Abstract
Objective:
Community of Hope Health Clinic (CHHC), a free and charitable clinic in Shelby County, Alabama, developed a screening protocol to identify patients with asymptomatic COVID-19 with the goal of minimizing infection risk for other patients, health care providers, and staff. We sought to determine whether the use of the CHHC screening protocol identified asymptomatic carriers of SARS-CoV-2 before their scheduled clinic visits.
Methods:
The CHHC screening protocol included the use of an adapted Centers for Disease Control and Prevention questionnaire via telephone 48-72 hours before a scheduled clinic appointment, coupled with a second administration of the questionnaire and a temperature check immediately before the patient’s scheduled visit. Patients with positive responses to any questions or whose temperatures were greater than 100.4 °F were refused entry to the clinic, their appointments were rescheduled, and a SARS-CoV-2 test was recommended. SARS-CoV-2 laboratory testing was conducted on all patients with an appointment from October 26, 2020, through February 18, 2021, to verify whether the screening protocol was effective.
Results:
Of 298 patient encounters, 20 patients screened positive on the SARS-CoV-2 patient screen. Another 278 patients screened negative, were seen for patient care, and received a SARS-CoV-2 test via nasal swab; 274 (98.6%) patients received a negative test result, and 4 (1.4%) patients received a positive test result.
Conclusions:
The CHHC SARS-CoV-2 screening protocol, as validated by SARS-CoV-2 assay, was effective in screening out asymptomatic patients infected with SARS-CoV-2 before they were seen in clinic. As clinics and office practices return to prepandemic volumes, the use of such a screening protocol can help mitigate the risk of serious COVID-19 infection, especially for unvaccinated patients, health care providers, and staff.
SARS-CoV-2, the virus that causes COVID-19 infection, is transmitted through airborne aerosols, which can travel for at least 6 feet. 1 The disease is characterized by an unpredictable course, ranging from asymptomatic to mild respiratory symptoms, to pneumonia, and even to acute respiratory distress syndrome and life-threatening illness. Diabetes, chronic lung disease, hypertension, heart disease, obesity, and increasing age are risk factors for severe disease. 2 In contrast, younger people (ie, aged <30 years) seem to have mild or even asymptomatic disease and, thus, might be crucial in further spreading the disease. 3 In addition, the COVID-19 pandemic has laid bare the health and social disparities in the United States, as disproportionate rates of exposure, illness, hospitalization, and death have occurred among racial and ethnic minority groups. 4 Racial and ethnic minority populations are often affected by underlying social determinants of health, such as socioeconomic status, reduced access to health care, and occupation-related exposures, all of which are factors leading to disparities. 4 Data from the Centers for Disease Control and Prevention (CDC) indicate that, in comparison with non-Hispanic White people, Hispanic people are 1.3 times as likely to contract SARS-CoV-2, 3.2 times as likely to be hospitalized for COVID-19, and 2.3 times as likely to die from the disease. 4 Similarly, compared with non-Hispanic White people, African American people are 1.1 times as likely to become infected, 2.9 times as likely to be hospitalized, and 1.9 times as likely to die from COVID-19. 4
The clinical guidelines for those suspected of infection with SARS-CoV-2 are based on the signs and symptoms present at illness onset. These symptoms may vary during the course of the disease, but those who are symptomatic often experience 1 or more of the following: fever or chills, cough, shortness of breath or difficulty breathing, fatigue, muscle or body aches, headache, new loss of taste or smell, congestion or runny nose, nausea or vomiting, and diarrhea. 5 Most recently, sore throat and upper respiratory symptoms have been signature concerns of those infected with the Omicron variant.6,7 Many people infected with SARS-CoV-2 never develop symptoms or develop mild symptoms and are substantial contributors to community SARS-CoV-2 transmission. 8 Such is the case for infection with the more contagious Omicron variant, for which symptoms tend to be mild or nonexistent.6,7
CDC has published several guidelines for health care providers, hospitals, clinics, nursing homes, and schools to help limit the spread of SARS-CoV-2.5,8,9 These recommendations have evolved as more information becomes available. The use of personal protective equipment (PPE) such as face masks, gowns, gloves, and face shields has been a cornerstone of many of the guidelines. In combination with questionnaire screening, temperature checks, and social distancing, the aim of these guidelines has been to reduce the spread of the SARS-CoV-2 virus among people in close contact with one another.
CDC recommends implementing an expanded screening testing strategy to rapidly identify people without symptoms (ie, asymptomatically or presymptomatically infected with SARS-CoV-2) who contribute to the silent spread of infection. 8 This expanded screening of asymptomatic people should focus on groups (eg, older adults, racial and ethnic minority groups, people with disabilities) that have been heavily affected by SARS-CoV-2 to reduce morbidity and mortality and to ensure health equity. 8 CDC recommended screening workers in high-density worksites or worksites with large numbers of close contacts (eg, restaurant workers, transportation workers, grocery store workers); government workers with public interactions as part of their duties (eg, post office); residents and staff in congregate settings (eg, shelters serving the homeless, correctional facilities, or residential settings serving people with disabilities); workplaces that provide congregate housing (eg, fishing vessels, offshore platforms, farmworker housing, wildland firefighter camps); students, faculty, and staff at institutions of higher education, including community colleges and technical schools; teachers and staff in kindergarten through 12th-grade schools and/or childcare settings; people who have recently traveled; those who attended mass gatherings; first responders (eg, police, firefighters, emergency medical technicians); and health care personnel. 8
Even as in-person clinic volumes have returned to prepandemic levels, pockets of the population are not yet fully vaccinated, and new variants of concern continue in the community. As a result, exposure to SARS-CoV-2–positive patients is inevitable. However, certain steps can mitigate the risk of transmission. CDC guidelines for outpatient facilities and ambulatory care practices include having staff ask symptomatic patients who require an in-person visit to call before they leave home, so that staff are ready to receive them using appropriate PPE and infection control practices. 9 Visual alerts, such as signs and posters in appropriate languages, at entrances and in strategic places should provide instructions on hand hygiene, use of face masks, and cough etiquette. 9 Other possible changes include arranging waiting rooms to allow patients to be socially distanced, using partitions or signs to create designated areas or waiting lines, and asking patients to remain outside or in their vehicles. 9 Screening patients for symptoms and recent close contacts with SARS-CoV-2 is essential. One option is to set up triage booths to screen patients safely. 9 Universal PPE for health care providers and their staff, ideally including N95 masks, is recommended. 10 Furthermore, it is important to be able to test all patients who present for care to determine their SARS-CoV-2 status.
Community of Hope Health Clinic (CHHC) is a free and charitable clinic located in Shelby County, Alabama, part of the Birmingham metropolitan area. CHHC has a small paid staff and uses volunteer health professionals (physicians, nurse practitioners, physician assistants, nurses) and volunteer staff to provide care to a medically underserved patient population, including many Hispanic patients, 12 hours per week.
As the COVID-19 pandemic hit during 2020, CHHC initially transitioned from in-person patient visits to telehealth visits via telephone and the clinic’s Health Insurance Portability and Accountability Act (HIPAA)–compliant telemedicine platform, doxy.me. When the clinic began to return to in-person visits in June 2020, it was clear that a protocol was needed to screen patients for SARS-CoV-2 infection to minimize the risk of transmission to other patients, clinic health care providers, and staff. This need for a screening protocol was especially important because the clinic patient population, which predominately comprised racial and ethnic minority groups and frontline workers, is at especially high risk for SARS-CoV-2 infection. Thus, the purpose of this quality improvement project was to determine whether screening for SARS-CoV-2 infection using an adapted CDC questionnaire administered 48-72 hours before a scheduled clinic appointment, coupled with a second administration of the same questionnaire and a temperature check immediately before the scheduled visit, could identify asymptomatic carriers of SARS-CoV-2 infection.
Methods
Starting in June 2020, patients scheduled for routine in-person visits at CHHC were contacted 48-72 hours before their appointment for a telephone screening questionnaire. Using an adaptation of the CDC Facilities SARS-CoV-2 Screening tool, 11 patients were asked if they had had a fever of ≥100.4 °F, chills, cough, sore throat, loss of smell or taste, shortness of breath, difficulty breathing, fatigue, muscle/body aches, headaches, congestion/runny nose, nausea/vomiting, or diarrhea in the past 14 days. They were also asked if they had been in close contact with anyone who had a confirmed SARS-CoV-2 diagnosis in the past 14 days. A positive response to any question on the screen resulted in a recommendation to seek testing for SARS-CoV-2 and rescheduling of the visit to a later date.
When patients who responded negatively to the initial telephone screen arrived at the clinic for their scheduled visits, they were met at the door by a staff member wearing a face mask and gloves. Patients were required to wear a face mask, and one was provided if necessary. Before being allowed entry into the clinic facility, patients were again surveyed using the SARS-CoV-2 patient screen, and their temperature was taken. Anyone with a temperature of ≥100.4 °F or who answered positively to any of the screening questions was refused entry to the clinic, and their appointment was rescheduled for a later date. It was also recommended that they be tested for SARS-CoV-2 infection. Those who screened negative for symptoms according to the questionnaire and temperature were allowed entry to the clinic. Patients were required to remain seated in the designated waiting room after being checked in and screened for SARS-CoV-2. Seating was arranged with a minimum of 6 feet between chairs, and hand sanitizer was available at the entrance and in the waiting room for patient use.
In October 2020, CHHC received Coronavirus Aid, Relief, and Economic Security Act 12 funding from the Alabama Statewide Area Health Education Center program. These funds were used to support SARS-CoV-2 testing of patients, allowing us to determine if our screening protocol was successful in identifying asymptomatic carriers of SARS-CoV-2. Thus, starting October 26, 2020, patients who screened negative and entered the clinic were offered a nasal swab for evaluation of the presence of SARS-CoV-2. Patients with a recent (within 3 days) negative SARS-CoV-2 test result or those seen in the clinic within 1 week with a negative test result were not required to be further tested. To be tested, each patient moved to a designated area, standing behind a clear plexiglass structure (6 feet tall by 3 feet wide). A staff member wearing appropriate PPE stood on the opposite side of the plexiglass, instructing and observing the patient self-swabbing the nasal nares. Patients were instructed to place the cotton-tipped applicator into each nostril and rotate for approximately 5 seconds. The swab was then removed and placed into a tube with medium. Once handed to the staff member, the applicator was snapped at a premarked line and the tube cap tightened. Samples were maintained in cold storage (2 °C to 8 °C) and transported to the clinical laboratory within 48 hours.
SARS-CoV-2 testing was conducted at the University of Alabama at Birmingham’s Division of Infectious Diseases Diagnostic Laboratory using the Aptima SARS-CoV-2 (Hologic) assay performed on the Hologic Panther system. The assay was granted Emergency Use Authorization by the US Food and Drug Administration in May 2020. This assay amplifies and detects SARS-CoV-2 RNA using transcription-mediated amplification (a process similar to polymerase chain reaction testing). This assay can detect as few as 50 viruses/1 mL with excellent specificity (ie, false-positive results are rare). The assay is suitable for use with nasopharyngeal, nasal, or oropharyngeal swabs. As of May 2022, all currently described mutations in the virus could be detected by the assay.
The average laboratory turnaround time was <24 hours. All patients were contacted by a staff member within 24-48 hours after the laboratory results were received. Patients with a positive test result were questioned again by telephone using the SARS-CoV-2 patient screen to determine if they had developed any symptoms. They were also instructed to self-quarantine for 14 days. Patients with positive results were contacted by telephone a second time, approximately 14 days after the initial test was completed, and again asked the questions on the SARS-CoV-2 patient screen. Test results and responses to the SARS-CoV-2 patient screen were recorded and filed in each patient’s medical record.
This quality improvement project was submitted for review to the University of Alabama at Birmingham Institutional Review Board, which determined this project to be not human subjects research.
Results
From October 26, 2020, through February 18, 2021, the CHHC SARS-CoV-2 screening protocol was used on 298 patient encounters scheduled for appointments at the clinic (Figure). Twenty patients (6.7% of the total scheduled visits) screened positive on the SARS-CoV-2 patient screen (18 screened positive by telephone when contacted 48-72 hours before their appointment, and 2 screened positive at the door of the clinic when they came for their appointment). These patients did not gain entry to the clinic, and all were recommended to receive a SARS-CoV-2 test. Seven of the 20 patients received a SARS-CoV-2 assay test conducted by CHHC, and all 7 received a positive test result. They included 6 Hispanic White people (4 women and 2 men) and 1 non-Hispanic White man, all aged 31 to ≥65 years. The remaining 13 patients who screened positive were either tested elsewhere or not tested at all; therefore, SARS-CoV-2 test results were not available for analysis for this group.
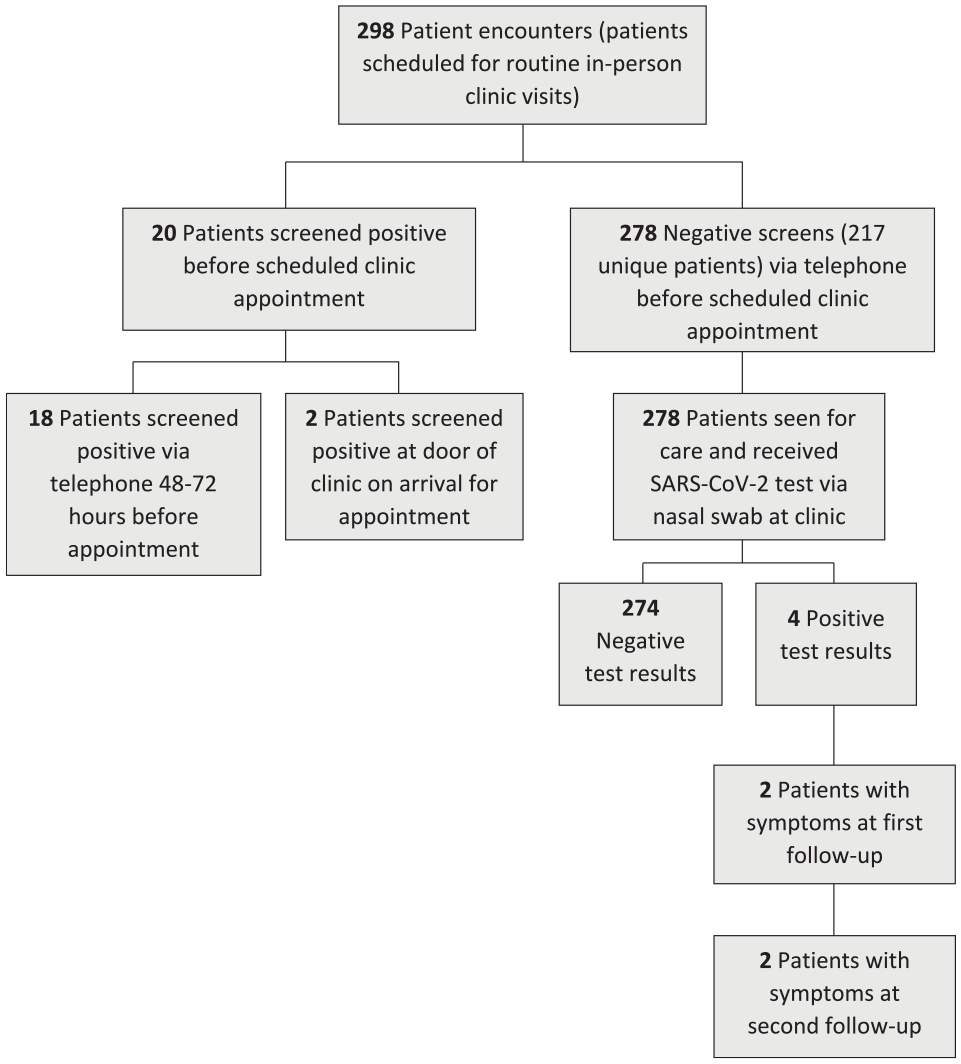
Patients screened via protocol for SARS-CoV-2 at Community of Hope Health Clinic, Shelby County, Alabama, October 26, 2020–February 18, 2021.
Another 278 patients screened negative via the 2 patient screens and temperature check and were seen in the clinic for patient care. These 278 patients received SARS-CoV-2 assay tests on site during their clinic appointment. A total of 61 patients had >1 clinic visit during the project time frame and, therefore, had >1 SARS-CoV-2 assay test, leaving 217 unique patients seen for care during the study period. The 217 patients ranged in age from 19 to ≥65 years, with most aged 41-60 years (n = 147, 67.7%). Most patients were female (n = 147, 67.7%) and Hispanic (n = 144, 66.4%) (Table 1).
Characteristics of unique patients screened for SARS-CoV-2 at Community of Hope Health Clinic (N = 217), Shelby County, Alabama, October 26, 2020–February 18, 2021
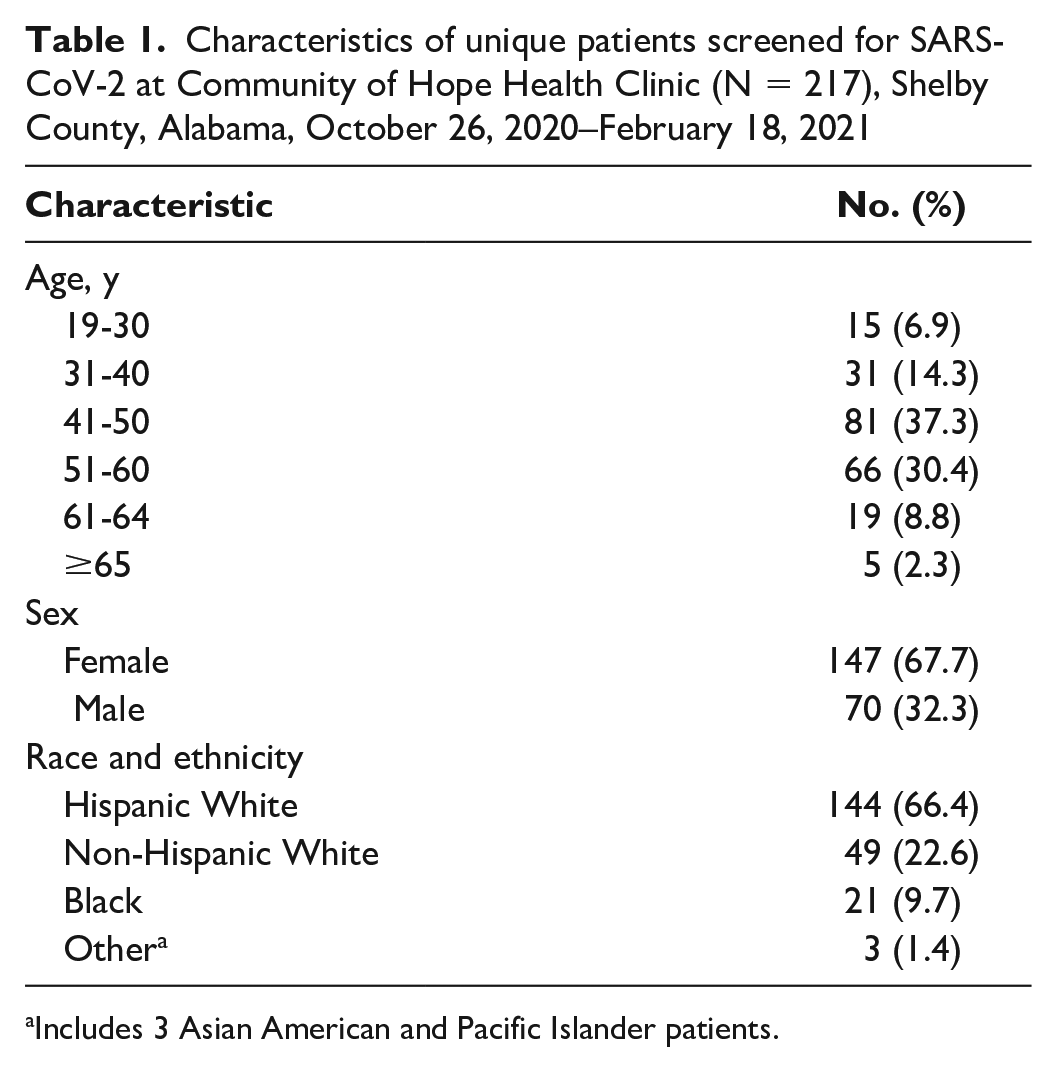
Includes 3 Asian American and Pacific Islander patients.
Of the 278 SARS-CoV-2 assay tests performed, 274 (98.6%) had negative results and 4 (1.4%) had positive results. The 4 patients who received a positive test result received 2 follow-up telephone calls from clinic staff. The first call, to notify the patient of the positive test result, occurred within 24-48 hours of their clinic visit. The second call took place approximately 14 days after the test was completed. During both calls, patients were again asked the questions on the SARS-CoV-2 patient screen. Two patients (1 Hispanic woman and 1 non-Hispanic White man) were symptomatic at both telephone calls, indicating they had been presymptomatic at the time of their clinic visit. The other 2 patients with positive test results (1 Hispanic woman and 1 Hispanic man) remained asymptomatic at both follow-up telephone calls.
Although we could not obtain SARS-CoV-2 test information from 13 patients who screened positive by the CHHC SARS-CoV-2 screening protocol, the performance statistics (sensitivity, specificity, positive predictive value, negative predictive value) of the screening protocol can be calculated based on some assumptions. The exact probabilities of observing 7 positive test results from 7 patients can be calculated using a hypergeometric probability distribution given various numbers of true positives among the 20 patients. For example, the probabilities of observing 7 positives from 7 patients are 0.01, 0.02, 0.04, and 0.08 given the true number of SARS-CoV-2 positive test results among 20 patients are 12, 13, 14, and 15, respectively. Therefore, we may assume that it is unlikely that there would be fewer than 15 SARS-CoV-2–positive cases if all 20 patients who screened positive had been tested. The performance statistics with accompanying 95% CIs assume 15, 18, and 20 patients are SARS-CoV-2–positive among the 20 patients screened by the CHHC SARS-CoV-2 screening protocol. These results suggest that the screening protocol is effective with high specificity and sensitivity (Table 2).
Performance statistics of the SARS-CoV-2 screening protocol at the Community of Hope Health Clinic (CHHC; N = 217), Shelby County, Alabama, October 26, 2020–February 18, 2021
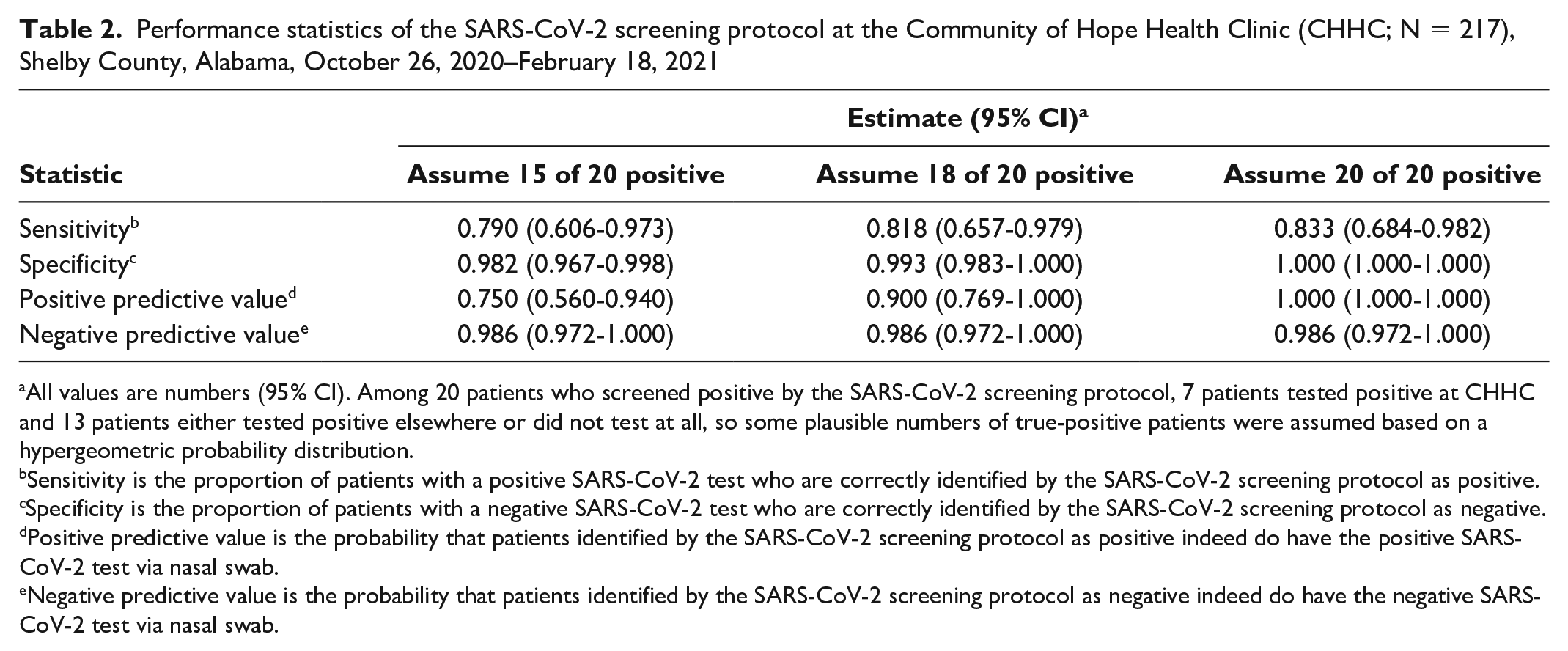
All values are numbers (95% CI). Among 20 patients who screened positive by the SARS-CoV-2 screening protocol, 7 patients tested positive at CHHC and 13 patients either tested positive elsewhere or did not test at all, so some plausible numbers of true-positive patients were assumed based on a hypergeometric probability distribution.
Sensitivity is the proportion of patients with a positive SARS-CoV-2 test who are correctly identified by the SARS-CoV-2 screening protocol as positive.
Specificity is the proportion of patients with a negative SARS-CoV-2 test who are correctly identified by the SARS-CoV-2 screening protocol as negative.
Positive predictive value is the probability that patients identified by the SARS-CoV-2 screening protocol as positive indeed do have the positive SARS-CoV-2 test via nasal swab.
Negative predictive value is the probability that patients identified by the SARS-CoV-2 screening protocol as negative indeed do have the negative SARS-CoV-2 test via nasal swab.
Discussion
Despite the CDC’s recommendation of screening patients for SARS-CoV-2, little published data are available on the use of screening protocols to identify asymptomatic SARS-CoV-2. We could find no published studies documenting the usefulness of a screening protocol to identify asymptomatic SARS-CoV-2 in primary, ambulatory, or outpatient care. However, as clinics transition to in-person visits, the use of a screening protocol to help identify patients with asymptomatic SARS-CoV-2 can help minimize the risk of transmission to others, including patients, health care providers, and staff. This need for a screening tool is especially true for free clinics such as CHHC that do not routinely have funds to support the cost of SARS-CoV-2 testing and whose patient populations are often among those most vulnerable to infection.
The CHHC SARS-CoV-2 screening protocol included a telephone screening using an adapted CDC Facilities SARS-CoV-2 screening tool 48-72 hours before the patient’s scheduled clinic appointment, followed by use of the same screening tool plus a temperature check when patients arrived for their appointment. This screening protocol, as validated by SARS-CoV-2 assay, was effective in screening out asymptomatic and presymptomatic patients infected with SARS-CoV-2 before they were seen in the clinic. The protocol can be replicated by others interested in implementing a screening process for identifying patients without symptoms who may contribute to the spread of infection.
Limitations
This study had several limitations. First, it was limited by the small number of patients seen during the project time frame. Second, the project was conducted before detection of the Omicron variant in the United States, which caused more asymptomatic infection. Lastly, while the screening protocol included numerous possible symptoms, patients with SARS-CoV-2 infection may have had other symptoms not included in the screening. It has been suggested that as variants and their associated symptoms change, screening questions may also need to change to better identify novel symptoms. 13
Conclusions
COVID-19 vaccines for the SARS-CoV-2 virus became available in the United States through an Emergency Use Authorization in late December 2020 14 and are now authorized and widely available to adults and children aged ≥6 months in every state. As more of the US population has been vaccinated, the numbers of SARS-CoV-2 cases, hospitalizations, and deaths have decreased. However, the Omicron variant has caused unprecedented numbers of new infections and increasing numbers of hospitalizations. A large number of the population has not yet been fully vaccinated because of lack of easy access, hesitancy, or refusal. Low vaccination rates are especially seen in the southeastern United States, where researchers fear continued outbreaks will occur because of low vaccination rates. 15 As clinics and office practices return to their prepandemic volumes, mostly via in-person visits, the risk of serious infection remains for unvaccinated patients, health care providers, and staff. Use of a sensitive and specific screening protocol, such as the one described here, can help mitigate that risk.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
