Abstract
Objectives
Racial and socioeconomic disparities in the incidence of Legionnaires’ disease have been documented for the past 2 decades; however, the social determinants of health (SDH) that contribute to these disparities are not well studied. The objective of this narrative review was to characterize SDH to inform efforts to reduce disparities in the incidence of Legionnaires’ disease.
Methods
We conducted a narrative review of articles published from January 1979 through October 2019 that focused on disparities in the incidence of Legionnaires’ disease and pneumonia (inclusive of bacterial pneumonia and/or community-acquired pneumonia) among adults and children (excluding articles that were limited to people aged <18 years). We identified 220 articles, of which 19 met our criteria: original research, published in English, and examined Legionnaires’ disease or pneumonia, health disparities, and SDH. We organized findings using the Healthy People 2030 SDH domains: economic stability, education access and quality, social and community context, health care access and quality, and neighborhood and built environment.
Results
Of the 19 articles reviewed, multiple articles examined disparities in incidence of Legionnaires’ disease and pneumonia related to economic stability/income (n = 13) and comorbidities (n = 10), and fewer articles incorporated SDH variables related to education (n = 3), social support (none), health care access (n = 1), and neighborhood and built environment (n = 6) in their analyses.
Conclusions
Neighborhood and built-environment factors such as housing, drinking water infrastructure, and pollutant exposures represent critical partnership and research opportunities. More research that incorporates SDH and multilevel, cross-sector interventions is needed to address disparities in Legionnaires’ disease incidence.
Keywords
Legionnaires’ disease is a severe respiratory illness resulting from breathing water droplets or aspirating water containing Legionella bacteria. Legionnaires’ disease is a public health challenge, with an estimated annual hospitalization cost of $433 million in the United States. 1 In 2018, 9933 Legionnaires’ disease cases were reported to the Centers for Disease Control and Prevention (CDC) National Notifiable Diseases Surveillance System (NNDSS), representing a nearly 900% increase in Legionnaires’ disease incidence in the United States since 2000. 2 However, Legionnaires’ disease is substantially underdiagnosed; the true number of cases has been estimated to range from 52 000 to 70 000 annually in the United States. 2,3 People at highest risk for Legionnaires’ disease include older adults, males, and people who smoke or who have immunocompromising conditions. National and state surveillance systems have found racial disparities in Legionnaires’ disease incidence. 4 -6
NNDSS data from 2014-2018 demonstrate racial disparities; in 2018, Legionnaires’ disease incidence was almost twice as high among Black people compared with White people (Figure 1). 5 Residents who were non-Hispanic Black and lived in high-poverty census tracts had the highest rates of Legionnaires’ disease in New York City. 6,7 Incidence of and hospitalization with other types of pneumonia among Black people and among people living in the most impoverished census tracts are also disproportionately high. 8 -12 Risk factors for Legionnaires’ disease are often described from an epidemiologic perspective; however, the reasons for racial and socioeconomic disparities in the incidence of Legionnaires’ disease and targeted interventions to address these gaps have not been as widely explored. 13,14 A literature-based analysis grounded in the social determinants of health (SDH) is needed to identify root causes of Legionnaires’ disease inequities and to inform future studies and comprehensive interventions.
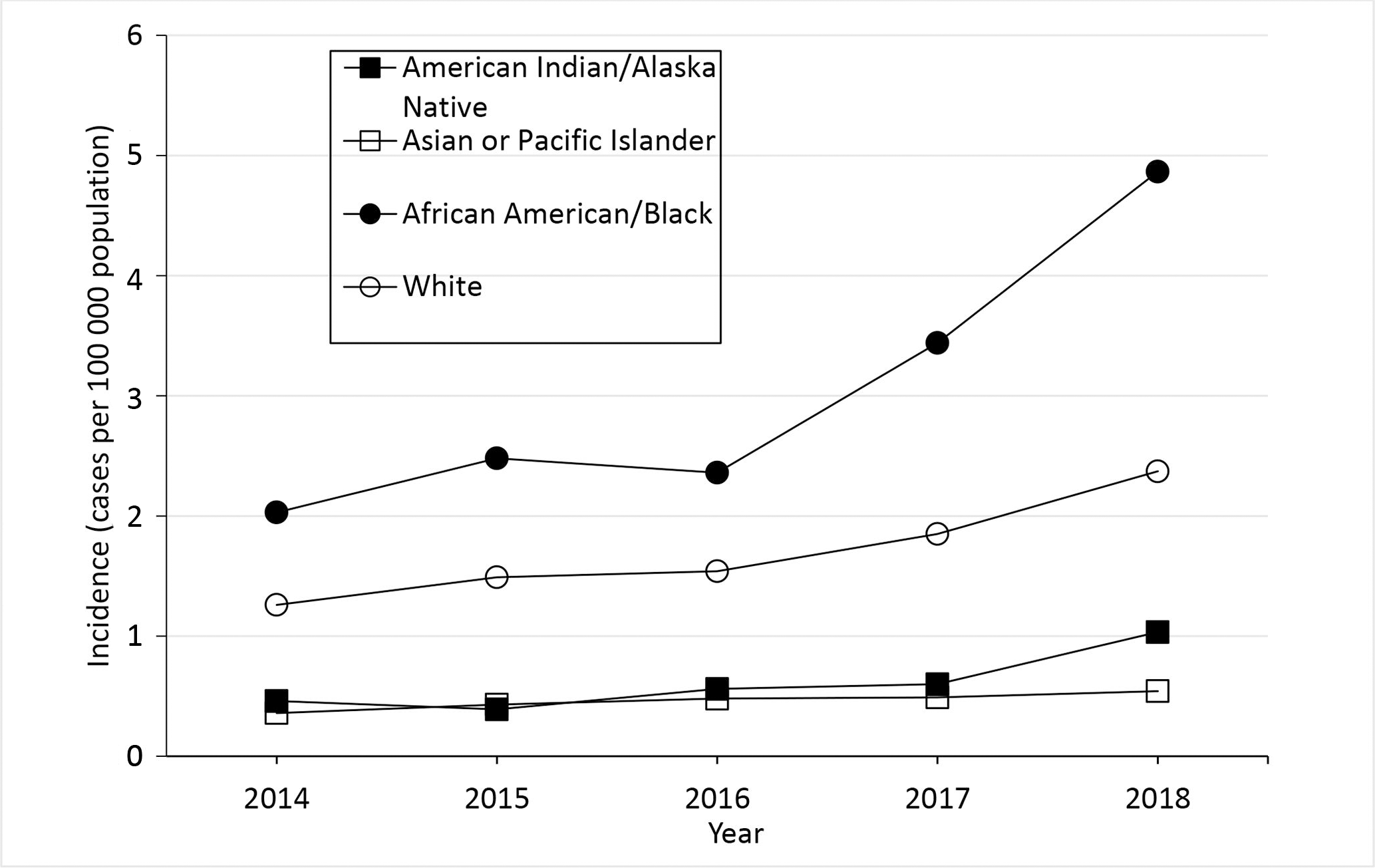
Crude incidence of reported confirmed cases of Legionnaires’ disease, by year and race, United States, District of Columbia, and New York City, 2014-2018. The crude incidence of cases per 100 000 population is the number of confirmed Legionnaires’ disease cases reported that year divided by the postcensal population estimate for that year x 100 000. Cases of disease due to Legionella are reported to the National Notifiable Diseases Surveillance System (NNDSS) as legionellosis, which includes Legionnaires’ disease, Pontiac fever, and disease due to Legionella infection at extrapulmonary sites, but is referred to as Legionnaires’ disease in this figure (because almost all legionellosis cases reported in the United States are due to Legionnaires’ disease). The year is when the case was reported to the Centers for Disease Control and Prevention. Data sources: 2014-2018 data are from NNDSS. 2
Social determinants of health are the conditions in which people are born, grow, live, work, worship, and age that can affect health outcomes and quality of life. 15,16 Healthy People 2030 organizes SDH into 5 domains: economic stability, education access and quality, social and community context, health care access and quality, and neighborhood and built environment.
A sentinel goal of the Healthy People program is “to achieve health equity, eliminate disparities, and improve the health of all groups.” 17 The objective of this topical review was to narratively characterize factors that may contribute to disparities in incidence of Legionnaires’ disease to inform efforts and research needed to prevent Legionnaires’ disease cases among populations disproportionately affected by Legionnaires’ disease.
Methods
We conducted a review of published literature from the PubMed and Scopus databases of journal articles related to health disparities associated with Legionnaires’ disease, bacterial pneumonia, and/or community-acquired pneumonia. Although Legionnaires’ disease differs from pneumonia of other etiologies by route of transmission, many underlying risk factors (eg, older age, chronic lung disease, smoking) for illness among adults are similar. 6 We expanded our review to include articles describing health disparities associated with other types of community-acquired or bacterial pneumonia to increase the breadth of studies available for analysis. Our search included articles published from January 1979 through October 2019. The Medical Subject Headings (MeSH) search terms included “Legionnaires’ disease,” “legionellosis” (which includes less common presentations of illness caused by Legionella bacteria, such as Pontiac fever and extrapulmonary infection), “racial and ethnic minority health,” “health care disparities,” “health disparities,” and “social determinants of health.” We used Endnote X9 software (Clarivate Analytics) to compile the final article records, and we identified and removed 4 duplicate articles.
We excluded articles upon review of the title, abstract, and full-text article based on the following criteria: non-original research; non-English publication; and research that did not examine Legionnaires’ disease or pneumonia (eg, bacterial pneumonia), health disparities, and SDH. The initial search resulted in 220 articles. After applying exclusion criteria to the abstracts, we reviewed 98 full-text articles. Of those 98 articles, we excluded 79 articles because they were unrelated to SDH or health disparities and 2 articles because the study population was limited to only people aged <18 years. Nineteen articles met the inclusion criteria and were included in this review (Figure 2, Table). 6 -11,18 -30 The most common reasons for exclusion of full-text articles were that the articles did not explicitly examine disparities in incidence of Legionnaires’ disease or pneumonia or did not characterize aspects of SDH.
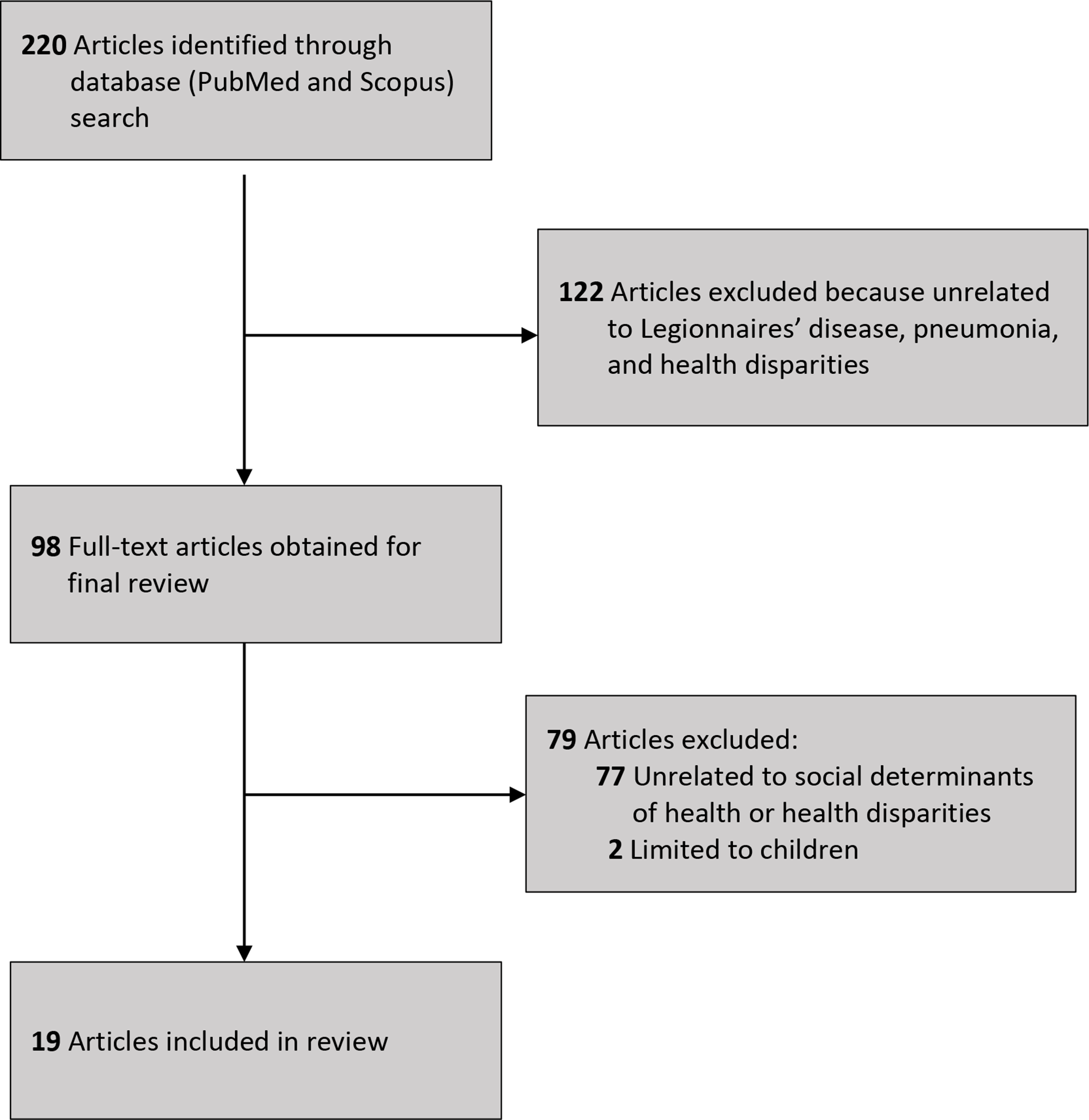
Flow diagram of selection of articles on racial disparities in Legionnaires’ disease incidence and social determinants of health published from January 1979 through October 2019.
Articles on health disparities in Legionnaires’ disease, by study period (N = 19)
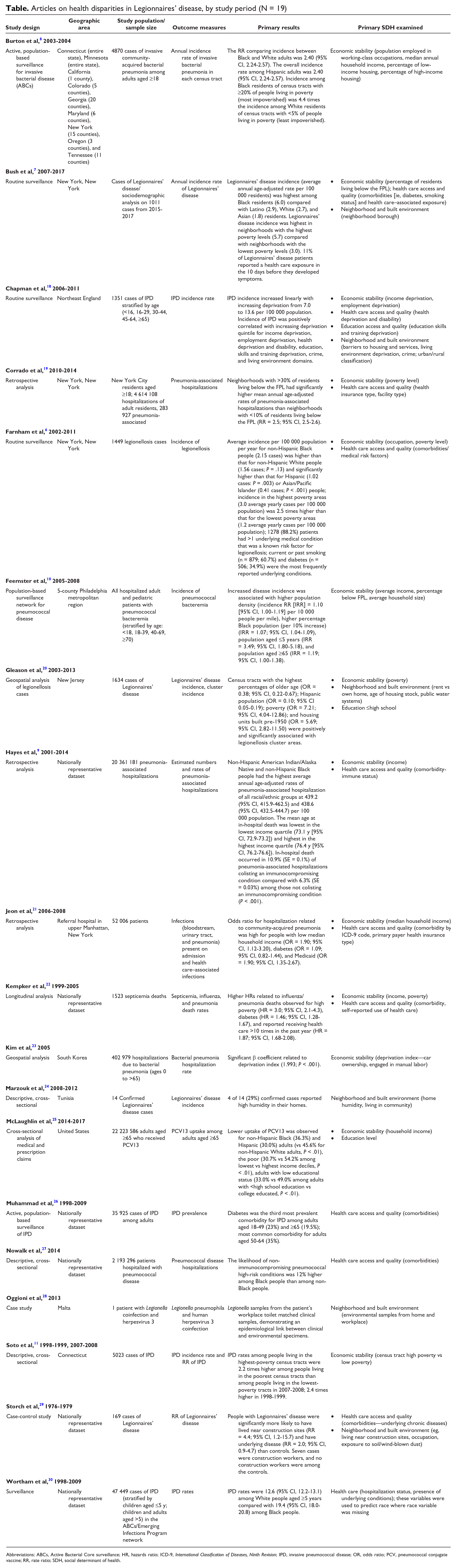
Abbreviations: ABCs, Active Bacterial Core surveillance; HR, hazards ratio; ICD-9, International Classification of Diseases, Ninth Revision; IPD, invasive pneumococcal disease; OR, odds ratio; PCV, pneumococcal conjugate vaccine; RR, rate ratio; SDH, social determinant of health.
Based on the results of eligible articles, we aimed to categorize the results into the 5 SDH domains as they related to disparities in incidence of Legionnaires’ disease or pneumonia: economic stability (income and working conditions), education access and quality (education level), social and community context (social capital, stress, discrimination), health care access and quality (comorbidities and health care access), and neighborhood and built environment (housing characteristics, drinking water).
Results
Economic Stability
Several multivariable models indicated that areas with the highest poverty levels have the highest incidence of Legionnaires’ disease. Among community-acquired cases in New York City during 2002-2011, Legionnaires’ disease incidence in the most impoverished areas was 2.5 times higher than in the least impoverished areas. 6 Surveillance data from 2015-2017 in New York City indicated that Legionnaires’ disease incidence in the highest-poverty census tracts was almost double the Legionnaires’ disease incidence in lower-poverty census tracts. 7 This trend was also reflected in the 2015 South Bronx Legionnaires’ disease outbreak, where most Legionnaires’ disease cases were found in impoverished areas. Percentage of the population with income below the federal poverty level was also strongly associated with a census tract being designated as a high-risk legionellosis cluster area in New Jersey (OR = 7.21; 95% CI, 4.04-12.86). 20
Publications on pneumonia also identified population income as being associated with disease incidence. Impoverished areas had the highest incidence of bacteremic pneumonia, invasive pneumococcal disease, and pneumonia deaths. 8,10,11,18,22 Incidence of pneumonia-associated hospitalizations was higher among people living in areas with lower household incomes than among people living in areas with higher household incomes. 9,19,23 In addition, lower accessibility and use of pneumococcal vaccines were associated with people experiencing poverty and racial/ethnic minority communities. 23
An examination of the role of occupation in 1279 cases of community-acquired legionellosis in New York City found most cases were among people employed in hazardous or service industries (ie, transportation, repair, protective services, cleaning, or construction). 6 In 1979, a case-control study of 100 sporadic Legionnaires’ disease cases across the United States also identified a significant occupational difference between cases and controls; 7 Legionnaires’ disease cases were construction workers and no controls were construction workers. 29
Education Access and Quality
Lower education level was associated with a higher census-tract incidence of Legionnaires’ disease in New Jersey, increased incidence of pneumococcal disease in North England, and decreased uptake of pneumococcal vaccine among adults in the United States. 18,20,25
Social and Community Context
No studies measured social and community context factors such as stress, discrimination, or social capital in the context of Legionnaires’ disease.
Health Care Access and Quality
Several studies examined the presence of comorbid illnesses and incidence of Legionnaires’ disease. In our review, surveillance data showed that patients with diabetes had a higher incidence of Legionnaires’ disease and invasive pneumococcal disease than people without diabetes. 6,7,26 In addition, immune-compromising comorbidities were associated with community-acquired pneumonia and pneumonia-related hospitalizations. 9,21,26,27 Two studies that examined invasive pneumococcal disease acknowledged that comorbidities could be a factor to disparities in incidence but did not extensively explore comorbidities. 10,30
Although several articles acknowledged lack of health insurance and medical care as factors contributing to disparities in adverse respiratory-related outcomes, most articles did not explore health care access or type of health insurance in their analyses. One study found that Medicare comprised the largest percentage of primary payer for pneumonia-associated hospitalizations in New York City, followed by Medicaid. 19
Neighborhood and Built Environment
Although residential housing can be a source of exposure to Legionella bacteria, 31,32 few articles examined patient housing characteristics. Gleason et al 20 identified housing characteristics such as percentage of vacant homes, rented homes, and older homes as potential factors associated with legionellosis incidence. Among census tracts in New Jersey, a higher percentage of older (pre-1950) housing was independently associated with increased odds of being a high-risk legionellosis cluster area. 20 Neighborhood density and household crowding were briefly discussed as potential risk factors or mediators for certain types of bacterial pneumonia, but these factors were not directly examined in the articles included in our review. 10,33
Certain housing and facility conditions may create environments conducive to Legionella growth. For example, an examination of Legionella samples in a Legionnaires’ disease patient’s home, workplace, and hotel found that the workplace was the most likely source of exposure. 28 High humidity in the home was listed as a risk factor for Legionnaires’ disease in Tunisia. 24 In England, incidence of invasive pneumococcal disease was correlated with impoverished living conditions. 18 In addition to housing quality, building features and proximity to certain industries were identified as potential risk factors for Legionnaires’ disease. Farnham et al 6 found proximity to cooling towers to be an environmental risk factor for Legionnaires’ disease outbreaks and suggested that continued maintenance of the towers and water systems could be warranted in poor neighborhoods. One study determined that patients with Legionnaires’ disease were more likely than patients without Legionnaires’ disease to reside near construction sites. 29
Only 1 article in our review incorporated drinking water and water infrastructure metrics in its analysis. This study found that drinking water source did not have a significant relationship with legionellosis incidence, although housing age and infrastructure were positively associated risk factors. 20
Discussion
Racial and socioeconomic disparities in Legionnaires’ disease incidence in the United States have been identified in the past 20 years. 5 -7 In addition, the incidence of Legionnaires’ disease has increased since 2000. 2 Numerous factors likely contribute to the increasing incidence of Legionnaires’ disease, including growth in the population aged ≥65; more people with underlying conditions or immunosuppressing medications; proliferation of Legionella in water systems because of warm water temperatures, aging infrastructure, and water conservation measures; and heightened awareness of Legionnaires’ disease among clinicians and public health staff members leading to increased testing and surveillance capacity. Reasons for underdiagnosis of Legionnaires’ disease are likely multifactorial as well. Because Legionnaires’ disease is not clinically distinguishable from pneumonia because of other etiologies, clinicians must order specific testing to confirm the diagnosis. Furthermore, selection of first-line antibiotics to treat community-acquired pneumonia often includes coverage for Legionella. Differences in diagnosis of Legionnaires’ disease by race/ethnicity have not been examined. Studies of racial differences in receipt of guideline-concordant antibiotics have not demonstrated disparities between Black and White patients. 34,35 Through examination of Legionnaires’ disease- and pneumonia-related articles, we identified dimensions of SDH that could contribute to understanding disparities in incidence. Many of these SDH disproportionately affect Black people and may explain some racial disparities. For example, 1 study indicated that Black people have had a higher percentage of household poverty and experienced a lower prevalence of employment, education, and homeownership than non-Hispanic White people. 36
Income was the most common SDH identified in our review, and Black people had the lowest median annual household income compared with other racial groups. 37 Lower income was associated with a higher incidence of Legionnaires’ disease and increased incidence and hospitalization for other respiratory diseases. However, a limitation to this review was that data on Legionnaires’ disease and income were from mostly urban areas and may not reflect data from rural areas. Poverty is a risk factor for increased morbidity and mortality and is correlated with SDH such as lack of medical care access, educational attainment, and inadequate housing conditions that put people at risk for adverse respiratory outcomes. 38 This finding underscores the need for targeted intervention strategies that would increase economic mobility, improve medical access, and further establish the importance of reporting census-tract poverty levels and other socioeconomic indicators in future Legionnaires’ disease research. Our review found that some occupations are associated with increased Legionnaires’ disease risk. Certain occupations have an overrepresentation of racial/ethnic minority groups that experience occupational respiratory exposures and illnesses. Inclusion of job characteristics and working conditions in Legionnaires’ disease research can identify potential occupational and environmental hazards and elucidate how employment status and occupational categories affect Legionnaires’ disease health disparities. 39
Although only a few studies in our review incorporated data on education levels in their analyses, lower levels of education are associated with increased incidence in Legionnaires’ disease, pneumonia, and other respiratory conditions. 20,40 Higher educational attainment is linked to improved employment opportunities and working conditions, higher income, and increased social support. Some racial/ethnic minority populations may have lower levels of educational attainment because of factors such as residential segregation, disinvestment in housing, and underresourced schools. 41,42 Multilevel interventions at the individual, interpersonal, community, and societal levels are most effective to address these educational inequalities. 43
The absence of studies examining community and social contextual factors in our review highlights the need for research to examine the role of these factors in attenuating Legionnaires’ disease health disparities. Two studies, one of which is not included in our review, acknowledge that stress caused by poverty could enhance mechanisms that alter immune function and susceptibility to respiratory illnesses, and these studies did not include variables to measure stress, social support, or social cohesion. 8,44 Social support may protect patients who have chronic obstructive pulmonary disease 44 and other conditions; therefore, exploratory studies could consider social support in future analysis.
Comorbidities of Legionnaires’ disease and other pneumonia-related diseases are frequently described in the literature. 45 -48 Examining the relationship between comorbid illnesses and Legionnaires’ disease may clarify the effect of existing conditions on various racial/ethnic groups. Comorbidity studies showed that Black people have higher rates of influenza and pneumonia morbidity and mortality than White people. Black people are also more likely than White people to have underlying health conditions and experience complications from these infections. 49 To address disparities in the incidence of influenza and pneumococcal disease among Black people, physicians should be proactive in treating comorbid conditions, and more interventions to provide vaccination could be made available. 27,49 In addition, health care access, health care quality, and type of health insurance were not frequently explored in the articles included in our review.
The causes of disparities in health care access and use are multifactorial. Racial/ethnic minority groups have historically encountered barriers to health care access and use. 50 Several studies indicated that the largest contributors to disparities in health care access among racial/ethnic minority groups are socioeconomic disadvantage, lower levels of education, higher rates of occupational hazards, and a lack of health insurance. 51 Exploration of effective interventions to address these disparities in health care access can help to identify potential solutions. For example, community health workers and local community organizations may be able to address challenges associated with health care access and provide additional support in managing comorbidities and respiratory conditions. 52
Neighborhood and built environment factors that may affect Legionnaires’ disease health disparities include housing, drinking water infrastructure, and pollutant exposures. Housing and neighborhood variables include housing vacancies, property ownership status, housing age, housing quality, and proximity to sources of air pollutants. For example, non-White people and poor communities have disproportionate levels of pollution from particulate matter–emitting facilities, which can lead to increased air pollution exposure and exacerbate respiratory conditions. 53 -55
Future studies could examine variables related to poor building infrastructure, building code violations, and air quality. For example, redlining has contributed to residential segregation and resulted in concentrated poverty centers, low-quality housing, decreased access to economic and educational opportunities, and high land-surface temperatures. 56,57 As solutions to address health disparities in neighborhoods and the built environment are explored, the support of community health workers in partnership with environmental health practitioners should also be considered. Their unique ability to identify community needs and provide support through shared experiences, language, and culture has proved to be successful. Community health workers are instrumental in closing gaps in health disparities by assessing community health risks and advocating for housing improvements.
The contribution of household water exposures and community drinking water infrastructure to sporadic, community-acquired Legionnaires’ disease is understudied. Older homes may have impaired premise-plumbing systems (ie, water piping systems in a building that connect the building to the water main line) that can affect water stagnation and quality. A Legionnaires’ disease case investigation in 2018 identified a Wisconsin home built in the 1910s as the most probable source of infection. 31 In addition, older homes may be in communities with overall aging water infrastructure that could contribute to decreased water quality. 20 Previous studies have indicated that potable water sources in the home could be an exposure source, and interventions such as point-of-use filters for homes with immunocompromised people have been recommended. 32,58 Building partnerships with healthy home initiatives to increase awareness about Legionnaires’ disease risks, water system flushing protocols, and drinking water quality may be beneficial.
Inadequate drinking water quality, access, and infrastructure are critical factors that have been posited to negatively affect environmental health disparities. 59 Water age, water pipe material, water quality, and drinking water source may influence growth of Legionella and other opportunistic pathogens. 39,60 Racial/ethnic minority groups and low-income populations are more likely than non-Hispanic White and high-income populations to be exposed to contaminated drinking water and have poor access to safe drinking water. Including drinking water quality, access, infrastructure information, and environmental justice indicators in research will help to identify possible associations among drinking water violations, socioeconomic factors, and Legionnaires’ disease incidence.
Public Health Implications
The disproportionately higher incidence of Legionnaires’ disease among Black people and people of low socioeconomic status warrants public health action. Our review revealed that income and comorbidities are commonly studied aspects of SDH, and SDH such as job characteristics, education, social support, stress, health care access, health insurance type, and built environment are areas for future study. Knowledge gaps related to built-environment factors (eg, housing, drinking water infrastructure, pollutant exposures) suggest that more research can elucidate the role of cumulative environmental risks in Legionnaires’ disease health disparities. We posit that an SDH perspective can reveal why some communities are more likely than other communities to have vacant buildings with poorly maintained water systems, older housing with deteriorating plumbing, and declining drinking water infrastructure, which could lead to conditions conducive for Legionella growth in pipes.
Future research and programs should incorporate SDH data sources with geocoded addresses from Legionnaires’ disease surveillance data. 61,62 Data sources such as the CDC Environmental Health Tracking Network, CDC/ATSDR Social Vulnerability Index, Robert Wood Johnson Foundation County Health Rankings, and other indices may provide links to environmental and socioeconomic variables that are often not addressed in Legionnaires’ disease research. 63 -65 The findings from our review illustrate the need for increased awareness about the SDH related to Legionnaires’ disease. Outreach to organizations that have an interest in addressing poor housing quality, comorbidities, air pollution, and conditions that disproportionately affect low-income and racial/ethnic minority populations should be prioritized.
In our review, we explored the disproportionate incidence of Legionnaires’ disease and pneumonia among Black people and low-income populations. A present concern is the COVID-19 pandemic and existing comorbidities associated with increased hospitalizations and a higher mortality rate in historically disenfranchised and racial/ethnic minority communities. 66,67 The COVID-19 pandemic is exacerbating the adverse health consequences of racial and socioeconomic disparities in health and health care. 68 Our research reflects a commonality of respiratory conditions and adverse outcomes as a result of longstanding health inequities.
Footnotes
Acknowledgments
The authors thank Jamila Eatman and Na’Taki Osborne Jelks, PhD, MPH, from Spelman College, and Jeffrey Hall, PhD, MSPH, from the Centers for Disease Control and Prevention (CDC) Office of Minority Health and Health Equity for initial conceptual ideas and discussions.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
