Abstract

The Commissioned Corps of the US Public Health Service (USPHS) is 1 of the 7 US uniformed services. Under the leadership of the US Surgeon General, USPHS has approximately 6800 commissioned officers, who span multiple categories of health service professionals and work for various agencies within the US Department of Health and Human Services. In addition to the US Air Force, US Army, US Navy, and US Department of Veterans Affairs, USPHS is recognized as an American Medical Association (AMA) Federal Services Member Organization and, therefore, holds a seat in the AMA House of Delegates (HOD).
The AMA HOD is the principal policymaking body of the AMA. The HOD follows parliamentary procedure, 1 with the bulk of the work conducted in committees and a final debate of resolutions occurring on the HOD floor (Figure). 2 HOD policy statements provide information and guidance that physicians, other health care providers, and policy makers seek from the AMA about a spectrum of health care and public health issues. In some cases (eg, when the HOD first defined obesity as a disease), they may attract considerable public attention. 3,4
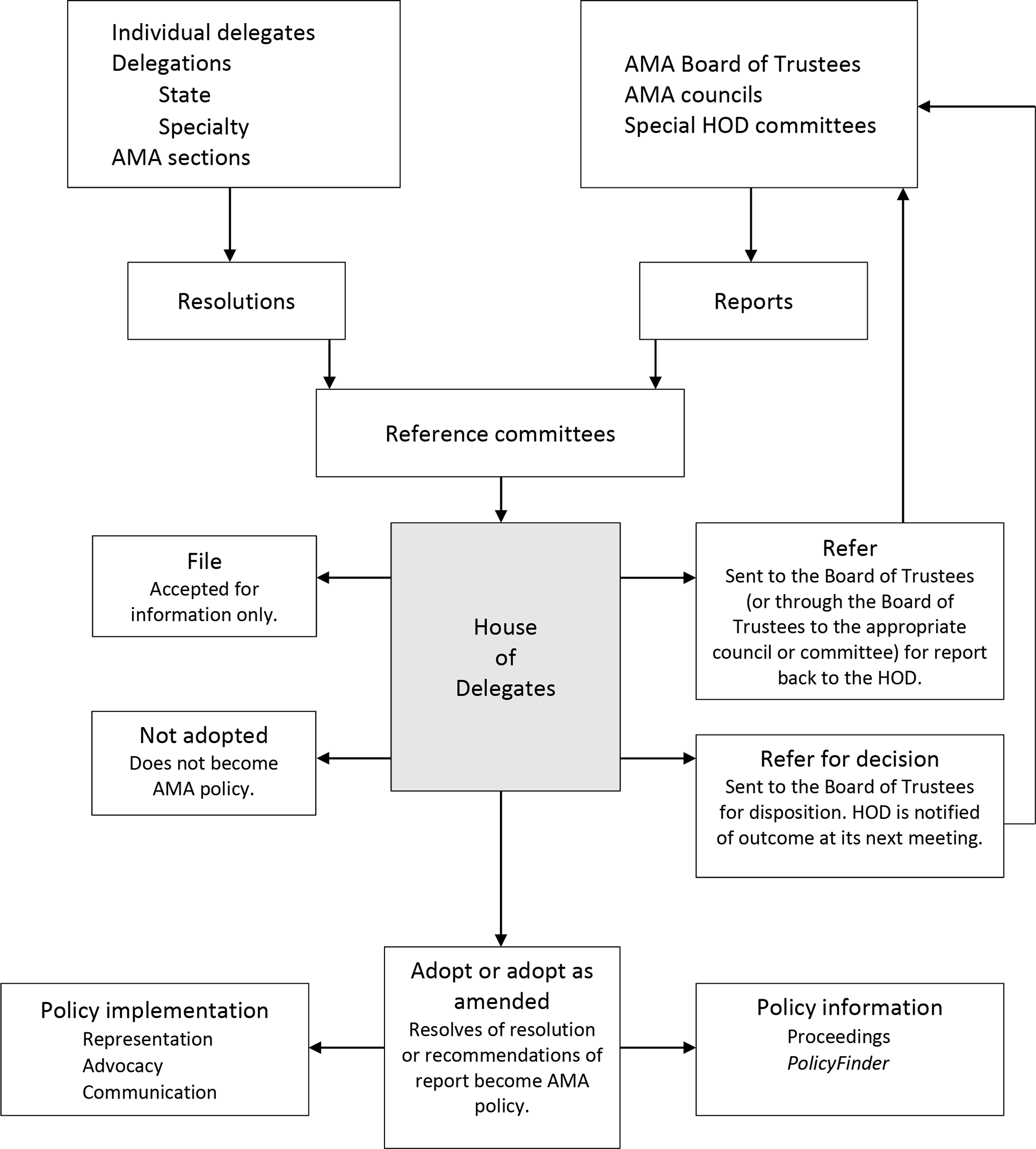
Schematic representation of the American Medical Association (AMA) policymaking process, as of November 2019. Abbreviation: HOD, House of Delegates. Adapted from American Medical News, June 11, 2007. 2
The HOD currently has more than 600 voting delegates. Delegates represent state medical societies, physician specialty groups, professional interest medical associations, and the AMA’s member sections. 5 -7 Although policy resolutions must be proposed to the HOD by an AMA delegate, both voting delegates and nonvoting members may testify.
USPHS has participated in the AMA policymaking process as a voting member since the inception of the HOD in 1901. The official delegate from USPHS is the Surgeon General. Historically, the Surgeon General assigns this role to a senior USPHS physician, such as its chief medical officer. The USPHS delegation to the HOD, which includes both a delegate and an alternate delegate, relies on volunteer officers from USPHS agencies to prepare and examine testimony on proposed AMA policies. In this commentary, we describe the role the USPHS delegation plays in helping the AMA make policy that reflects current and accurate public health knowledge.
In proposed policies in which federal health agencies have evidence for health-related impact of the proposed policy, the USPHS delegation reviews resolutions and develops testimony based on this evidence for discussion during reference committees (Figure). Each resolution has 2 parts. The whereas parts, or preambles of the resolutions, are considered informational and explanatory only. Only the resolved parts of the resolutions are considered by the HOD and, if adopted, will become AMA policy. Because development of USPHS testimony focuses on the whereas parts of resolutions rather than on the resolved parts, the emphasis in developing testimony is on ensuring that resolutions contain information about public health issues that is scientifically accurate, evidenced based, and current and that reflects applicable federal laws and regulations. This approach is consistent with a “science first” policy posture 7 and leaves the debating of policy to the broader HOD.
Collaboration and alignment of priorities in the development of testimony occur both within and between federal agencies. Each agency in which USPHS officers serve follows a separate internal process. For example, in the Centers for Disease Control and Prevention (CDC), the Office of the Associate Director for Policy and Strategy is the organizational location for reviewing AMA resolutions and developing testimony. This model of AMA testimony development exemplifies the engagement of public health policy professionals, CDC subject matter experts, and persons outside USPHS in a high-profile policy process, demonstrating the “science first” approach. 8 Because key agencies may have various perspectives on the same resolution, testimony development before the annual meeting of the HOD in June includes cross-agency conference calls conducted by the USPHS delegation.
Testimony may be provided before the meeting in written form as comments or as proposed edits to draft resolutions. However, testimony also may be provided through in-person verbal attestation during Reference Committee hearings for changes to be considered before a resolution’s referral to the HOD floor for vote. Based on USPHS testimony, sponsors of the proposed policy or the Reference Committee itself may revise, withdraw, or postpone a resolution from being considered by the full HOD. For example, one of those actions may occur if substantial inaccuracies in the whereas portion are elucidated.
At the November 2018 interim meeting of the HOD, CDC, the US Food and Drug Administration, the National Institutes of Health, and the Health Resources and Services Administration presented testimony on 22 resolutions and reports. The full HOD voted in concurrence with USPHS testimony in 16 cases and in opposition with USPHS testimony in 4 cases; in 2 cases, reports were filed without discussion (USPHS, unpublished data). Notable examples included:
A resolution titled “Common sense strategy for tobacco control and harm reduction” encouraged the use of e-cigarettes for smoking cessation. USPHS commented on 2 statements in the preamble, based on expert conclusions from the US Food and Drug Administration and CDC that evidence was insufficient to recommend this category of nicotine-containing products for tobacco cessation. The resolution was not adopted by the HOD.
On a resolution relating to prevention of suicides among physicians and medical students, USPHS shared CDC guidance on several evidence-based suicide prevention resources. 9 This resolution was adopted by the HOD.
On a report relating to medical tourism, the USPHS delegation clarified that no evidence exists to support the idea that medical tourists (ie, US residents who travel abroad for medical care) pose a danger to their communities when they return. This report was 1 of 2 reports that was filed but not discussed.
On a resolution involving drug pricing transparency that called for the AMA to “advocate to the US Surgeon General” for federal legislation, the USPHS delegation clarified that although the Surgeon General can draw attention to important public health issues, the Surgeon General has no direct authority to regulate or propose legislation.
At the 2018 annual and interim meetings of the HOD, the USPHS delegation commented on resolutions involving topics such as drug pricing, payment systems, and physician maintenance of certification programs. USPHS feedback was most frequently reflected in adoption of resolution language that reflected up-to-date evidence.
Both the role of the Surgeon General overall and the role of USPHS in the AMA policy process are types of meta-leadership 10,11 in the sense that USPHS does not have direct policy authority but can inform policy creation. Reports released by the Surgeon General are perhaps the most prominent example of meta-leadership in the field of public health, as they do not themselves make law or policy but draw attention to their subject matter and, ultimately, may affect public opinion. Prominent examples include former Surgeon General Luther Terry’s groundbreaking report in 1964 on the adverse health consequences of smoking and VADM C. Everett Koop’s report in 1986 on AIDS. 12,13 With more than 70 000 persons in the United States dying from a drug overdose in 2017, the Surgeon General’s report in 2018 advanced a vision of addiction as a brain disease and challenged Americans to end the stigma associated with addiction. 14
The participation of USPHS in the AMA HOD has created opportunities for additional partnerships among USPHS officers, the agencies they serve, and parts of the AMA. Beyond the scope of HOD activities, the current USPHS alternate delegate serves as the CDC liaison to the AMA. This role is exemplified by work involving the CDC Radiation Annex Team and the AMA Council on Public Health and Science, which seek to identify clinician responders who are likely to encounter radiation casualties, to review interim federal guidelines for radiation incident response, and to help communities address radiation incidents nationally, regionally, and locally. Opportunities also exist for the USPHS delegation to work informally with delegates from other uniformed services and those who share common interests (eg, members of the AMA Public Health Caucus and the American Academy of Preventive Medicine).
USPHS participation in the AMA HOD on behalf of USPHS agencies exemplifies better health through better partnerships. Within the US Department of Health and Human Services, CDC, the US Food and Drug Administration, and other federal public health agencies follow the resolutions as they emerge and participate in drafting and assessing USPHS testimony. This coordinated process ensures that the voice of federal public health—representing diverse agencies, professions, and personnel systems—is heard in a unified manner at one of the most important policy forums of organized medicine. The policy positions of the AMA have important implications not just for physicians but also for broad areas of the health care system, including electronic health information and financing. 15,16
USPHS representation in the AMA HOD fosters dialogue, which might not otherwise occur, between 2 important organizations in medicine and public health. An important outcome of this process is the inclusion of up-to-date public health data and knowledge in resolutions debated, and passed, by the HOD. Because of this wide-ranging impact, USPHS engagement with the AMA HOD warrants ongoing leadership and agency support. Members of the HOD, as active representatives of organized medicine, should continue to hear directly from federal representatives to access relevant data about emerging public health issues on which they are preparing to debate or vote. With this support from USPHS, the AMA can continue to craft accurate and effective policy.
Footnotes
Acknowledgments
The authors acknowledge the following for their contributions to resolution review and testimony preparation: Anna Fine, Kimberly Lindsey, Deborah Belsky, John Hariadi, Laura Polakowski, Toya Kelley, Kristie E.N. Clarke, Steve L. Morin, Stuart Nightingale, Kara Jacobs Slifka, and Elizabeth Van Dyne. The authors also thank US Surgeon General VADM Jerome M. Adams, MD, MPH, for his contributions to and leadership support of the work described here.
Authors’ Note
John Iskander and Dana Thomas contributed equally to this commentary. The views presented in this commentary are those of the authors and do not necessarily represent the policy of the Centers for Disease Control and Prevention, the US Public Health Service, or the US Department of Health and Human Services.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared no financial support with respect to the research, authorship, and/or publication of this article.
