Abstract
Objectives:
Opioid-related overdoses are commonly attributed to prescription opioids. We examined data on opioid-related overdose decedents in Massachusetts. For each decedent, we determined which opioid medications had been prescribed and dispensed and which opioids were detected in postmortem medical examiner toxicology specimens.
Methods:
Among opioid-related overdose decedents in Massachusetts during 2013-2015, we analyzed individually linked postmortem opioid toxicology reports and prescription drug monitoring program records to determine instances of overdose in which a decedent had a prescription active on the date of death for the opioid(s) detected in the toxicology report. We also calculated the proportion of overdoses for which prescribed opioid medications were not detected in decedents’ toxicology reports.
Results:
Of 2916 decedents with complete toxicology reports, 1789 (61.4%) had heroin and 1322 (45.3%) had fentanyl detected in postmortem toxicology reports. Of the 491 (16.8%) decedents with ≥1 opioid prescription active on the date of death, prescribed opioids were commonly not detected in toxicology reports, specifically: buprenorphine (56 of 97; 57.7%), oxycodone (93 of 176; 52.8%), and methadone prescribed for opioid use disorder (36 of 112; 32.1%). Only 39 (1.3%) decedents had an active prescription for each opioid detected in toxicology reports on the date of death.
Conclusion:
Linking overdose toxicology reports to prescription drug monitoring program records can help attribute overdoses to prescribed opioids, diverted prescription opioids, heroin, and illicitly made fentanyl.
In the United States in the 1990s and early 2000s, annual increases in opioid-related overdose deaths and entries into treatment for opioid addiction paralleled increases in prescriptions of opioid medications for pain. 1 This correlation appears to have been causal, as the expansion of opioid prescribing for pain led to more persons overdosing on opioids and more persons seeking treatment for opioid use disorder. 2,3 Since 2010, opioid prescribing leveled and then declined. 4 However, the number of opioid-related overdose deaths has surged, largely driven by increases in the use of heroin and illicitly made fentanyl in states such as Florida, 5,6 Kentucky, Maryland, Massachusetts, 7,8 New Hampshire, Ohio, 6 Rhode Island, 9 -11 and West Virginia. 12,13 Heroin and illicit fentanyl have become increasingly available at the same time as the most commonly used prescription opioids (eg, oxycodone and hydrocodone) have been prescribed less and become more difficult to obtain legally. 14,15 As overdose deaths attributed to fentanyl and law enforcement seizures of illicitly made fentanyl have surged, fentanyl prescription rates have remained relatively low and stable. 12
Before the widespread availability of illicitly made fentanyl, several state- and county-level studies linked data from prescription drug monitoring programs (PDMPs) and postmortem toxicology reports. The studies reported that ≥60% of overdose deaths involved prescription opioids and one-third or more of the decedents had filled a prescription for an opioid within 2 months before the overdose death. 3,16 -20 A 2012 study of overdose deaths in North Carolina found that 74% of unintentional overdoses involved prescription opioids, and 47% of decedents whose postmortem toxicology report indicated the presence of oxycodone had received a prescription for oxycodone within 30 days before death. 17 A study of prescription-related deaths in San Diego in 2013 found that 39% of decedents had received prescriptions for the opioids found in the toxicology reports within 2 months before death. 18 A similar study of overdoses in Kentucky in 2013 and 2014 found that 60% of overdoses involved prescription opioids and 33% of persons who had an overdose had ≥1 opioid prescription filled, with the supply covering the day of death. 19 Lastly, a study of overdoses in Tennessee during 2013-2016 found that 68% of opioid-related overdoses involved prescription opioids, and 42% of decedents had an active opioid prescription on the day of the overdose death. 20
Medical examiners in many jurisdictions do not precisely or consistently use postmortem opioid toxicology reports to determine causes of overdose deaths. 21 -25 Even though heroin overdose has its own International Classification of Diseases, Tenth Revision (ICD-10) code (T40.1), 26 heroin is rapidly metabolized by the body to morphine, and heroin-specific metabolites are not commonly detected in toxicology specimens at death. Thus, heroin-related overdoses may be miscoded as ICD-10 T40.2 (natural and semi-synthetic opioid-related overdose), a category that should include pharmaceutical morphine, oxycodone, and other pharmaceutically produced opioids. 22,23,25,27 Furthermore, fentanyl overdoses are classified under ICD-10 code T40.3 (synthetic opioids, excluding methadone), which does not distinguish pharmaceutical fentanyl from illicitly made fentanyl on death certificates. Thus, fentanyl-related deaths may be classified as “prescription opioid–related,” although evidence from multiple states demonstrated that the type of fentanyl that is causing an increase in overdoses is made outside the pharmaceutical fentanyl distribution system (ie, illicitly made). 7,12,13
Misclassification of overdoses caused by heroin and illicitly made fentanyl has important policy implications. Policy makers may too narrowly focus efforts on preventing the misuse of prescription opioids and devote inadequate resources to addressing heroin and illicit fentanyl use. Massachusetts was an early adopter of safer opioid prescribing education, legislative requirements for checking PDMPs, restrictions on large-volume prescribing, and, more recently, strict limits on the days’ supply permitted with initial opioid prescriptions. 28 Massachusetts also expanded distribution of naloxone rescue kits in the community 29 and increased the number of providers of medication for opioid use disorder, including methadone, buprenorphine, and naltrexone, especially through community health centers. 30 Despite these efforts, which fulfill the key public health strategies recommended by the federal government, 31 the number of opioid-related deaths in Massachusetts tripled from 656 in 2011 to 2050 in 2017. 8
To better understand these trends, the Massachusetts Legislature, through Chapter 55 of the Acts of 2015, permitted the Massachusetts Department of Public Health (MDPH) to assemble a data warehouse of individually linked data sets. In this study, we examined data on persons in Massachusetts who died of an overdose to determine whether they were prescribed an opioid, which opioid they were prescribed, whether that medication was dispensed, and which opioids—prescribed or illicit—were detected in medical examiner toxicology specimens at death. By examining data from 2013-2015—the period during which illicitly made fentanyl was introduced into the opioid supply—we can better understand the relationship among prescription opioids, illicit opioids, and overdose deaths.
Methods
Population and Setting
We conducted a retrospective cohort study of Massachusetts residents aged ≥11 years who died of an opioid-related overdose from June 1, 2013, through December 31, 2015. Opioid-related deaths were defined by ICD-10 codes indicating unintentional or undetermined intentional poisoning (X40-X44, Y10-Y14) in the underlying cause-of-death field and an opioid-specific T code of T40.0-T40.4 and/or the narcotic T code T40.6 in any of the multiple cause-of-death fields.
Death certificates on fatal poisonings in Massachusetts are required by law to be reported through the single, centralized, statewide Office of the Chief Medical Examiner. We included data from the Chapter 55 database, which linked 16 administrative databases at the individual level and included 98% of the Massachusetts population aged ≥11 years. 32 Children aged <11 years were not included in the database because their risk of overdose is low. We used a multistep deterministic matching algorithm based on name, birth date, social security number, and street address. Chapter 55 of the Acts of 2015 permitted MDPH to analyze linked data from several Massachusetts government agencies to identify and report on trends among persons who died from an opioid-related overdose. Linkage across data sets at the individual level allowed analysts to understand the circumstances that influence opioid-related overdoses.
Our study focused on the following data sources from the Chapter 55 database: deaths recorded in the Registry of Vital Records and Statistics, postmortem toxicology reports of the Office of the Chief Medical Examiner, the PDMP, licensed treatment encounters recorded by the Bureau of Substance Addiction Services, and claims reported in the Massachusetts All-Payer Claims Database. We included decedents who died of opioid-related overdose from June 1, 2013, through December 31, 2015. Although the Chapter 55 database extends to January 1, 2011, toxicology reports were not consistently available before June 1, 2013, and linked data were not available after December 31, 2015.
Toxicology testing was conducted by the Massachusetts State Police Crime Laboratory or was sent to NMS Labs. Both laboratories ran routine substance-specific gas chromatography mass spectroscopy testing for 6-monoacetylmorphine (6-MAM), buprenorphine, codeine, hydrocodone, hydromorphone, morphine, oxycodone, methadone, and tramadol. We categorized samples that had 6-MAM or a combination of morphine and codeine in the same sample as “heroin.” We categorized samples with morphine detected, but no 6-MAM or codeine, as “morphine.” 27 We excluded decedents for whom the postmortem toxicology reports were available but incomplete. Toxicology reports may not have been complete because tissue samples were not available for testing or because the medical examiner did not order toxicology testing. This work was mandated by Massachusetts law and conducted by a public health authority; as such, institutional review board review was not required. 32
We searched for all Schedule II-V opioid drug products in the PDMP, including oxycodone, hydrocodone, hydromorphone, morphine, codeine, buprenorphine, methadone, tramadol, meperidine, and oxymorphone. We did not include in our analyses data on meperidine or oxymorphone from the PDMP or postmortem toxicology reports because we did not find any prescriptions or toxicology reports for meperidine among decedents during the study period and <5 persons had a prescription for oxymorphone within 1 year before their death. Oxymorphone was indicated on toxicology reports, but oxymorphone is a metabolite of oxycodone, routinely detected among persons taking oxycodone, 33 and was thus counted as oxycodone. For patients treated with methadone for opioid use disorder, information was available by month and determined by (1) a claim for methadone administration from the Massachusetts All-Payer Claims Database (code HCPCS H0020) or (2) indication of treatment in a methadone maintenance program through the state’s Bureau of Substance Addiction Services. Data were linked among databases via the All-Payer Claims Database.
We defined a case of an overdose related to a prescribed opioid as an overdose in which a decedent had ≥1 prescription that was active on the date of death for the opioid indicated in the postmortem toxicology report. To determine “active on the date of death,” we used the fill date and the days’ supply fields in the PDMP. For those decedents with an active prescription on the date of death, we calculated the mean daily morphine milligram-equivalent on the date of death. We distinguished between methadone prescribed for pain found in the PDMP and methadone administered for opioid use disorder found in the All-Payer Claims Database or Bureau of Substance Addiction Services. We defined an overdose related to administered methadone for opioid use disorder as an overdose in which a decedent had methadone in a postmortem toxicology report and was enrolled in a methadone maintenance program during the month before death. We also calculated the proportion of overdoses for which opioids prescribed in the PDMP were not detected in toxicology specimens on the date of death. We determined an opioid to be “not detected” if a person filled a prescription with days’ supply overlapping the date of death but the toxicology report at death did not include that opioid.
Analysis
We calculated descriptive statistics to characterize the cohort and the pool of decedents from which the cohort was derived. We determined what proportion of the decedents had active opioid prescriptions within 12 months before death, 6 months before death, 3 months before death, 1 month before death, 1 week before death, and on the date of death. We calculated the proportion of decedents for whom the opioids indicated on the postmortem toxicology report were also the opioids prescribed at the time of death. We calculated the frequency that each substance was detected in toxicology specimens at death individually and in combination with other opioids. For the combinations, we reported fentanyl, heroin, and other opioids into mutually exclusive categories. We calculated the proportion of decedents who had an active prescription at the date of death. We also calculated the frequency with which a prescription for an opioid overlapped the date on which the decedent died and whether these prescribed opioid medications were indicated in the postmortem toxicology report. We conducted all analyses by using SAS Studio version 3.5. 34
Results
Of 3710 Massachusetts residents in the Chapter 55 database who died of an opioid-related overdose during the study period, 2916 (78.6%) had complete postmortem toxicology reports. The mean and median ages were lower among decedents who had complete toxicology results (mean, 39.4 years; median, 38.0 years) than among decedents who did not have complete toxicology results (mean, 40.5 years; median, 41.0 years), and most decedents in each group (87.0% and 86.0%, respectively) were non-Hispanic white (Table 1).
Characteristics of Massachusetts residents aged ≥11 years who died from an opioid-related overdose, June 1, 2013, through December 31, 2015a
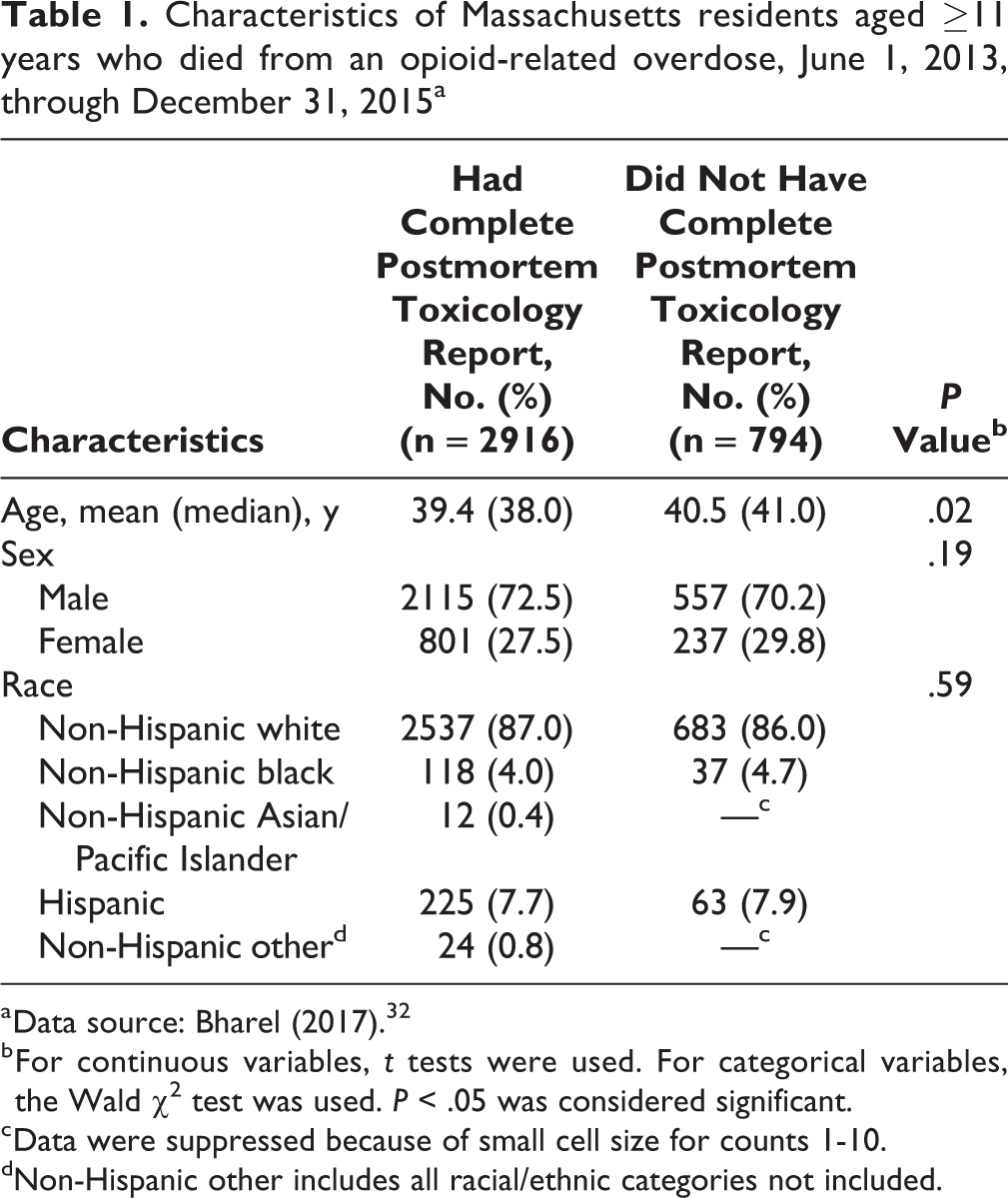
a Data source: Bharel (2017). 32
b For continuous variables, t tests were used. For categorical variables, the Wald χ2 test was used. P < .05 was considered significant.
c Data were suppressed because of small cell size for counts 1-10.
dNon-Hispanic other includes all racial/ethnic categories not included.
Of the 2916 decedents with a complete report, prescription opioids were detected in the postmortem toxicology specimens of 1171 (40.2%). The percentage of decedents with an active opioid prescription in the PDMP database decreased as the time between active opioid prescription and death decreased: 57.6% (n = 1681) at 12 months, 45.0% (n = 1311) at 6 months, 35.7% (n = 1041) at 3 months, 26.8% (n = 781) at 1 month, 20.6% (n = 600) at 1 week, and 16.8% (n = 491) on the date of death.
Of the 2916 decedents with a complete report, oxycodone was the most commonly prescribed opioid that was active on the date of death (n = 176; 6.0%), followed by methadone administered for opioid use disorder in a methadone maintenance program (n = 112; 3.8%), buprenorphine (n = 97; 3.3%), and prescribed methadone (n = 38; 1.3%) (Table 2). Tramadol, morphine, hydrocodone, and hydromorphone were prescribed and dispensed to <1% of decedents who died of an opioid-related overdose. Among decedents with an active opioid prescription, excluding buprenorphine, the mean daily morphine milligram-equivalent was 134 mg (median, 90 mg) on the date of death. Only 1.3% (n = 39) of decedents had an active prescription for each opioid indicated in their postmortem toxicology report.
Decedents who had ≥1 active opioid prescription at and before opioid-related overdose death among 2916 residents aged ≥11 years who died of an opioid-related overdose and had a complete postmortem toxicology report in Massachusetts, June 1, 2013, through December 31, 2015a
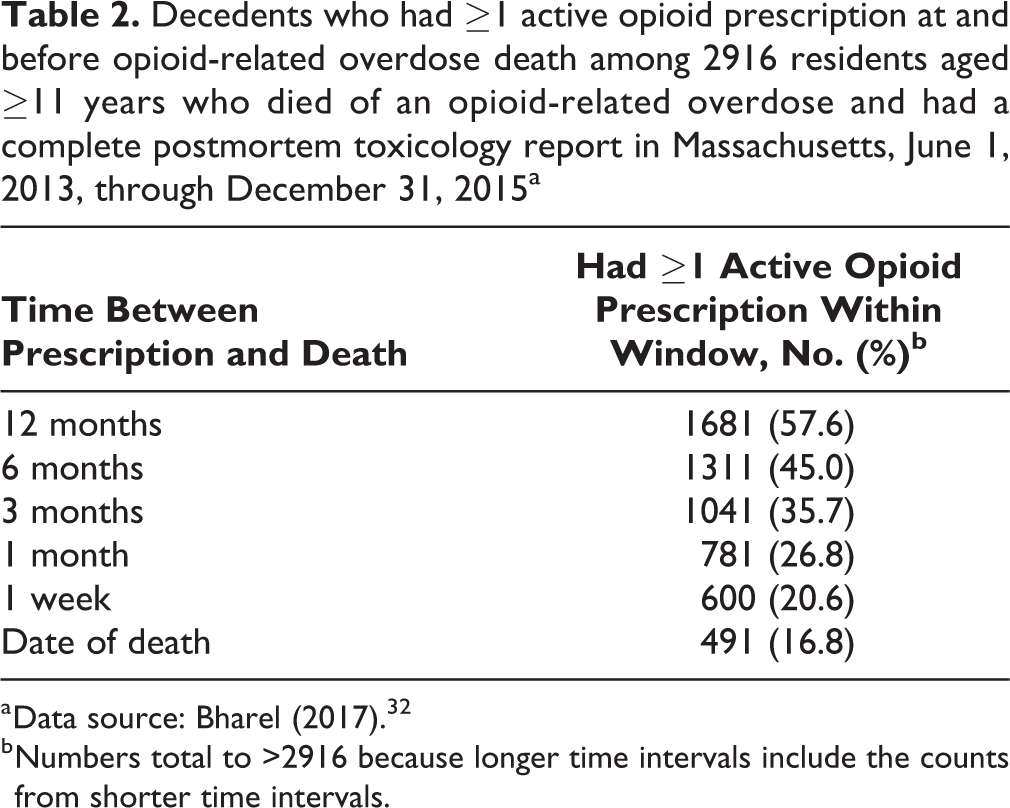
a Data source: Bharel (2017). 32
b Numbers total to >2916 because longer time intervals include the counts from shorter time intervals.
The most commonly detected opioids in toxicology reports were heroin (n = 1789; 61.4%) and fentanyl (n = 1322; 45.3%) (Table 3). Prescription opioids without heroin or fentanyl were indicated in 481 (16.5%) toxicology reports, and prescription opioids with heroin, fentanyl, or both were indicated in 690 (23.7%) toxicology reports.
Decedents whose postmortem toxicology reports indicated presence of opioids individually or in combination among 2916 residents aged ≥11 years who died of an opioid-related overdose and had a complete postmortem toxicology report in Massachusetts, June 1, 2013, through December 31, 2015a
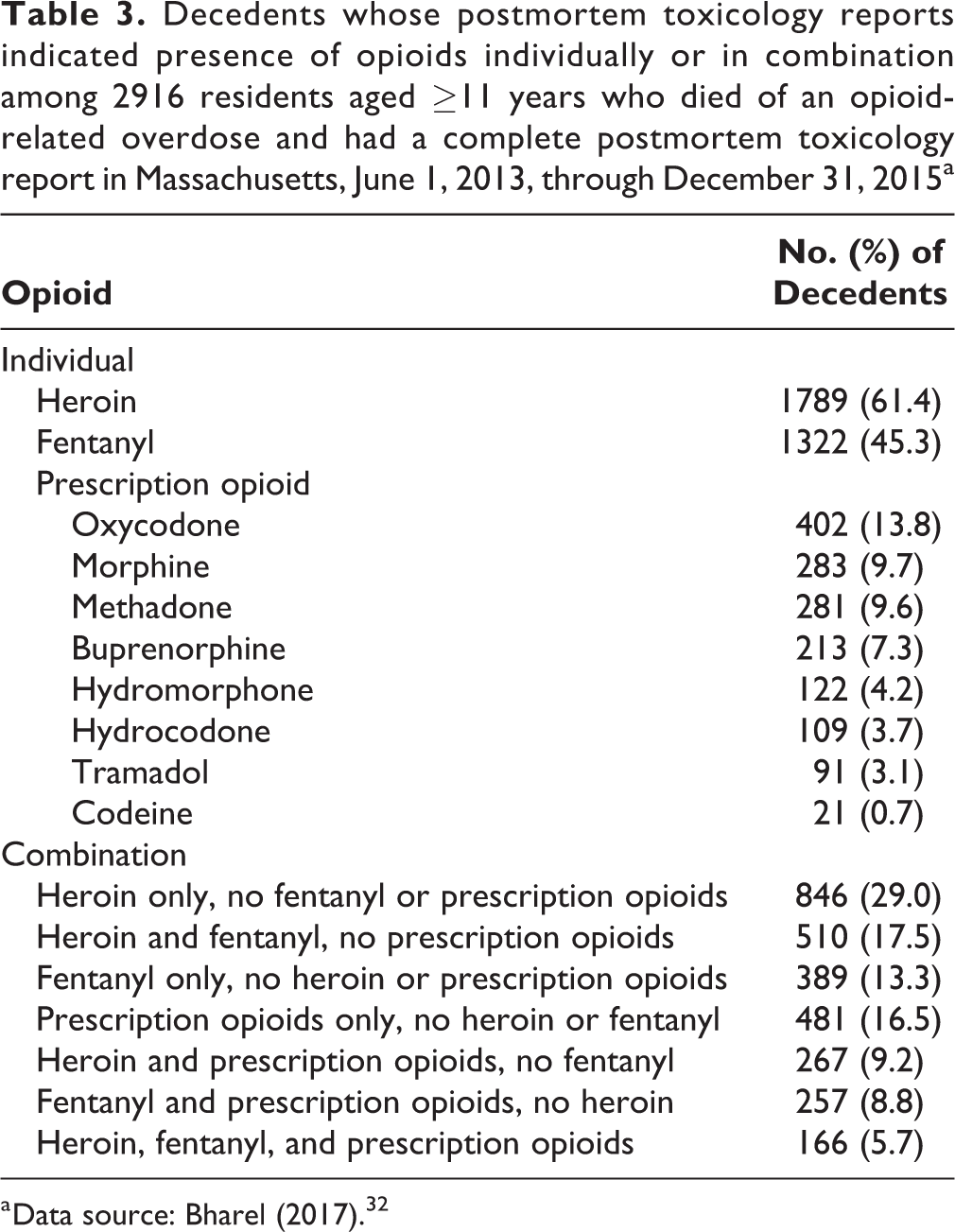
a Data source: Bharel (2017). 32
Eighty-three of 402 (20.6%) decedents with oxycodone and 41 of 213 (19.2%) decedents with buprenorphine indicated in postmortem toxicology reports had an active prescription on the date of death (Table 4). Thirty-three of 281 (11.7%) decedents with methadone indicated in postmortem toxicology reports had an active prescription for methadone to treat pain on the date of death, and 76 (27.1%) decedents were being treated for opioid use disorder in a methadone maintenance program within the month of death. Only 17 of 1322 (1.3%) decedents with fentanyl indicated in postmortem toxicology reports had an active prescription for fentanyl on the date of death.
Decedents whose postmortem toxicology reports indicated presence of opioids and percentage with ≥1 active opioid prescription on the date of death among 2916 residents aged ≥11 years who died of opioid-related overdose and had a complete postmortem toxicology report, Massachusetts, June 1, 2013, through December 31, 2015a
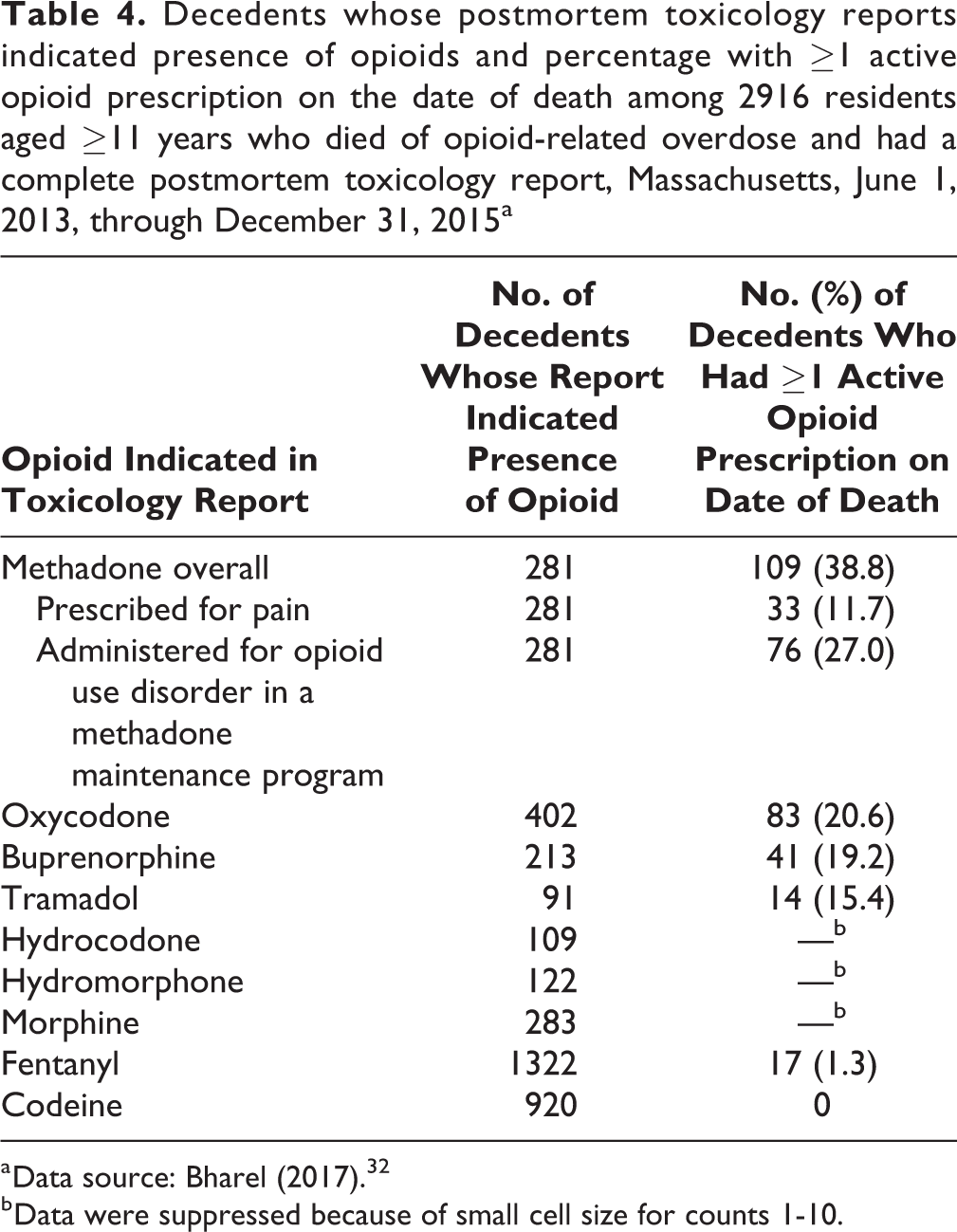
a Data source: Bharel (2017). 32
b Data were suppressed because of small cell size for counts 1-10.
Opioids that were actively prescribed at the time of death were commonly not detected in the toxicology specimen, indicating that the decedent had not taken the prescribed medication on or a few days before the date of death. Prescribed opioid medications were not detected in toxicology specimens as follows: 57.7% (56 of 97) of buprenorphine, 52.8% (93 of 176) of oxycodone, and 32.1% (36 of 112) of methadone administered for opioid use disorder in a methadone maintenance program (Table 5).
Opioid prescriptions on the date of death and percentage of opioids not present in postmortem toxicology reports of decedents who died of opioid-related overdose, Massachusetts, June 1, 2013, through December 31, 2015 (n = 2916)a
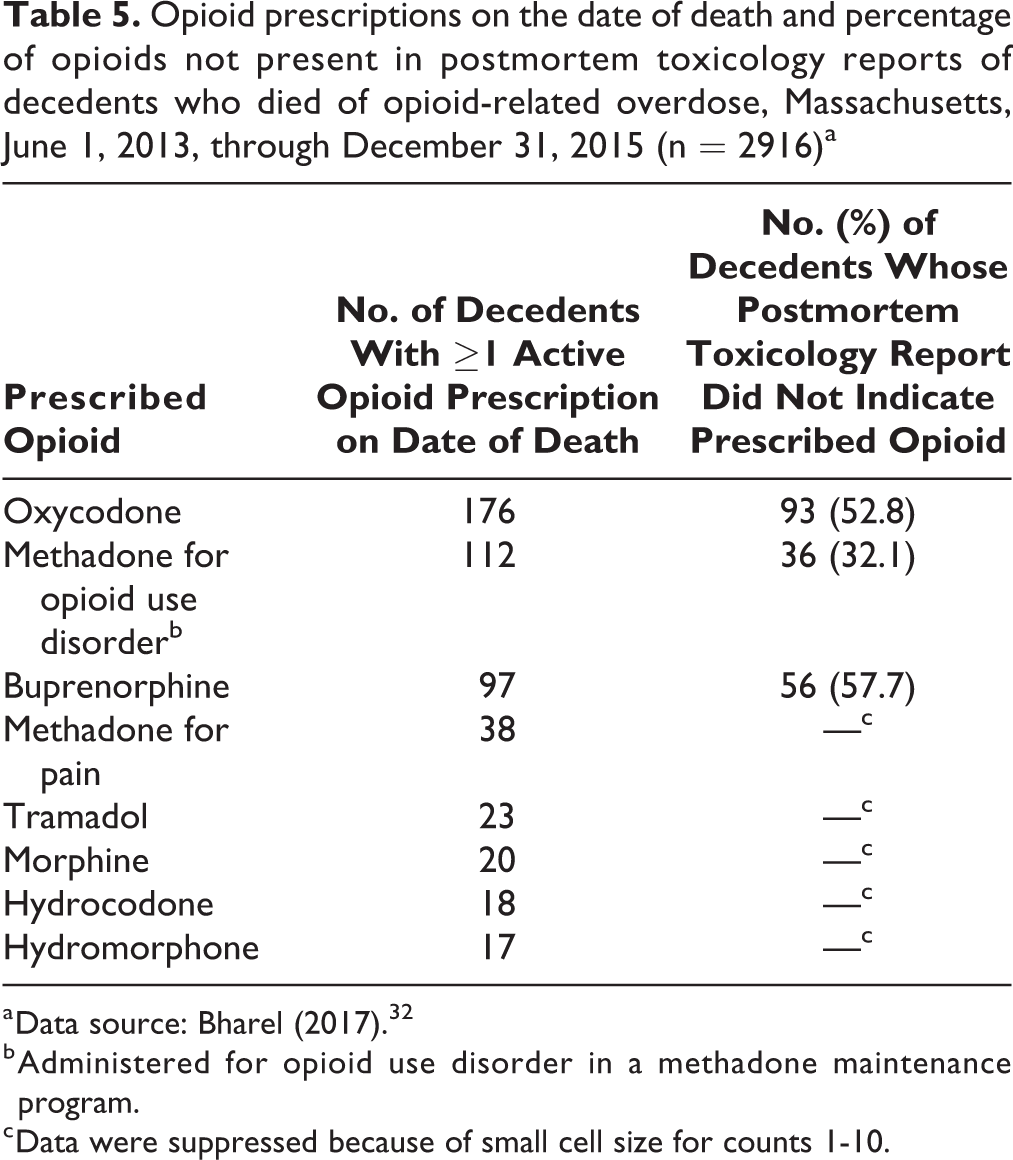
a Data source: Bharel (2017). 32
b Administered for opioid use disorder in a methadone maintenance program.
c Data were suppressed because of small cell size for counts 1-10.
Discussion
This study was conducted during a time when the incidence of fentanyl-involved deaths was increasing. Illicit opioids—heroin, fentanyl, or both—were involved in most opioid-related overdose deaths. 7,8 Prescription opioids were detected in postmortem toxicology reports of fewer than half of the decedents; when opioids were prescribed at the time of death, they were commonly not detected in postmortem toxicology reports.
In Rhode Island, a bordering state with substantial fentanyl exposure, a study of 536 overdose deaths occurring from 2012 through 2014 found that only 13% of deaths involved fentanyl. 10 In our study, during an overlapping time period but 1 year later, we found a substantially higher proportion of postmortem toxicology specimens tested positive for fentanyl (45.3%; 1322 of 2916). A study of national opioid-related overdose rates in 2015 and 2016 found that deaths involving synthetic opioids other than methadone, which were primarily fentanyl-related, increased in every subgroup examined and accounted for 45.9% (n = 19 413 of 42 249) of all opioid-involved deaths in 2016, more than any other drug examined. 15 By the end of 2016, fentanyl was involved in more than 70% of opioid-related overdose deaths in Massachusetts. 8
Oxycodone was the most commonly prescribed opioid among persons who died of an opioid-related overdose and the third most common opioid detected in toxicology specimens, after heroin and fentanyl. Oxycodone is commonly used to treat chronic pain and is dispensed in high-dose formulations. About one-fifth of those who died and whose postmortem toxicology report indicated oxycodone had an active prescription for it; therefore, about four-fifths were taking oxycodone diverted from someone else or leftover from a previous prescription. Fewer than half of persons who died and had an active prescription for oxycodone had oxycodone in their postmortem toxicology report. Decedents who died of an opioid-related overdose and were prescribed oxycodone but did not have oxycodone detected in toxicology specimens may have lost their prescription, not taken it as directed, or diverted their prescribed oxycodone to others.
The next most commonly prescribed opioids were methadone and buprenorphine. We distinguished between methadone prescribed for pain and methadone administered for opioid use disorder. More than one-quarter of persons who died and whose postmortem toxicology report indicated methadone had received methadone administered for opioid use disorder within 1 month of death. We were not able to determine exactly when persons received their last dose of methadone within the month; therefore, this number could represent persons who were discharged from methadone maintenance treatment within the month. In other studies, few methadone-involved overdose deaths occurred while patients were actively receiving methadone treatment. 16,35,36 The immediate period after stopping methadone or buprenorphine treatment is well established as a particularly high-risk time for overdose death. 37
More than half of persons who were prescribed hydromorphone, oxycodone, or buprenorphine at the time of death did not have these opioids detected in their toxicology specimens, which may indicate that a person had lost the medication, not taken it as directed, or diverted it, and overdosed on another opioid, most likely fentanyl or heroin. For decedents who received methadone either for pain or for addiction treatment, more than half had methadone indicated in their postmortem toxicology specimens. This greater relative presence of methadone in the toxicology report may be attributed to the fact that methadone has a longer duration of action and elimination half-life than most other opioids, and methadone may be less commonly diverted than prescription opioids because it is monitored more closely. 38 Among opioids, methadone is particularly dangerous when combined with other sedating medications. 39 However, when patients with opioid use disorder are treated with methadone or buprenorphine, mortality rates overall and from overdose are substantially reduced. 37
Other opioids—morphine, hydromorphone, hydrocodone, tramadol, and codeine—were not commonly indicated in toxicology reports and not commonly prescribed to persons who died of an overdose. As with fentanyl, <25 decedents had prescriptions for any of these medications at the time of death. Because hydrocodone is the most commonly prescribed short-acting outpatient pain medication, it was surprising how infrequently persons died with an active prescription.
In Massachusetts, prescribed opioids do not appear to be the major proximal cause of opioid-related overdose deaths. However, prescriptions may introduce persons to opioids, leading to a subsequent opioid use disorder that transitions to use of illicit opioids. Thus, prescribed opioid medications may exert their influence as a gateway drug or via diversion, but they alone do not cause many fatal overdoses among the persons to whom they are prescribed. Legal requirements instituted in 2016 included limiting opioid prescriptions for acute pain to 7 days and checking the PDMP before prescribing Schedule II and Schedule III opioids. Substantial decreases in opioid prescribing statewide were observed starting in 2015, before the new legal requirements, and continued through 2018. 9
Limitations
This study had several limitations. First, we did not incorporate opioid-specific data from death certificates because they were not systematically available or consistent. Second, toxicology reports were not available for 794 (21%) decedents during the study period; as such, our data do not apply to these decedents. Third, data on methadone maintenance were available only at the month level; as such, we could not determine whether decedents were stable on methadone or had stopped taking it within the month. Fourth, opioid prescriptions obtained from out-of-state pharmacies would not be included in the Massachusetts PDMP unless a pharmacy delivered the medication via mail to the patient. Fifth, prescriptions would not be attributed to the decedent if identifying data reported to the PDMP were not accurate, either intentionally (because of patient behavior) or unintentionally (because of pharmacy error). Sixth, we focused on prescribed opioids and opioid toxicology reports and did not include non-opioid substances (eg, cocaine, benzodiazepines, or alcohol), so conclusions about polysubstance use beyond opioids could not be drawn from this study. Lastly, emerging non-prescribed opioids (eg, carfentanil, furanyl fentanyl) were not routinely tested for and, thus, were not reported during the study period.
Conclusions
The major proximal contributors to opioid-related overdose deaths in Massachusetts during the study period were illicitly made fentanyl and heroin. Efforts to address opioid-related overdose death should focus on educating, engaging, and treating persons who use illicitly made fentanyl and heroin. The effect of prescribed opioid exposure appears to be more distal rather than proximal to overdose risk. Linking overdose toxicology reports to PDMP records can help better attribute overdoses to prescribed opioids, diverted prescription opioids, heroin, and illicitly made fentanyl.
Footnotes
Authors’ Note
Preliminary data from this project were presented at the College on Problems in Drug Dependence 2017 Annual Conference in Montreal, Quebec, Canada. The authors acknowledge the Massachusetts Department of Public Health for creating the unique cross-sector database used for this project and for providing technical support for the analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Larochelle was supported by award no. K23 DA042168 from the National Institute on Drug Abuse and a Boston University School of Medicine Department of Medicine Career Investment Award.
