Abstract
Objectives:
In May 2021, the Health Resources and Services Administration Health Center COVID-19 Vaccine Program (HCCVP) began supporting the national adolescent vaccination rollout for a safe return to in-person learning for children and adolescents from medically underserved communities. To understand the initial implementation of adolescent vaccinations, we estimated the number of vaccines administered through the HCCVP at the national and state level to adolescents aged 12-17 years, and we examined challenges and solutions in vaccine deployment.
Methods:
We analyzed data on vaccine administration, challenges, and solutions from the Health Center COVID-19 Survey during May 14–August 27, 2021, and we analyzed data on patients served from the 2019 Uniform Data System. National adolescent COVID-19 vaccination and population data came from CDC’s COVID Data Tracker and the US Census Bureau’s 2019 Current Population Survey.
Results:
HCCVP health centers administered >485 000 COVID-19 vaccine doses to adolescents during the study period, with variations across states. Health centers in 13 states and territories (Arizona, California, Colorado, Connecticut, Delaware, Maine, Massachusetts, Missouri, Nebraska, Nevada, Oregon, Virginia, and Puerto Rico) vaccinated more adolescents than their share of prepandemic adolescent patients. The most frequently reported challenges in vaccine administration were vaccine confidence and staffing availability.
Conclusions:
This assessment of the initial months of COVID-19 vaccine administration among adolescent health center patients suggests rapid response by health centers in several states, reaching beyond their adolescent patient population to support state-level pandemic response. Future research could examine processes to optimize strategic activation of health centers in public health emergency responses.
Keywords
During the first year of the COVID-19 pandemic, adolescents were hospitalized for COVID-19 at nearly 3 times the typical hospitalization rate for influenza, with nearly one-third of cases requiring intensive care. 1 The pandemic also significantly affected adolescents’ mental and emotional health, social well-being, and educational experiences.2,3 A July 2020 survey of young people aged 14-24 years in the United States found that most (90%) believed that contracting COVID-19 would negatively affect their lives and most (86%) were concerned about spreading the virus. 4 COVID-19 vaccinations for adolescents are a critically important public health tool in protecting against potential COVID-19 infections, medical complications, and long-lasting symptoms that may influence adolescent health, mental health, and quality of life as well as reducing transmission in the general population.5-8
On May 10, 2021, the US Food and Drug Administration (FDA) authorized the Pfizer-BioNTech COVID-19 vaccine for use in children and adolescents aged 12-15 years through Emergency Use Authorization (EUA). 9 Two days later, the Centers for Disease Control and Prevention (CDC) Advisory Committee on Immunization Practices (ACIP) made an interim recommendation for use of the vaccine in that age group. 10 With the EUA and ACIP recommendations for children and adolescents aged 12-15 years, approximately 17 million people in this age group became eligible for vaccination, in addition to almost 9 million adolescents aged 16-17 years who were previously eligible when the Pfizer-BioNTech vaccine first received EUA.11,12
Health centers supported by the Health Resources and Services Administration (HRSA) serve more than 3 million children and adolescents aged 12-17 years from medically underserved communities, representing 1 in 10 US children and adolescents nationally, through more than 1400 health centers with >15 000 primary care service delivery sites in every US state, US territory, and the District of Columbia. 13 To ensure equitable vaccine distribution, HRSA launched the federal Health Center COVID-19 Vaccine Program (HCCVP) in February 2021, in collaboration with CDC, to directly distribute an allocation of limited supply of COVID-19 vaccines to health centers serving populations disproportionately affected by COVID-19, including individuals experiencing homelessness, public housing residents, migrant/seasonal agricultural workers, and patients with limited English proficiency.14,15 From January through May 2021, health centers administered more than 10 million doses of the COVID-19 vaccine through vaccine allocations from the HCCVP as well as health departments in states and jurisdictions. Health centers made important advances in equitable distribution of COVID-19 vaccines among adults: from January through May 2021, people from racial and ethnic minority groups made up nearly two-thirds (64%) of people receiving the first dose and 61% of people receiving the second/final dose of the vaccine at health centers. 16 Subsequently, the HCCVP was asked to expand its efforts beyond the original target populations and support the national rollout of COVID-19 vaccines for adolescents from May through August 2021 to enable a safe return to in-person learning in fall 2021 for children from medically underserved communities who experienced disparate access to school (eg, technological barriers) and language barriers during the pandemic. 17
The objective of this study was to understand health centers’ progress and challenges during COVID-19 vaccine administration to adolescent populations in the summer months before the 2021-2022 school year. To ascertain progress, we estimated national and state-level COVID-19 vaccine doses administered to adolescents aged 12-17 years from May through August 2021, for health centers participating in the HCCVP and for the general US adolescent population. We also compared the state-level distribution of vaccine administrations with the distribution of adolescent health center patients served prior to the pandemic in 2019. To understand challenges, we examined vaccine deployment difficulties and solutions reported by health centers.
Methods
Health Center Vaccine Administration and Patient Data
During the study period, 832 health centers, including 798 HRSA-funded health centers and 34 look-alikes, participated in the HCCVP. Look-alikes are HRSA-designated federally qualified health centers that provide primary health care services but do not receive Health Center Program funding. 18 Data on health center COVID-19 vaccine administration to adolescents came from the Health Center COVID-19 Survey, which is a survey administered to health centers on a rolling basis that launched in spring 2020 to understand the effect of COVID-19 on health centers’ operations, patients, and staff as well as the COVID-19 response in medically underserved communities. The survey contains an addendum for health centers participating in the HCCVP, which includes fields to report the number of children and adolescents (aged <18 y) who initiated their COVID-19 vaccination series (first dose) and the number of children and adolescents who received their second dose. 19 The survey also includes yes or no questions about specific types of challenges (ie, vaccine confidence, staffing, financial reimbursement, vaccine supply, storage capacity, other) faced by health centers in deploying the COVID-19 vaccine and an optional open-ended question for additional qualitative responses about challenges. All health centers participating in the HCCVP are required to respond to the survey and addendum, and respondents include health center personnel supporting the overall COVID-19 response (eg, chief executive officer, director of nursing, director of quality in health care). The survey and addendum were administered weekly from May 14 through July 2, 2021, with respondents reporting on activities in the previous week. The survey then transitioned to biweekly administration from July 16 through August 27, 2021, and respondents reported on activities in the previous 2 weeks. The survey response rates ranged from 86% to 94% throughout the study period (May 14–August 27, 2021).
Data on the total number of adolescent patients (aged 12-17 y) served by health centers participating in the HCCVP came from the 2019 Uniform Data System (UDS). 13 The 2019 UDS was selected as the source for patient population counts because it more accurately reflected prepandemic health center operations than the 2020 UDS. Additional analyses of the effect of the pandemic during 2020 on the adolescent patient population served (eTable 1 in Supplemental Material) provide further rationale for using the 2019 UDS. The UDS is a standardized core set of annual reporting requirements that includes data on patient characteristics, primary care services provided, clinical processes and health outcomes, patients’ use of services, staffing, costs, and revenues from health centers. 13 Of the 832 health centers participating in the HCCVP, 11 health centers were new, did not have 2019 UDS data available, and were excluded from subsequent analyses. Because these 11 health centers administered only 63 COVID-19 vaccine doses to adolescents during the study period (0.01% of the total number of vaccine doses administered to adolescents), this exclusion had minimal impacts on the analyses.
General Population Data
Data on population counts of US adolescents came from the US Census Bureau’s 2019 Current Population Survey, and data on COVID-19 vaccinations among adolescents in the general population came from CDC’s COVID Data Tracker, which includes downloadable real-time data on vaccine doses administered in each state.20,21 We downloaded CDC state-level data as of August 29, 2021, to align with the last wave of the Health Center COVID-19 Survey used in the study. 21 Per the US Department of Health and Human Services Policy for Protection of Human Research Subjects, the study did not qualify as human subjects research; therefore, the requirements specified by the Common Rule (including institutional review board review and determination) were not applicable.
Statistical Analysis
From the Health Center COVID-19 Survey, we obtained the weekly (and later biweekly) number of first and second COVID-19 vaccine doses administered by HCCVP-participating health centers to adolescents aged 12-17 years from May 14 through August 27, 2021. We summed first and second doses to calculate the cumulative number of doses administered by the HCCVP. We estimated the cumulative percentage of adolescent health center patients receiving first and second COVID-19 vaccine doses during this period, using the total number of adolescent patients served in health centers during 2019 as the denominator.
At the national and state levels, we compared health centers participating in the HCCVP with the general US population with respect to (1) the total number of COVID-19 vaccinations administered to adolescents (first and second doses), (2) the percentage of adolescents vaccinated by the end of the study period, and (3) the share of all vaccinations administered to US adolescents by the HCCVP. We also examined the state-level distribution of COVID-19 vaccine doses administered by HCCVP health centers, by calculating each state’s share of the total vaccines administered through the HCCVP. We compared this distribution with the state-level distribution of adolescent patients served by health centers by calculating the ratio of first vaccine dose proportions to adolescent patient proportions in each state. We used these ratios to identify states as “high performers” within the HCCVP, where the share of states’ vaccine administrations was equal to or higher (ratio ≥ 1.0) than their share of adolescent health center patients.
Finally, we examined trends in types of vaccine deployment challenges and optional qualitative narrative comments. We searched for terms in comments related to adolescent vaccinations as follows: adolescents, youth, teens, children, pediatric, 12-17 years, 12-15 years, <18 years, and ≥12 years. We identified a total of 160 write-in responses describing challenges in adolescent vaccination administration as well as examples of solutions. We reviewed and grouped the responses to identify themes.
Results
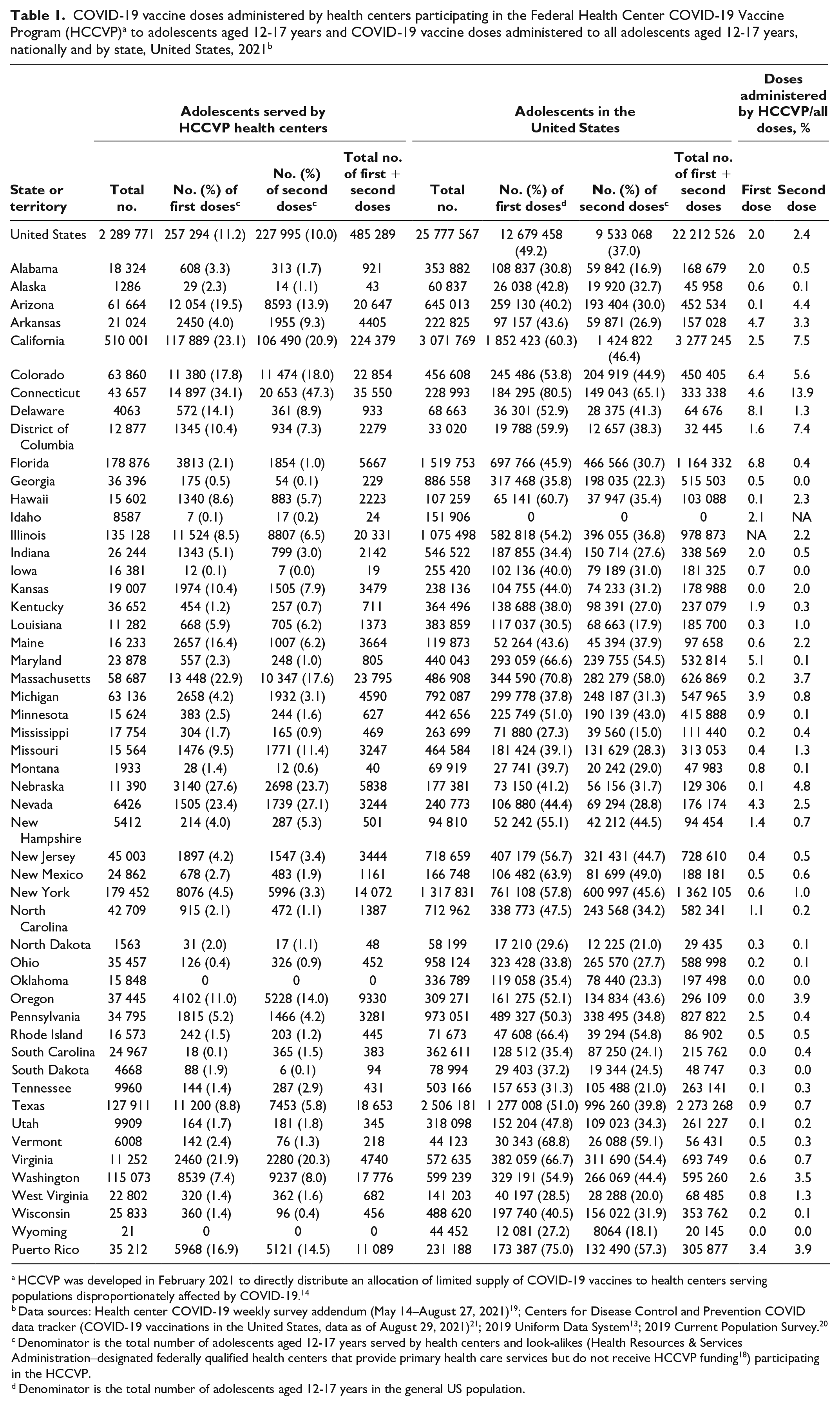
More than 485 000 COVID-19 vaccine doses were administered to adolescents by health centers participating in the HCCVP from May through August 2021, including 257 294 first doses and 227 995 second doses across the 50 states, the District of Columbia, and Puerto Rico (Table 1). Nationally, an estimated 11.2% of adolescent health center patients received their first dose and 10.0% received their second dose through the HCCVP vaccine supply.
HCCVP was developed in February 2021 to directly distribute an allocation of limited supply of COVID-19 vaccines to health centers serving populations disproportionately affected by COVID-19. 14
Data sources: Health center COVID-19 weekly survey addendum (May 14–August 27, 2021) 19 ; Centers for Disease Control and Prevention COVID data tracker (COVID-19 vaccinations in the United States, data as of August 29, 2021) 21 ; 2019 Uniform Data System 13 ; 2019 Current Population Survey. 20
Denominator is the total number of adolescents aged 12-17 years served by health centers and look-alikes (Health Resources & Services Administration–designated federally qualified health centers that provide primary health care services but do not receive HCCVP funding 18 ) participating in the HCCVP.
Denominator is the total number of adolescents aged 12-17 years in the general US population.
The estimated percentage of adolescent health center patients receiving their first dose from HCCVP health centers ranged from 0% in Oklahoma and Wyoming to 34.1% in Connecticut. The estimated proportion of adolescent health center patients receiving the first dose of COVID-19 vaccine was 20% or more at HCCVP health centers in 6 states (California, Connecticut, Massachusetts, Nebraska, Nevada, and Virginia) (Figure 1). Thirty-seven states administered HCCVP first doses to 10% or fewer adolescent health center patients. We observed similar patterns in the distribution of second doses.
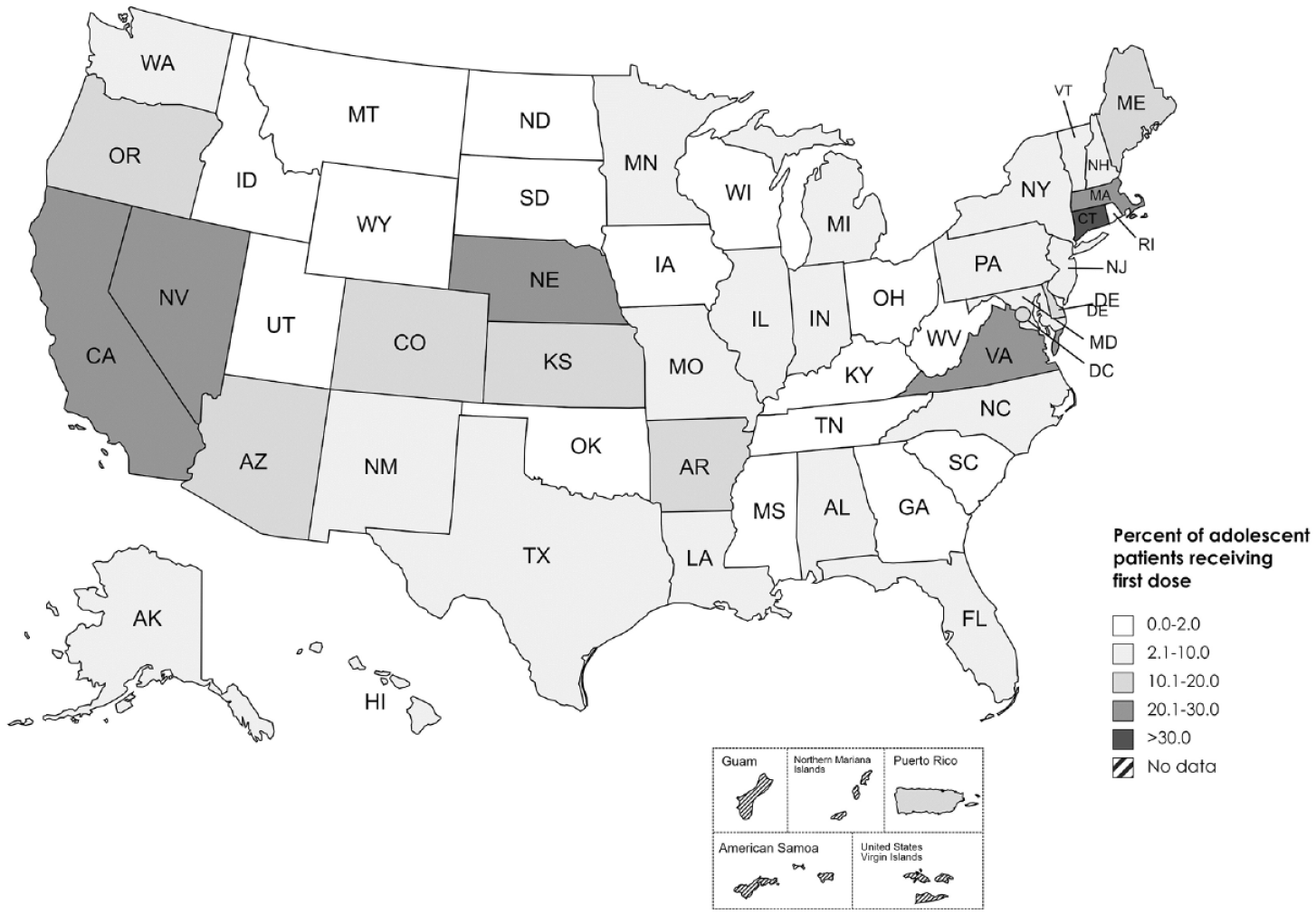
Percentage of health center patients aged 12-17 years who received a first dose of COVID-19 vaccine through health centers participating in the federal Health Center COVID-19 Vaccine Program (HCCVP), by state, May–August 2021. HCCVP was developed in February 2021 to directly distribute an allocation of limited supply of COVID-19 vaccines to health centers serving populations disproportionately affected by COVID-19. 14
Across the US general population, a total of 22 212 526 doses were administered to adolescents during the study period (Table 1). As of August 29, 2021, 49.2% of all US adolescents had received a first dose and 37.0% of all adolescents had received a second dose of COVID-19 vaccine. Health centers participating in the HCCVP administered 2.0% of all first doses and 2.4% of all second doses of COVID-19 vaccine administered to US adolescents. HCCVP-participating health centers in 4 states administered >5% of total first doses to adolescents in their state (California [6.4%], Connecticut [8.1%], District of Columbia [6.8%], and Maine [5.1%]) and 4 states administered >5% of total second doses to adolescents in their state (California [7.5%], Colorado [5.6%], Connecticut [13.9%], and the District of Columbia [7.4%]).
In an examination of state-by-state ratios of the percentage of all HCCVP first vaccine doses delivered by health centers to the percentage of all adolescents served by health centers in 2019, we identified 13 states as “high performers” in the HCCVP (Figure 2). For example, health centers in California served 22.3% of all adolescent health center patients across the nation but administered 45.8% of all first doses administered through the HCCVP (ratio = 2.1). Similarly, health centers in Connecticut served 1.9% of all adolescent health center patients but administered 5.8% of all first doses through the HCCVP (ratio = 3.0). The remaining high-performing states/territories for adolescent COVID-19 vaccinations administered to adolescents through the HCCVP were Arizona, Arkansas, Colorado, Delaware, Maine, Massachusetts, Nebraska, Nevada, Oregon, Virginia, and Puerto Rico.
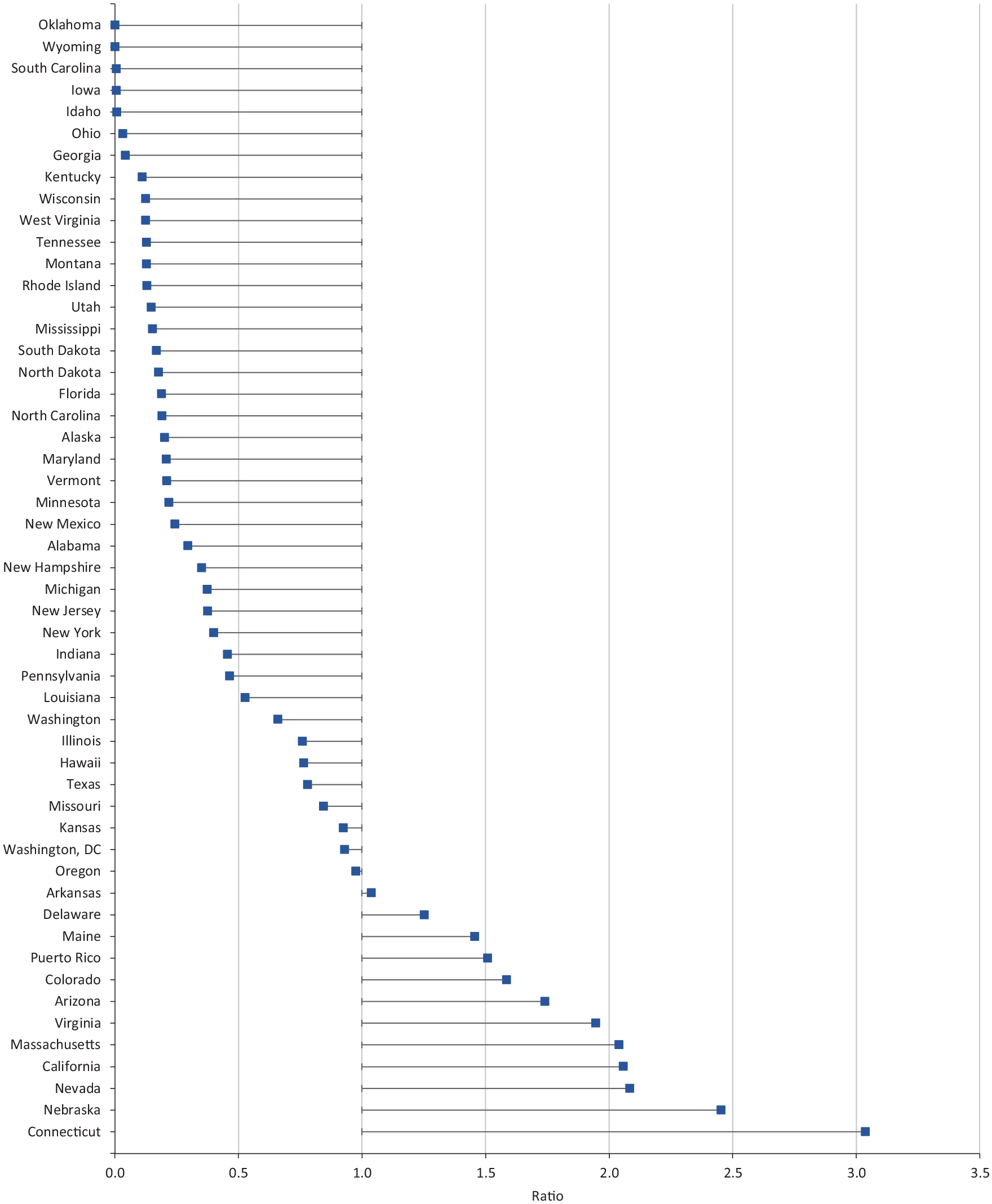
Ratio of proportion of COVID-19 vaccine first doses delivered to children and adolescents aged 12-17 years by health centers participating in the federal Health Center COVID-19 Vaccine Program (HCCVP) to the proportion of adolescents served by health centers in 2019. HCCVP was developed in February 2021 to directly distribute an allocation of limited supply of COVID-19 vaccines to health centers serving populations disproportionately affected by COVID-19. 14
The most frequently reported challenge to vaccination was vaccine confidence, reported by 29.9% of health centers on May 14 and increasing to 43.9% by July 30, before dipping slightly by the end of the study period (Figure 3). The second most frequently reported challenge was staffing to administer COVID-19 vaccines, affecting 29.3% of health centers at the beginning of the study period and dropping to 17.1% by July 16, before increasing again to 25.6%. Other challenges were less frequently reported (financial reimbursements, range: 5.2%-8.9%; vaccine supply, range: 3.5%-4.8%; vaccine storage, range: 2.1%-5.2%). For each reporting period from May through August 2021, approximately one-third of health centers reported no vaccine deployment challenges.
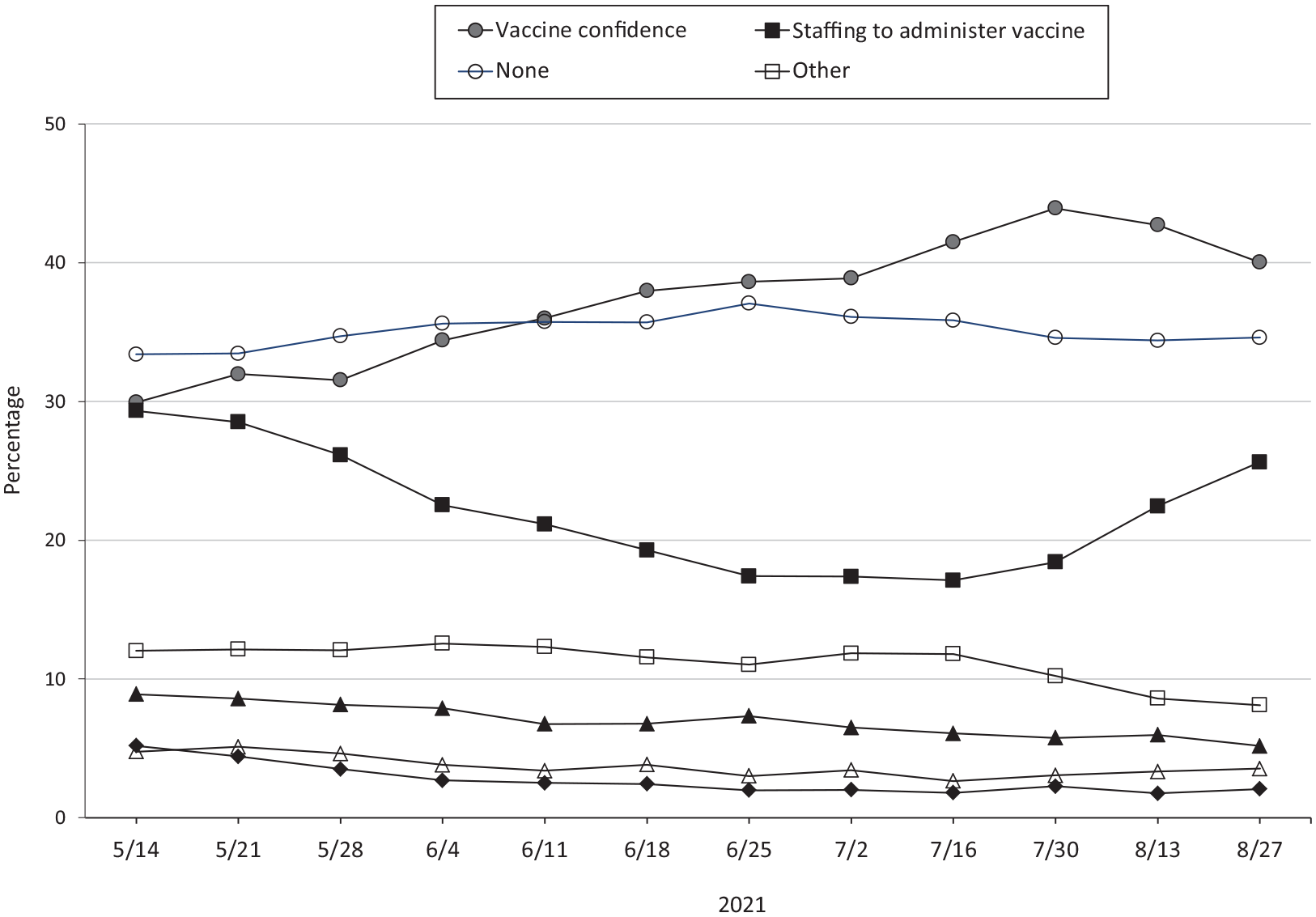
Trends in vaccine deployment challenges reported by health centers/look-alikes participating in the federal Health Center COVID-19 Vaccine Program (HCCVP), May–August 2021. HCCVP was developed in February 2021 to directly distribute an allocation of limited supply of COVID-19 vaccines to health centers serving populations disproportionately affected by COVID-19. 14 Look-alikes are Health Resources and Services Administration–designated federally qualified health centers that provide primary health care services but do not receive HCCVP funding. 18 Data source: Health center COVID-19 weekly survey addendeum (May 14–August 27, 2021). 19 The number of reporting health centers differed for each reporting period and ranged from 716 to 782. States with a ratio >1.0 delivered a greater share of the first doses of HCCVP COVID-19 vaccines to adolescents than their share of health center adolescents served in 2019.
Six themes that emerged from the survey’s qualitative comments revealed additional details about the challenges faced by health centers and solutions that health centers pursued (Table 2). Challenges reported by health center staff included parents’ safety concerns about their children getting vaccinated, staff being stretched thin due to quarantining/isolation requirements among exposed or infected staff, health centers not using Pfizer vaccines as part of their vaccination activities due to storage requirements and large vaccine allotments, adolescents getting vaccinated at other locations (eg, schools, pharmacies, mass vaccination sites), and low demand during the summer break. Health centers described several approaches to counter challenges, including developing social media campaigns to address vaccine misinformation and hesitancy, collaborating with community partners (eg, schools, children’s hospitals, health departments, youth organizations) to promote vaccination and conduct pediatric vaccine clinics, and capitalizing on every interaction with patients and their families to administer COVID-19 vaccines during sports physicals, back-to-school visits, and routine primary care visits.
Challenges and solutions in administering COVID-19 vaccines to children and adolescents aged 12-17 years through health centers participating in the federal Health Center COVID-19 Vaccine Program, United States, May through August 2021 a
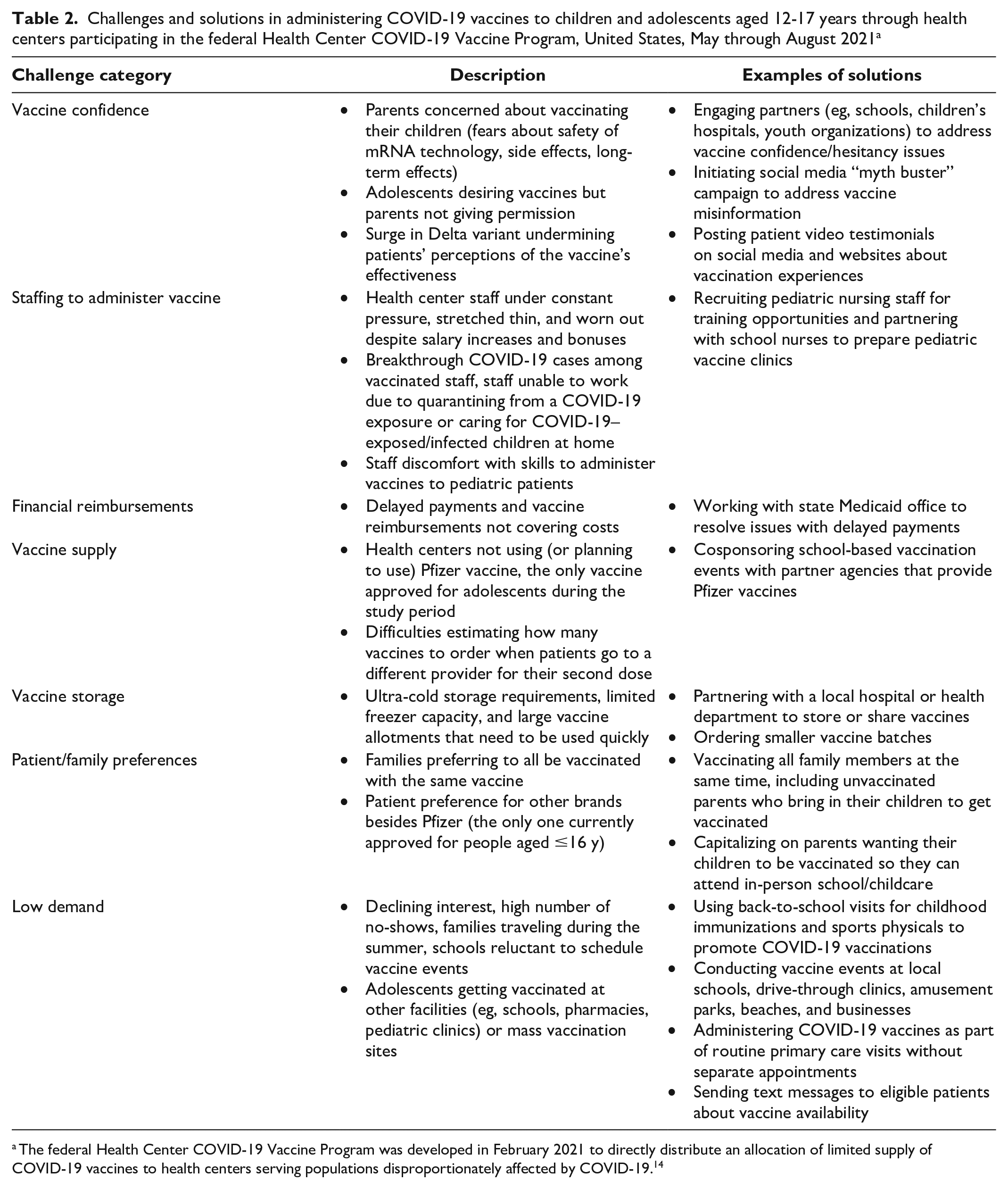
The federal Health Center COVID-19 Vaccine Program was developed in February 2021 to directly distribute an allocation of limited supply of COVID-19 vaccines to health centers serving populations disproportionately affected by COVID-19. 14
Discussion
Nationally, 37% of children and adolescents aged 12-17 years completed the primary COVID-19 vaccine series by the end of August 2021, with the HCCVP providing 2.0% of all first doses and 2.4% of all second doses. Our study identified state-level differences in the role of health centers in administering COVID-19 vaccines to adolescents in the United States, with the estimated proportion of adolescent health center patients receiving their first dose from HCCVP health centers ranging from 0% in Oklahoma and Wyoming to 34.1% in Connecticut. During the study period, health centers reported conducting adolescent vaccination efforts through community partnerships and by setting up vaccine clinics at school-based sites, mobile clinics, pop-up clinics in places of worship, community centers, or other community events. Health centers in 13 states and territories (namely, Arizona, California, Colorado, Connecticut, Delaware, Maine, Massachusetts, Missouri, Nebraska, Nevada, Oregon, Virginia, and Puerto Rico) vaccinated higher proportions of adolescents relative to their share of the adolescent health center patient population, suggesting a relatively greater role for health centers in the COVID-19 public health emergency response for adolescents in these states compared with other states.
Vaccine confidence was the most frequently reported barrier to vaccination against COVID-19. To overcome this barrier and address vaccine hesitancy, health centers strategically delivered COVID-19 messaging through social media and relevant community partners. As reported in previous research in medically underserved communities, health centers in our study reported that parents of adolescents questioned vaccine efficacy and expressed concerns about potential adverse effects, despite evidence of vaccine safety from COVID-19 vaccine studies, strong vaccine efficacy in adolescent hospitalization prevention, and high hospitalization rates among unvaccinated adolescents.22-24 Efforts to overcome vaccine hesitancy and misinformation are complex and especially challenging within a time-limited primary care visit.25-27 To improve vaccine confidence, health centers developed and used culturally appropriate communication strategies to “meet people where they are,” including door-to-door visits by community health workers, outreach visits to businesses throughout the community, town halls to address vaccine safety concerns, and the provision of culturally competent care and communication to community members with limited English proficiency.28-31
The second most frequently reported challenge was staffing for vaccine administration. Prior research described the stress and burnout among health care workers from the extended COVID-19 pandemic response and increased patients’ needs from deferred primary preventive and chronic care in 2020 because of the pandemic.32-35 Health centers also reported staffing shortages due to COVID-19 cases among staff, staff caring for children at home who tested positive for COVID-19, and staff needing to quarantine after COVID-19 exposures. While some health centers described creative partnerships to meet the demand, the COVID-19 pandemic magnified how clinical workforce challenges can negatively affect health care in a public health crisis, especially in geographic areas with health disparities that predate the pandemic.36,37 Substantial efforts will be required for thoughtful reshaping of health care workforce policies and clinical training programs to better prepare for future public health emergencies.38,39
In addition, few health centers reported problems with vaccine supply, which suggests the positive impact of access to HCCVP and plentiful national COVID-19 vaccine supply by late spring 2021. 40 Furthermore, vaccine storage challenges were mitigated by the FDA’s amendment to Pfizer’s EUA allowing for undiluted, thawed Pfizer COVID-19 vaccine vials to be stored in the refrigerator, which alleviated the previous ultra-cold storage requirements. 41
Finally, HCCVP health centers reported challenges in financial reimbursement less frequently than they reported other challenges. The 2021 American Rescue Plan Act provided more than $6 billion in one-time funding for 2 years to support the work of health centers in preventing, mitigating, and responding to the COVID-19 pandemic, including COVID-19 vaccine administration activities. 42 However, that funding expired in federal fiscal year 2023, which will pose substantial financial challenges in sustaining COVID-19 response efforts among health centers moving forward.
Limitations
This study had several limitations. First, the Health Center COVID-19 Survey and addendum are self-reported by health centers’ key personnel supporting the COVID-19 response, which are subject to potential biases and social desirability. Second, the addendum data reflect only vaccine administrations through the HCCVP supply and do not include health centers’ vaccine administrations that use state and jurisdictional health departments’ supply, which may have resulted in underreporting of health center vaccine administration efforts. Third, health center patients may also have received vaccination from the federal pharmacy program or public health mass vaccination sites instead of accessing COVID-19 vaccination services at health centers. In accounting for these limitations jointly, vaccine administration rates are likely underestimates of the true rates in adolescent health center patients. Furthermore, the Health Center COVID-19 Survey did not collect granular data on adolescents; therefore, we were unable to assess disparities (eg, by age group, race, ethnicity, sex) in vaccine administration among adolescents. Finally, individuals received CDC COVID-19 vaccination cards directly from COVID-19 vaccination providers, and CDC does not maintain vaccination records. 43 The lack of an organization that maintains vaccination records across the United States presents policy and technical challenges for collecting data on vaccination surveillance; therefore, comparisons of CDC data with data from the US general population should be interpreted with caution. 44
Conclusion
To our knowledge, our study provides the first assessment of and lessons learned from COVID-19 vaccine administration among adolescents in health centers during the initial months of vaccine implementation. Vaccine confidence and health care workforce were key challenges in vaccine administration. Results suggest that the rapid response and strategic contribution of health centers in several states reached beyond their share of the health center adolescent patient population to support state-level pandemic response. Future research could further explore the process that enabled them to vaccinate more than their share of adolescent patients. Adolescents in medically underserved communities had experienced unique challenges in accessing care prior to the pandemic.45,46 To better prepare for future public health emergencies, health centers need to be engaged as strategic partners in support of public health vaccination efforts, to tailor outreach and communication strategies, to serve as trusted messengers for those who are disproportionately affected by public health emergencies, and to leverage community partnerships to achieve vaccine equity in medically underserved communities.
Supplemental Material
sj-docx-1-phr-10.1177_00333549231218723 – Supplemental material for Lessons Learned From Adolescent COVID-19 Vaccine Administration in Medically Underserved Communities
Supplemental material, sj-docx-1-phr-10.1177_00333549231218723 for Lessons Learned From Adolescent COVID-19 Vaccine Administration in Medically Underserved Communities by Sue C. Lin, Julie Fife Donney and Lydie A. Lebrun-Harris in Public Health Reports
Footnotes
Disclaimer
The views expressed in this article are solely the opinions of the authors and do not necessarily reflect the official policies of the US Department of Health and Human Services or the Health Resources and Services Administration, nor does mention of the department or agency names imply endorsement by the US government.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online. The authors have provided these supplemental materials to give readers additional information about their work. These materials have not been edited or formatted by Public Health Reports’s scientific editors and, thus, may not conform to the guidelines of the AMA Manual of Style, 11th Edition.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.