Abstract
Objectives:
From September 2015 through March 2016, Hawaii had the largest outbreak of locally transmitted dengue since 1944. We report on the Hawaii Department of Health’s (HDOH’s) investigation, findings, and response to the outbreak.
Methods:
We defined cases of dengue using a modified version of the Council of State and Territorial Epidemiologists’ case definition for dengue virus infections. We conducted epidemiologic investigations, including interviews with case-persons, review of medical records, laboratory testing, genetic sequencing of specimens, and geographic information system (GIS) data analysis. Outbreak response included community outreach and vector-control activities.
Results:
We identified 264 confirmed cases of dengue; illness onset dates ranged from September 11, 2015, to March 17, 2016, all with reported travel to or residence on the Island of Hawaii. Of 264 persons with confirmed dengue, 238 (90.2%) were Hawaii residents. Thirty-seven (14.0%) persons required hospitalization; no cases of severe dengue or death were reported. GIS hot-spot analysis identified a cluster of cases on the western side of the island. Established risk factors for dengue exposure included holes in window or door screens, presence of standing water, and not using insect repellent or wearing protective clothing.
Conclusions:
To prevent or mitigate the spread of future arboviral introductions and outbreaks, the public health response should focus on behavioral and cultural attitudes, emphasizing personal mosquito protection and mosquito control at the community level. Outbreak responses can also be enhanced through the use of advanced GIS techniques, such as hot-spot analysis, to provide situational awareness and guide response efforts.
Dengue is a mosquito-borne disease caused by any of 4 dengue viruses, dengue virus type 1 (DENV-1) through dengue virus type 4 (DENV-4). It is primarily transmitted by day-biting Aedes (Stegomyia) mosquitoes. Ae. aegypti is one of the most competent vectors, but Ae. albopictus has been implicated in nonendemic regions as a major outbreak driver. 1 -5 As the world’s climate changes, the range of habitats suitable for Ae. albopictus and Ae. aegypti are expected to expand; as a result, the regions at risk for dengue are also expected to expand. 6 Dengue is found most frequently in tropical and subtropical regions worldwide, causing approximately 50 million infections annually in more than 100 countries. 7
Dengue virus infection commonly results in high fever, arthralgias, myalgias, severe headache, retro-orbital eye pain, maculopapular rash, and mild hemorrhagic manifestations after an incubation period ranging from 3-14 days. Infected persons generally recover in 3-7 days, although some may progress to severe dengue (previously designated dengue hemorrhagic fever or dengue shock syndrome), characterized by plasma leakage leading to shock and/or fluid accumulation, severe bleeding, and/or severe organ involvement. Most cases of severe dengue occur in persons with repeated dengue infection, although it can also occur in persons infected for the first time. 7
Dengue was endemic in Hawaii until the mid-1940s; several large outbreaks were recorded during 1840-1944. 8 -10 Eradication efforts (through the use of dichlorodiphenyltrichloroethane [DDT]) in the 1940s and the 1960s eliminated Ae. aegypti from most of the state and greatly contributed to the eventual elimination of dengue endemicity. 11 Since then, until 2015, only 2 recognized outbreaks had occurred, both within the past 20 years: 1 in 2001-2002 with 122 confirmed cases 1 and the other in 2011 with 5 confirmed cases (Hawaii State Department of Health [HDOH]), unpublished data). Routinely, 4-14 cases per year of imported dengue have been identified in Hawaii since 2006. 12
On October 21, 2015, HDOH received an electronic laboratory report of a positive result for dengue immunoglobulin M (IgM) antibody in a specimen from a resident of the Island of Hawaii. This person reported no travel history, and 2 family members had a similar illness: 1 with the same onset date and 1 with illness onset approximately 2 weeks earlier. The timing of onsets and lack of history of travel strongly suggested local arboviral transmission. In response, HDOH launched an investigation to determine the scope of the outbreak and engaged county partners in response and control activities. On October 24, 2015, HDOH received a report of febrile illness, later confirmed as dengue, among US mainland travelers who had visited the Island of Hawaii in mid-October, further strengthening suspicion of local transmission. In addition, HDOH received a laboratory report on October 27, 2015, of another positive result for dengue IgM for an Island of Hawaii resident epidemiologically unrelated to the previous cases.
Methods
Case Definition
We developed an outbreak-specific case definition based on the Council of State and Territorial Epidemiologists’ case definition for DENV infection. 13 A person was considered a suspected case if he or she had illness onset on or after September 1, 2015, with an acute onset of fever; at least 2 of the following symptoms: headache/retro-orbital pain, nausea, myalgias, and/or arthralgias or a generalized maculopapular rash, without an alternative explicative etiology; and a history of travel to or residence on Hawaii Island on or after September 1, 2015, with no history of recent travel to other regions with active dengue transmission. A case was considered to be confirmed when (1) it met the suspected case criteria and (2) any of the following laboratory tests in an appropriate clinical specimen confirmed infection: DENV polymerase chain reaction (PCR), detection of anti-DENV IgM without evidence of other flavivirus infection (or vaccination), or plaque reduction neutralization test (PRNT). This case definition differed from the standard case definition used by HDOH case investigation, for which all serologically positive (IgM) cases are confirmed by PRNT.
Case Finding
To identify additional cases, on October 29, 2015, HDOH distributed a medical advisory to clinicians statewide to alert them of local dengue transmission on the Island of Hawaii and ask if they had reported any patients meeting the case definition. On the same day, HDOH also posted a call for cases on Epi-X, the Centers for Disease Control and Prevention’s (CDC’s) web-based communications program for public health professionals, asking health departments nationwide to report any potential dengue cases with recent travel to Hawaii. HDOH distributed 2 additional medical advisories on November 5, 2015, and January 13, 2016, to advise clinicians of the ongoing disease activity and appropriate diagnosis and management. HDOH issued press releases during the outbreak to raise awareness statewide and encourage persons with symptoms to consult their health care provider for clinical evaluation and management and to aid in the identification of cases.
Data Collection
We administered a standard HDOH questionnaire by telephone or in person, collecting data on demographic characteristics, symptoms, travel history, and exposure histories (eg, place of work, school, and locations visited), to all potential case-persons. The questionnaire also asked about other previously established dengue risk factors, such as the presence and condition of screened windows or doors in homes, the presence of standing water on properties, and mosquito protection practices. We recorded information on potential exposure locations (ie, areas where persons had been and could have been exposed to mosquitoes) for the 14 days before illness onset until 7 days after illness onset, encompassing the time persons were likely to have been exposed to dengue through the time when they would be viremic and infectious. When available, we also collected data on medical records and test results for suspected case-persons. Because this outbreak investigation was considered public health practice, institutional review board review was not needed.
Mapping
We mapped and analyzed case-person geographic data by using ArcGIS version 10.3 to identify areas of activity and potential disease transmission patterns. 14 We mapped case-person residences and the locations that case-persons reported visiting 3-14 days before illness onset as the outbreak progressed. We used these maps to track the outbreak progression and help inform decisions on public health response. We mapped reported locations of potential exposure or case-person residences surrounded by a mile-wide buffer; we coalesced overlapping sites into a single zone. Coalescing overlapping sites protected persons’ privacy and prevented underestimation of the areas of potential disease transmission or activity. For public health messaging and transmission tracking purposes, the color of the area reflected the most recent onset of case-persons with a reported potential exposure in the area: onsets that occurred within 2 weeks of the published map were red, within 3 weeks were orange, and within 4 weeks were yellow. We conducted hot-spot analyses at the census-block group level to identify clustering of case-person residences and reported potential exposure locations.
Laboratory Testing
Dengue IgM serology and reverse transcription PCR (RT-PCR) testing were performed at the HDOH State Laboratories Division (SLD) on the island of Oahu. SLD performed testing using the CDC DENV-1–4 real-time RT-PCR assay for the detection and serotype identification of DENV 15 and InBios DENV Detect IgM CAPTURE ELISA (InBios International, Inc, Seattle, WA). We asked clinicians and commercial laboratories to submit serum specimens from patients who met the case definition for confirmatory testing. We tested specimens by RT-PCR if collected within the first 5 days of illness or by serological testing if collected after day 7. Specimens collected within 5-7 days of illness onset and specimens with equivocal results were tested by both RT-PCR and serological testing. We submitted selected specimens to CDC’s reference laboratories in Fort Collins, Colorado, for PRNT and San Juan, Puerto Rico, for genetic sequencing.
Vector Control
Because of program reductions in 2009, HDOH vector control staff (VCS) member resources were limited. The few remaining VCS members coordinated and conducted mosquito activity surveys and abatement of larval sites around case-person residences within 48 hours of a suspected case of dengue being reported to HDOH. Report of suspected dengue infection was sufficient to take action, including surveying the place of residence to determine the presence of mosquito activity, followed by spraying adulticide (Aqua-Reslin, Bayer Environmental Science, Research Triangle Park, NC) over a 25-yard radius around the residence to reduce the number of mosquitoes present. VCS members also surveyed and applied larvicide (VectoBac G, Valent BioSciences, Libertyville, IL), when consent was provided by residents or owners, to standing water on any neighboring properties within a 200-yard radius of the case-person’s residence. During these activities, VCS members also distributed information about how to protect oneself from mosquitoes and how to reduce mosquito breeding areas.
Public Outreach
We established a web page to provide outbreak information, including case counts and maps of recent activity, to the public and updated it at least weekly. Throughout the outbreak, HDOH staff members conducted community outreach meetings on the Island of Hawaii. Other outreach methods included triaging telephone calls and using social media to disseminate information and identify and monitor potential public health messaging needs. The state launched a “Fight the Bite” campaign to raise awareness of mosquito-borne disease risks and advise persons on how to protect themselves from mosquitoes, including eliminating standing water around homes, fixing leaky outdoor faucets and hoses, treating plants that hold water (eg, bromeliads) with larvicide, clearing storm gutters and drains, repairing screens and windows, and disposing of old tires and other detritus that may hold standing water.
Statistical Analysis
We conducted statistical analyses by using SAS version 9.4 and R software. 16,17 We generated descriptive statistics (frequencies, proportions, medians, and ranges) for epidemiologic data. We conducted cluster and hot-spot analyses using ArcGIS version 10.3 and used Moran’s I, which measures spatial autocorrelation of data, and Getis-Ord Gi*, which identifies where high or low numbers of cases cluster spatially, to determine significant results. 14 We considered results to be significant at P < .05.
Results
As of April 26, 2016, HDOH had evaluated 2056 reports of potential dengue cases, including suspected cases from all counties in Hawaii and from out of state. We confirmed 264 of the 2056 (12.8%) reports as cases associated with the outbreak. Illness onset of the confirmed cases ranged from September 11, 2015, to March 17, 2016 (Figure 1). The median age of case-persons was 35 years (range, 9 months–85 years), 218 (82.6%) case-persons were aged >18, and 133 (50.4%) were male. Of the 258 case-persons who reported their race, most were white (n = 148; 57.4%) followed by Native Hawaiian/Pacific Islander (n = 65; 25.2%) and Asian (n = 24; 9.3%). Of 264 case-persons, 238 (90.2%) were Island of Hawaii residents and 26 (9.8%) were visitors from outside Hawaii; visitors’ median length of stay on Hawaii Island was 8.5 days (Table 1). All case-persons reported residing on or having recently traveled to the Island of Hawaii.
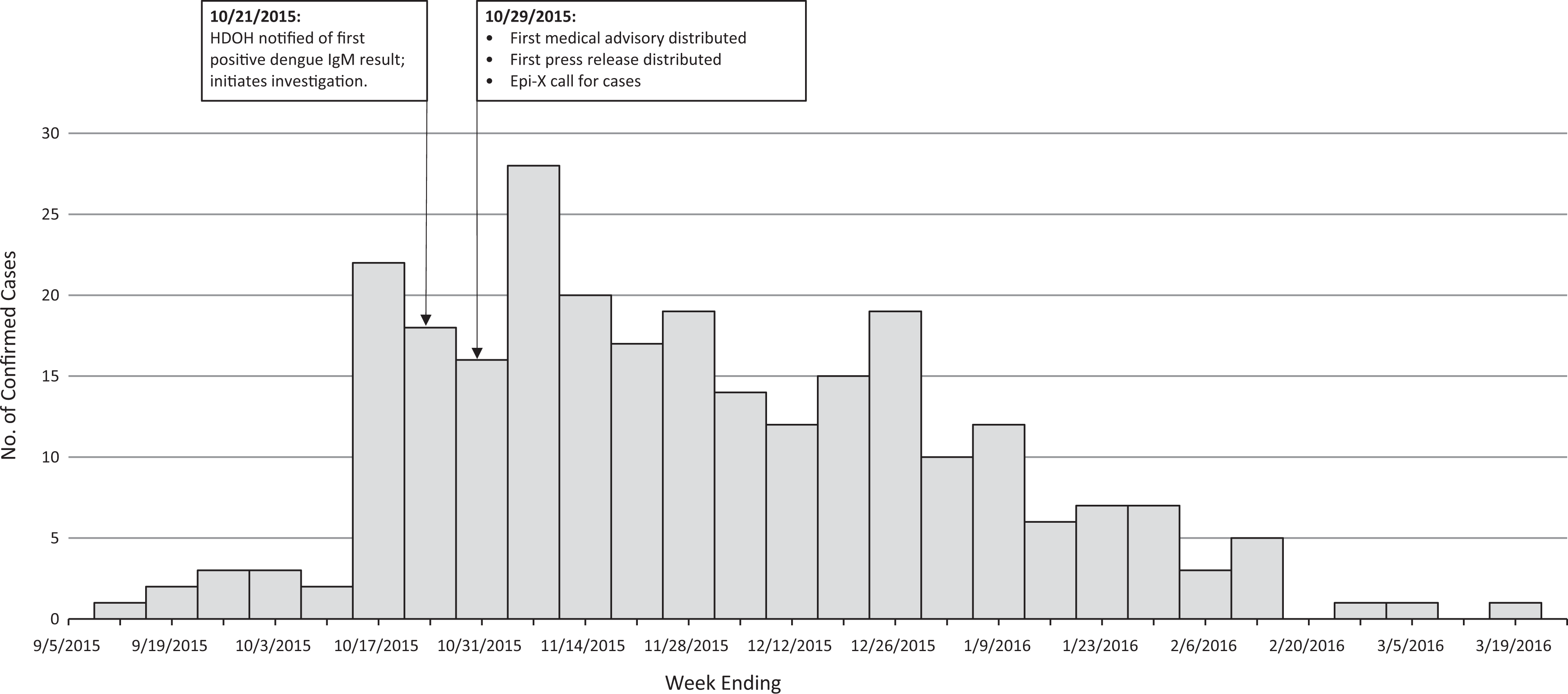
Number of outbreak-associated confirmed dengue virus infections, by date of onset, Island of Hawaii, 2015-2016. Abbreviations: HDOH, Hawaii State Department of Health; IgM, immunoglobulin M. Data collected from a questionnaire administered by HDOH and from medical records. Epidemic Information Exchange (Epi-X) is the Centers for Disease Control and Prevention’s secure, web-based information exchange system for public health professionals.
Characteristics of persons with an outbreak-associated confirmed dengue virus infection (N = 264), Island of Hawaii, 2015-2016a
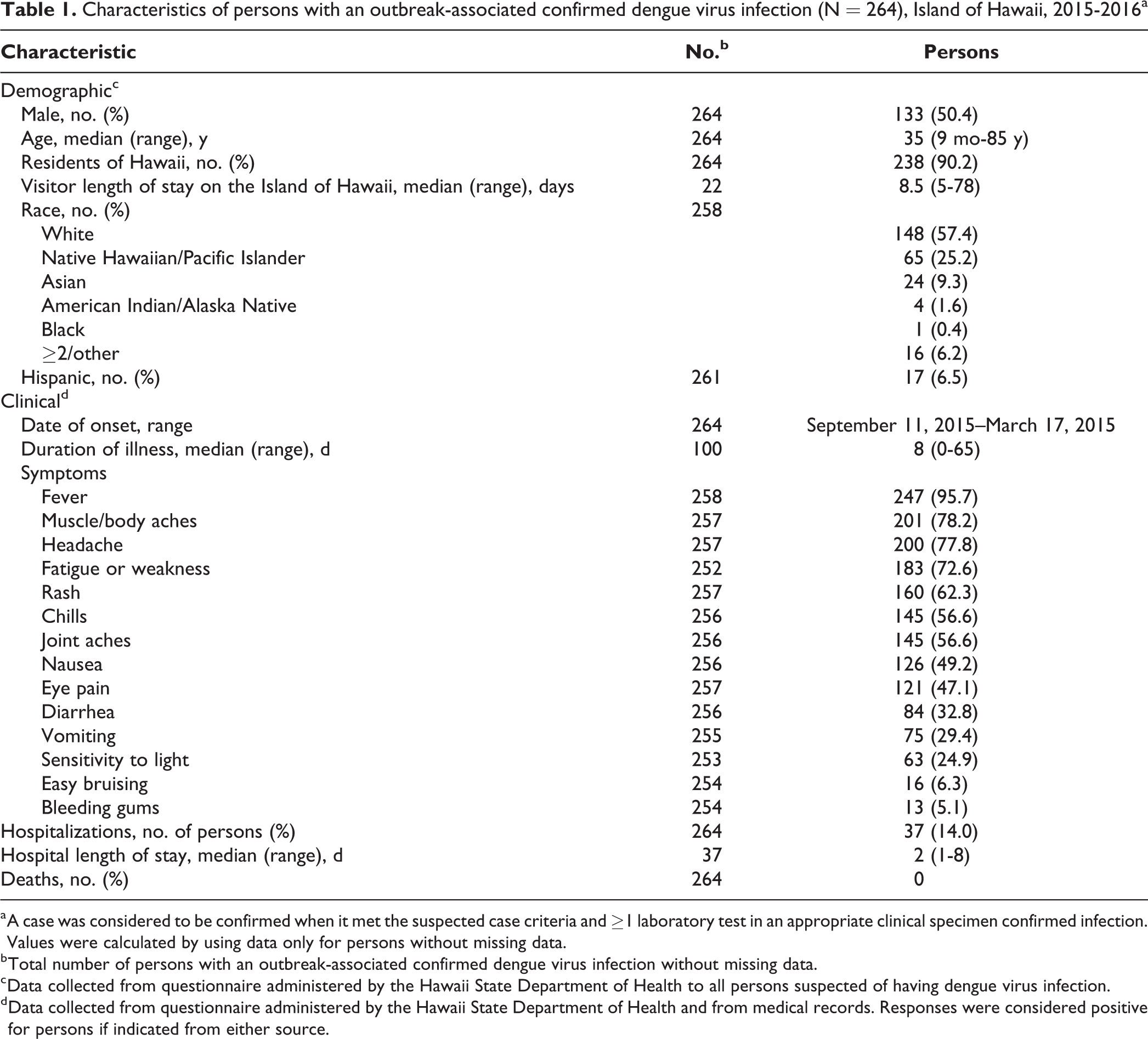
a A case was considered to be confirmed when it met the suspected case criteria and ≥1 laboratory test in an appropriate clinical specimen confirmed infection. Values were calculated by using data only for persons without missing data.
b Total number of persons with an outbreak-associated confirmed dengue virus infection without missing data.
c Data collected from questionnaire administered by the Hawaii State Department of Health to all persons suspected of having dengue virus infection.
d Data collected from questionnaire administered by the Hawaii State Department of Health and from medical records. Responses were considered positive for persons if indicated from either source.
Of case-persons who reported symptoms, the most commonly reported symptoms were fever (247 of 258, 95.7%), muscle/body aches (201 of 257, 78.2%), headache (200 of 257, 77.8%), and fatigue or weakness (183 of 252, 72.6%). The median duration of illness was 8 days (range, 0-65 days). No cases of severe dengue or death were reported. Of all 264 confirmed cases, 37 (14.0%) required hospitalization, and the median hospital length of stay was 2 days (range, 1-8 days; Table 1).
One hundred fifteen of 243 (47.3%) case-persons reported noticing mosquitoes inside their home within 7 days before illness onset (Table 2). Most case-persons (209 of 241, 86.4%) had screened windows or doors on their homes, of which 81 (38.8%) reported having holes or tears in the screen. Also, 28.9% (74 of 246) of case-persons reported having sources of standing water on their property. In the 7 days before illness onset, 63 of 254 (24.8%) case-persons indicated using insect repellent, and 25 of 253 (9.9%) indicated wearing long sleeves and long pants when outdoors (Table 2).
Risk factors reported by persons with an outbreak-associated confirmed dengue virus infection (N = 264), Island of Hawaii, 2015-2016a,b

a Data collected from questionnaire administered by the Hawaii State Department of Health to all persons suspected of having dengue virus infection.
b A case was considered to be confirmed when it met the suspected case criteria and ≥1 laboratory test in an appropriate clinical specimen confirmed infection. Values were calculated by using data only for persons who responded to the question; not all respondents answered all questions.
c Total number of persons with an outbreak-associated confirmed dengue virus infection who responded to the question.
Mapping
Case-person residences and potential exposure locations were geographically distributed across the Island of Hawaii. At the census-block group level, Moran’s I demonstrated positive spatial autocorrelation in both case-person residences per 1000 population (Moran’s I = 0.41, z score = 11.02, P < .001) and reported potential exposure locations (excluding residences) per square mile (Moran’s I = 0.11, z score = 4.63, P < .001; ie, their distributions were clustered rather than randomly spread across the island). Hot-spot analysis found significant hot spots (z scores = 2.23-7.56; P value range, <.001-.01) of case-person residences per 1000 population on the western side of the Island of Hawaii and significant cold spots (z scores = –1.97 to –3.31; P value range, <.001-.01) on the eastern side. Significant hot spots (z scores = 2.84-3.63; P value range, <.001-.01) of reported potential exposures (excluding residences) were identified on the western side (Figure 2). During the course of the outbreak, case-persons consistently reported residences and potential exposures on the western side of the Island of Hawaii, although pockets of case-persons were identified elsewhere around the island (Figure 3).
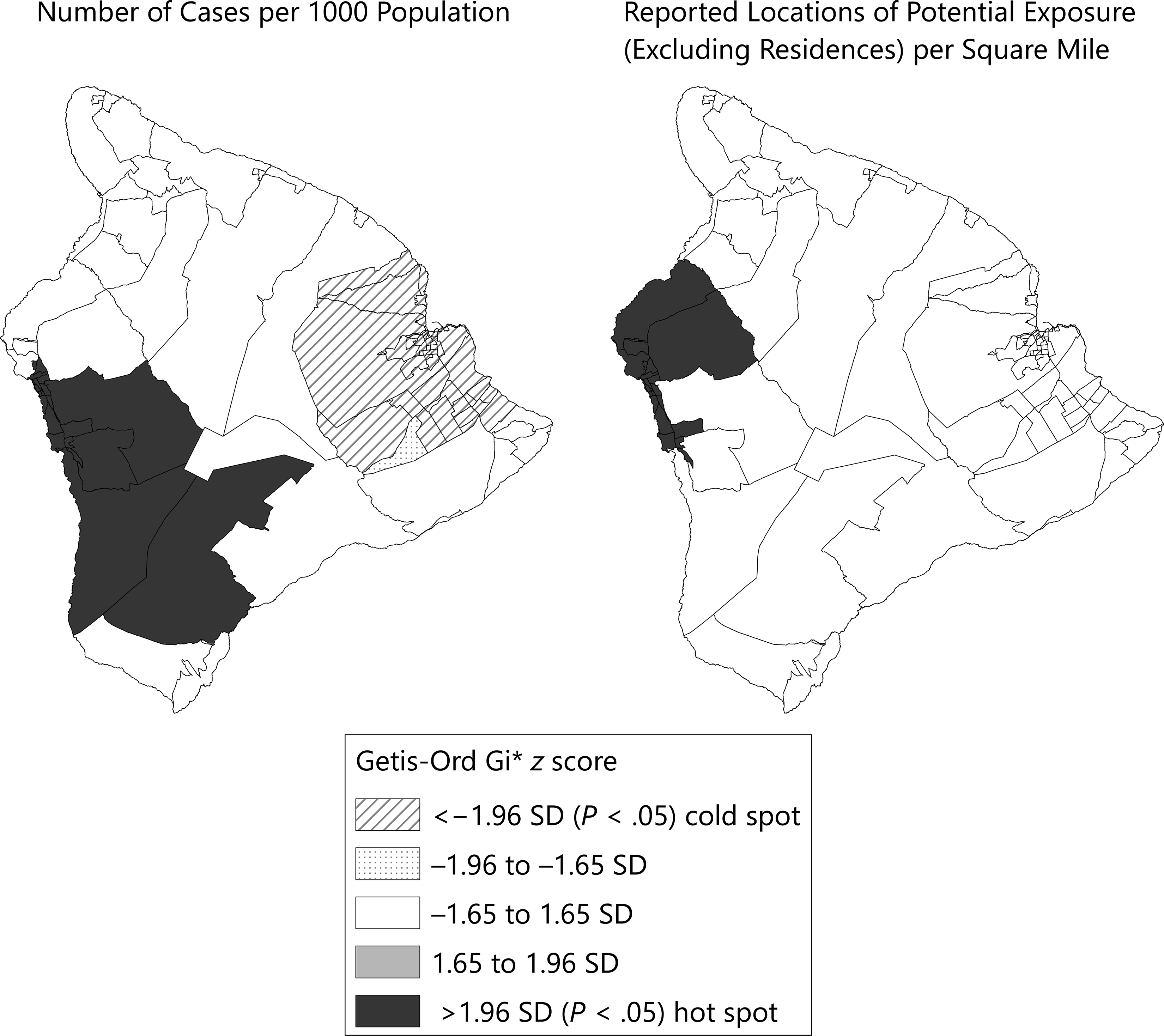
Hot-spot analysis (Getis-Ord Gi*) of residences of persons with outbreak-associated confirmed dengue virus infection and reported potential locations of exposure, by census-block group, Island of Hawaii, 2015-2016. Potential exposure locations are sites that case-persons reported visiting during the 3-14 days before illness onset. Abbreviation: SD, standard deviation.
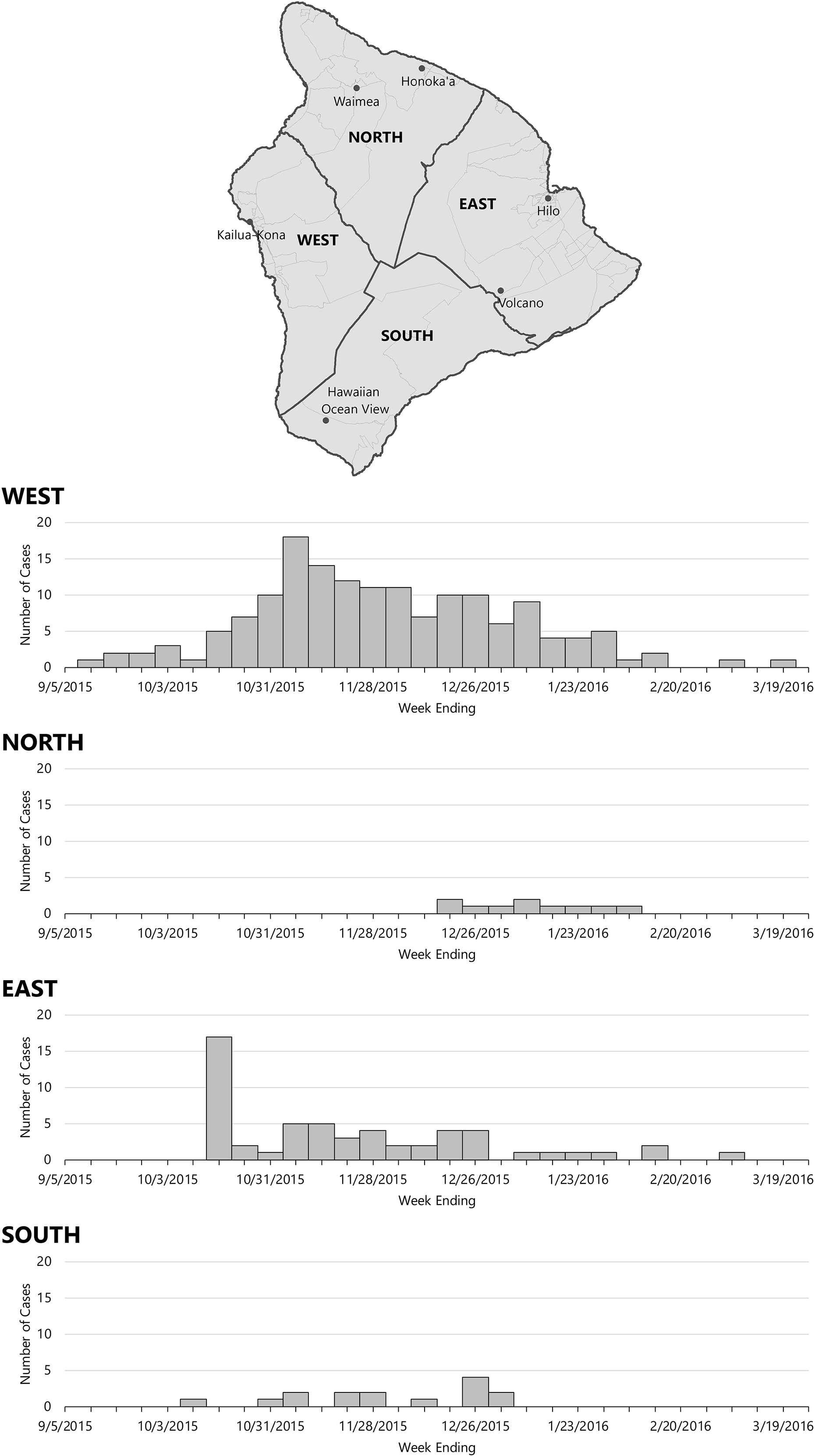
Onset dates of outbreak-associated confirmed dengue virus infection, by region of residence, Island of Hawaii, 2015-2016. Data collected from questionnaire administered by the Hawaii State Department of Health and from medical records.
Laboratory Results
Of the 264 outbreak specimens tested, 184 (69.7%) were tested by RT-PCR, and all 184 were identified as serotype DENV-1. Sequencing results were available for 6 case-person specimens. These case-persons were ill at various points in time, from the start through the end of the outbreak, and varied in exposure histories and locations. Per a report from the CDC Dengue Branch, sequencing demonstrated that the specimens were closely related as a genotype originating in Asia. However, an exact area of origin was not identified (CDC Dengue Branch, unpublished written and oral communication, March 2017).
Vector Control
Not all owners allowed VCS members access to their properties or allowed adulticide spraying. Reasons for refusal included a lack of understanding of the intent or purpose of the abatement, organic farms concerned about endangering their organic certification, potential negative effects on apiaries, and other apprehensions (VCS, unpublished written and oral communication, February 2017). Resource constraints prevented data on refusals from being collected systematically. Therefore, only qualitative data were available.
Discussion
Since 1944, endemic dengue in Hawaii has been eliminated through aggressive and widespread mosquito control, involving numerous personnel (the response to the 1943-1944 outbreak required the help of nearly 1 person per 1000 population 9 ) and DDT, a pesticide that is now recognized as having detrimental effects on the environment and on persons and is banned in the United States. 11,18 Today, such mosquito eradication measures, using alternative pesticides, would be costly in terms of personnel and resources, challenging to execute, and could have substantial negative effects on Hawaii’s plants and animals. Focusing efforts on behavioral and cultural changes in attitudes, such as shifting persons’ perceptions about mosquitoes from one of a nuisance to being disease carriers or emphasizing the importance of practicing personal protection from mosquitoes and community-level mosquito control, would seem more feasible than focusing on mosquito eradication to address mosquito-borne disease risk. 19 -21 However, effecting these changes is an ongoing challenge and may be more effective as a prevention strategy than as an outbreak response. Targeting response activities, such as mosquito abatement, with the aid of GIS methods may prove timelier and more effective during an outbreak response through improving situational awareness.
Genetic sequencing has suggested that this outbreak resulted from a single imported introduction. GIS analyses revealed general patterns of outbreak progression in consistent hot spots on the western side of the island. Because the eastern side of the island has higher annual amounts of rainfall than the western side of the island, it would be reasonable to anticipate a higher concentration of disease vectors and, in turn, a higher level of disease activity on the eastern side. 22 However, the western side is more densely populated, and much of the island’s tourist and other economic industries are also focused there; some of the largest increases in daytime populations are related to commuting. 23 -25 Drier-than-normal weather may facilitate persons spending more time outdoors, potentially contributing to an increase in mosquito exposure. Other potential factors that would affect the type and concentration of mosquitoes present, including altitude, amount of vegetation, and land use, could also affect mosquito exposure risk.
Knowledge of how to prevent mosquito exposure did not appear to correlate with the practice of preventing mosquito exposure. Our findings suggested that although persons may know how to protect themselves from mosquitoes, they do not necessarily practice these behaviors, thereby greatly increasing their exposure risk. Differences in socioeconomic status, geography, or other factors may also affect this discordance. Further study is required to more fully evaluate these factors.
Limitations
Our outbreak investigation and response had several limitations. First, precise identification of transmission areas was complicated by the 14-day potential exposure window, which increased the likelihood of inaccuracies in the recall of potential exposure locations among case-persons. Case-persons may have been unaware of when mosquito bites occurred or if they encountered Aedes or another genus, adding more uncertainty. Second, not having a control group with which to compare reported risk factors of case-persons meant we were unable to determine whether an association with infection existed for any behavior. Third, VCS member efforts to limit exposure risk through mosquito abatement were limited by some owners denying access to properties. Finally, an extended assessment of Aedes (Stegomyia) mosquitoes on the Island of Hawaii had not been conducted in more than 6 years 26 ; as such, knowledge of prevalence and relative density of the species in affected areas was lacking. Because of the lack of an active vector-control program, data on Aedes species distribution were unavailable during the outbreak, and this lack of data limited our ability to focus response activities where needed.
Conclusions
Although dengue is not endemic in Hawaii, given the number of visitors to Hawaii each year (8.7 million visitors in 2015 compared with 1.4 million population 27,28 ), the number of residents traveling worldwide, and the presence of competent vectors, Hawaii is constantly at risk for the introduction of mosquito-borne diseases such as dengue. The failure to identify the index case of the outbreak highlights difficulties associated with identifying and preventing cases among incoming travelers. Using multiple prevention activities routinely would be more feasible and effective than relying solely on identifying and controlling an outbreak. Although we should attempt to effect a change in behavior and attitudes about mosquito control and ensure that personal protection is routinely implemented, we could also increase public education and outreach and focus mosquito eradication efforts. Enhanced outbreak response methods, such as GIS hot-spot analyses, could provide more accurate and timely tracking of outbreak progression, identify potential areas of transmission, and better target both education and vector control efforts. Ongoing surveillance is vital to identify the introduction of cases and understand areas that have a high risk of transmission. With the increasing range of Aedes (Stegomyia) mosquitoes worldwide, an increasing number of public health jurisdictions will need to consider more aggressive efforts to reduce the risk of mosquito-borne diseases.
Footnotes
Acknowledgments
The authors thank the following for their contributions to the response to the outbreak: Dengue Response Team (Hawaii State Department of Health: Hawaii Island District Health Office, Vector Control, Sanitation, Public Health Nursing, Administration, Epidemiology, Family Health Services, Chronic Disease Section, Public Health Preparedness, State Laboratories Division [Rebecca Kanenaka, Roland H. K. C. Lee, Remedios Gose, Derek Harauchi, Lei Inouye-Ching, Mark Nagata], Medical Reserve Corps; County of Hawaii: Civil Defense, Fire Department, Emergency Medical Services, Public Works, Parks and Recreation); CDC Dengue Branch Laboratory.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. However, staff and resources funded by the CDC Epidemiology and Laboratory Capacity (ELC) grant (ELC grant No. NU50CK000415, Epidemiology and Laboratory Capacity for Infectious Diseases) were used in the outbreak response.
