Abstract
Objectives
Routine surveillance for streptococcal toxic shock syndrome (STSS), a severe manifestation of invasive group A
Methods
Active Bacterial Core surveillance (ABCs) conducts active population-based surveillance for invasive GAS disease in selected US counties in 10 states. We categorized invasive GAS cases with a diagnosis of STSS made by a physician as STSS–physician and cases that met the Council of State and Territorial Epidemiologists (CSTE) clinical criteria for STSS based on data in the medical record as STSS–CSTE. We evaluated agreement between the 2 methods for identifying STSS and compared the estimated national incidence of STSS when applying proportions of STSS–CSTE and STSS–physician among invasive GAS cases from this study with national invasive GAS estimates for 2017.
Results
During 2014-2017, of 7572 invasive GAS cases in ABCs, we identified 1094 (14.4%) as STSS–CSTE and 203 (2.7%) as STSS–physician, a 5.3-fold difference. Of 1094 STSS–CSTE cases, we identified only 132 (12.1%) as STSS–physician cases. Agreement between the 2 methods for identifying STSS was low (κ = 0.17; 95% CI, 0.14-0.19). Using ABCs data, we estimated 591 cases of STSS–physician and 3618 cases of STSS–CSTE occurred nationally in 2017.
Conclusions
We found a large difference in estimates of incidence of STSS when applying different surveillance methods and definitions. These results should help with better use of currently available surveillance data to estimate the incidence of STSS and to evaluate disease prevention efforts, in addition to guiding future surveillance efforts for STSS.
Invasive group A
Guidelines published in 1993 classify GAS infections as STSS on the basis of the presence of hypotensive shock and clinical criteria indicative of multiorgan failure. 8 The National Notifiable Diseases Surveillance System (NNDSS) follows a public health surveillance case definition from the Council of State and Territorial Epidemiologists (CSTE) in 2010 that is based on these 1993 guidelines. 9 Because STSS is so often fatal, recognition and appropriate diagnosis are critical for instituting treatment, such as clindamycin and, in some instances, intravenous immunoglobulins. 10
Consistent application of clinical criteria for defining STSS may help avoid misclassification or underenrollment of cases in studies evaluating therapeutic options for STSS or exploring pathogenesis of the disease. Accurate identification of STSS cases is also needed for epidemiological surveillance used to generate estimates of disease incidence and to inform studies on the cost-effectiveness of potential interventions, including vaccines. However, the frequency with which clinicians use the CSTE case definition to categorize invasive GAS cases as STSS is unknown and likely low. 5,11 It is possible that clinicians frequently label cases of STSS as septic shock because of an overlap in presenting features such as hypotension with multiple organ failure.
We compared the number and characteristics of invasive GAS cases captured in a large surveillance study during 2014-2017 identified as STSS or septic shock on the basis of physician diagnosis and those that met the CSTE–STSS case definition on the basis of clinical details obtained from medical records. We quantified the misclassification of STSS and discuss potential clinical and epidemiologic implications.
Methods
Surveillance Methods and Case Definitions
Active Bacterial Core surveillance (ABCs) conducts active population- and laboratory-based surveillance for invasive GAS disease in selected counties in 10 states (California, Colorado, Connecticut, Georgia, Maryland, Minnesota, New Mexico, New York, Oregon, and Tennessee) covering a population of about 34 million in 2017. 1 From 2014 to 2017, ABCs conducted surveillance for invasive GAS disease consistently in the 3-county Bay Area in California, 5-county Denver area in Colorado, 20-county Atlanta area in Georgia, 6-county Baltimore area in Maryland, 7-county Rochester area and 8-county Albany area in New York, 3-county Portland area in Oregon, 20 counties in Tennessee, and statewide in Connecticut, Minnesota, and New Mexico. ABCs methods for case ascertainment and data collection are detailed elsewhere. 12 Briefly, ABCs staff members work with all clinical microbiology laboratories in the surveillance catchment area to identify cases. An invasive GAS case is defined as illness with isolation of GAS from a normally sterile site (eg, blood or cerebrospinal fluid) or from a wound specimen accompanied by necrotizing fasciitis or STSS, occurring in a surveillance area resident. For all cases, ABCs staff members review medical records to complete a standardized case report form that captures demographic and clinical data, including diagnoses of clinical syndromes such as septic shock and STSS. Case patients can have more than 1 syndrome identified. We categorized cases for which the medical record mentioned “STSS” or “toxic shock” as STSS–physician.
Beginning in 2014, surveillance staff members completed a supplemental form for each case of invasive GAS, capturing data from the medical record on clinical criteria needed to categorize cases as STSS as per the CSTE case definition. 8,9 Staff members examined the medical records for clinical findings or laboratory information that was documented or collected within 2 days of admission or collection of the culture that grew GAS. Hypotension was defined as a systolic blood pressure ≤90 mm Hg for adults. In children, hypotension was defined on the basis of their age using Pediatric Advanced Life Support guidelines. 13 For applying the CSTE case definition, clinical criteria were defined as follows on medical record review:
Renal impairment: creatinine levels ≥2 mg/dL for adults or ≥2 times the upper limit of normal for age. In patients with preexisting renal disease, renal impairment was defined as a greater than 2-fold increase over the baseline level of creatinine. 14
Coagulopathy: a platelet count ≤100 000/mm3 or disseminated intravascular coagulation listed in the medical record.
Liver involvement: alanine aminotransferase, aspartate aminotransferase, or total bilirubin levels ≥2 times the upper limit of normal for the patient’s age. In patients with preexisting liver disease, a more than 2-fold increase over the baseline level was considered as liver involvement. 14
Adult respiratory distress syndrome (ARDS): ARDS or acute onset of generalized edema or pleural or peritoneal effusions with hypoalbuminemia as defined by a serum albumin <3 g/dL.
Generalized erythematous rash: presence of a generalized erythematous rash.
Soft-tissue necrosis: presence of necrotizing fasciitis, necrotizing myositis, or necrotizing gangrene.
For each case, we assigned a clinical criteria score of 1 to 6 based on the number of clinical criteria present. Following CSTE guidelines, we defined a case as STSS–CSTE if hypotension was present and the clinical criteria score was ≥2. Although the CSTE definition classifies cases as probable STSS when illness is associated with GAS isolation from a normally nonsterile site and as confirmed STSS when illness is associated with GAS isolation from a normally sterile site, we did not make this distinction in our analysis.
We included data on all invasive GAS cases captured by ABCs from January 1, 2014, through December 31, 2017, for which a supplemental form was available. We compared the demographic and clinical characteristics of STSS–physician, septic shock, and STSS–CSTE cases. For STSS–CSTE cases, we calculated the clinical criteria scores and frequencies of the contributing clinical criteria. We also compared the proportion of cases that were identified as STSS–physician, septic shock, and STSS–CSTE at each ABCs site during the study period.
The Centers for Disease Control and Prevention (CDC) publishes national estimates of invasive GAS disease annually by applying race- and age-specific rates of invasive GAS disease from ABCs areas to age and racial distribution of the United States. 1 We applied the proportion of STSS–physician and STSS–CSTE cases among invasive GAS cases in ABCs to the national estimates of invasive GAS cases for 2017, the most recent year for which data were available, to estimate the incidence of STSS in the United States.
Statistical Analysis
We assessed agreement between STSS–physician cases and STSS–CSTE cases by using the Cohen κ coefficient. We reviewed the ABCs case reporting and isolate collection in accordance with CDC human research protection procedures and determined this study to be nonresearch public health surveillance. The ABCs sites reviewed the protocol and obtained institutional review board approval as required.
Results
ABCs identified 7612 invasive GAS cases from January 1, 2014, through December 31, 2017, of which 7572 (99.5%) cases had a supplemental form completed (range by site, 97.3%-100.0%). Of these 7572 cases, 7479 (98.8%) cases had a sterile-site GAS isolate.
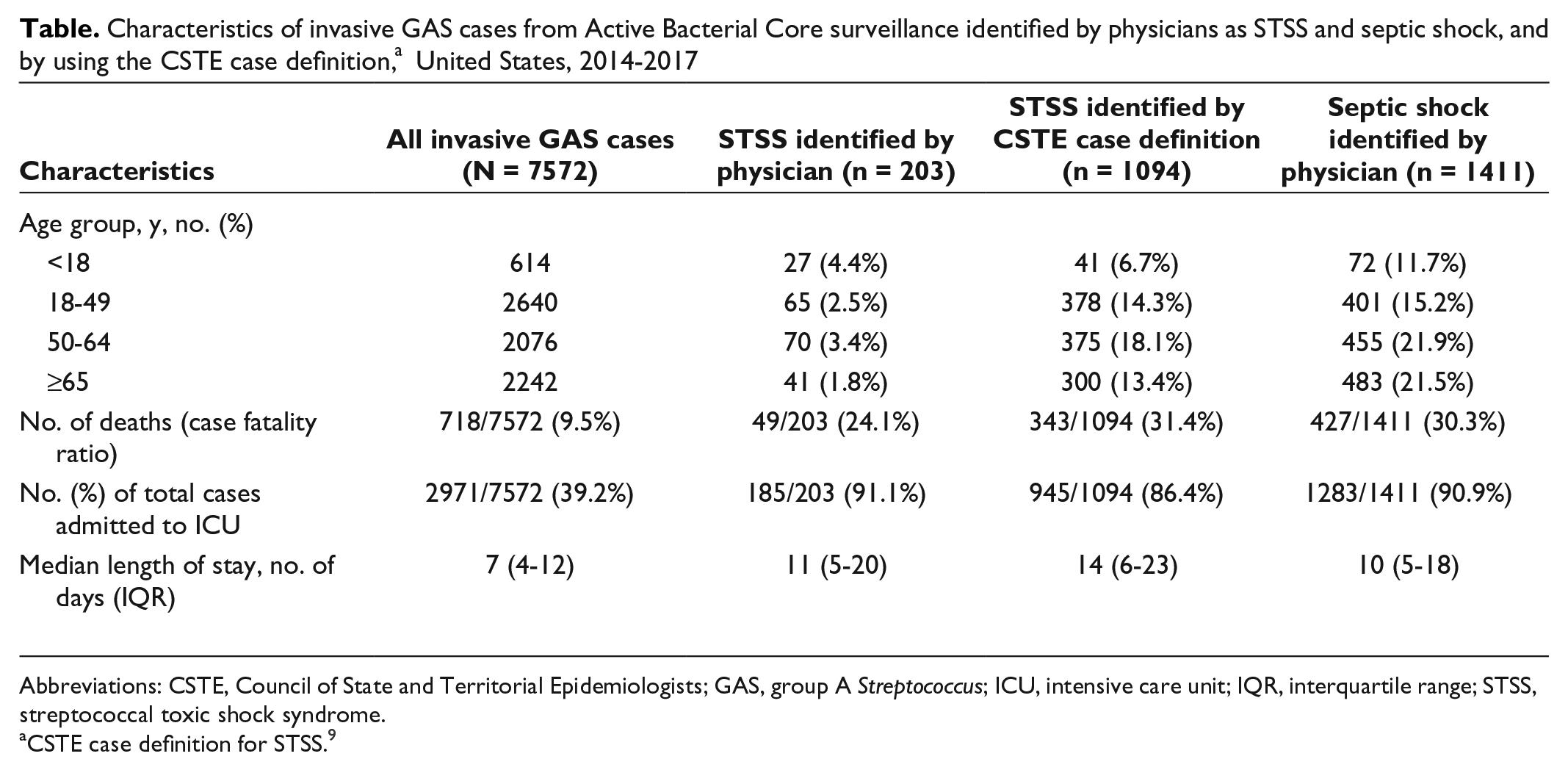
Of 7572 invasive GAS cases with supplemental form completed, 1094 (14.4%) were categorized as STSS–CSTE, 203 (2.7%) as STSS–physician, and 1411 (18.6%) as septic shock (Table). Of 203 STSS–physician cases, 132 (65.0%) met the criteria for categorization as STSS–CSTE (Figure 1). Of 1094 cases categorized as STSS–CSTE, only 132 (12.1%) were identified as STSS–physician. Agreement between the 2 methods for identifying STSS cases was low (κ = 0.17; 95% CI, 0.14-0.19). Of 1094 STSS–CSTE cases, 654 (59.8%) were also identified by physicians as septic shock cases; 692 (63.3%) were identified as either STSS–physician or septic shock cases.
Characteristics of invasive GAS cases from Active Bacterial Core surveillance identified by physicians as STSS and septic shock, and by using the CSTE case definition, a United States, 2014-2017
Abbreviations: CSTE, Council of State and Territorial Epidemiologists; GAS, group A
aCSTE case definition for STSS. 9
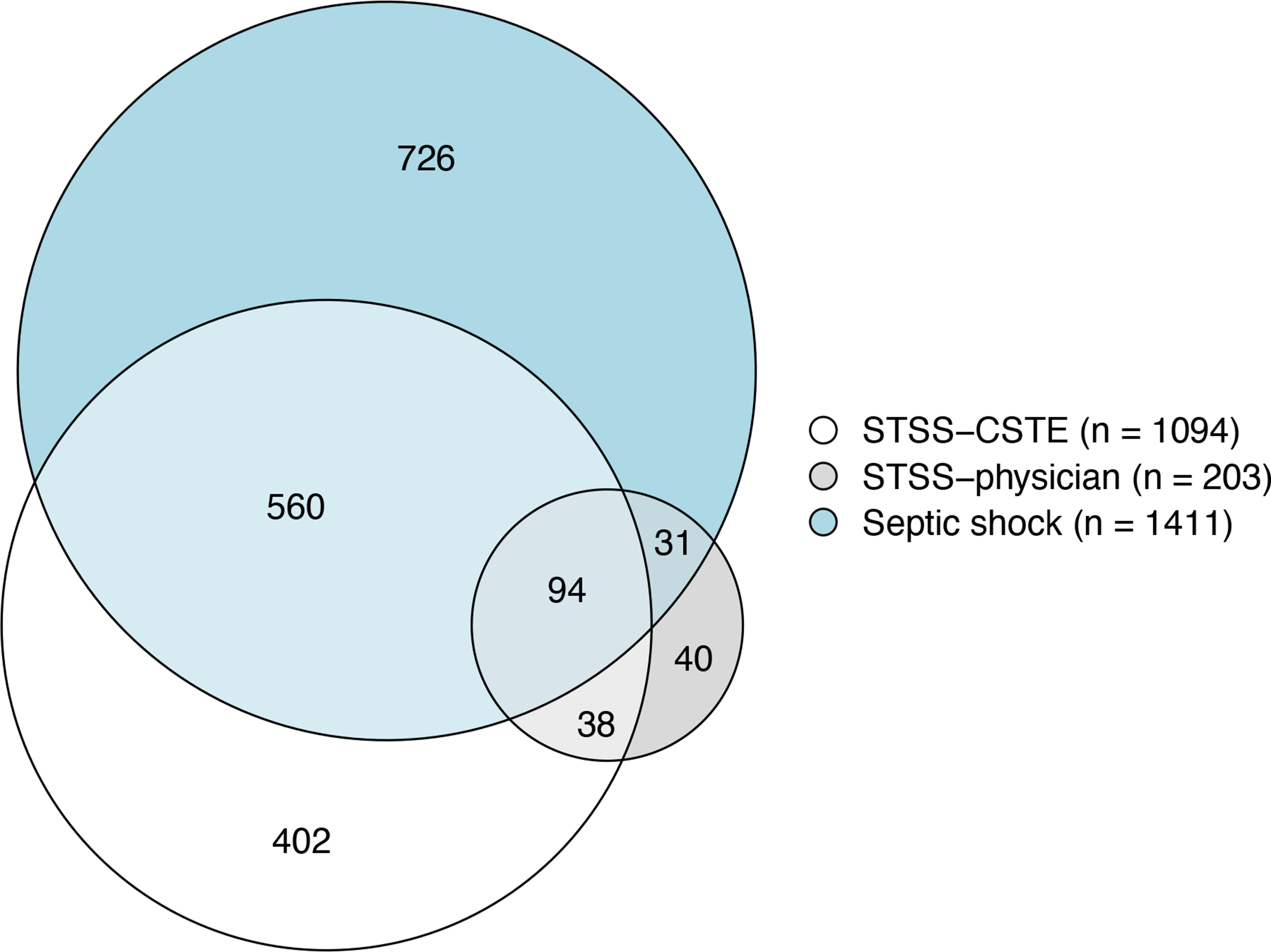
Venn diagram describing the overlap among cases of STSS identified by physicians (STSS–physician), STSS identified by applying CSTE case definitions
9
(STSS–CSTE), and septic shock identified by physicians among all invasive GAS cases from 10 Active Bacterial Core surveillance sites, 2014-2017. The sites were in California, Colorado, Connecticut, Georgia, Maryland, Minnesota, New Mexico, New York, Oregon, and Tennessee. Abbreviations: CSTE, Council of State and Territorial Epidemiologists; GAS, group A
The percentage of cases that were admitted to the intensive care unit (ICU) and the case-fatality ratio among cases categorized as STSS (by either CSTE criteria or physician) and as septic shock were similar (Table). Compared with STSS–CSTE cases that were not identified as septic shock or as STSS–physician, STSS–CSTE cases identified also as septic shock or STSS–physician cases were nearly 1.4 times more likely to be admitted to the ICU (96.7% vs 68.7%) and nearly twice as likely to have died (38.3% vs 19.4%).
The most common clinical manifestations of severity among cases categorized as STSS–CSTE were renal impairment (n = 681; 62.2%), ARDS (n = 664; 60.7%), and coagulopathy (n = 624; 57.0%). Of all STSS–CSTE cases, 545 (49.8%) met the CSTE case definition based on the presence of only 2 clinical indicators of severity; generalized erythematous rash was the least frequently identified clinical criterion (62 of 545 cases [11.4%]). In our sensitivity analysis, of 1094 STSS–CSTE cases, 1032 (94.3%) would continue to be categorized as STSS–CSTE even if generalized erythematous rash were not considered as a qualifying criterion.
Across the ABCs sites, the proportion of invasive GAS cases identified as STSS–physician ranged from 0.4% at the Colorado site to 7.1% at the New York site (Figure 2). The proportion of invasive GAS cases identified as STSS–CSTE ranged from 7.5% at the Oregon site to 21.0% at the New Mexico site. The ratio of the number of cases identified as STSS by CSTE guidelines to the number of STSS cases identified by physicians ranged by ABCs site from a 2.3-fold increase in New York to a 26.5-fold increase in Colorado.
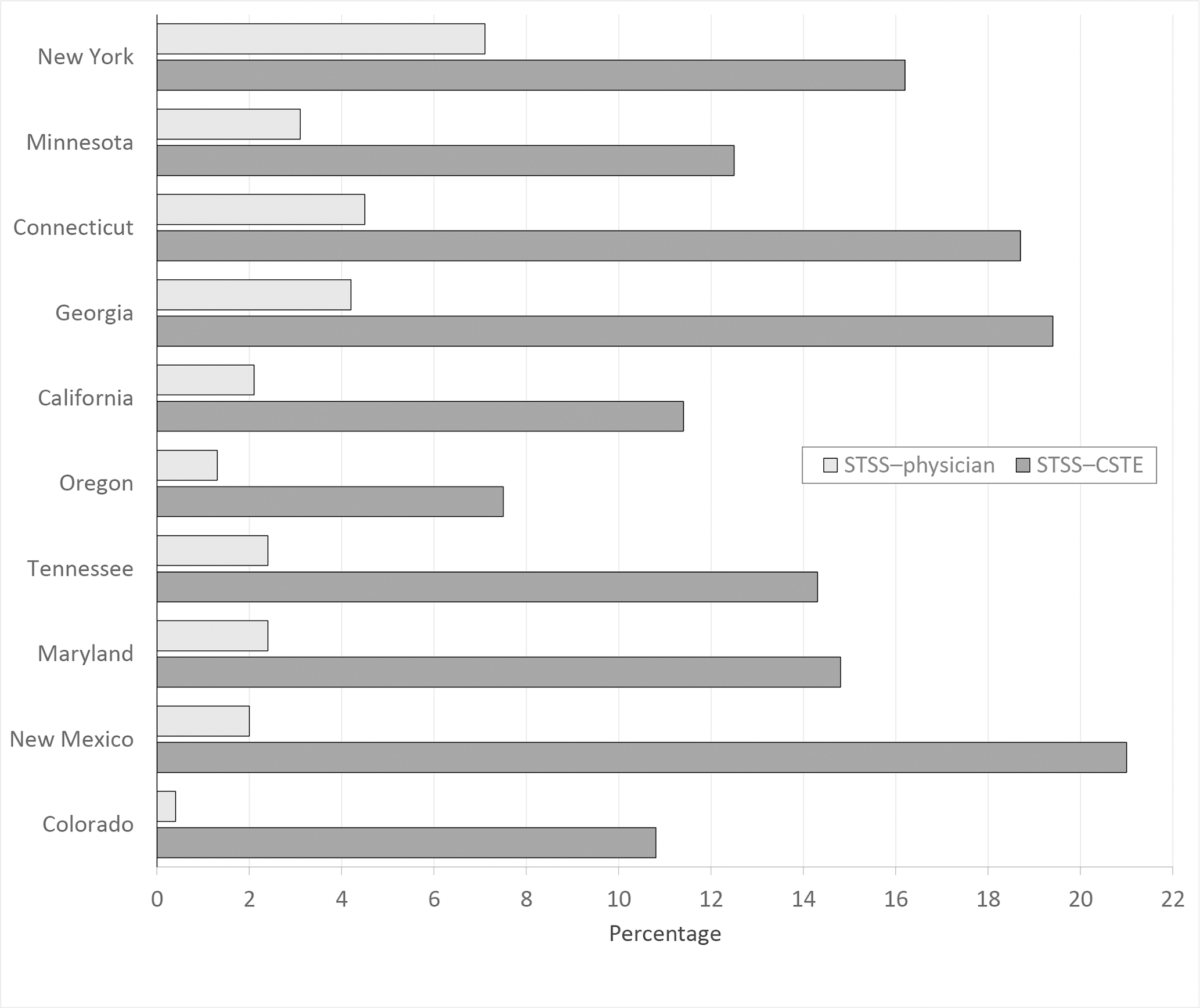
Proportion of invasive GAS cases identified as STSS when comparing proportion of cases identified as STSS by applying CSTE case definitions
9
(STSS–CSTE) with cases identified as STSS by physicians (STSS–physician), by ABCs site, 10 states, 2014-2017. ABCs conducts active population- and laboratory-based surveillance for invasive GAS disease in selected counties of 10 states (California, Colorado, Connecticut, Georgia, Maryland, Minnesota, New Mexico, New York, Oregon, and Tennessee). Abbreviations: ABCs, Active Bacterial Core surveillance; CSTE, Council of State and Territorial Epidemiologists; GAS, group A
In 2017, 2.5% of invasive GAS cases were identified as STSS–physician and 15.3% as STSS–CSTE. Applying these proportions to national estimates of invasive GAS disease for 2017, we estimated that 591 cases of STSS–physician and 3618 cases of STSS–CSTE occurred that year.
Discussion
Of all invasive GAS cases detected through active population- and laboratory-based surveillance in the United States during 2014-2017, we identified 14.4% of cases as STSS using clinical data and the CSTE case definition. This percentage was 5.3 times higher than the 2.7% of invasive GAS cases that were explicitly identified as STSS in medical records by physicians. The poor agreement between the 2 methods for identifying STSS cases has implications for estimates of STSS incidence that use ABCs data and other surveillance systems that depend on capturing physician-diagnosed STSS cases. Previous surveillance reports from ABCs in the United States (2000-2012) and from Alaska’s Arctic Investigations Program (2001-2013) that categorized cases as STSS based on a recorded diagnosis in medical records estimated that approximately 3% to 6% of invasive GAS cases manifested as STSS. 5,15 However, studies from Europe (2003-2004), the United Kingdom (2003-2004), and Australia (2003-2004) that categorized invasive GAS cases as STSS based on clinical criteria reported higher proportions of 8% to 14%. 2,16,17 These proportions were closer to the proportion of invasive GAS cases identified as STSS–CSTE (14.5%) in our study when using a similar approach of categorizing cases as STSS using previously established clinical criteria.
The CSTE case definition for STSS requires the presence of well-defined clinical features and is likely more specific than sensitive. 18 Despite being designed for specificity, use of these criteria resulted in a far greater proportion of invasive GAS cases being classified as STSS than based on explicit diagnosis by health care providers in the medical records in our study. We suspect that, in clinical practice, the distinction between STSS and septic shock as a diagnosis could be minimal; both are characterized by severe hypotension and underlying organ failure in the presence of an infection. The STSS definition is stringent, requiring isolation of GAS and evidence of multiorgan failure. 19 In our study, nearly 60% of STSS–CSTE cases, which were identified as STSS by meeting established clinical criteria, had a diagnosis of septic shock associated with GAS isolation in their medical records, indicating that surveillance for STSS cases solely based on an explicit diagnosis of clinician-defined STSS from medical records leads to an underestimation of STSS incidence because of physicians’ misclassification of STSS cases as septic shock.
Although an
Although clinical definitions for sepsis and septic shock have evolved over time, the CSTE case definition for STSS is based on a consensus definition that has not changed since its publication since 1993 and, thus, may be used to measure STSS incidence, monitor disease trends, and evaluate the effect of new therapies for STSS. 8,26 -28 At least 1 criterion from the consensus definition for STSS—generalized erythematous rash—is subjective and nonspecific for physicians to observe and, although it is a GAS-associated toxin-mediated manifestation, it is not indicative of underlying organ failure. However, eliminating rash as a defining criterion would have excluded only 5.7% of the STSS–CSTE cases in our study. Furthermore, the most common criteria leading to categorization of cases as STSS–CSTE in our study were renal impairment, ARDS, and coagulopathy—all overt and specific criteria. The high proportion of invasive GAS cases identified as STSS–CSTE in our study was not the result of high frequency of any single or less severe clinical criterion.
Regardless of the method used to identify STSS, the proportion of invasive GAS cases identified as STSS by different ABCs sites varied widely. This variation may represent local differences in strains that cause invasive disease and their propensity to cause severe manifestations such as STSS. However, we also noted large variation in the difference in proportion of STSS–CSTE and STSS–physician cases by ABCs site ranging from proportion of STSS–CSTE cases being 2.3 times higher than the proportion of STSS–physician cases at the New York site to 26.5 times higher at the Colorado site, suggesting some differences in physician practices in diagnosing STSS across sites. Whether these differences are associated with differences in clinical management, however, is unknown.
Limitations
Our study had several limitations. First, ABCs relies on manual abstraction of medical record data; as such, human error in capturing data on syndromes or clinical characteristics cannot be ruled out. However, ABCs has been conducting surveillance for decades using refined protocols and annual refresher trainings for surveillance staff members, thereby reducing the likelihood of errors. Second, because ABCs does not collect data on treatment, we were unable to ascertain differences in clinical management among cases classified as STSS–CSTE, STSS–physician, or septic shock. It is possible that although physicians do not use the term STSS explicitly in their diagnosis, treatment of these cases is appropriate. We do not know if lack of diagnosis of STSS resulted in delayed recognition of severe associated syndromes, such as necrotizing fasciitis, and delayed GAS-specific treatment. Finally, relying solely on laboratory confirmation of the etiology of STSS for any case definition will miss cases in which diagnostic specimens are not collected or are culture-negative because of previous use of empiric antibiotics. Also, physician-based reporting of syndromes such as septic shock can be poor. Therefore, even our revised estimates likely underestimate the true incidence of STSS in the United States.
Conclusion
Although STSS is a nationally notifiable condition, underreporting to the NNDSS is likely. In 2017, a total of 372 STSS cases were reported to the NNDSS. 29 However, applying proportions derived from our study to the national estimates of invasive GAS disease and using the CSTE-supported case definition, we estimated that nearly 3600 STSS–CSTE cases occurred in 2017. Our data also suggest differences in physician practices with identifying STSS cases across ABCs sites that need to be characterized further. In summary, the wide differences in estimates of STSS with differing surveillance methods and definitions highlight the difficulties in using currently available surveillance data to evaluate policy or guidelines for STSS. Our study suggests that using established clinical criteria may provide more accurate estimates of the incidence of morbidity and mortality resulting from this severe manifestation of GAS infection. Accurate and consistent categorization of STSS may also improve capacity to use medical records to evaluate and monitor use of therapeutic interventions, such as intravenous immunoglobulins, and preventive measures, such as GAS vaccines, in observational studies.
Footnotes
Acknowledgments
The authors thank everyone in Active Bacterial Core surveillance (ABCs) who are involved in surveillance and maintenance of the system at the 10 sites. We also thank the laboratorians and technicians who isolate the ABCs pathogens and make it possible to track these infections, and the surveillance and laboratory personnel at the Centers for Disease Control and Prevention (CDC) for their careful work characterizing the isolates. We acknowledge the following members of the ABCs team and others for their contributions: Kathy Angeles, Lisa Onischuk, and Salina Torres (New Mexico); Kathleen Shutt, Rosemary Hollick, Terresa Carter, and Vijitha Lahanda Wadu (Maryland); Joelle Nadle, Maria Rosales, Pam Daily-Kirley, and Susan Brooks (California); and Glenda Smith, Jillian Karr, Nancy Spina, and Rachel Wester (New York).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Nisha Alden reported receiving funding from a cooperative agreement with CDC during the conduct of this study. Dr William Schaffner reported consulting for VBI vaccines outside the submitted work. Dr Lee Harrison reported consulting in the area of epidemiology and vaccine prevention of bacterial infections with Merck, GSK, Sanofi Pasteur, and Pfizer outside the submitted work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CDC provided financial support for the Active Bacterial Core surveillance program through cooperative agreements with the Active Bacterial Core surveillance sites.
