provided updated contact information for some patients. Consequently, nonresponse may be attributed to relocation, waning interest in the study without request to disenroll, or death. Authorization to access CDC’s National Death Index is in progress to identify and compare, for example, the sociodemographic characteristics of opioid-related overdose and all-cause mortality for any patients without an 18-month interview. Therefore, bias associated with nonresponse may have affected our results. Sixth, the lowest participation rates were for the requested patient health records. Much of these data are available from the patient questionnaires, albeit without corroboration from test results. Patient questionnaire responses were consistent with the submitted health record information, but this association may not apply to all study patient data. Consequently, errors in the self-reported information may have biased our univariate statistics.
Conclusion
The MOUD Study was designed to provide longitudinal observational data on OUD treatment facilities and adults receiving outpatient treatment for OUD to identify correlates of positive treatment outcomes. The study is poised to meet these objectives with results obtained from future multilevel, multivariable, model-based analyses—including the complex patterns of OUD treatment termination and re-initiation—that can inform evidence-based clinical care practice and discussions by policy makers, health care providers, and others involved in the treatment of OUD.
Supplemental Material
sj-docx-1-phr-10.1177_00333549231222479 – Supplemental material for The Medications for Opioid Use Disorder Study: Methods and Initial Outcomes From an 18-Month Study of Patients in Treatment for Opioid Use Disorder
Supplemental material, sj-docx-1-phr-10.1177_00333549231222479 for The Medications for Opioid Use Disorder Study: Methods and Initial Outcomes From an 18-Month Study of Patients in Treatment for Opioid Use Disorder by Jill A. Dever, Marci F. Hertz, Laura J. Dunlap, John S. Richardson, Sara Beth Wolicki, Bradley B. Biggers, Mark J. Edlund, Michele K. Bohm, Didier Turcios, Xinyi Jiang, Hong Zhou, Mary E. Evans and Gery P. Guy in Public Health Reports
Footnotes
Acknowledgements
The authors thank staff at the following federal health agencies for their review of the design and results from this study: the US Food and Drug Administration, the Health Resources & Services Administration, the National Institute on Drug Abuse, the Office of the Assistant Secretary for Planning and Evaluation, and the Substance Abuse and Mental Health Services Administration. The authors also acknowledge the invaluable contributions of the following team members from RTI: Victoria Albright, MA, Matthew Bensen, MA, Derick Brown, BS, Michelle Casper, BA, Cindy O. McClintock, BA, Mary McMillan, PMP, Stephanie Terry, MA, Thomas S. Walker, BA, Dorothy Wu, BA, and many others.
Disclaimer
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention, RTI International, or Stop Soldier Suicide.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: RTI International conducted the study under contract no. HHSD2002013M53964B with the Centers for Disease Control and Prevention.
Supplemental Material
Supplemental material for this article is available online. The authors have provided the supplemental material to give readers additional information about their work. These materials have not been edited or formatted by
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
