Abstract
The overlapping epidemics of hepatitis C virus (HCV) and HIV infection stem from underlying behaviors and health disparities among disproportionately affected populations, especially people who inject drugs (PWID). Characterizing the prevalence of HCV–HIV coinfection offers improved data to address these underlying determinants of health. We performed a literature search for articles that describe US populations, were published during 2005-2021, and summarized evidence of the prevalence of HCV infection in recent HIV clusters and outbreaks among PWID. In population- and community-based studies, HCV antibody prevalence among PWID with HIV ranged from 10.7% to 71.4%, depending on the setting and study design. HCV–HIV coinfection ranged from 70% to 94% among 5 larger HIV clusters or outbreaks among PWID during 2014-2021; where characterized, HCV diagnosis preceded HIV detection by a median of 4 to 5 years. Robust modernized surveillance is needed to support and measure the progress of city, state, and national activities for ending the HIV epidemic and eliminating hepatitis C. Developing and leveraging surveillance systems can identify missed opportunities for prevention, evaluate care, and build capacity for outbreak investigation. In addition, improved data on injection drug use are crucial to inform efforts for improved HCV and HIV testing, prevention, and treatment in settings that serve PWID. By providing data in a wholistic, integrated manner, public health surveillance programs can support efforts to overcome inefficiencies of disease-specific silos, accelerate delivery of preventive and clinical services, and address the excess disease burden and health disparities associated with HCV–HIV coinfection.
Keywords
Syndemics can occur when diseases, social determinants of health, and disparities overlap, concentrate in populations, and interact synergistically to produce excess morbidity. 1 The epidemics of viral hepatitis, HIV, and sexually transmitted disease in the United States intersect with long-standing disparities and poor health outcomes among disproportionately affected populations: people who inject drugs (PWID), people experiencing homelessness, sexual and gender minority groups, and people with a history of incarceration. Understanding and addressing these interactions concurrently, rather than through disease-specific approaches, is crucial for successful disease elimination efforts.
In 2018, the prevalence of current injection drug use (IDU) was estimated at 1.5% of the US population, representing nearly 3.7 million people, 2 a 4-fold increase from 2011 estimates. 3 The number of overdose deaths steadily increased during the past 2 decades 4 and rose 46% from 2019 through 2021, when the estimated number surpassed 100 000 for the first time. 5 Rates of acute hepatitis C virus (HCV) infection, for which IDU is the primary reported risk, increased >400% from 2010 to 2020, with 66 700 estimated infections in 2020. 6 The number of new HCV infections has been highest in US regions most affected by the opioid crisis, particularly the central Appalachian region (Kentucky, Tennessee, Virginia, and West Virginia), although similar increases in IDU and HCV infection have been documented in states such as Massachusetts, Wisconsin, and New York.7 -12 Approximately 2.2 million civilian, noninstitutionalized adults had HCV infection in the United States during January 2017–March 2020.13 -16 From 2015 through 2019, at least 7 HIV outbreaks among PWID were identified in the United States.17,18 At the end of 2019, an estimated 1.1 million people in the United States had diagnosed HIV infection, with recent increases in the number of new HIV diagnoses among PWID. IDU (including IDU and male-to-male sexual contact) was reported among 11% of new HIV diagnoses in 2019. 19
Hepatitis C is one of the primary causes of chronic liver disease in the United States, 20 and the American Association for the Study of Liver Diseases/Infectious Diseases Society of America joint guidelines recommend that all people with hepatitis C receive curative treatment. 21 HCV-related liver injury progresses more rapidly among people who are coinfected with HIV than among other populations, with accelerated rates of progression to cirrhosis, end-stage liver disease, hepatocellular carcinoma,22 -27 and death. 28
In addition to being at increased risk for HIV and hepatitis C, PWID are at increased risk for serious invasive bacterial and fungal infections, 29 hepatitis A,30 -32 hepatitis B,33,34 and other sexually transmitted infections, such as syphilis,35 -37 because of unsafe injection behaviors, condomless sexual practices, and limited access to sanitation, which is sometimes associated with homelessness and unstable housing.31,32,38 A substantial unmet need exists for integrated treatment of IDU-associated infectious diseases and substance use disorders.39,40 The prevalence of HCV–HIV coinfection among PWID highlights the urgent need for improved data to inform interventions, including comprehensive syringe service programs (SSPs) and substance use disorder treatment, that address the underlying behaviors and health disparities.
The US Viral Hepatitis National Strategic Plan: A Roadmap to Elimination set forth goals for the elimination of viral hepatitis as a public health threat, including prevention of new infections and improvement of viral hepatitis–related health outcomes and health equity. 41 In addition to promoting testing and linkage to hepatitis C care and treatment, current strategies for hepatitis C elimination include substance use disorder treatment, improving hepatitis A and B vaccination coverage, and providing access to evidence-based care—including medication for opioid use disorder—in correctional facilities and SSPs.40,42,43 Underpinning the success of this roadmap is the need to improve viral hepatitis surveillance and data use to bolster data collection and reporting, enhance sharing and use of clinical viral hepatitis data, and conduct routine analysis of data for public health action. Many of these strategies are critical to the Ending the HIV Epidemic initiative, which also consists of goals for improving surveillance, reducing disparities, and improving preventive services for disproportionately affected populations, including PWID.44,45
Here, we describe the overlapping epidemics of HCV and HIV infection in the United States with a focus on PWID, who are particularly vulnerable to infection. We reviewed data to summarize estimates of HCV–HIV coinfection prevalence among PWID and discuss gaps in data needed to support efforts to end the HCV and HIV epidemics in this population. 41
Methods
We identified articles on HCV–HIV coinfection by conducting the following PubMed searches on January 22, 2022, restricted to articles written after January 1, 2005, in English that involved humans: “HIV HCV coinfection,” “hepatitis C prevalence among people living with HIV,” “hepatitis C, HIV, match, coinfection,” and “HIV, hepatitis C, registry.” After reviewing 3213 abstracts, we excluded articles that described non–US populations or did not report on coinfection prevalence, resulting in 103 abstracts for further consideration. Review of these articles resulted in additional exclusion of articles that did not include primary data on coinfection prevalence or were health care setting–based studies for which the denominator could not be clearly ascertained. We abstracted data from the 36 references that met criteria for inclusion (eTable in Supplemental Material).46 -83
We selected additional studies that reported coinfection prevalence among PWID that were known to the authors but were not identified in the literature search.84 -88 These studies included articles that summarized the prevalence of HCV infection in HIV clusters and outbreaks among PWID reported to CDC through 2021 or articles that were summarized in a recently published CDC literature review for 2000-2020,17,89 -99 where the denominator for HCV infection prevalence was clearly described.
Results
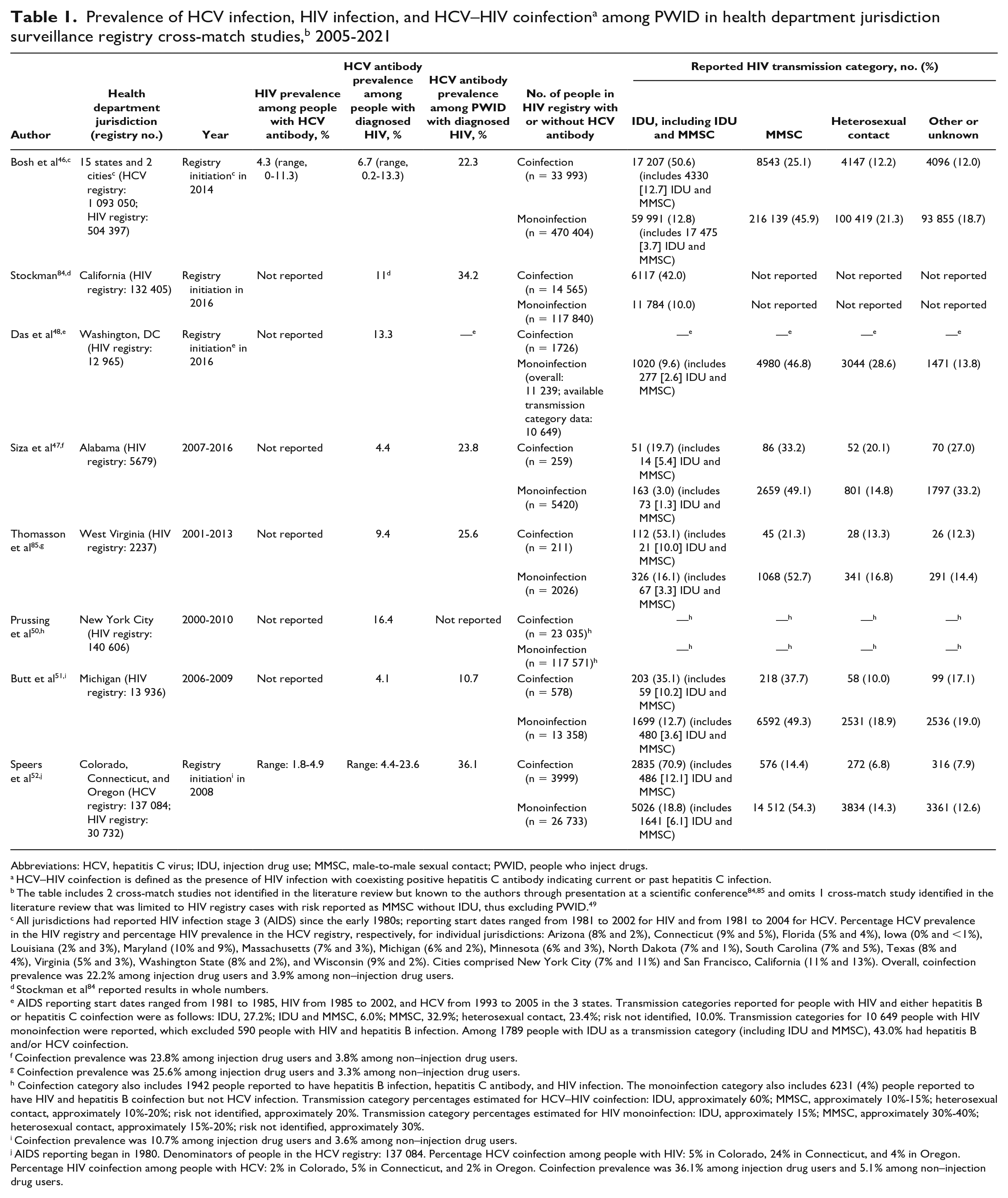
Health Department Jurisdiction Surveillance Registry Cross-Match Studies
We identified 9 population-based health jurisdiction surveillance registry cross-match studies that met the criteria for inclusion (Table 1),46 -52,84,85 but we omitted 1 study that was limited to HIV registry cases that excluded PWID. 49 The 8 studies described regional rates of HCV antibody prevalence among PWID with HIV (10.7%-36.1%) that were generally higher than the prevalence rates among all people with HIV (4.4%-23.6%). Rates of HCV–HIV coinfection among people with HCV antibodies ranged from 0% to 11.3%. Among all HIV registry cases, the prevalence of coinfection (defined as the presence of HCV antibody and HIV infection) was higher among those reporting IDU (10.7%-36.1%) than among those not reporting IDU (3.3%-5.1%).46,47,51,52,85 Similarly, the prevalence of reported IDU among people with HCV–HIV coinfection (19.1%-70.8%) was higher than among those with HIV monoinfection (3.0%-18.8%).
Abbreviations: HCV, hepatitis C virus; IDU, injection drug use; MMSC, male-to-male sexual contact; PWID, people who inject drugs.
HCV–HIV coinfection is defined as the presence of HIV infection with coexisting positive hepatitis C antibody indicating current or past hepatitis C infection.
The table includes 2 cross-match studies not identified in the literature review but known to the authors through presentation at a scientific conference84,85 and omits 1 cross-match study identified in the literature review that was limited to HIV registry cases with risk reported as MMSC without IDU, thus excluding PWID. 49
All jurisdictions had reported HIV infection stage 3 (AIDS) since the early 1980s; reporting start dates ranged from 1981 to 2002 for HIV and from 1981 to 2004 for HCV. Percentage HCV prevalence in the HIV registry and percentage HIV prevalence in the HCV registry, respectively, for individual jurisdictions: Arizona (8% and 2%), Connecticut (9% and 5%), Florida (5% and 4%), Iowa (0% and <1%), Louisiana (2% and 3%), Maryland (10% and 9%), Massachusetts (7% and 3%), Michigan (6% and 2%), Minnesota (6% and 3%), North Dakota (7% and 1%), South Carolina (7% and 5%), Texas (8% and 4%), Virginia (5% and 3%), Washington State (8% and 2%), and Wisconsin (9% and 2%). Cities comprised New York City (7% and 11%) and San Francisco, California (11% and 13%). Overall, coinfection prevalence was 22.2% among injection drug users and 3.9% among non–injection drug users.
Stockman et al 84 reported results in whole numbers.
AIDS reporting start dates ranged from 1981 to 1985, HIV from 1985 to 2002, and HCV from 1993 to 2005 in the 3 states. Transmission categories reported for people with HIV and either hepatitis B or hepatitis C coinfection were as follows: IDU, 27.2%; IDU and MMSC, 6.0%; MMSC, 32.9%; heterosexual contact, 23.4%; risk not identified, 10.0%. Transmission categories for 10 649 people with HIV monoinfection were reported, which excluded 590 people with HIV and hepatitis B infection. Among 1789 people with IDU as a transmission category (including IDU and MMSC), 43.0% had hepatitis B and/or HCV coinfection.
Coinfection prevalence was 23.8% among injection drug users and 3.8% among non–injection drug users.
Coinfection prevalence was 25.6% among injection drug users and 3.3% among non–injection drug users.
Coinfection category also includes 1942 people reported to have hepatitis B infection, hepatitis C antibody, and HIV infection. The monoinfection category also includes 6231 (4%) people reported to have HIV and hepatitis B coinfection but not HCV infection. Transmission category percentages estimated for HCV–HIV coinfection: IDU, approximately 60%; MMSC, approximately 10%-15%; heterosexual contact, approximately 10%-20%; risk not identified, approximately 20%. Transmission category percentages estimated for HIV monoinfection: IDU, approximately 15%; MMSC, approximately 30%-40%; heterosexual contact, approximately 15%-20%; risk not identified, approximately 30%.
Coinfection prevalence was 10.7% among injection drug users and 3.6% among non–injection drug users.
AIDS reporting began in 1980. Denominators of people in the HCV registry: 137 084. Percentage HCV coinfection among people with HIV: 5% in Colorado, 24% in Connecticut, and 4% in Oregon. Percentage HIV coinfection among people with HCV: 2% in Colorado, 5% in Connecticut, and 2% in Oregon. Coinfection prevalence was 36.1% among injection drug users and 5.1% among non–injection drug users.
HCV and HIV Coinfection in US Studies Among PWID
We identified 5 studies of PWID recruited for HIV and HCV testing (Table 2),56,57,86 -88 4 of which used respondent-driven sampling, in which a small number of initial participants (“seeds”) were selected to complete the survey and recruit their peers to participate, which may have reached PWID not routinely engaged in health care. Across all 5 studies, the HCV antibody prevalence among PWID living with HIV ranged from 28.6% to 71.4%, and the HIV prevalence among HCV antibody–positive PWID ranged from 2.3% to 5.7% (9% in 1 study reporting HIV prevalence among HCV RNA–positive people). Among PWID overall, the prevalence of HIV infection ranged from 2.3% to 9%, HCV antibody prevalence from 27.8% to 67.7%, and HCV (antibody)–HIV coinfection from 1.2% to 2.2% (4% in 1 study that reported HIV and HCV [RNA] coinfection).
Prevalence of HCV infection, HIV infection, and HCV–HIV coinfectiona among PWID in US studies
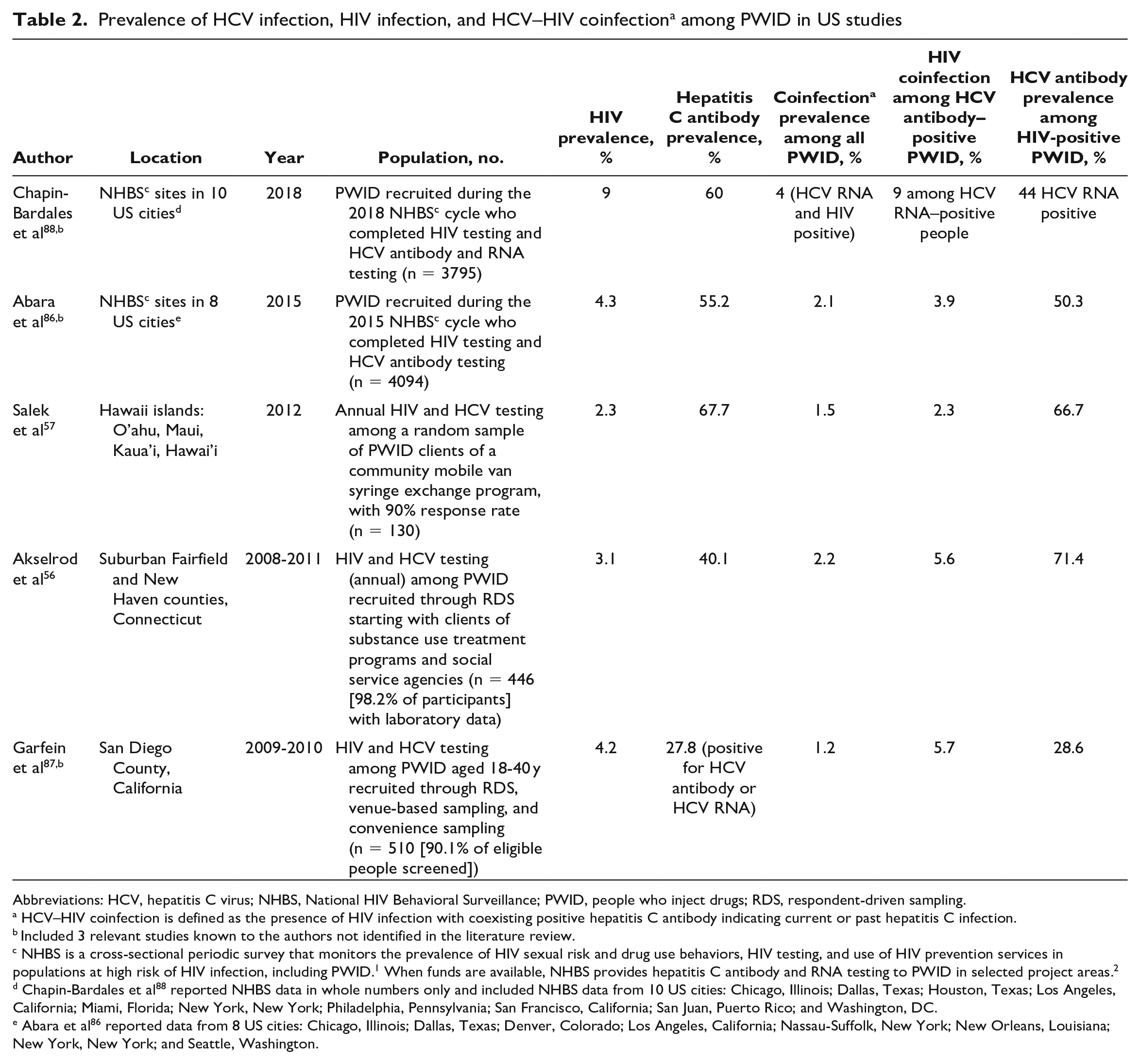
Abbreviations: HCV, hepatitis C virus; NHBS, National HIV Behavioral Surveillance; PWID, people who inject drugs; RDS, respondent-driven sampling.
HCV–HIV coinfection is defined as the presence of HIV infection with coexisting positive hepatitis C antibody indicating current or past hepatitis C infection.
Included 3 relevant studies known to the authors not identified in the literature review.
NHBS is a cross-sectional periodic survey that monitors the prevalence of HIV sexual risk and drug use behaviors, HIV testing, and use of HIV prevention services in populations at high risk of HIV infection, including PWID. 1 When funds are available, NHBS provides hepatitis C antibody and RNA testing to PWID in selected project areas. 2
Chapin-Bardales et al 88 reported NHBS data in whole numbers only and included NHBS data from 10 US cities: Chicago, Illinois; Dallas, Texas; Houston, Texas; Los Angeles, California; Miami, Florida; New York, New York; Philadelphia, Pennsylvania; San Francisco, California; San Juan, Puerto Rico; and Washington, DC.
Abara et al 86 reported data from 8 US cities: Chicago, Illinois; Dallas, Texas; Denver, Colorado; Los Angeles, California; Nassau-Suffolk, New York; New Orleans, Louisiana; New York, New York; and Seattle, Washington.
HCV Infection in Recent HIV Clusters and Outbreaks Among PWID
From 2014 to 2021, HCV coinfection was observed in 69.8%-93.8% of newly diagnosed HIV cases among the 5 largest HIV outbreaks among PWID that incorporated HCV antibody or RNA testing (Table 3).89 -92,95,98,99 In contrast, smaller HIV cluster and outbreak investigations93,94,96,97 reported more varied HCV–HIV coinfection rates (28.6%-64.3%). In 2 large outbreaks for which the temporal sequence of infection was reported, diagnosis of HCV preceded diagnosis of HIV by a median of 4 to 5 years.89,95 In a large HIV outbreak among PWID in Scott County, Indiana, HCV–HIV coinfection was identified in 92% of new HIV cases. The largest HCV molecular cluster (n = 130) in this outbreak consisted of 4 cases of hepatitis C previously diagnosed in 2010, 5 years prior to recognition of the HIV outbreak. 99
US clusters and outbreaks of HIV among PWID with prevalence of HCV coinfection a reported, 2014-2021
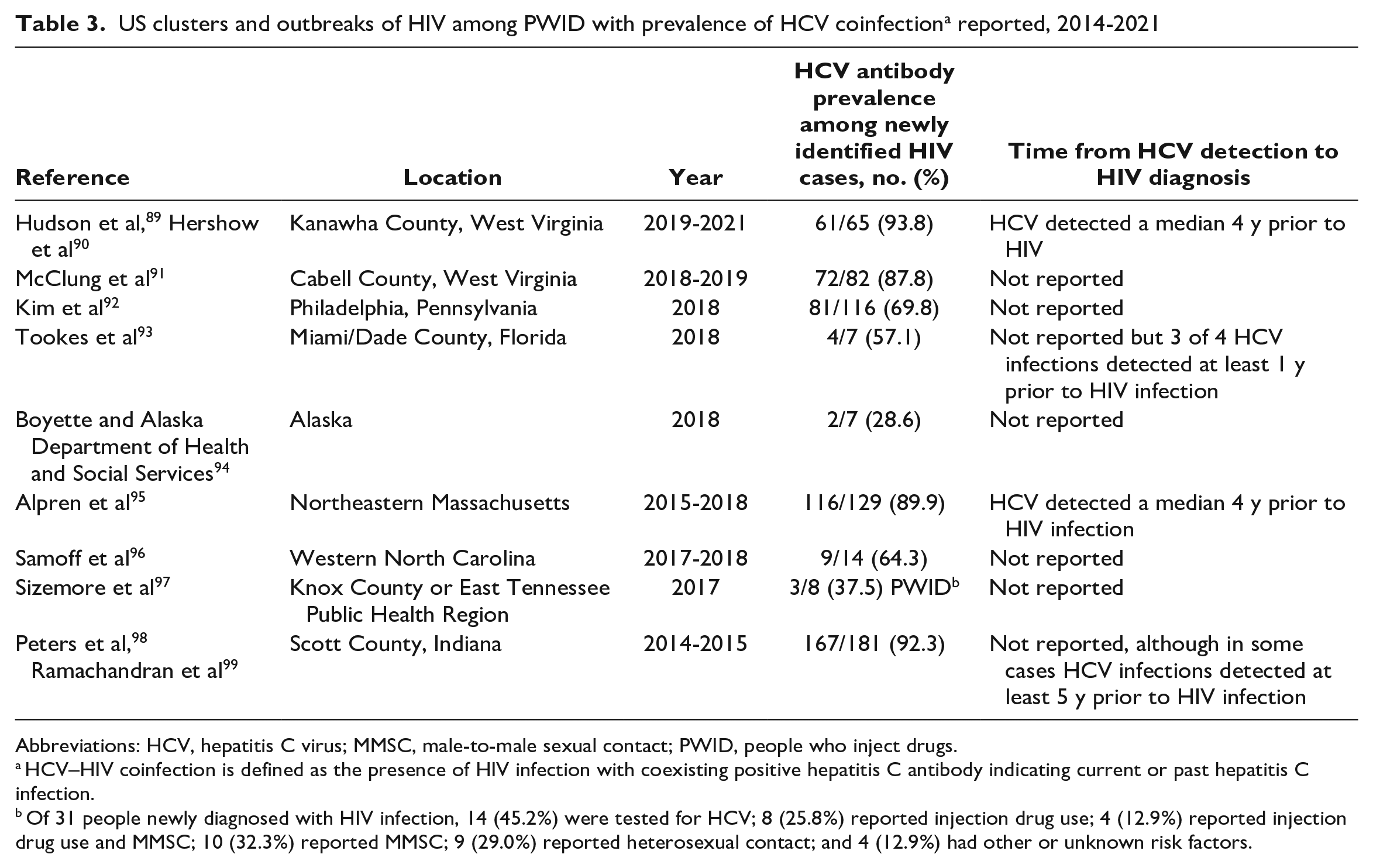
Abbreviations: HCV, hepatitis C virus; MMSC, male-to-male sexual contact; PWID, people who inject drugs.
HCV–HIV coinfection is defined as the presence of HIV infection with coexisting positive hepatitis C antibody indicating current or past hepatitis C infection.
Of 31 people newly diagnosed with HIV infection, 14 (45.2%) were tested for HCV; 8 (25.8%) reported injection drug use; 4 (12.9%) reported injection drug use and MMSC; 10 (32.3%) reported MMSC; 9 (29.0%) reported heterosexual contact; and 4 (12.9%) had other or unknown risk factors.
A comprehensive literature review of HCV–HIV coinfection studies not focused on PWID is summarized (eTable in Supplemental Material).
Discussion
Hepatitis C and HIV interact in predictable patterns: the prevalence of HCV infection is higher than the prevalence of HIV infection,56,57,86 -88 and the prevalence of IDU is higher among people coinfected with HIV and HCV than among people infected with HIV only.46,47,51,52,85 In our review, HCV antibody prevalence among PWID with HIV ranged from 10.7% to 71.4% in population- and community-based studies (Tables 1 and 2) and from 69.8% to 93.8% among the 5 larger HIV outbreaks or clusters (Table 3). Where the question has been evaluated, HCV infection preceded HIV infection in outbreaks among PWID by a median of 4 to 5 years. This relationship is supported by an ecologic association between the population prevalence of HCV infection and HIV in a review of US and international population-level studies. 100 The positive association between prevalence of hepatitis C and HIV suggests that HCV infection may be an important marker of IDU and other behavioral or social determinants of health that could increase the risk for future HIV infection among PWID.
While these studies demonstrate a substantial prevalence of infection among PWID, testing is likely performed for a low percentage of people in this high-risk group. For example, in an analysis of nationwide health insurance claims data during 2010-2017, among people identified as PWID through a clinical diagnosis algorithm, approximately 90% missed opportunities for HIV or HCV testing. 101 Many previously missed opportunities for HIV testing among PWID in the year prior to diagnosis were identified in one of the larger recent HIV outbreaks. 102 Because PWID may be difficult to reach in routine surveys 10 as well as for clinic-based prevention services, 18 the prevalence of HCV–HIV coinfection may be underestimated.
The relationship among HCV infection, HIV infection, and IDU has several implications for disease prevention and control. First, prevention pathways for HCV and HIV are similar. The combination of sufficient sterile injection paraphernalia for all injections and medication for opioid use disorder reduces hepatitis C transmission by 74%, 103 and these measures are critical for preventing HIV transmission. 104 Comprehensive SSPs can address not only the need for sterile injection equipment and safe disposal, but also provision of or referral to social, mental health, and medical services including substance use disorder treatment, testing and treatment for infectious diseases, and nalaxone distribution. 105 While SSPs are a critical link for testing, prevention, and treatment for HIV and HCV infection, they can also help PWID address the broader range of syndemic social, mental health, and medical challenges. National plans for ending the HIV epidemic and eliminating viral hepatitis as a public health problem share key strategies and populations, specifically targeting improvement of the distribution and quality of SSPs.41,44,45
Second, active surveillance is important for HCV and HIV infection, as well as infectious diseases such as hepatitis A and B.106,107 Improvements in HCV surveillance systems could allow for earlier identification of populations with a high incidence or prevalence of hepatitis C; that information could be used to mitigate or prevent subsequent infections associated with IDU, including HIV. 17 Identifying and investigating outbreaks among PWID leads to a better understanding of patterns of coinfection and appropriate prevention and control measures. Robust data are needed to estimate disease prevalence and measure progress toward goals of ending the HIV epidemic and eliminating viral hepatitis as a public health threat.41,44 In addition, understanding vulnerabilities at the population level is critical to assessing the potential impact of HCV–HIV coinfection on communities. Many factors, such as poverty, employment, race, and physician capacity to offer buprenorphine treatment, have been used to predict county-level vulnerability to the rapid spread of HCV and HIV. 108 Better integration of surveillance data with data on social determinants of health, including composite indexes such as the Social Vulnerability Index,109,110 can provide important context and guide disease elimination planning. Readily accessible data-driven estimates are powerful advocacy tools that can motivate the allocation of resources for hepatitis C and HIV programs; however, further evaluation is needed to understand the impact of these tools on reducing the prevalence of disease. 111
Lastly, lessons learned from HCV–HIV outbreaks among PWID can inform prevention, detection, and responses to future outbreaks. Lack of access to evidence-based care for PWID, including SSPs, medication for opioid use disorder, and HIV preexposure prophylaxis, has been documented during investigations of outbreaks of HIV among PWID; contributing social factors have included homelessness, exchanging sex for drugs, incarceration, and discrimination and stigma.17,18,89-92,95,98,99,112 In response to HIV outbreaks among PWID, state and local health departments have expanded testing for HIV90 -93,95,96,98,112 and hepatitis C90,93,96,98; expanded access to HIV preexposure prophylaxis90 -92; offered training of service providers on HIV and stigma reduction 90 ; enhanced linkage to HIV90 -93,95,96,98 and substance use disorder90,91,95,98 treatment services; expanded SSP access91 -93,95,98,112; and hired additional partner services staff members (eg, disease intervention specialists) to reach people who may be medically underserved, experiencing homelessness, or using drugs.90,91,95,98 Given that HCV infection might frequently precede HIV infection,89,95,99 implementation or intensification of these evidence-based prevention activities should ideally take place at the first sign of increasing HCV infections.
This literature review was subject to several limitations that highlight data gaps to be addressed in future programs and studies. First, relatively few studies included data on the prevalence of current HCV–HIV coinfection among PWID. When such data were provided, they were primarily based on measures of the prevalence of HCV antibody (an indicator of past or current HCV infection), although some studies also described the prevalence of HCV RNA (an indicator of current HCV infection). However, among untreated HCV antibody–positive PWID, the prevalence of current infection is typically ≥70%.113 -115 Second, our search approach was not intended to be a systematic review of the literature; thus, some articles on the prevalence of HCV–HIV coinfection prevalence may not have been identified, as evidenced by the inclusion of some studies known to the authors that did not appear in the search results.
Data Needs and Opportunities for the Future
The accelerated progression of HCV-related liver disease among people with HIV and the substantial coinfection prevalence across populations have prompted American Association for the Study of Liver Diseases/Infectious Diseases Society of America guidance 21 and the World Health Organization 116 to urge diagnosis and treatment of HCV infection among all people living with HIV. Successful HCV elimination efforts among people with HIV have been initiated in other industrialized nations, such as Australia, Scotland, and the Netherlands.117 -124 To support this goal in the United States, integrated data sharing between HIV and HCV surveillance and clinical systems is necessary to better understand the prevalence of HCV–HIV coinfection and develop HCV and HIV care cascades 125 —a sequence of steps that follow progression from testing and treatment to viral clearance for hepatitis C or viral suppression for HIV. 126 A need exists to develop methods of simple and secure data sharing among HIV and HCV surveillance programs in health departments. Cascades are important data tools because they can identify missed opportunities for HCV and HIV prevention and linkage to care and inform population-wide interventions for those in need.
To this end, the Health Resources and Services Administration is supporting a demonstration project to link people with HCV–HIV coinfection in the Ryan White HIV/AIDS Program to HCV care by leveraging existing public health surveillance with clinical data systems. Ryan White HIV/AIDS programs and federally qualified health centers are well positioned to facilitate HCV testing and treatment given the strong health advocacy and patient–provider relationships fostered in these HIV care environments.117,127,128 Existing HIV care strategies, such as using retention specialists and adherence support, monitoring viral suppression, counseling on harm reduction, and addressing stigma and other barriers to care, can also be leveraged to provide comprehensive HCV care in a co-localized setting for both infections.127,128
To address the health disparities related to IDU, better data are needed on the prevalence of IDU, risk behaviors, and access to evidence-based services for PWID. CDC is supporting multiple programs to describe the infectious consequences of IDU and improve access to SSPs.129 -131 The National HIV Behavioral Surveillance system is a comprehensive system for biobehavioral surveillance in metropolitan areas among populations with a high prevalence of HIV, including PWID. 132 Data on risk factors, testing, and receipt of prevention services are collected. All participants are offered HIV testing and, in some cases, HCV testing. 133 To expand surveillance of PWID, CDC is piloting the Injection Drug Use Surveillance Project, which recruits PWID from SSPs in urban, suburban, and rural settings and assesses injection behavior, use of harm reduction services, and overdose prevention. HIV and HCV testing are offered, as well as linkage to care, as needed.134,135 Lastly, CDC has implemented a national SSP survey that measures service delivery capacity, service gaps, and operational challenges faced.136,137 Together, these data will provide a comprehensive picture of the public health burden of IDU, infectious complications, and access to evidence-based preventive services.
The Viral Hepatitis National Strategic Plan prioritizes integrating the efforts of hepatitis, HIV, and sexually transmitted infections surveillance systems to better address data quality and interrelated risk factors. 41 Efforts to further integrate activities include developing interoperable surveillance systems by standardizing electronic case reporting and data collection strategies and removing barriers to sharing data between surveillance programs. The efficient exchange of surveillance data will reduce data entry errors and expedite the transfer of data to public health policy makers and other partners. These efforts are aligned with the goals of CDC’s Data Modernization Initiative, a multiyear national effort to modernize core data and surveillance infrastructure across the federal and state public health landscape. 138 A long-term outcome of the initiative is to develop a state-of-the-art workforce in the areas of data science, health information technology, and cybersecurity to deploy approaches that move the public health community from “siloed and brittle public health data systems to connected, resilient, adaptable, and sustainable ‘response-ready’ systems capable of meeting today’s and tomorrow’s health challenges.” 138
Public Health Implications
National plans for ending the HCV and HIV epidemics among PWID share important strategies for prevention, including test-and-treat, comprehensive SSPs, and treatment of substance use disorder.41,44,45 Robust integrated surveillance of HCV–HIV disease incidence and prevalence, IDU, and social determinants of health is needed to inform public health programs at city, state, and national levels. By providing data on these overlapping epidemics in a wholistic, integrated manner, public health surveillance programs can overcome the inefficiencies of disease-specific silos, accelerate the delivery of preventive and clinical services, and address the health disparities and social drivers associated with HCV–HIV coinfection.
Supplemental Material
sj-xlsx-1-phr-10.1177_00333549231181348 – Supplemental material for Hepatitis C Virus–HIV Coinfection in the United States Among People Who Inject Drugs: Data Needed for Ending Dual Epidemics
Supplemental material, sj-xlsx-1-phr-10.1177_00333549231181348 for Hepatitis C Virus–HIV Coinfection in the United States Among People Who Inject Drugs: Data Needed for Ending Dual Epidemics by Anne C. Moorman, Danae Bixler, Eyasu H. Teshale, Megan Hofmeister, Henry Roberts, Johanna Chapin-Bardales and Neil Gupta in Public Health Reports
Footnotes
Acknowledgements
The authors acknowledge contributions from Nathan Furukawa, MD, Division of Viral Hepatitis, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, Centers for Disease Control and Prevention.
Disclaimer
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online at ![]() . The authors have provided these supplemental materials to give readers additional information about their work. These materials have not been edited or formatted by Public Health Reports’s scientific editors and, thus, may not conform to the guidelines of the AMA Manual of Style, 11th edition.
. The authors have provided these supplemental materials to give readers additional information about their work. These materials have not been edited or formatted by Public Health Reports’s scientific editors and, thus, may not conform to the guidelines of the AMA Manual of Style, 11th edition.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.