Abstract
Objectives:
Public health laboratories (PHLs) are essential components of US Public Health Service operations. The health information technology that supports PHLs is central to effective and efficient laboratory operations and overall public health response to infectious disease management. This analysis presents key information on how the Nebraska Public Health Laboratory (NPHL) information technology system evolved to meet the demands of the COVID-19 pandemic.
Materials and Methods:
COVID-19 presented numerous, unforeseen information technology system challenges. The most notable challenges requiring changes to NPHL software systems and capability were improving efficiency of the laboratory operation due to high-volume testing, responding daily to demands for timely data for analysis by partner systems, interfacing with multiple testing (equipment) platforms, and supporting community-based specimen collection programs.
Results:
Improvements to the NPHL information technology system enabled NPHL to perform >121 000 SARS-CoV-2 polymerase chain reaction tests from March 2020 through January 2022 at a sustainable rate of 2000 SARS-CoV-2 tests per day, with no increase in laboratory staffing. Electronic reporting of 62 000 rapid antigen tests eliminated paper reporting and extended testing services throughout the state. Collection of COVID-19 symptom data before specimen collection enabled NPHL to make data-driven decisions to perform pool testing and conserve testing kits when supplies were low.
Practice Implications:
NPHL information technology applications proved essential for managing health care provider workload, prioritizing the use of scarce testing supplies, and managing Nebraska’s overall pandemic response. The NPHL experience provides useful examples of a highly capable information technology system and suggests areas for additional attention in the PHL environment, including a focus on end users, collaboration with various partners, and investment in information technology.
Public health laboratories (PHLs) have been challenged with multiple emerging pathogens during the past 15 years. The evolution and rapid spread of SARS-CoV-2 are the most recent example of the immense impact of emerging diseases on PHLs. Laboratory testing of entire communities for COVID-19 required mobilization of large quantities of resources, specimen collection materials, and personnel. The role of the PHL in testing services extended beyond routine assays to extensive support of epidemiologic investigations and high-volume COVID-19 diagnostic testing on multiple platforms.
New and unforeseen needs that emerged for PHLs included mobile, drive-through screening centers; participation by National Guard or third-party contractors; online test scheduling; electronic consent to treat and report; management of point-of-care testing by public health departments; large-scale specimen management; laboratory automation; and heightened demands for data analysis. Laboratory information systems (LISs) and PHL information technology systems were not designed to address these challenges, and PHLs needed to rapidly modify and adjust their operations to meet the needs of their epidemiology partners. This situation resulted in the development of innovative informatics applications within the Nebraska Public Health Laboratory (NPHL). We describe the information technology gaps faced by NPHL during the COVID-19 pandemic and how technology innovations were used to meet the laboratory’s needs.
Background
NPHL is administered by the University of Nebraska Medical Center (UNMC) and the Department of Pathology and Microbiology. It provides testing and analytic services under contract to the Nebraska Department of Health and Human Services (DHHS). The laboratory is located on the UNMC campus and is adjacent to Nebraska Medicine, the academic hospital for the university. In addition to the UNMC and Nebraska Medicine infrastructure, NPHL developed and operates a web-based electronic test, order, and result system named NUlirt (Nebraska University Laboratory Information Reporting Tool) based on the Centers for Disease Control and Prevention (CDC) National Electronic Disease Surveillance System (NEDSS) and recommendations of the Association of Public Health Laboratories electronic laboratory reporting.1-5
Since 2017, NUlirt has been central to the daily operations of NPHL. The system serves as a web portal to access laboratory services. Health care providers, local health departments (LHDs), and their designees have access to NUlirt to order NPHL tests, collect epidemiologic information, and receive test results. NUlirt supports full, bidirectional electronic interfaces with the Nebraska Medicine LIS for purposes of test, order, and result management (Box). Bidirectional interfaces with NPHL clients’ electronic health record (EHR) systems are routinely provided, and electronic results reporting to the Nebraska DHHS disease surveillance system, NEDSS, and CDC are managed by NUlirt. An overview of the NUlirt and NPHL information technology system architecture at the beginning of the COVID-19 pandemic is shown in Figure 1.
Glossary of key information technology terms used in the public health laboratory

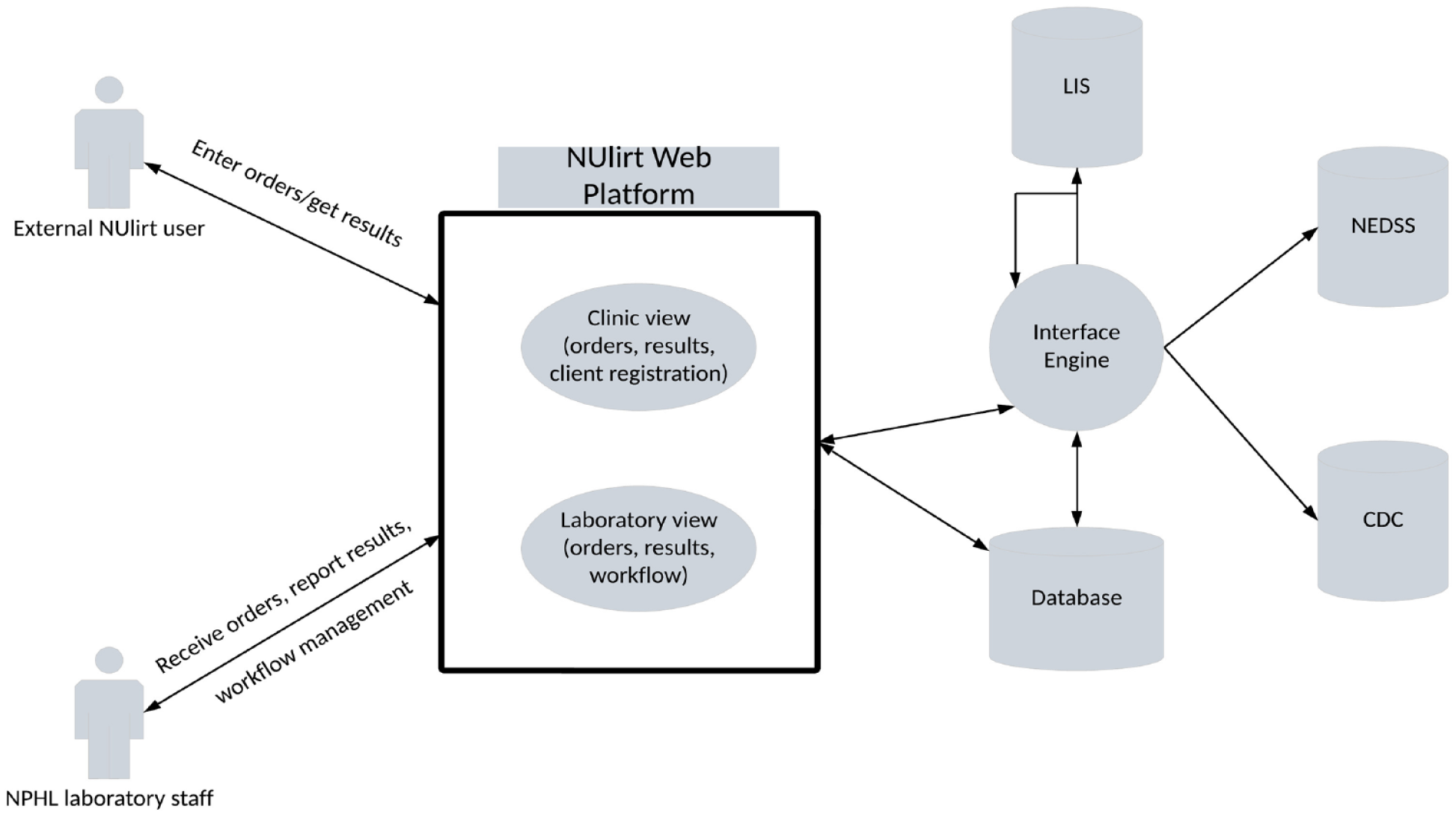
Overview of the Nebraska University Laboratory Information Reporting Tool (NUlirt) at the beginning of the COVID-19 pandemic, Nebraska Public Health Laboratory (NPHL), March 2020. Functionality entailed web-based laboratory test and test supply ordering, collection of epidemiologic data, results reporting, laboratory specimen management, Health Level Seven interfacing, and Nebraska Department of Health and Human Services and Centers for Disease Control and Prevention (CDC) reporting. Abbreviations: LIS, laboratory information system; NEDSS, National Electronic Disease Surveillance System.
Health care providers at LHDs, federally qualified health centers (FQHCs), or other care settings access the system through a password-protected web portal or from their EHR system. High-volume NPHL client sites are provided label printers to allow for direct printing of specimen labels from the NUlirt application. After specimens are collected, they are sent to NPHL for testing. Upon physical receipt at NPHL, specimens are electronically received through NUlirt, and laboratory orders are transmitted to the Sunquest LIS by Health Level Seven (HL7) messaging. 6 After analytic testing is performed, results are transmitted by HL7 message to NUlirt. NUlirt transmits these results by HL7 message to NEDSS, CDC, and/or interfaced EHR systems. Noninterfaced clients—those without the capacity to send or receive laboratory test orders or results electronically—can review test results via web portal function and/or by faxed report if the local health care provider has limited information technology support. This health information technology functionality and workflow define the base information technology capabilities that NPHL had developed after the H5N1 and Ebola pandemics and before onset of the COVID-19 pandemic.
On February 17, 2020, 13 passengers from the Diamond Princess Cruise Ship exposed to SARS-CoV-2 arrived in Nebraska for quarantine and potential treatment at the National Quarantine Unit. 7 During the next several weeks, community-acquired COVID-19 infections became prevalent, with outbreaks at meat-processing facilities and in Nebraska populations with a high risk for SARS-CoV-2 exposure and development of COVID-19. Laboratory services in support of efforts to screen for and contain COVID-19 through CDC Emergency Use Authorization test kits were launched at NPHL. We describe the challenges and scenarios faced by NPHL and the response by NPHL information technology during the pandemic.
Materials and Methods
Early Pandemic and Initial Public Health Response
Localized COVID-19 outbreaks began in communities proximal to meat-processing facilities along the Interstate 80 corridor in the central and western portions of Nebraska. Before the pandemic, NUlirt was developed to provide standard patient registration. Consent to treat was a manual, in-person process. During the pandemic, Nebraska DHHS requested that NPHL support epidemiologic investigations and provide diagnostic testing for multiple clients, including National Guard–operated mobile collection centers and county/city–managed pop-up collection facilities in community centers, tents, and drive-through sites. These efforts focused on medically underserved populations, central urban sites, and meat-processing facilities. These sites allowed community members to have specimens collected for COVID-19 testing at no cost. Core NUlirt functionality readily accommodated these remote collection sites by using mobile hotspots to obtain internet access. The UNMC Institutional Review Board determined that this project did not constitute human subjects research as defined by 45 CFR 46.102. Therefore, it did not require institutional review board review.
Expansion of Testing Locations, Testing Administrators, and Legal Considerations
As the pandemic persisted and outbreaks became more widespread, the number of mobile and new physical specimen collection site locations increased. Operational staffing and management for collections were extended to include non–US Public Health Service organizations. Client support services offered by UNMC’s outreach laboratory, Regional Pathology Services, helped support specimen collection, labeling, shipping, and/or receiving at NPHL. Specimen collection by Regional Pathology Services for COVID-19 testing was offered to other essential organizations to include the Omaha police and fire departments and US Strategic Command (Offutt Airforce Base) at their respective locations. With the expansion of screening sites, additional staffing provided by commercial organizations, and site logistics of specimen collection, new challenges for the NPHL information technology systems were encountered.
To avoid congestion at the drive-through site and to maintain social distancing, NPHL informatics staff developed the capacity for electronic, web-based patient registration within the NUlirt application. Registration was linked with patient self-scheduling to reduce congestion at the specimen collection site. The ability to capture electronic signatures conformant to requirements in Nebraska was developed to capture legal electronic signatures for patient consent for testing, acknowledgment of risks, and consent for communication of test results. Patients were given web links to NUlirt registration and scheduling sites to allow them to access the link on any internet-capable device prior to arrival at the collection site. After check-in, specimen labels were printed from NUlirt that contained all the necessary patient identifiers and collection information as directed by the Clinical Laboratory Improvement Amendments 8 regulatory standard.
During the pandemic, the medical provider who ordered NPHL COVID-19 testing was the state epidemiologist or local public health official. The increased number of tests with subsequent contact tracing of cases placed local health officials into roles of frontline health care providers. These local health officials were quickly overwhelmed with reporting of test results directly to patients. To address this issue, NPHL used UNMC’s secure email services for electronic communication of COVID-19 test results directly to patients.
Integration With EHR Systems and FQHCs
As COVID-19 cases increased during summer 2020, particularly in medically underserved communities, drive-through clinics were established at FQHCs. Drive-through specimen collection sites in the clinic’s catchment areas were open to the entire community regardless of patient status in the FQHC’s census. To ensure proper patient follow-up in the event of a positive test result, results obtained from drive-through sites were transmitted by HL7 to the clinic EHR. Results for patients registered at the clinic were moved into those patients’ EHRs, and positive test results of patients not registered at the clinics were flagged and communicated to the LHD for follow-up.
School-Based Screening for Students and Staff: Saliva Polymerase Chain Reaction Testing
In the latter months of 2020 and into 2021, requests were received by local school districts to provide support for weekly saliva polymerase chain reaction (PCR) testing of faculty and students at public schools. To support this effort, a series of consents to test, treat, and communicate test results was developed with test accessioning and reporting services provided by NUlirt. 9 Consent forms written in English and Spanish were provided to school staff as well as for parents and guardians of students. The results of testing were interfaced to the EHR systems used by the FQHCs whose catchment areas were included in the school district.
Rapid Antigen Testing, LHDs, and Athletic Programs
While Emergency Use Authorization–approved PCR assays remained the gold standard for SARS-CoV-2 detection, rapid antigen tests were introduced in the state to supplement PCR testing for screening purposes in urban and rural areas with limited access to it. Antigen testing became a common testing platform for the long-term care facilities where Nebraska DHHS provided rapid test kits to LHDs and participating community clinics. NUlirt was configured to allow sites to order a rapid antigen test, document the test kit used, perform the test under Clinical Laboratory Improvement Amendments–waived certifications, and manually record the test result. Antigen test results were transmitted electronically to NEDSS for contact tracing and epidemiologic purposes. Prior to development of this functionality within NUlirt, these sites were required to fax COVID-19 antigen test results to adhere to state-reportable disease-testing regulations.
While the Nebraska DHHS used these rapid antigen tests to supplement PCR testing, local universities and collegiate athletic associations also used this testing platform to screen athletes, staff, and game officials prior to competitions. In support of an effort to screen athletes, staff, and game officials, 10 the NPHL informatics team configured NUlirt to manage the testing of athletes, coaching staff, and game officials.
Results
Throughout the COVID-19 pandemic, NUlirt addressed critical needs for NPHL, DHHS, UNMC, Nebraska Medicine, and multiple LHDs. NUlirt functionality as of June 2022 is illustrated in Figure 2. A core lesson learned was the importance of robust and flexible information technology resources necessary to mount effective responses to issues faced by PHLs during emergencies.
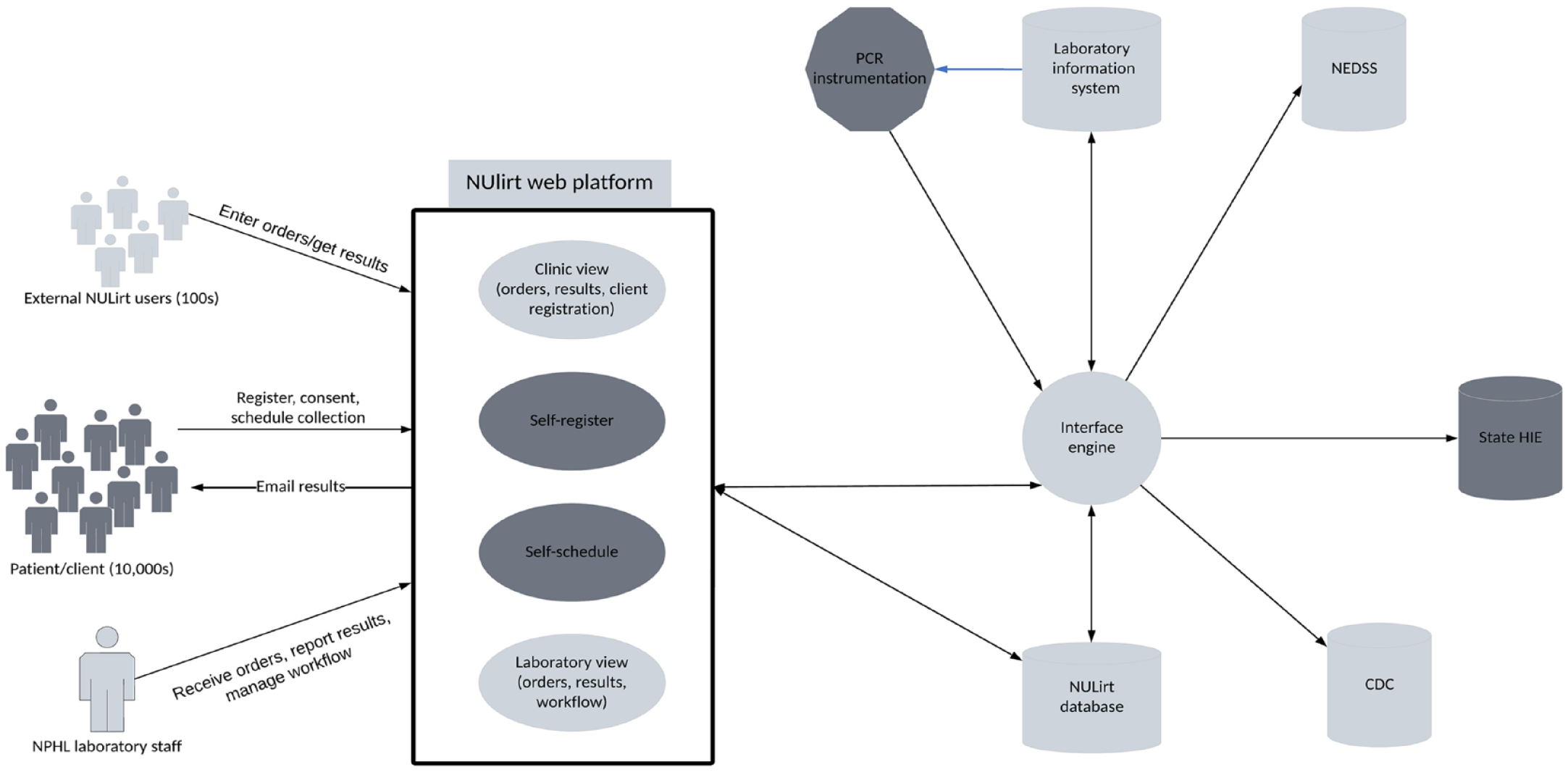
Functionality of the Nebraska University Laboratory Information Reporting Tool (NUlirt), Nebraska Public Health Laboratory (NPHL), June 2022. Functionality expanded to include patient self-registration and scheduling, secure results reporting directly to patients, and laboratory automation/device-level integration. New functions and data communication are in dark gray. Abbreviations: CDC, Centers for Disease Control and Prevention; HIE, health information exchange; NEDSS, National Electronic Disease Surveillance System; PCR, polymerase chain reaction.
Number of Tests and Impact on NPHL and Regional Pathology Services Operations
NPHL completed >121 000 PCR COVID-19 tests using NUlirt as the central health information technology platform from March 2020, the beginning of the pandemic, through January 2022. In addition to nasopharyngeal PCR test management, >62 000 rapid antigen tests performed by LHDs were managed and recorded through NUlirt, as well as 17 000 saliva PCR tests in the public school system and among UNMC faculty, staff, and students.
Because state law required that all COVID-19 test results be reported to the Nebraska DHHS, results recorded in NUlirt regardless of test method (ie, PCR or rapid antigen test) were electronically transmitted by HL7 messaging and ingested into NEDSS for epidemiologic use and contact tracing. Messages were also communicated via the state health information exchange, CyncHealth, so that test results could be accessed by any authorized health care provider in the state for purposes of providing patient care.
User Implementation and Adoption
Implementation and adoption of NUlirt was rapid and straightforward for most users. Questions about NUlirt use from end users were typically limited to new users and/or sites unfamiliar with NUlirt functionality. General comments from end users were positive, without substantial user issues. For example, one LHD manager said, “NUlirt was the best thing about the pandemic.”
Use of NUlirt software has expanded throughout Nebraska. The software is now used for student and staff COVID-19 testing and screening at UNMC and Nebraska Medical and at the University of Nebraska–Omaha campus in congregate living areas. It was used at major athletic events during summer 2021. NUlirt has been instrumental to the success of asymptomatic COVID-19 screening programs, with diverse partners that include theater and music venues, migrant and American Indian communities, and critical infrastructure operations (eg, airport authority), who may not otherwise have had access to traditional health care facility–based testing.
While successes were many, several hurdles had to be overcome. Time pressure was the primary issue. The NPHL information technology team often had <48 hours to develop, test, and deploy a NULirt feature. These urgent requests were made in addition to ongoing software enhancements. Therefore, trade-offs were made, including programming shortcuts and delaying important software improvements. Shortcuts that ensured a minimally viable product worked in the short term, but long-term software performance cannot be achieved via shortcut. As a result, some aspects of NULirt could not be easily extended for other uses without causing software failures. In essence, a technical deficit was created. Technical deficits in software applications are not uncommon, but when they persist and are not corrected, the result is a nonsustainable product. The NPHL information technology team was able to address these technical issues with the help of extramural funding from CDC and a lull in the pandemic.
Discussion
NPHL operations were designed primarily to support epidemiologic services for Nebraska DHHS and not as a high-volume diagnostic laboratory. The pandemic required a paradigm shift for the PHL operation to function as a diagnostic laboratory. NUlirt enabled this operational evolution to be successful. Laboratory staffing was a limiting factor for testing capacity. Integration of specimen collection and handling, laboratory equipment interface and integration, and broad use of HL7 electronic messaging allowed the laboratory to increase testing throughput by 400%, support up to 2000 SARS-CoV-2 PCR tests per day, and maintain normal PHL testing services with no additional laboratory staffing, while increasing information technology staff by only 2 full-time equivalents. High levels of health system interfaces with client EHRs, the LIS, epidemiology systems, and the state health information exchange essentially eliminated paper processes from the laboratory.
The transition from testing in traditional health care settings to testing at community locations throughout the state pushed the information technology platforms used to order, accession, and result tests to evolve. NUlirt adapted to meet community testing needs that were rapidly growing and changing alongside accelerating disease transmission throughout the state. From an epidemiologic and public health operations perspective, NUlirt gave Nebraska DHHS an edge through 3 primary advantages, which grew from close collaboration among NPHL information technology, NPHL laboratorians, and Nebraska DHHS epidemiologists.
The first advantage entailed the incorporation of ask at order entry (AOE) questions, which assisted in intelligent use of limited testing supplies. Specifically, AOEs addressed 3 critical functions early in the pandemic: targeted use of limited testing supplies, symptom/infection association, and support of pooled tests. When an order for a SARS-CoV-2 test was placed in NUlirt, the ordering health care provider answered questions about the patient’s symptom status, date of illness onset, type of symptoms, type of exposure (eg, direct contact, travel, congregate living), illness severity (eg, hospitalized, intensive care unit), and occupation. 11 By selecting patient factors at the time of order entry, limited testing capacity could be prioritized toward specimens with the highest pretest probability of positivity. Prioritization of specimens with a higher likelihood of testing positive facilitated higher rates of COVID-19–positive patients identified for isolation and allowed the limited supply of personal protective equipment to be used more efficiently.
Throughout the pandemic, case investigations have been relied on to uncover new associations between COVID-19 and symptom type as well as conditions that put people at high risk for hospitalization and death. Case investigation workloads were partially mitigated by completing components of the investigation at the time of order entry through AOE questions as a requirement to receive testing. Associations were able to be identified without additional, direct patient contact. Moreover, associations were more rigorously identified by comparing responses between those who received positive and negative test results. AOE questions in the software program allowed for Nebraska DHHS to identify loss of taste and loss of smell as the most specific criteria associated with receiving a positive test result early in the pandemic response.
AOE questions also guided a process to group test specimens in pools when reagents were in short supply. 12 Nebraska DHHS analyzed AOE questions and identified the symptom and exposure characteristics most associated with a positive test result. Specimens identified as having a high likelihood of testing positive were tested individually, while specimens with a low likelihood of testing positive were grouped in a pool of 5 to test. If that pool then tested negative, reagents for 4 tests were saved. This practice allowed the laboratory to continue to test a large number of specimens when critical supply shortages existed. Because NUlirt contained the AOE questions and was capable of flagging specimens for pooling, the workflow between field entry and laboratory pooling practice was streamlined.13-16
The second advantage for Nebraska DHHS is that NUlirt supported the transition from health care facility testing to community testing in places where traditional administrative support functions were not available, such as patient scheduling, patient registration, acquisition of consent to test, and reporting of personal test results. 17 NUlirt functionality supported the change in testing and specimen collection settings to support high-throughput community collection sites, which were rapidly pulled together during the emergency response, thus creating greater access to testing and subsequent care to the general population.
Finally, support for antigen test result reporting and AOE gathering was used to address the known reduced accuracy of the various antigen tests. 18 If a patient was symptomatic or exposed and tested negative on an antigen test, a prompt appeared in the software program to immediately reflex to a PCR order. Similarly, if a patient was not symptomatic and not exposed but received a positive test result, a prompt appeared to immediately reflex to a PCR order. By building reflex testing into NUlirt, more confirmatory testing was completed, and more false-negative and false-positive antigen results were identified.
Many of the successes realized by NPHL are based on the importance that information technology has played in NPHL for decades. During many years, NPHL and Nebraska DHHS leadership consistently decided to invest in information technology services as a mechanism to extend NPHL services for Nebraska. The web-portal approach used for NPHL testing services and communication of testing results during the pandemic is one example of the return on this investment. The approach predated the COVID-19 pandemic by >15 years and will continue to play an important role in NPHL. Investment in electronic interfaces between NULirt and multiple client and data consumer sites and retention of the internal expertise to develop and support such interfaces further demonstrate NPHL’s and Nebraska DHHS’s commitment to sustainable and useful information technology services. As a result, NPHL had the necessary information technology infrastructure and expertise to develop, expand, and support NULirt before, during, and after the pandemic.
Practice Implications
The NULirt set of software was developed by using open-source software with liberal licensing agreements. As noted, some features were developed de novo even when commercial options were available to allow NPHL to distribute the NULirt program to other PHLs. NPHL staff are aware of the financial stresses and limits experienced by the PHL community and desired to develop an asset that could benefit other PHLs without substantial commitments to capital expenditures. While some PHLs may choose alternative directions in some instances (eg, the use of a data broker or interface consolidator vs in-house staff), the NULirt platform is flexible and can be incorporated into any number of information technology configurations.
The advent of and reliance on genomic sequence data of SARS-CoV-2 to inform public health officials was evident during the pandemic. Genomics will play an ever-increasing role in public health as now observed in clinical medicine. 19 Architecting genomic data management systems that can serve public health, research, and clinical medicine will be foundational for future health emergencies but also for known issues, such as multidrug-resistant organisms. A continued focus on laboratory operations and adoption of data communications—including laboratory automation; LIS-to-device integration; use of instrument-ready, bar-coded specimen labels to streamline laboratory operations; and broad adoption of electronic data exchange between PHLs and clients—will be necessary to leverage scarce resources for high-value use.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received support from the Centers for Disease Control and Prevention through CK19-1904 ELC Enhancing Detection Through Coronavirus Response and Relief.
