Abstract
During 2014-2016, the largest outbreak of Ebola virus disease (EVD) in history occurred in West Africa. The New York City Department of Health and Mental Hygiene (DOHMH) worked with health care providers to prepare for persons under investigation (PUIs) for EVD in New York City. From July 1, 2014, through December 29, 2015, we classified as a PUI a person with EVD-compatible signs or symptoms and an epidemiologic risk factor within 21 days before illness onset. Of 112 persons who met PUI criteria, 74 (66%) sought medical care and 49 (44%) were hospitalized. The remaining 38 (34%) were isolated at home with daily contact by DOHMH staff members. Thirty-two (29%) PUIs received a diagnosis of malaria. Of 10 PUIs tested, 1 received a diagnosis of EVD. Home isolation minimized unnecessary hospitalization. This case study highlights the importance of developing competency among clinical and public health staff managing persons suspected to be infected with a high-consequence pathogen.
The largest outbreak of Ebola virus disease (EVD) in history occurred in West Africa from 2014 through March 2016, resulting in more than 28 600 cases and 11 300 deaths. 1 In September 2014, a traveler from Liberia received a diagnosis of EVD in a Dallas, Texas, hospital; 2 health care workers who cared for the patient subsequently developed EVD. 2 This event emphasized the need for preparedness among health care workers and public health agencies and prompted a national effort to enhance surveillance for imported cases of EVD.
The New York City Department of Health and Mental Hygiene (DOHMH) began EVD preparedness activities in July 2014, after reports of the outbreak in West Africa. Efforts built upon existing partnerships with New York City health care facilities and emergency response agencies. 3,4 In early August 2014, DOHMH identified Bellevue Hospital Center as the city’s designated EVD treatment center. DOHMH worked closely with Bellevue and the New York City Fire Department Bureau of Emergency Medical Services to prepare for EVD cases, 3 leveraging lessons learned from the initial 2 patients with EVD treated in the United States. 5 New York State subsequently designated 3 additional hospitals to serve as EVD treatment centers in New York City 6 and mandated that all acute care facilities have the capacity to isolate and stabilize patients suspected of having EVD. 7 DOHMH developed and routinely updated a protocol for health department on-call physicians to use when evaluating suspected EVD cases and referring patients to a designated EVD treatment center.
This case study summarizes our approach to persons under investigation (PUIs) for EVD in New York City from July 1, 2014, through December 29, 2015.
Methods
We classified as a suspected EVD case any patient for whom a New York City provider had a concern about EVD, regardless of whether the patient had a clinical presentation or an epidemiologic risk factor consistent with EVD.
Case Definition of a Person Under Investigation for EVD
From July 1, 2014, through December 29, 2015, we classified suspected EVD cases as PUIs according to the Centers for Disease Control and Prevention’s (CDC’s) case definition. On July 28, 2014, a CDC alert established the initial case definition for a PUI: temperature ≥101.5°F, EVD-compatible symptoms (eg, headache, myalgias, vomiting, diarrhea, abdominal pain, or unexplained hemorrhage), and travel to an EVD-affected country within 21 days before illness onset. 8 EVD-affected countries with widespread transmission were Guinea, Liberia, and Sierra Leone. We retrospectively classified suspected cases as PUIs if they met the criteria but were reported from July 1 through July 27, 2014, before the case definition was created, and we continued to classify cases according to this definition through October 8, 2014. On October 9, 2014, after learning of an imported case of EVD in a patient in Dallas who initially presented with a temperature of 100.3°F, we revised the case definition temperature criterion to >100.0°F or self-report of fever and expanded the epidemiologic criteria to include contact with a patient with EVD within 21 days before illness onset. We used this case definition through December 29, 2015, when DOHMH ended active monitoring of travelers from EVD-affected countries.
Triage, Management, and Laboratory Testing of Patients With Suspected EVD
On October 25, 2014, DOHMH began actively monitoring persons arriving from EVD-affected countries and health care workers who had contact with a patient with confirmed EVD in the United States. 9 -11 Monitoring entailed daily telephone contact with these persons to inquire about their temperature and presence of symptoms potentially compatible with EVD. To manage this effort, DOHMH established an active-monitoring call center with staff members who contacted persons at least once daily for the maximum EVD incubation period of 21 days after their last exposure to an EVD-affected area. 10 CDC provided local jurisdictions with information on each traveler’s arrival date in the United States, airport of arrival, and level of risk of EVD exposure. If staff members at the DOHMH active-monitoring call center, health care providers at New York City facilities, or CDC Quarantine Station staff members at the John F. Kennedy International Airport (the main New York City airport receiving passengers from the affected areas) identified a case of suspected EVD, they contacted the DOHMH on-call physician through a telephone line staffed 24 hours per day. This direct access to DOHMH physicians existed before the EVD outbreak and continues to be used by New York City providers to report patients suspected to have illnesses due to reportable diseases, including EVD.
In late October 2014, after EVD was diagnosed in a New York City resident, 12 DOHMH augmented its existing on-call team, adding an additional physician dedicated to responding exclusively to EVD-related telephone calls received during nonbusiness hours; 23 DOHMH physicians with communicable disease experience participated in the EVD on-call rotation. DOHMH developed and continuously updated a protocol and ongoing trainings for on-call physicians that addressed the clinical presentation of EVD, initial triage of suspected cases, infection prevention and control, home monitoring, transport, testing, and notification of external agencies. DOHMH physicians determined whether suspected EVD cases met the PUI case definition based on signs and symptoms reported by individuals to active-monitoring call center staff members or reported by health care providers and managed next steps, including whether the PUI should be referred to an acute care facility for medical evaluation or monitored at home. For PUIs with mild illness who preferred to stay at home, DOHMH implemented a home-monitoring protocol to minimize the burden of infection prevention and control required for EVD on the health care system. When a PUI needed referral to an acute care facility, DOHMH directed the individual to 1 of 4 EVD treatment centers in New York City, when possible, and alerted the facility, arranged patient transport, made infection prevention and control recommendations, reviewed alternate diagnoses with hospital physicians, and, if indicated, arranged EVD testing through the DOHMH Public Health Laboratory. DOHMH followed all PUIs until, for individuals monitored at home, the symptoms resolved and, for patients who were hospitalized, the diagnosis of EVD was no longer under consideration. For each PUI, DOHMH made the decision to test for EVD in consultation with the hospital’s clinical team and based on clinical presentation and severity, epidemiologic risk, and need to move a patient out of isolation to optimize care.
The DOHMH Public Health Laboratory performed EVD testing using a CDC-developed Ebola virus real-time reverse transcriptase polymerase chain reaction (PCR) assay, with approval from CDC. 13 CDC conducted EVD testing for 1 New York City PUI. Health care providers collected 2 blood specimens at the time of testing. If the first specimen had a positive result by PCR, the Public Health Laboratory considered this a presumptive positive for Ebola and submitted the second specimen to CDC for confirmatory testing.
In general, if the clinical team at the hospital collected an initial specimen for EVD testing within 72 hours of symptom onset and the result was negative, DOHMH requested a subsequent specimen to rule out EVD because low virus levels early in the course of disease could yield a false-negative result in an EVD-infected person. If the clinical team collected the initial specimen 72 hours after illness onset, or if the patient was improving clinically and had an alternative diagnosis at the time of the initial negative result, DOHMH physicians did not always request a second specimen. For each PUI, DOHMH physicians recorded clinical descriptions reported by health care providers and laboratory diagnoses but did not conduct a medical record review. For PUIs without laboratory-based diagnoses, DOHMH defined categories of syndromes to classify PUIs by clinical presentation.
The DOHMH Institutional Review Board categorized this activity as public health surveillance that was nonresearch.
Outcomes
Surveillance
One hundred twelve persons in New York City met PUI criteria from July 1, 2014, through December 29, 2015. From July 1 through October 24, 2014, before active monitoring was instituted, health care providers reported 20 (18%) persons to DOHMH. After active monitoring was instituted in New York City, from October 25, 2014, through December 29, 2015, 5359 persons were monitored, of whom 92 (2%) met PUI criteria. Of these 92 persons, active-monitoring call center staff members determined that 69 (75%) persons had symptoms requiring referral to DOHMH physicians. The remaining 23 (25%) persons were reported directly to DOHMH physicians by CDC Quarantine Station staff members at John F. Kennedy International Airport (n = 2), New York City Fire Department Bureau of Emergency Medical Services (n = 6), and New York City Medical Providers (n = 15).
Epidemiologic Risk
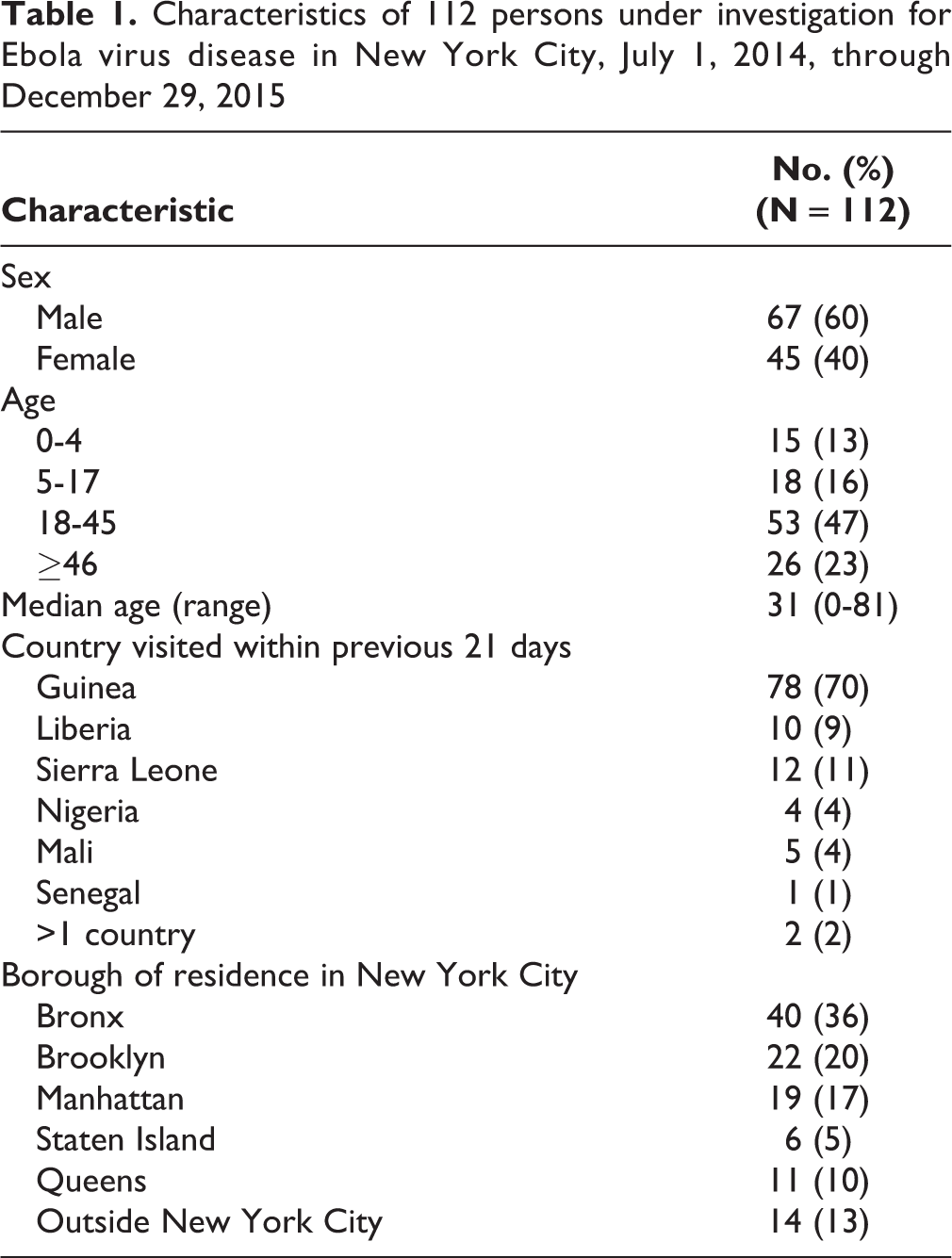
All 112 PUIs identified in New York City had traveled from EVD-affected countries; 78 (70%) had traveled from Guinea (Table 1). Only 1 PUI had direct exposure to a patient with known EVD while in a country with widespread transmission; this person (the 1 confirmed case of EVD diagnosed in New York City) had an elevated risk of EVD from serving as a health care worker in Guinea, where the person had had direct contact with patients diagnosed with EVD while wearing appropriate personal protective equipment (PPE). None of the New York City health care workers caring for the person with EVD in New York City developed signs or symptoms compatible with EVD.
Characteristics of 112 persons under investigation for Ebola virus disease in New York City, July 1, 2014, through December 29, 2015
Clinical and Laboratory Findings
Of the 112 persons meeting criteria for PUI, 74 (66%) sought medical care and 38 (34%) with mild illness did not seek medical care and were monitored by DOHMH at home. Of the 74 who sought medical care, 49 (44%) required hospitalization, including 13 patients treated in an intensive care unit (ICU). Thirty-five (47%) PUIs who sought medical care were first examined at 1 of 4 New York State–designated EVD treatment centers in New York City. The remaining 39 (53%) PUIs who sought medical care were first examined at 21 outpatient facilities, emergency departments, or hospitals throughout New York City. The 38 PUIs who did not seek medical care were managed in home isolation, and DOHMH staff members contacted them daily by telephone until illness resolved. Of these, 26 patients reported a single episode of elevated temperature; some reported drinking a hot beverage before taking their temperature or not knowing how to use the digital thermometer provided upon arrival in the United States. Repeat measurements for these 26 PUIs yielded normal temperatures.
Clinical presentation of PUIs varied from a single elevated temperature without additional signs and symptoms to critical illness. The most common presenting sign or symptom among the 112 PUIs was fever, which was present in 90 (80%) persons; other common symptoms included headache and weakness (Table 2). The most common laboratory-based diagnosis was malaria, which was identified in 32 of 112 (29%) PUIs, of whom 28 were hospitalized and 9 received treatment in an ICU. However, most PUIs did not have a laboratory-diagnosed illness; these PUIs were aggregated by clinical syndrome, based on presenting signs and symptoms (Table 3).
Presenting signs and symptoms among 112 persons under investigation for Ebola virus disease in New York City, July 1, 2014, through December 29, 2015
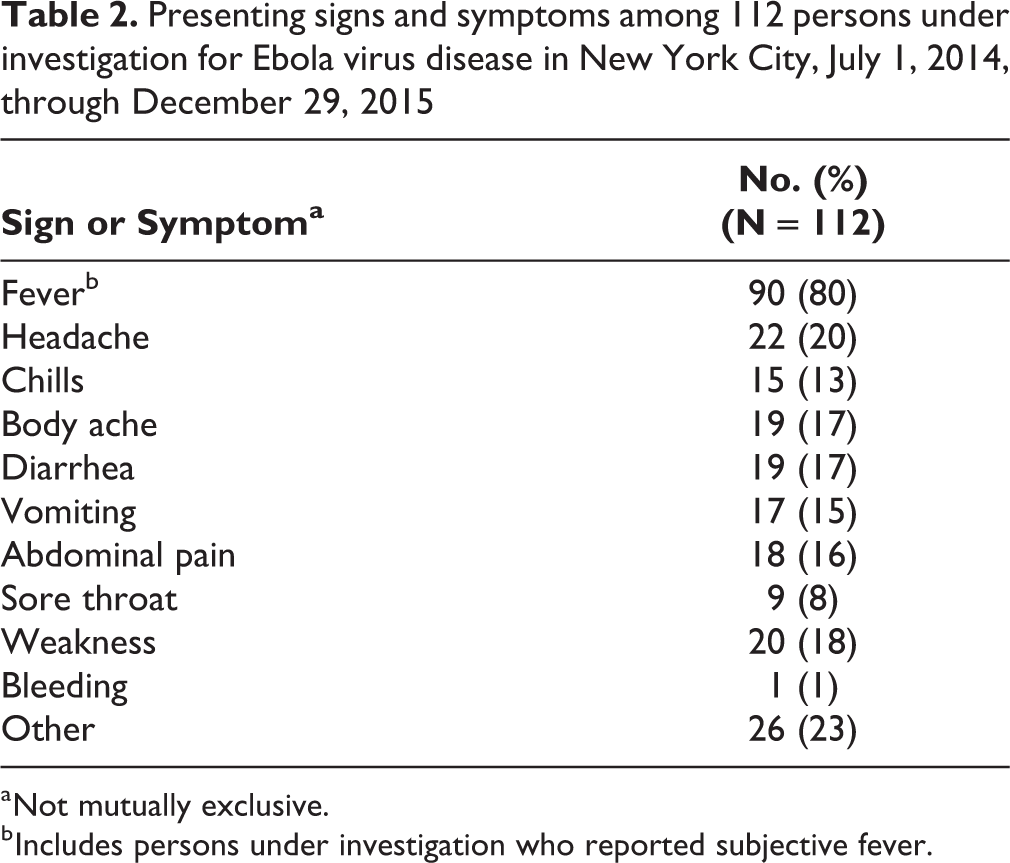
a Not mutually exclusive.
b Includes persons under investigation who reported subjective fever.
Syndromic illness and diagnoses of 112 persons under investigation for Ebola virus disease (EVD) in New York City, July 1, 2014, through December 29, 2015
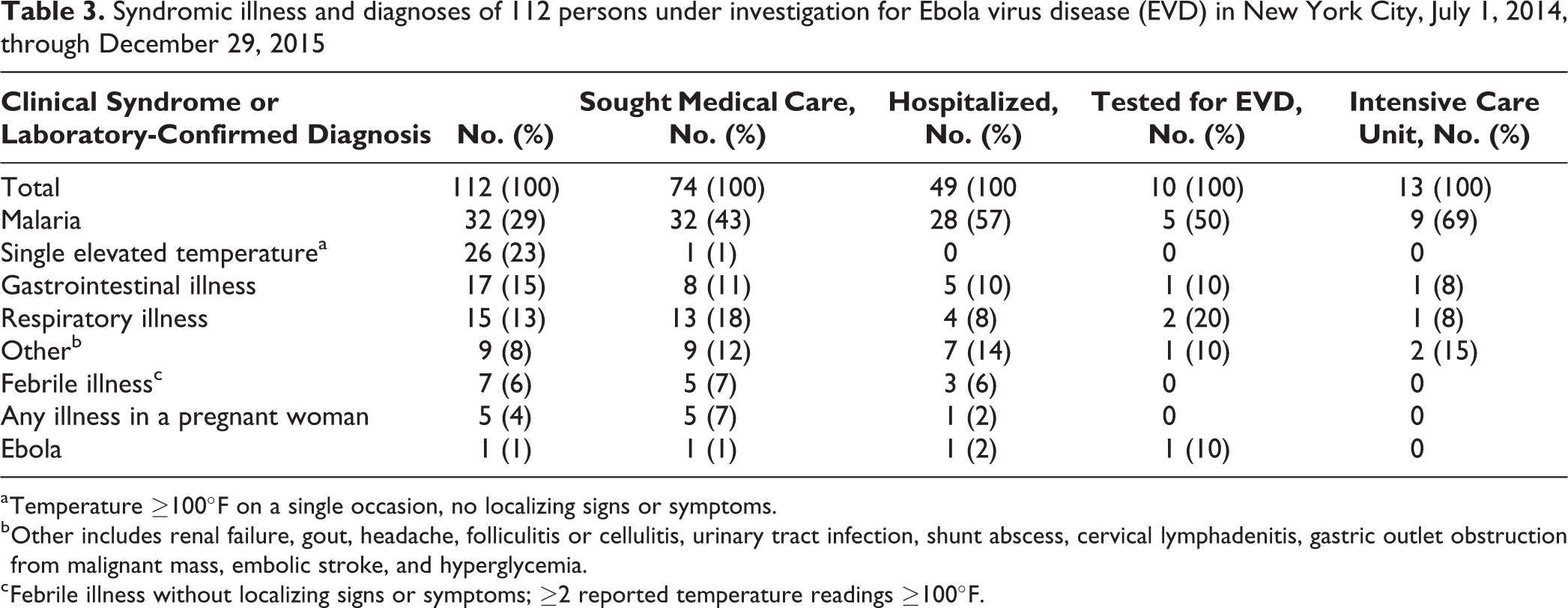
a Temperature ≥100°F on a single occasion, no localizing signs or symptoms.
b Other includes renal failure, gout, headache, folliculitis or cellulitis, urinary tract infection, shunt abscess, cervical lymphadenitis, gastric outlet obstruction from malignant mass, embolic stroke, and hyperglycemia.
c Febrile illness without localizing signs or symptoms; ≥2 reported temperature readings ≥100°F.
Ten of 11 New York City patients who had EVD testing met PUI criteria. One person who received EVD testing despite not meeting the PUI criteria died suddenly within 21 days after returning from an EVD-affected country; no EVD-compatible symptoms preceded the death, but the DOHMH Public Health Laboratory performed testing because of heightened concern about EVD among travelers to EVD-affected countries and the inability to collect information about potential exposures while in the affected country. CDC tested specimens from 1 individual before the test assay was available at the DOHMH Public Health Laboratory; subsequently, the DOHMH Public Health Laboratory tested specimens from 10 others, with confirmatory testing performed at CDC for the 1 patient who received a diagnosis of EVD.
Case Reports: Hospitalized PUI Cases
Several PUIs had illnesses that required medical management using equipment not readily available in high-containment EVD treatment units. Use of PPE by health care providers had the potential to introduce barriers to and delays in care because donning and doffing PPE was time-consuming and required a trained observer to be present. 14 The following case reports illustrate challenges related to PUI management in health care facilities.
Case 1
In December 2014, a 58-year-old man had confusion and diarrhea 2 days after arriving in New York City from Liberia. An ambulance unit was dispatched after his friend found him in distress and surrounded by vomitus and stool. On examination, his blood pressure was 99/71 mm Hg, heart rate was 116 beats per minute, and respiratory rate was 28 breaths per minute. The patient’s skin was pale and cool, and he showed rapid, shallow breathing. He was responsive only to painful stimuli. After transport to a designated EVD treatment center, he was taken immediately to the EVD treatment unit. Laboratory testing revealed severe acidosis and a positive malaria antigen, with 3.5% Plasmodium falciparum parasitemia on thin smear. He was managed with mechanical ventilation for airway protection and received antimalarial medication. Comprehensive neurologic assessment could not be conducted in the EVD treatment unit because of limited access to imaging and other diagnostic tools. Urgent interviews by DOHMH of family members in Liberia revealed no additional EVD risk exposures in Liberia and suggested the patient felt ill during the week before traveling to New York City. After a negative Ebola real-time PCR result, isolation precautions were discontinued, and the patient was transferred to the ICU for management of cerebral malaria, which included computed tomography of the brain and electroencephalogram monitoring for seizure activity—modalities that were unavailable in the EVD treatment unit. DOHMH did not perform a second PCR test for EVD because the patient had an alternative diagnosis of malaria and because his illness onset was more than 72 hours before the first EVD test.
Case 2
In July 2015, an 81-year-old man had onset of fever and weakness 2 days after arriving from Guinea; his family called an ambulance, and he was transported by emergency medical services to a designated EVD treatment center, then transferred to the hospital’s EVD treatment unit. His family denied EVD risk exposures in Guinea. His white blood cell count was 18 × 103/μL (normal is 3.5-10.5 × 103/μL), and his platelets were 138 × 103/μL (normal is 150-450 × 103/μL). Malaria antigen and thin smear were negative. Based on a clinical examination, treating physicians suspected pneumonia but were not able to confirm the diagnosis with chest radiography in the EVD treatment unit; the hospital did not have approved methods for disinfecting large equipment, such as portable radiography, after use in the EVD treatment unit. Consequently, the patient was treated empirically with antibiotics. The patient had a negative Ebola real-time PCR test and was transferred to a general medical floor, where a portable unit radiograph revealed right lower lobe pneumonia. DOHMH did not request a second specimen for EVD testing because the patient had a clinical presentation that was consistent with bacterial pneumonia and improved after receiving antibiotic therapy.
Lessons Learned
DOHMH conducted active surveillance for approximately 5300 individuals and identified 112 persons who met EVD PUI criteria. Clinical and epidemiological features of EVD had to be reconciled with the recognition that most illness among travelers from EVD-affected countries would likely be attributable to other causes, such as malaria, 15 and that delays in care for non-EVD illness might lead to adverse outcomes. 16 DOHMH facilitated the evaluation for PUIs at area hospitals and communicated frequently with health care providers managing hospitalized PUIs. A consistent approach to PUI triage that used a core team of on-call physicians was crucial to directing patients to appropriate care while minimizing unnecessary burden to health care facilities.
The large number of health care facilities (n = 25) that managed at least 1 PUI and the relatively few PUIs (n = 10) who were tested for Ebola were 2 distinctive characteristics of triage and management of PUIs in New York City. Although there were 4 designated EVD treatment units in New York City, PUIs were evaluated at 21 other New York City facilities because several patients went to their nearest or preferred health care facility before DOHMH was aware of their illness. In some other jurisdictions, PUIs were assessed at a single facility by using standardized procedures. 17 In New York City, because of the large number of acute care facilities, it was challenging to ensure that all health care facilities were prepared to isolate and stabilize patients suspected of EVD according to the New York State Health Commissioner’s mandate. 7
Only 10 of 112 (9%) PUIs were tested for EVD in New York City. Testing every patient who met criteria for PUI would have imposed a substantial strain on health care facilities, emergency medical services, and the DOHMH Public Health Laboratory, particularly when most travelers did not have direct contact with a person with EVD and most illnesses were likely due to more common causes of fever in persons returning from sub-Saharan Africa, such as malaria. Our experience of testing a relatively small proportion of PUIs is similar to that described in Minnesota, where 2 of 29 (7%) patients with signs or symptoms consistent with EVD were tested. 18 However, among EVD-related clinical inquiries received by CDC from July 9 through November 15, 2014, 61 of 118 (52%) persons were tested (although not all patients tested met PUI criteria). 16 In Spain, 4 of 11 PUIs were tested. 19 In a US academic medical center, 8 of 25 (32%) PUIs were tested. 17 In New York City, before offering EVD testing, especially for patients who had illnesses compatible with other more common pathogens and no known direct exposure to EVD, DOHMH worked with providers to facilitate timely evaluation for alternate diagnoses or to encourage observation of patients under appropriate infection control precautions to determine the course of illness. Having a rapid point-of-care EVD test administered in the hospital might improve preparedness for future EVD outbreaks.
Our experience has implications for the nation’s strategy in preparedness for management of high-consequence pathogens such as Ebola virus. This strategy relies on a small subset of regional centers with high-level expertise in biocontainment and a larger number of designated treatment centers that are capable of isolating and stabilizing patients until they can be transferred to a regional center. The EVD outbreak in the Democratic Republic of Congo that began in August 2018 and is ongoing is a reminder that the clinical and public health communities need to strengthen partnerships and prepare health care workers to promptly recognize and respond to PUIs who initially may present to any health care facility in the United States. Therefore, a crucial element of preparedness for high-consequence pathogens is the community and frontline setting, where consistent screening protocols and clear procedures for patient isolation and notification of public health are critically important. Our finding that 92 of 5359 (2%) persons under active monitoring met PUI criteria provides a useful estimate for public health officials trying to anticipate how many travelers from Ebola-affected countries may have febrile illness requiring triage and possible medical evaluation.
Limitations
This case study had several limitations. DOHMH did not review most medical records; as such, details of PUI clinical presentation and outcomes were limited in some cases. It is possible that not all persons who met the PUI case definition in New York City were identified by DOHMH if they did not know how to measure their temperature using the thermometers provided or if they did not report signs and symptoms to health care providers or active-monitoring staff members. Barriers and delays in care for PUIs were anecdotal and not quantifiable.
Conclusion
DOHMH partnerships with CDC, the New York City Fire Department Bureau of Emergency Medical Services, and New York City health care facilities were crucial to preparedness and response. Our cadre of on-call physicians enabled us to respond to PUIs and have a core on-call team that facilitated communication and consistent processes. Active monitoring identified many but not all PUIs; even after initiation of active monitoring, 25% of patients under monitoring presented to health care facilities or called 911 without notifying the active-monitoring call center first, emphasizing the importance of direct and frequent outreach to community practitioners. Home isolation of PUIs with mild illness allowed us to maintain contact with patients while minimizing the need for unnecessary and costly interaction with the health care system. Although New York City had designated EVD treatment centers, all New York City hospitals had to be prepared to receive and offer initial care for PUIs. Close collaboration between public health and frontline health care workers is essential for future scenarios in which a disease is both highly lethal and highly transmissible in health care settings.
Footnotes
Authors’ Note
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention (CDC).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication was supported (in part) by the Epidemiology and Laboratory Capacity (ELC) for Infectious Diseases Cooperative Agreement (grants 2014 ELC: 3U50CK000407-01; 2015 ELC: NU50CK000407-02) funded by CDC and the Public Health Emergency Preparedness Cooperative Agreement (grants 2014: 5U90TP000546-03; 2015: 5U90TP000546-04) funded by CDC. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of CDC or the US Department of Health and Human Services.
