Abstract
Objectives
The Utah Study of Associated Risks of Stillbirth (SOARS) collects data about stillbirths that are not included in medical records or on fetal death certificates. We describe the design, methods, and survey response rate from the first year of SOARS.
Methods
The Utah Department of Health identified all Utah women who experienced a stillbirth from June 1, 2018, through May 31, 2019, via fetal death certificates and invited them to participate in SOARS. The research team based the study protocol on the Pregnancy Risk Assessment Monitoring System surveillance of women with live births and modified it to be sensitive to women’s recent experience of a stillbirth. We used fetal death certificates to examine survey response rates overall and by maternal characteristics, gestational age of the fetus, and month in which the loss occurred.
Results:
Of 288 women invited to participate in the study, 167 (58.0%) completed the survey; 149 (89.2%) responded by mail and 18 (10.8%) by telephone. A higher proportion of women who were non-Hispanic White (vs other races/ethnicities), were married (vs unmarried), and had ≥high school education (vs <high school education) responded to the survey. Differences between responders and nonresponders by maternal age, gestational age of the fetus, or month of delivery were not significant. Among responders, item nonresponse rates were low (range, 0.6%-5.4%). The question about income (4.8%) and the questions about tests offered and performed during the hospital stay had the highest item nonresponse rates.
Conclusions:
The response rate suggests that a mail- and telephone-based survey can be successful in collecting self-reported information about risk factors for stillbirths not currently included in medical records or fetal death certificates.
Stillbirths, or fetal deaths at ≥20 weeks of gestation, are associated with profound and long-lasting adverse effects on families, the health care system, and society.1,2 In 2017, the national rate of stillbirth was 5.89 stillbirths per 1000 live births plus stillbirths. 3 In Utah, 251 stillbirths occurred in 2017, for a rate of 5.14 stillbirths per 1000 live births plus stillbirths. 4 Although stillbirths are one of the most common adverse pregnancy outcomes, occurring in approximately 1 in 160 deliveries in the United States, they are not well studied. 5 Recognized causes of stillbirths include maternal infection and obstetric complications, but the etiology of many stillbirths is unclear. 6
The Pregnancy Risk Assessment Monitoring System (PRAMS) is a surveillance system of selected maternal behaviors and experiences that occur before, during, and shortly after pregnancy. 7 It began in 1987 and is conducted jointly by participating state, territorial, or local health departments and the Centers for Disease Control and Prevention (CDC). Women are randomly selected for participation based on birth certificate records 2-6 months after delivery. Sampled women are contacted by mail, with follow-up telephone calls for nonresponders. 7 However, this methodology does not capture data on women whose infant was not born alive and did not receive a certificate of live birth.
In 2012-2013, a pilot study to investigate the feasibility of expanding the PRAMS methodology to include stillbirths was conducted for 3 months in Georgia. 8 The research team identified women who experienced a stillbirth through fetal death records. The results showed that sampling women based on the fetal death certificate, as opposed to the live birth certificate, was possible, although the response rate was lower than the regular Georgia PRAMS response rate. Based on the Georgia pilot study, CDC developed a model stillbirth surveillance protocol for implementation and refinement.
In 2016, Utah received funding from CDC to conduct an extended pilot study of this revised PRAMS methodology to sample women who experienced a stillbirth. The objectives of this study, the Study of Associated Risks of Stillbirth (SOARS), were to evaluate the expansion of the PRAMS methodology in Utah to women who experience stillbirths and to collect data about stillbirths that are not included in medical records or on fetal death certificates. We describe the methods and response rates, overall and by maternal characteristics, from the first year of Utah SOARS.
Methods
The PRAMS team at the Utah Department of Health initiated the SOARS project by requesting the full list of fetal death certificates for a 1-year period from Utah Vital Records and Statistics. We included all women who were Utah residents and experienced a fetal death in Utah with a reported gestational age of ≥20 weeks from June 1, 2018, through May 31, 2019, in the study. We sampled women who had experienced multiple losses from a multiple-gestation pregnancy once because the study focused on the experiences of the woman, not the infant.
The Utah Department of Health used a modified version of the pilot survey from Georgia as the starting point for questionnaire development. 8 The Utah SOARS advisory committee reviewed the survey instrument, and the research team revised it based on committee feedback. The Utah team invited members of the regular PRAMS advisory committee, maternal–fetal medicine physicians specializing in stillbirth, pathologists, infant loss support groups, and grief support groups to participate on the SOARS advisory committee. During the pre-implementation phase, the committee developed partnerships through in-person meetings and ongoing email and telephone communication. Through this process, several committee members became champions of the SOARS program, which was integral for its successful launch.
The final SOARS instrument included 65 questions about women’s life experiences and medical problems before and during pregnancy, health behaviors, clinical testing, experiences in the hospital around the time of delivery, and bereavement support. 9 The team used many questions based on the regular PRAMS survey for live births.
The Utah Department of Health field-tested the instrument with 14 English-speaking women who had experienced a stillbirth to ensure that the questions were clear and sensitive to the grieving process. The women completed the draft survey and the team assessed the women’s understanding of the questions. Then the team asked the women how the survey questions made them feel and gave the women the opportunity to provide additional personal feedback on the survey questions.
Because data from the Utah fetal death file showed that 13% of fetal deaths occur among Hispanic women, the final survey was translated to Spanish. The research team also created a separate version of the survey for women aged <18 years, which excluded questions about physical abuse. In Utah, it is mandatory to report physical abuse of minors, and a statement about this requirement would need to be included in the informed consent document. Because of concerns that this language would deter minors from responding, Utah SOARS chose not to include the physical abuse questions for women aged <18 years.
The PRAMS team at the Utah Department of Health developed the SOARS protocol based on the model stillbirth surveillance protocol developed by CDC. Although this protocol is similar to the regular PRAMS protocol, adapted from Dillman et al, 10 which includes multiple contacts by mail and telephone for women with a recent live birth, 7 the Utah team made several important modifications to be sensitive to women’s recent experience of a stillbirth, including fewer contacts with the women. About 2 to 4 months after delivery, the Utah team mailed a card to women that expressed sympathy for their loss and explained that they would receive a survey about their experience about a week later. About 11 days after the team mailed the sympathy card, the team mailed the first questionnaire packet. The packet included a cover letter describing the goals of the study, participation and consent information, the questionnaire booklet, a pen, a list of support resources, the study incentive, and a self-addressed stamped envelope for returning the survey. A 3-year calendar to aid recall of experiences before and during pregnancy appeared inside the back cover of the questionnaire booklet. Hispanic women received materials in both English and Spanish. Women who did not return the survey within 10 days were sent a follow-up letter that highlighted the importance of their responses and asked them to please complete the survey. Women who did not return the survey within 10 days of the follow-up letter received a second questionnaire packet, which included the same items as the first questionnaire packet, other than a pencil instead of a pen and no additional incentive. Telephone follow-up began 14 days after the second questionnaire packet was mailed. Women who were reached by telephone were given the option of completing the survey by telephone with a trained interviewer in English or Spanish. The team made a maximum of 8 telephone call attempts. This research was approved by the institutional review boards at the Utah Department of Health and CDC.
Caring Connections, a program at the University of Utah College of Nursing, provided bereavement training at the Utah Department of Health for telephone interviewers and other staff members. Telephone interviewers learned techniques to comfort the interviewee without compromising data collection, as well as motivational interviewing techniques to gently encourage an interviewee to postpone participation instead of declining. All SOARS staff members also learned self-care techniques that could be used if they became overwhelmed by the sadness of the topic of stillbirth.
To encourage responses by mail, the Utah Department of Health developed customized, professional, coordinated mailing materials that included official SOARS letterhead for cover letters, envelopes that matched the letterhead to mail all correspondence, personal salutations on cover letters (instead of “Dear mother”), and a signature in blue ink of the Utah SOARS project coordinator on the cover letters printed in color. In addition, women received a necklace with a swallow charm, hand crafted by a Utah artist, as an incentive (Figure). Furthermore, a maternal–fetal medicine practice in Salt Lake City offered a free medical consultation, in person or via telephone, to all women who received a survey. Completion of the SOARS survey was not required for the free consultation. Women received the contact information for this medical practice in the first questionnaire packet and had the opportunity to contact the office directly. Women who completed the survey were not given rewards.

Necklace with a swallow charm, hand crafted by a Utah artist, which was provided as an incentive for the Utah Study of Associated Risks of Stillbirth (SOARS).
The Utah team developed Excel spreadsheets to track operational data such as mailing dates and call attempts. The Utah team used Epi Info software (developed by CDC) for data entry for mail and telephone surveys. 11 To describe the characteristics of the women in the study, as well as variation in response rates, we assessed data on maternal age (<20, 20-29, 30-39, ≥40), race/ethnicity (non-Hispanic White, non-Hispanic Black, Hispanic, “other” [ie, American Indian/Alaska Native, Asian, Native Hawaiian/Other Pacific Islander, and mixed race/ethnicity]), marital status (married or unmarried), educational attainment (<high school diploma, high school diploma/general educational development, some college, associate’s degree, bachelor’s degree, master’s or doctoral degree), gestational age of the fetus in weeks (20-27, 28-32, 33-36, ≥37), and month in which the loss occurred (June 2018–May 2019). We used Pearson χ2 tests to test for differences between responders and nonresponders, with P < .05 considered significant. The demographic variables used for the analysis were from the fetal death certificate obtained through the Utah Vital Records and Statistics office. We also examined item nonresponse to determine whether women were more likely to skip the most sensitive survey questions or the questions at the end of the survey.
Results
The initial list of fetal death certificates included 288 Utah residents who experienced a stillbirth from June 1, 2018, through May 31, 2019. A total of 167 women completed surveys, for a response rate of 58.0%. Of these, 149 (89.2%) responded by mail and 18 (10.8%) responded by telephone. Ten women (3.5%) refused participation: 3 contacted the SOARS program and asked to be removed, and 7 refused to participate when contacted by telephone. The other 111 women did not respond by mail or by telephone.
The average number of days from first contact to survey completion was 36.5 days. About half of women (n = 84, 50.3%) completed the survey more than 1 month after initial contact. Only 2 women completed the Spanish version of the questionnaire. A higher proportion of women who were non-Hispanic White, were married, and had ≥high school diploma responded to the survey than women who were of other races/ethnicities, were unmarried, and had <high school diploma, respectively (Table). We found no significant differences between responders and nonresponders by maternal age, gestational age of the fetus, or month of delivery.
Characteristics of women eligible a to participate in the Utah SOARS survey, stratified by survey response status, 2018-2019
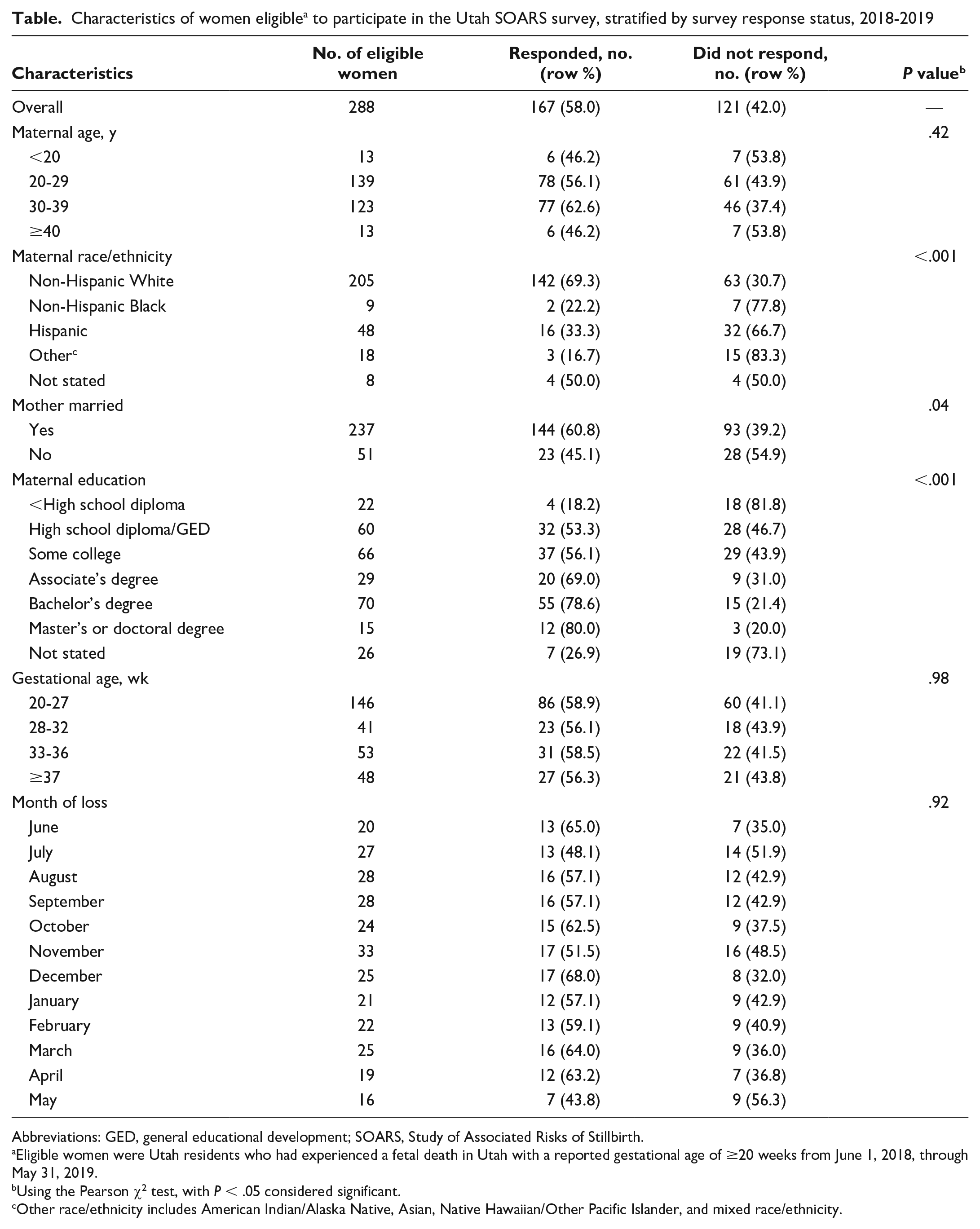
Abbreviations: GED, general educational development; SOARS, Study of Associated Risks of Stillbirth.
Eligible women were Utah residents who had experienced a fetal death in Utah with a reported gestational age of ≥20 weeks from June 1, 2018, through May 31, 2019.
Using the Pearson χ2 test, with P < .05 considered significant.
Other race/ethnicity includes American Indian/Alaska Native, Asian, Native Hawaiian/Other Pacific Islander, and mixed race/ethnicity.
Item nonresponse rates were low (range, 0.6%-5.4%). The questions with the highest item nonresponse rates were the question about income (4.8%) and the questions about tests offered (2.4%) and tests performed (5.4%) during the hospital stay. Nonresponse rates for questions at the end of the survey were not higher than nonresponse rates for questions at the beginning of the survey.
Discussion
In this study, the Utah Department of Health invited all Utah women who experienced a stillbirth from June 1, 2018, through May 31, 2019, identified via fetal death certificates, to participate in SOARS. The SOARS unweighted response rate (58.0%) was higher than the response rate of the previous stillbirth surveillance pilot study in Georgia (40%) 8 and the 2018 Utah PRAMS unweighted response rate (54.0%) (personal communication, Nicole Stone, Utah Department of Health, May 2020).
Item nonresponse was also low, demonstrating that most women who participated in Utah SOARS were willing to respond to the survey questions. The question about income, which typically elicits the highest item nonresponse rate in PRAMS, 7 had a slightly lower item nonresponse rate in SOARS (4.8%) than in Utah PRAMS (5.0%). The most sensitive survey questions had low item nonresponse rates: questions about tests that were offered (2.4%) and tests that were performed (5.4%) during the hospital stay. Item nonresponse at the beginning of the survey did not differ from item nonresponse at the end of the survey, suggesting that survey responders completed the full survey.
As with the Georgia pilot study, our response rate varied significantly by maternal race/ethnicity and marital status. 8 The response rate also varied by maternal educational attainment, which was not assessed in the Georgia pilot study. Because of response differences, the final Utah SOARS analytic data set will include weights to adjust for nonresponse.
Most (89.2%) SOARS participants responded by mail, compared with 68.3% of participants in the 2018 Utah PRAMS survey. A SOARS telephone interviewer anecdotally reported that women often acknowledged receiving the packet in the mail and said they were interested in completing the survey, but not in person or by telephone. The telephone calls may have served as a gentle reminder that prompted the women to complete the survey by mail.
The SOARS team used materials and lessons learned from the pilot study in Georgia to refine the methodology for conducting stillbirth surveillance at the state level. 8 As with the experience in Georgia, our study found that expanding PRAMS methodology to include women who have experienced stillbirths was feasible. 8 Several valuable lessons from the first year of Utah SOARS can be used to inform similar studies in the future. First, including the advisory committee in every step of the development process was critical, as the committee’s expertise was invaluable. The questionnaire development required multiple areas of expertise. The state advisory committee and the PRAMS team from CDC’s Division of Reproductive Health worked closely together to finalize the wording of questions. The advisory committee also helped participate in media coverage about the study in Utah, which may have been beneficial in encouraging participation.
Second, field testing before implementing the survey instrument helped both finalize the questions and inform the research approach. For example, Utah SOARS originally planned for the first contact to be about 4 months after the stillbirth, but the timing was changed to 2 months because field-testing participants said they would be less likely to respond at 4 months than at 2 months. Furthermore, field-testing participants said they would not likely respond to a mailed survey if they were not familiar with the sender. Based on this feedback, Utah SOARS worked with a local grief support group to provide SOARS information when visiting these women after delivery in the hospital. Staff members were careful in their recruitment of women and facilitation of field testing not to inflict additional pain to participants and to minimize the potential for participants to self-blame. Field testing demonstrated that although the subject is painful, participation seemed to provide some level of comfort to women, especially the idea of turning their loss into something that could help others.
Third, translating the questionnaire into Spanish was difficult because of sensitive language used about stillbirths and took longer than anticipated. It was useful that the director of the Utah Office of Health Disparities was a member of the SOARS advisory committee and could provide expertise in question development.
Fourth, the timeline for the study needed to be adjusted. The team had to delay data collection because the team did not allot ample time for study development and the pre-implementation stage. Programming the PRAMS Integrated Data Collection System could not be completed in time for stillbirth data collection, which necessitated the development of a separate data entry system. It took 3 to 4 months to program and test questions in Epi Info. Furthermore, the timing of the follow-up letter was too early. At times, Utah had not received any surveys before the date of the follow-up letter (ie, 10 days after the first questionnaire packet was mailed); when this occurred, mailing of the follow-up letter was pushed back a couple of days.
Finally, Utah staff members felt that bereavement training was essential. The techniques that telephone interviewers learned may have helped improve the response rate for such a sensitive topic. The self-care techniques that all SOARS staff members learned helped them cope with the sadness of the topic of stillbirth in a healthy way and appropriately process their own grief as they interacted with mothers.
A strength of Utah SOARS was the attention that was placed on the needs of women who had experienced a stillbirth. The SOARS team at the Utah Department of Health was sensitive and responsive to the needs of the women they surveyed. At the beginning of the process, the SOARS team included on the SOARS advisory committee women who had experienced a stillbirth. Women who had experienced a stillbirth tested the survey questions to ensure they were sensitive to the grieving process. The team also prioritized bereavement training for telephone interviewers and other staff members involved in SOARS. When women were contacted, the first piece of mail they received was a handwritten sympathy card. The swallow charm incentive necklace was deliberately chosen because the swallow is a symbol of hope and new beginnings. All women contacted were offered both a list of support resources and a free consultation with a maternal–fetal medicine practice, regardless of survey completion. This focus on the needs of women who have experienced a stillbirth may have helped improve the survey response rates and data quality.
Limitations
This study had 2 limitations. First, results might not be generalizable outside Utah because we included in the study only residents who had experienced a stillbirth in Utah. Second, our results depend on the quality of data on the fetal death record, which has previously documented limitations.12 -18 Although a previous study in Salt Lake County, Utah, found high-quality data for selected variables on the fetal death certificate, it highlighted concerns about misreporting of maternal race/ethnicity and gestational age, 19 which were 2 variables of interest in our study. One possible way to address this limitation is to add questions about maternal race/ethnicity and gestational age to the survey instrument for validity purposes.
Conclusion
The Utah SOARS unweighted response rate of 58.0% suggests that a mail- and telephone-based survey can be successful in collecting self-reported information about risk factors for stillbirths not currently included in medical records or fetal death certificates. Results from Utah alone may not be generalizable to the entire United States but can inform the work of jurisdictions interested in establishing their own stillbirth surveillance by adopting the SOARS methodology.
Footnotes
Disclaimer
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention (CDC).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the research was provided by CDC under cooperative agreement #1U01DP006216.
