Abstract
Objectives:
Supplemental federal funding is allocated to state and local tuberculosis (TB) programs using a formula that considers only countable cases reported to the National Tuberculosis Surveillance System (NTSS). Health departments submit reports of cases, which are countable unless another (US or international) jurisdiction has already counted the case or the case represents a recurrence of TB diagnosed ≤12 months after completion of treatment for a previous TB episode. Noncountable cases are a resource burden, so in 2009, NTSS began accepting noncountable case reports as an indicator of program burden. We sought to describe the volume and completeness of noncountable case reports.
Methods:
We analyzed 2010-2014 NTSS data to determine the number and distribution of noncountable cases reported. We also surveyed jurisdictions to determine the completeness of noncountable case reporting and obtain information on jurisdictions’ experience in reporting noncountable cases. In addition, we prepared a hypothetical recalculation of the funding formula to evaluate the effect of including noncountable cases on funding allocations.
Results:
Of 54 067 TB case reports analyzed, 1720 (3.2%) were noncountable; 47 of 60 (78.3%) jurisdictions reported ≥1 noncountable case. Of 60 programs surveyed, 34 (56.7%) responded. Of the 34 programs that responded, 24 (70.6%) had not reported all their noncountable cases to NTSS, and 11 (32.4%) stated that reporting noncountable cases was overly burdensome, considering the cases were not funded.
Conclusions:
Complete data on noncountable TB cases help support estimates of programmatic burden. Ongoing training and a streamlined reporting system to NTSS can facilitate noncountable case reporting.
The Centers for Disease Control and Prevention’s (CDC’s) National Tuberculosis Surveillance System (NTSS) 1 is the repository of tuberculosis (TB) surveillance data from throughout the United States. NTSS receives TB case reports electronically from 60 reporting areas (50 US states, the District of Columbia, New York City, and 8 US-affiliated island areas) through a 6-page standard data collection form, the Report of Verified Case of Tuberculosis (RVCT). 2
The RVCT allows CDC to obtain an accurate count of US TB cases and to describe epidemiologic characteristics of patients. In addition, other items on the RVCT allow CDC to evaluate data on diagnostic testing, risk factors, treatment initiation, and completion, which can identify aspects of the quality of care and the difficulty of case management for these TB patients. RVCT data are particularly important in capturing the burden of noncountable TB cases because no other source for this information exists.
Supplemental federal funding to state and local jurisdictions is allocated on the basis of the relative programmatic burden as measured by data reported on the RVCT and ancillary data sources. 3 The factors considered by the funding formula are reevaluated periodically to ensure that the formula continues to accurately approximate relative programmatic burden among the funded jurisdictions.
A verified case of TB reported in NTSS must meet the joint Council of State and Territorial Epidemiologists’ TB surveillance case definition. 4 Cases of verified TB disease are included in the official NTSS case counts unless they are noncountable. 2 A case is noncountable if (1) it was counted by another US reporting area (cases already counted in 1 US reporting area among persons who have moved to another jurisdiction cannot be counted again as a separate case), (2) the patient received TB treatment initially in another country and therefore is presumed to have been counted in that country, or (3) the patient had recurrent TB disease ≤12 months after completing TB treatment (in the United States, these cases are considered a continuation of the originally reported TB disease episode).
Funding of state and local TB programs comes from multiple sources, including state appropriations, reimbursements from private and government-sponsored health insurance, and supplemental federal funding appropriated by Congress and disbursed by CDC. Federal funding supports state and local prevention and control activities, laboratory capacity, and human resource development. CDC allocates federal funding to each jurisdiction on the basis of a proportional formula that considers the official NTSS TB case count in that jurisdiction and the number of cases that had certain characteristics reported on the RVCT that are associated with greater costs for case management. CDC also incorporates 2 performance measures by using data reported on the RVCT as part of the funding formula for prevention and control activities. These performance measures evaluate the effectiveness of the jurisdiction in interrupting disease transmission and ensuring treatment with an adequate regimen of anti-TB drugs. 5 -9
State and local TB program staff members have expressed concern that resources spent on managing noncountable TB cases are not represented in the funding formula. An analysis conducted by CDC staff members determined that the cost to the health department of treating noncountable TB cases was $2000-$3000 per noncountable case. 10 That study did not provide an estimate of the cost of treating counted cases; however, the estimated direct medical cost in 2014 dollars for a non–multidrug-resistant (resistant to at least isoniazid and rifampin) TB case was $17 000. 11 To better track the programmatic burden of noncountable cases without inflating official case counts by double-counting cases, CDC revised the RVCT in 2009 to allow reporting of noncountable TB cases. CDC’s cooperative agreement with state and local TB programs does not distinguish between countable and noncountable TB cases in its surveillance data reporting requirements. 12
In 2015, the RVCT Quality Assurance work group and the TB data users group, with representatives from selected reporting jurisdictions, requested an evaluation of the completeness of noncountable TB case reporting in NTSS. These groups wanted to determine the financial burden for TB control programs of treating TB among these patients, along with patients with suspected TB, to guide them in improving resource allocation. We sought to evaluate the completeness and accuracy of noncountable case reports and ascertain the experiences of NTSS reporting areas with reporting noncountable cases to identify any reporting barriers.
Materials and Methods
We analyzed data on noncountable TB cases reported to NTSS from January 1, 2010, through December 31, 2014, by using SAS version 9.3. 13 For 1 reporting area, which included in its noncountable case reports out-of-state cases diagnosed at its regional laboratory, we limited noncountable cases to those patients who were actually managed in the state. We calculated the total number of TB cases reported to NTSS during the study period and the number and proportion of those cases that were countable vs. noncountable. For noncountable cases, we determined the frequency of each of the 3 reasons that a case could be noncountable, as reported on the RVCT. In addition, we determined the number of noncountable cases that were multidrug resistant and the reasons for the case being noncountable for each category.
Survey
We also distributed a questionnaire via email during spring 2015 to the surveillance data coordinators of each reporting area. We followed up by email and telephone to confirm reported data. The objectives of the questionnaire were (1) to define the program’s number of noncountable TB cases, (2) to identify the number and proportion of each type of noncountable TB case not already reported to NTSS, (3) to identify barriers or challenges to reporting noncountable TB data to NTSS, and (4) to obtain input for improving reporting.
To reduce the burden of response on the reporting jurisdictions, we asked only 4 questions in the questionnaire. What are your challenges and barriers to reporting noncountable cases to CDC? Which of the following approaches would you prefer for reporting noncountable TB cases? A simplified/condensed version of the RVCT The full RVCT form with all questions as asked for countable cases Other approach (respondents were asked to describe their preferred approach) If we are able to develop a simplified form for reporting, please rank the following according to importance in determining your TB burden on a scale of 1-5, with 1 = most important and 5 = least important: Diagnostic test results (eg, sputum culture) Risk factor data (eg, history of homelessness) Case management data (eg, initial drug regimen) Outpatient health care provider (eg, health department, private provider) Other data (respondents were asked to specify what other data are important) Please provide the number of noncountable TB cases in your current system for each of these noncountable case types: Verified cases counted in another US area Verified cases but treatment initiated in another country Verified cases but had recurrent TB within 12 months after completion of therapy Other (respondents were asked to describe the type of noncountable cases being reported)
For each reporting jurisdiction, we included a table showing the number of noncountable cases received by CDC for each year of the analysis and a blank table in which the reporting area could specify the number of noncountable cases in its reporting system if its number of cases by year differed from what was reported by CDC. We also allowed respondents to add their own comments in free text.
Funding Allocation
The absolute amount of CDC’s funding for state and local TB programs is established by congressional appropriation; however, CDC can alter the funding formula that governs how funds are allocated to each program (eg, by including noncountable cases in the formula). We estimated the effect on potential funding allocations to TB programs if noncountable TB cases were included in national TB funding formula calculations in which the total funding amount was not changed. 5 -9 We considered a scenario in which countable and noncountable cases were included in the federal fiscal year 2017 funding formula. The funding formula assigns weights to each component on the basis of needs or performance and considers each jurisdiction’s most recent 3-year average counts. The cases that are most expensive to treat and that provide the most funding per case are multidrug resistant. CDC reviewed this project and determined it to be a public health evaluation activity; as such, institutional review board approval was not required.
Results
Reported Noncountable TB Cases, 2010-2014
Of 54 067 verified TB cases reported to NTSS during 2010-2014, 1720 (3.2%) were noncountable (Table 1), ranging from 303 in 2011 to 393 in 2014. The percentage of noncountable cases of TB varied among the reporting areas. For 202 of 1720 (11.7%) cases, no reason for being noncountable was reported (Table 2). Of 1518 cases with reported reasons for being noncountable, 1106 (72.9%) were counted by another US jurisdiction, 328 (21.6%) had initiated treatment outside the United States, and 84 (5.5%) had recurrent TB ≤12 months after completion of initial TB therapy.
Case reporting of noncountable tuberculosis (TB), United States, 2010-2014a
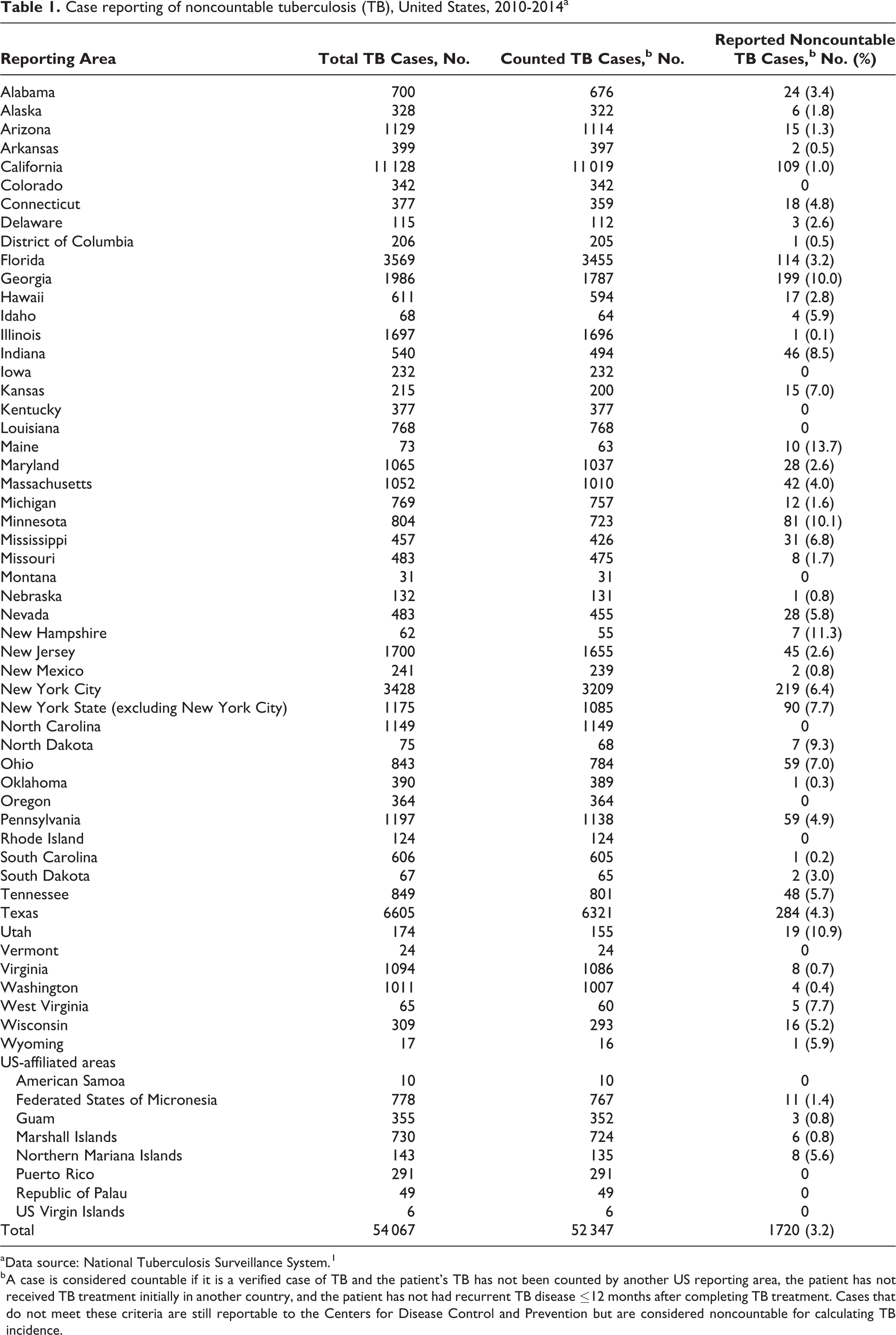
a Data source: National Tuberculosis Surveillance System. 1
b A case is considered countable if it is a verified case of TB and the patient’s TB has not been counted by another US reporting area, the patient has not received TB treatment initially in another country, and the patient has not had recurrent TB disease ≤12 months after completing TB treatment. Cases that do not meet these criteria are still reportable to the Centers for Disease Control and Prevention but are considered noncountable for calculating TB incidence.
Noncountable tuberculosis (TB)a cases, by category and selected risk factors, United States, 2010-2014b
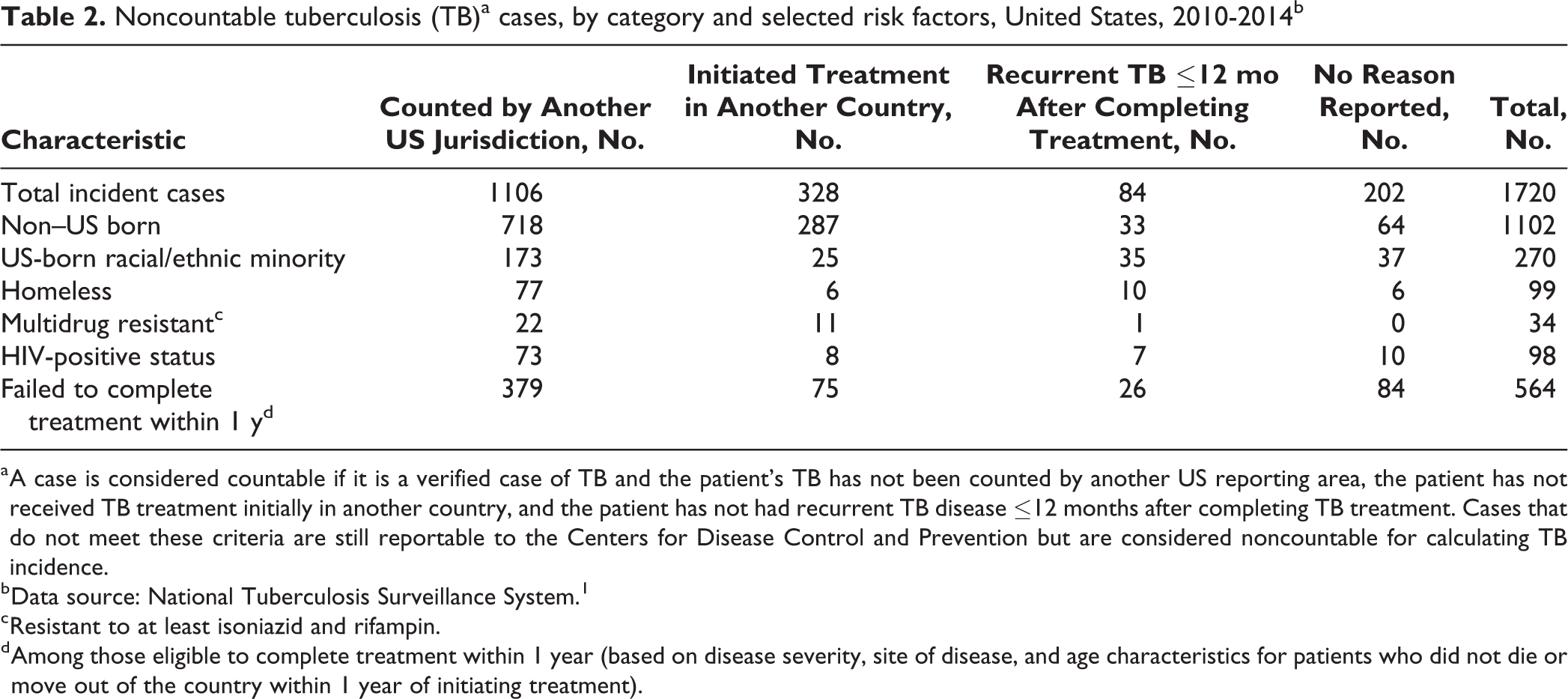
a A case is considered countable if it is a verified case of TB and the patient’s TB has not been counted by another US reporting area, the patient has not received TB treatment initially in another country, and the patient has not had recurrent TB disease ≤12 months after completing TB treatment. Cases that do not meet these criteria are still reportable to the Centers for Disease Control and Prevention but are considered noncountable for calculating TB incidence.
b Data source: National Tuberculosis Surveillance System. 1
c Resistant to at least isoniazid and rifampin.
d Among those eligible to complete treatment within 1 year (based on disease severity, site of disease, and age characteristics for patients who did not die or move out of the country within 1 year of initiating treatment).
On the basis of drug-susceptibility test results reported on the RVCT, 34 noncountable TB cases were multidrug-resistant TB. Of the 34 noncountable multidrug-resistant TB cases, 22 (64.7%) were counted by another US jurisdiction, 11 (32.4%) had initiated treatment outside the United States, and 1 (2.9%) had recurrent TB (Table 2). These noncountable multidrug-resistant TB cases accounted for 34 of 552 (6.2%) verified multidrug-resistant TB cases during the study period.
Survey Results
From the 60 reporting areas, we received 34 (56.7%) survey responses. Of 31 respondents who provided the number of noncountable cases in their local surveillance databases, 24 (77.4%) had more noncountable cases in their systems than were submitted to NTSS (range, 1-132; median, 35). When addressing challenges and barriers to reporting noncountable cases to CDC, some respondents indicated that they believed that CDC did not require reporting noncountable cases as part of the cooperative agreement. Other respondents attributed underreporting to a lack of resources to submit the reports to CDC. Still others stated the need for training when reporting data on noncountable cases of TB.
Overall, 11 of 34 reporting areas commented that completing all data elements of the RVCT for noncountable cases was excessively time-consuming, especially the reporting on patients who had moved from another US reporting area because of the delay in receiving information from the transferring area. Eighteen (52.9%) respondents recommended allocation of additional CDC funding to defray the cost of managing noncountable TB cases.
Seventeen (50.0%) reporting areas supported creating a simplified reporting form for noncountable cases instead of completing the full RVCT. Among possible data elements to include on a streamlined noncountable case reporting form, respondents ranked case management data as most important, followed by diagnostic test results, TB risk factor data, and type of patient health care provider.
Funding Formula Analysis
After calculating the 2017 funding allocations while including noncountable cases in the funding formula, 13 recipients would receive increased funding (ranging from an increase of 1.2%-17.1%), 17 recipients would have approximately level funding (from a 0.9% increase to a 0.9% decrease), and 31 would have received less funding (from a decrease of 1.1%-3.4%).
Discussion
To our knowledge, our study is the first analysis of noncountable TB cases in the United States since NTSS began collecting data for these cases in 2009. We determined that reporting of noncountable cases varied by jurisdiction; some programs did not report noncountable cases at all, and other programs did not report all noncountable cases. Among jurisdictions that did report noncountable cases, not all of the requested data were available to include on the RVCT, as noted by certain programs that responded to the survey. A lack of communication with the state from which the case originated or incomplete data available for those patients who had initiated treatment in another country were some of the explanations provided in the survey. Differences in TB incidence among reporting areas were not associated with the reporting frequency of noncountable cases.
Although relatively rare in the United States, a single occurrence of multidrug-resistant TB has been estimated to impose a direct cost to the public sector of $134 000 per case. 14 If such a case were to be noncountable and therefore not included in the funding formula, which is designed to help offset the extreme cost of managing a multidrug-resistant TB case, the ability of the affected program to provide TB services could be severely strained.
The major reason jurisdictions gave for not reporting noncountable cases was the perception that it was not cost-effective to expend the time needed to complete and submit the RVCT for noncountable cases when these case reports would not affect subsequent funding allocations. We also identified multiple barriers to reporting noncountable TB cases. We grouped these responses into categories that included software concerns, incomplete documentation from other jurisdictions, limited resources, and other challenges. Our analysis determined that inclusion of noncountable cases in the CDC funding formula would alter the funding allocations for most NTSS reporting areas, although the degree of the change in funding varied.
Limitations
This study had several limitations. First, the 57% response rate to the questionnaire may have limited the generalizability of the survey results. Second, the hypothetical funding formula scenario used the actual noncountable cases reported to NTSS. If all reporting areas consistently and completely reported noncountable cases, the effect on the funding allocations could have differed.
Practice Implications
Reporting of all verified TB cases to NTSS, both countable and noncountable, is necessary for capturing the true burden of TB and providing high-quality data for TB surveillance. Complete data on noncountable TB cases can help guide estimates of resources used by health departments to manage these cases (eg, providing dates that cases were managed in each jurisdiction to facilitate estimates of duration of treatment burden). Consideration of additional funding for managing these cases can provide incentives for noncountable case reporting. Ongoing training 15 -17 and a streamlined reporting system to NTSS might also motivate TB control partners to report noncountable cases and provide a more complete picture of the extent of TB treatment and case management in the United States.
Footnotes
Acknowledgments
The authors thank the following for their valuable input: Thomas Navin, MD; Carla Winston, PhD; Clarisse Tsang, MPH; and C. Kay Smith, MEd, from the Centers for Disease Control and Prevention (CDC); and Ms. Cynthia Adams, CDC Information Management Systems Contractor.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This evaluation was conducted by CDC employees using intramural CDC funding.
