Abstract
Antiretroviral treatment has greatly improved the survival of people living with diagnosed HIV (PLWDH), but little information is available on the time since diagnosis among them. Using New York City HIV surveillance data, we described the trend in the number of years since diagnosis among PLWDH during 2010-2019 and reported the mean, median, and interquartile range (IQR) of years since diagnosis among PLWDH in New York City in 2019, overall and by gender, race and ethnicity, and transmission risk. The median number of years since diagnosis among PLWDH in New York City increased from 10.5 years (IQR, 6.3-15.6) in 2010 to 16.3 years (IQR, 8.9-22.1) in 2019. By gender, transgender people had the shortest time since diagnosis, with a median of 11.4 years (IQR, 5.6-17.9), compared with men (median = 15.2 years; IQR, 8.1-21.6) and women (median, 18.5 years; IQR, 12.0-23.0). By race and ethnicity, non-Hispanic White people had been living with the diagnosis for the longest time (median = 17.4 years; IQR, 9.5-23.5), and Asian/Pacific Islander people had been living with the diagnosis for the shortest time (median = 10.1 years; IQR, 4.7-17.0). With an expected and continuing increase in the number of years since HIV diagnosis among PLWDH, programs that provide treatment and support services will need to be expanded, updated, and improved.
Keywords
In 1981, the Centers for Disease Control and Prevention (CDC) reported 5 cases of Pneumocystis carinii pneumonia in Los Angeles among previously healthy men who have sex with men (MSM). 1 In 1982, CDC named the illness acquired immunodeficiency syndrome (AIDS). 2 During the AIDS epidemic in the 1980s, approximately half of people diagnosed with AIDS died within 1 year of diagnosis.2,3 Many people with AIDS in the United States had been living with their diagnosis for <1 year, and their primary needs were for treatment and prophylaxis of opportunistic infection and hospice care. 4
In 1983, Barré-Sinoussi et al 5 reported the discovery of a retrovirus, later named human immunodeficiency virus (HIV), as the cause of AIDS. 6 In 1985, the US Food and Drug Administration licensed the first commercial blood test to detect antibody to HIV. 7 With the availability of HIV testing, people were increasingly diagnosed with HIV before developing AIDS. Before the introduction of highly active antiretroviral therapy (HAART), most people living with diagnosed HIV (PLWDH) in the United States had been living with the diagnosis for several days to several years. 8
Since its introduction in 1995-1996, HAART has greatly improved the survival of people with HIV.9-14 PLWDH in the United States have now been living with the diagnosis for several days to several decades, and their needs are broad, including clinical care and services to support medication adherence and address needs related to comorbid medical conditions, housing, substance use, mental health, and aging. 15
Despite these advances, little current information is available on the time since diagnosis among PLWDH. We describe the time since diagnosis among PLWDH in New York City at the end of 2019.
Methods
Data Source
We used data from the New York City HIV Surveillance System reported to the New York City Department of Health and Mental Hygiene by March 31, 2020, for the analysis. 16 The New York City HIV registry contained information on a cumulative total of 252 027 unique individuals (both living and dead) and more than 12 million laboratory test results. The vital status of PLWDH is obtained by quarterly matching against New York City Vital Statistics data and annual matches against the National Death Index and the Social Security Death Master File.
Analysis Population
PLWDH include both in-care and out-of-care patients. In-care is classified as having had ≥1 CD4/viral load test in the calendar year in the HIV registry, 17 but this method cannot be used to identify out-of-care patients because patients without a CD4/viral load test can either be out-of-care patients or patients who have migrated out of the jurisdiction. In our analysis, we included only in-care patients as the analysis population and used a special group of in-care patients who were previously out of care and recently returned for care to represent out-of-care patients, using a previously described statistical weighting method.18,19
Briefly, we treated the New York City HIV laboratory data reporting system as a special annual population-based survey, lasting from January 1 through December 31 every year. We considered patients who had ≥1 CD4/viral load test in the calendar year as participants in the survey, 17 and we assigned each participant a weight equal to the inverse of the probability that a patient had a CD4/viral load test in New York City during the year (ie, had received HIV-related care from a New York City health care provider in that year). We calculated the probability based on the time interval between the last care visit before the year of interest, or date of diagnosis if the patient had no care visits before the year, and the first care visit in the year. If a patient’s interval was ≤1 year, meaning that the patient was in regular care and included in the survey with a probability of 100%, the patient received a weight of 1. If a patient’s interval was >1 year, the patient received a weight equal to the time interval in years with each unit above 1 representing an out-of-care patient. For example, if a patient had his last care visit exactly 3 years before his first care visit in the year, he received a weight of 3, and the patient represented not only himself but also 2 out-of-care patients.
Statistical Analysis
We created 10 analytic datasets of PLWDH, 1 dataset for each calendar year from 2010 through 2019. Using all 10 analytic datasets, we described the trend in time since HIV diagnosis among PLWDH in New York City during 2010-2019. Using the 2019 analytic dataset, we reported the mean (SD), median (interquartile range [IQR]), and range of time since HIV diagnosis through December 31, 2019, among PLWDH in New York City in 2019. We reported time since HIV diagnosis overall and by gender (men, women, and transgender), race and ethnicity (non-Hispanic Black, Hispanic/Latino, non-Hispanic White, Asian/Pacific Islander, American Indian/Alaska Native, and multiracial), age, and transmission risk (MSM, injection drug use [IDU], MSM/IDU, heterosexual, perinatal, and other/unknown). Transmission risk is classified using the CDC definition by selecting the transmission risk most likely to have resulted in HIV transmission. One exception is MSM/IDU; this group comprises a separate transmission risk category. 20 The analysis used surveillance data and was a public health practice activity, not human subjects research.
Results
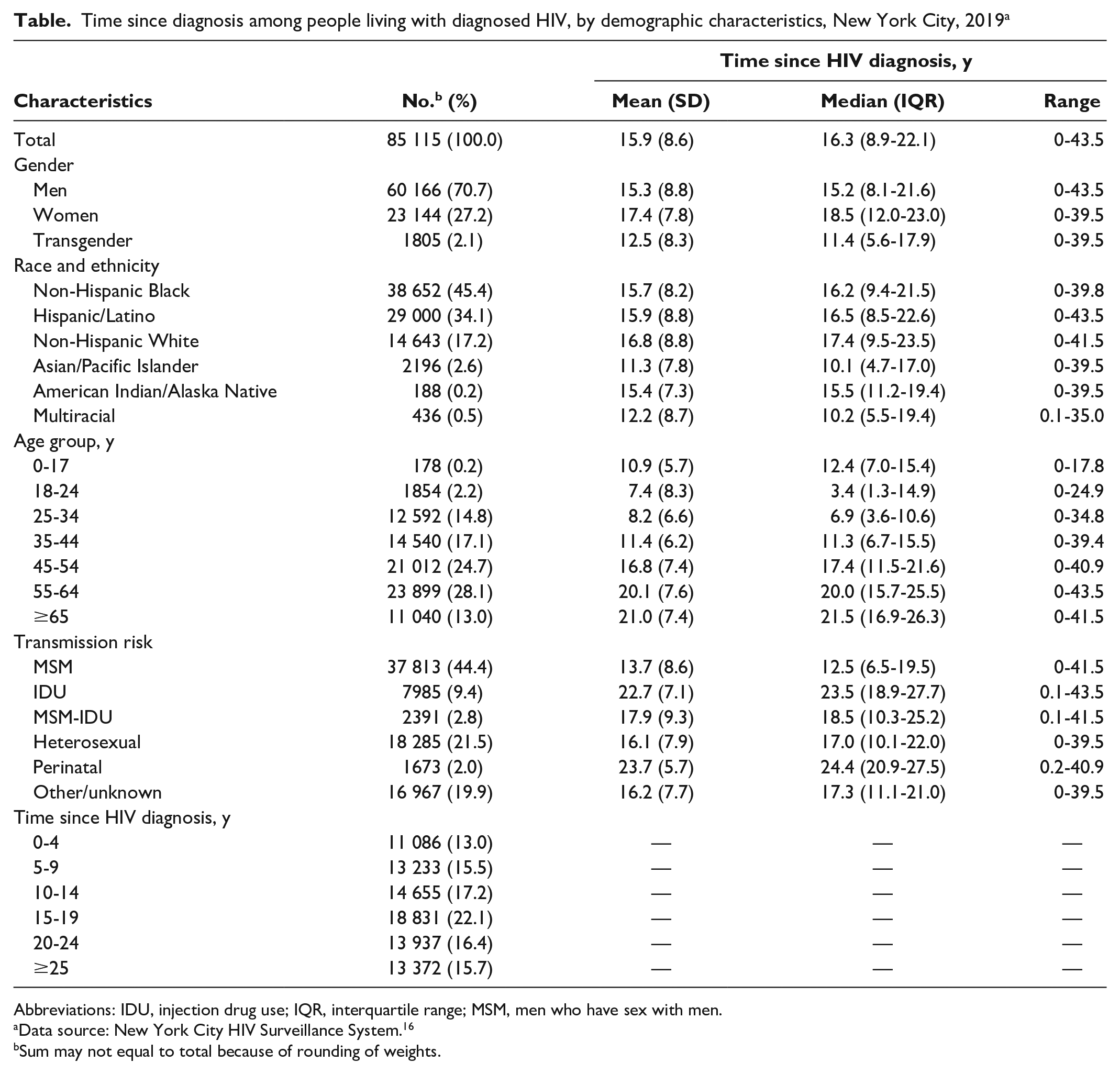
At the end of 2019, a total of 85 115 PLWDH were living in New York City. Among them, 70.7% self-identified as men, 27.2% as women, and 2.1% as transgender; 79.5% were either non-Hispanic Black (45.4%) or Hispanic/Latino (34.1%); and 44.4% were MSM (Table).
Time since diagnosis among people living with diagnosed HIV, by demographic characteristics, New York City, 2019 a
Abbreviations: IDU, injection drug use; IQR, interquartile range; MSM, men who have sex with men.
Data source: New York City HIV Surveillance System. 16
Sum may not equal to total because of rounding of weights.
By gender, transgender people had the shortest time since HIV diagnosis, with a median of 11.4 years (IQR, 5.6-17.9), compared with men (median = 15.2 years; IQR, 8.1-21.6) and women (median = 18.5 years; IQR, 12.0-23.0) (Table). By race and ethnicity, non-Hispanic White people had been living with the diagnosis for the longest time (median = 17.4 years; IQR, 9.5-23.5), and Asian/Pacific Islander people had been living with the diagnosis for the shortest time (median = 10.1 years; IQR, 4.7-17.0). By transmission risk, people with a history of IDU (median = 23.5 years; IQR, 18.9-27.7) and people with perinatal transmission risk (median = 24.4 years; IQR, 20.9-27.5) had the longest time since HIV diagnosis, and MSM had the shortest time since HIV diagnosis (median = 12.5 years; IQR, 6.5-19.5).
The median time since HIV diagnosis among PLWDH in New York City increased from 10.5 years (IQR, 6.3-15.6) in 2010 to 16.3 years (IQR, 8.9-22.1) in 2019 (Figure).
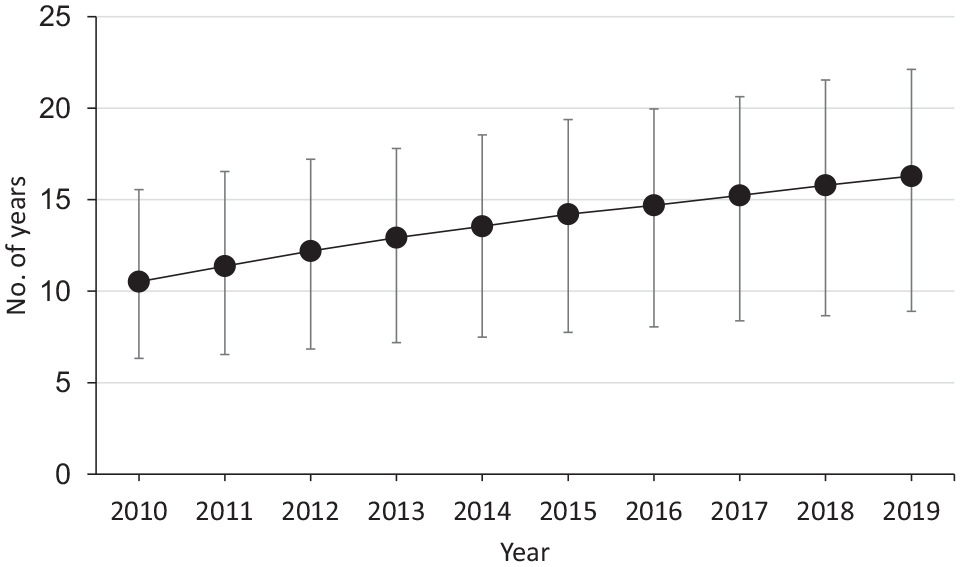
Median time since diagnosis among people living with diagnosed HIV in New York City, 2010-2019. Error bars indicate interquartile range.
Discussion
Antiretroviral treatment (ART) substantially reduces HIV-related mortality.10,21 One study that examined data from 88 504 PLWDH who started ART during 2008-2010 reported that a 20-year-old person with a CD4 count >350 cells/mm3 1 year after starting ART was expected to live to age 78, which was close to the life expectancy of the general US population. 22 In any jurisdiction, there are PLWDH who have been diagnosed for days, months, years, or decades, and they face different challenges and have different needs.11,23 For example, people who have been diagnosed with HIV for days or months need psychological support dealing with HIV diagnosis and initiation of treatment; people who have been diagnosed for years need ongoing support for treatment adherence; and people who have been diagnosed for decades need support dealing with their comorbidities and long-term side effects of HIV treatment. To more efficiently allocate resources, it is necessary to know the distribution of years since HIV diagnosis among PLWDH (ie, the proportions of PLWDH who have been diagnosed for <5, 5-9, 10-14, 15-19, 20-24, and ≥25 years).
The distribution of years since HIV diagnosis in a PLWDH population is driven by 3 factors: (1) HIV incidence among HIV-negative people, (2) time from HIV infection to diagnosis among those infected, and (3) time from HIV diagnosis to death among those diagnosed. Lower HIV incidence among HIV-negative people, a longer time from HIV infection to diagnosis, or a longer time from HIV diagnosis to death can lead to a higher proportion of people with a longer time since HIV diagnosis in a PLWDH population.16,24-26 In our analysis, we found that half of PLWDH in New York City in 2019 had been living with the diagnosis >15 years. Years living with HIV diagnosis varied by gender, race and ethnicity, age, and transmission risk. By gender, a larger proportion of women than men had been living with an HIV diagnosis for >15 years, because HIV-negative women had lower HIV incidence than HIV-negative men, and HIV-positive women had a similar time from HIV infection to diagnosis but a longer time from HIV diagnosis to death.16,24-26 The short time living with HIV among transgender people was likely the result of their high HIV incidence 27 but was also attributed in part to changes in our classification of gender; that is, not all people’s transgender identity was known to surveillance, and people who were diagnosed before New York City began to systematically collect information on gender were more likely to be misclassified as nontransgender. 28 By transmission risk, people who inject drugs had the longest time living with an HIV diagnosis because of the continuing decrease in HIV incidence in this population in New York City. 29
The increasing time living with an HIV diagnosis among PLWDH in New York City reflects the success of treatment that prolongs their lives21,30 and of prevention that has reduced HIV incidence.24,25 With such a large proportion (54.2%) of people having lived with an HIV diagnosis for >15 years, and with this proportion expected to continue to increase, we will need to allocate more resources to serve this enlarging population. We will also need to expand, update, and improve programs that provide treatment and support services, including ART adherence support, sexual and reproductive health services, and services to address non–HIV-related medical conditions. To better serve this population, additional research, both qualitative and quantitative, will be needed to detail the psychological, clinical, and social needs of PLWDH, by time since HIV diagnosis and other characteristics (eg, gender, race and ethnicity, neighborhood).
Footnotes
Acknowledgements
The authors thank Kent Sepkowitz, Anisha Gandhi, Neil Vora, and Hannah Helmy for their review and comments on this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
